Abstract
Tumor metastasis is a key factor of therapeutic failure in tumor patients, but the underlying molecular mechanism remains to be explored and novel effective curative strategies are urgently required. Emerging evidence suggests that sporoderm-removed Ganoderma lucidum spore powder can suppress tumor growth and metastasis. However, the molecular mechanisms of action remain elusive. In the present study, we investigated the effects and mechanisms of sporoderm-removed Ganoderma lucidum spore powder against esophageal squamous cell carcinomas (ESCC). The expression of MCP-1 in esophageal squamous cell carcinoma cells was detected by Western blotting. The MTS assay was used to assess the esophageal squamous cell carcinoma cells viability. The clone formation assay was used to evaluate to the proliferation ability of KYSE140 and KYSE510 cells. Apoptosis and the cell cycle were analyzed by flow cytometry. Wound healing and Transwell assays were used to analyze the migration of KYSE140 and KYSE510 cells. Invasion was also analyzed by the Transwell assay. The expressions of PI3K, AKT/p-AKT, Erk/p-Erk, JNK1, and mTOR were detected by Western blotting. We found that the MCP-1 protein was highly expressed in KYSE140 and KYSE510. In addition, sporoderm-removed Ganoderma lucidum spore powder treatment was found to inhibit esophageal squamous cell carcinoma cell proliferation, to block the cell cycle, to induce cell apoptosis and to inhibit cell migration and invasion. Finally, we found that sporoderm-removed Ganoderma lucidum spore powder decreased the expression of PI3K/AKT/mTOR and Erk signaling pathways. Taken together, these findings demonstrate that sporoderm-removed Ganoderma lucidum spore powder suppresses esophageal squamous cell carcinomas by involving MCP-1, regulated by PI3K/AKT/mTOR and Erk signal pathways.
Keywords
Introduction
Esophageal squamous cell carcinoma (ESCC) is one of the most diagnosed cancers and the sixth leading cause of cancer-associated deaths worldwide. ESCC is the dominant histological type of esophageal cancer and is a major public health burden in China. 1 ESCC is a severe malignancy owing to its aggressive nature and very poor survival rate, with a 5-year survival rate of less than 30%. 2 Although the last 2 decades have witnessed significant progress in screening and curative treatments techniques such as surgical resection and chemoradiotherapy, the prognosis for patients with ESCC remains poor. There is dire need to identify potential biomarkers with higher prognostic accuracies and new drugs for patients with ESCC.
Traditional Chinese medicine (TCM) is an important resource for anti-cancer drug research. Traditional Chinese medicine may effectively remove tumor cells from the body without disturbing the normal function of the body organs. Ganoderma lucidum (Curtis) P. Karst is a medicinal and edible fungus and also a traditional Chinese herb with high medicinal value. Recently, Ganoderma lucidum has been reported to possess anticancer activities. 3 According to Li et al 4 Ganoderma lucidum spores, which are the essence of Ganoderma lucidum, reduced the migration of cholangiocarcinoma TFK-1 cells by inhibiting TGF-1-induced epithelialization and targeting the NF-κB signaling pathway. Sporoderm-broken Ganoderma lucidum spores inhibited protein kinase B / mammalian target of rapamycin (AKT/mTOR) signal transduction. In addition, lung cancer A549 cells were blocked in the G2/M phase, and the expressive activity of cyclin and anti-apoptotic proteins were inhibited, which promoted cell apoptosis. 5 Moreover, the bioactive substances of Ganoderma lucidum include polysaccharides, triterpenoids, amino acids, peptides, fatty acids, oligosaccharides, and trace elements. Among these active components, triterpenoids and polysaccharides were major active components to exert anticancer activities.6,7 Interestingly, sporoderm-removed Ganoderma lucidum spore powder, which contains much more polysaccharide and triterpenoids than Ganoderma lucidum spore and sporoderm-broken Ganoderma lucidum spores, has demonstrated antitumor activities in both in vivo and vitro studies.8,9 However, the anti-cancer effect and mechanism of sporoderm-removed Ganoderma lucidum spore powder in esophageal squamous cell carcinoma has not been clarified.
In our previous study, it was found that the expression of monocyte chemotactic protein-1 (MCP-1) in non-small cell lung cancer was associated with sporoderm-removed Ganoderma lucidum spore powder treatment. Monocyte chemotactic protein-1 (MCP-1/CCL2) is a 76 amino-acid peptide, secreted by fibroblasts, endothelial and epithelial cells, monocytes, and various tumor cells. 10 Accumulating evidence suggests that MCP-1 may be involved in tumor growth, metastasis and angiogenesis. 11 Various studies have shown that MCP-1 promotes tumor growth by regulating the PI3K/AKT/mTOR, MAPK/ERK, and STAT3 signaling pathways.10,12 In addition, MCP-1 has been shown to correlate with head and neck cancer (HNC) metastases through regulating PI3K/AKT/mTOR and MAPK/ERK pathways. 13 Esophageal squamous cell carcinoma (ESCC) is a primary squamous cell carcinoma of the head and neck, which often metastasizes to the neck and forms HNC. It has been reported that the expression of MCP-1 is significantly increased in esophageal squamous cell carcinoma compared with normal esophageal mucosal tissue. 14 MCP-1 promotes angiogenesis of esophageal squamous cell carcinoma through recruitment of macrophages, which produce angiogenic factors. In addition, the expression of MCP-1 in vascular endothelial cells is also involved in angiogenesis of ESCC. 15 MCP-1 can also increase the expression of deoxypurine/pyrimidine endonuclease-1, which promotes the expression of vascular endothelial growth factor and angiogenesis by upregulation of COX-2, and contributes to the development of ESCC. 16 Furthermore, the 5-year survival rate of ESCC patients with high MCP-1 expression were significantly lower than that of patients with low MCP-1 expression. 14 In our previous study, we also found that MCP-1 was highly expressed in ESCC cell lines, including KYSE70, KYSE140, KYSE410, KYSE450, KYSE510, and KYSE680. Therefore, we hypothesized that sporoderm-removed Ganoderma lucidum spore powder might exert an anti-esophageal squamous cell carcinoma effect by regulating the expression of MCP-1.
Materials and Methods
Cell Culture, Reagents and Treatments
The human esophageal squamous cell carcinoma cell lines YES2, KYSE30, KYSE70, KYSE140, KYSE150, KYSE180, KYSE410, KYSE450, KYSE510, and KYSE680 were kindly provided by Dr. Y. Shimada (Kyoto University). All the cells were cultured in RPMI 1640 (Lonza, Switzerland), supplemented with 10% fetal bovine serum (Gibco, USA) and 1% penicillin/streptomycin (Gibco, USA). Both types of ESCC cells were maintained in a humidified incubator at 37°C with 5% CO2.
Sporoderm-removed Ganoderma lucidum spore powder was provided by Zhejiang Shouxiangu Pharmaceutical Co., Ltd. The main active components in sporoderm-removed Ganoderma lucidum spore powder were polysaccharides (1.81%) and triterpenoid acid (1.31%). First, we measured and transferred 2.0 g sporoderm-removed Ganoderma lucidum spore powder to a clean flask, added 50 mL RPMI 1640 (Lonza, Switzerland) into the flask, then sealed it with a cap in a 37°C water bath for 30 minutes. After this, we sonicated the spore powder suspension at 135 W, 20 kHz for 60 minutes and transferred the sonicated suspension into proper clean tubes and centrifuged at 4000 r/minute for 2 minutes. Finally, we took the supernatant fluid and stored it at −20°C. During the experiments, we diluted the sporoderm-removed Ganoderma lucidum spore powder with RPMI 1640 (Lonza, Switzerland) at 400, 800, and 1200 mg/µL, with RPMI 1640 as control media.
Cell Proliferation and Apoptosis Assay
Cells seeded in 96-well plates were treated with sporoderm-removed Ganoderma lucidum spore powder of different concentrations. Cell proliferation was assessed after 24, 48, and 72 hours after treatment using the MTS assay (Promega) according to the manufacturer’s instructions. Apoptosis was assessed after 72 hours after treatment using an Annexin V-FITC apoptosis detection kit (Beyotime, China) according to the manufacturer’s instructions.
Colony Formation Assay
ESCC cells were seeded in 6-well plates at a density of 5000 cells per well. These cells were cultured in RPMI 1640 supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin with different concentrations of sporoderm-removed Ganoderma lucidum spore powder. After 2 weeks, the cultures were washed with pre-cooled PBS, fixed with methanol and stained with a 0.1% crystal violet solution for 30 minutes. The colonies were examined and calculated automatically by Image-Pro Plus.
Cell Cycle Assay
After treatment with different concentrations of sporoderm-removed Ganoderma lucidum spore powder for 72 hours, 1 × 106 cells were collected, trypsinized, and fixed in 70% ethanol overnight. Then the cells were washed 3 times with pre-cooled PBS and incubated with a PI-staining solution with RNase A (BD Biosciences, USA) for at least 15 minutes at room temperature before analysis. The cells were run on a FACS can cytometer (BD Biosciences, USA) in accordance with the manufacturer’s guidelines.
Cell Migration Assays
The wound healing assay is one of the earliest methods developed to study directional cell migration in vitro. Cells were seeded in a 6-well plate and allowed to attach overnight to 80% confluence. Subsequently, cell monolayers were wounded by 200 µL pipette tips and washed with 1 × PBS 3 times to remove floating cells. Cells treated with different concentrations of sporoderm-removed Ganoderma lucidum spore powder were then incubated in RPMI 1640 medium. Cells migrated into the wound surface and the number of migrating cells was determined under an inverted microscope at 0, 12, and 24 hours. Three randomly chosen fields were analyzed for each well. The percentage of migration was expressed using untreated wells as 100%. All the independent experiments were run in triplicate.
Transwell Migration Assays
Migration of cells was assessed in Transwell cell culture chambers with 6.5 mm diameter polycarbonate membrane filters containing 8 μm pore size (Neuro Probe, Gaithersburg, MD, United States). In total, 5 × 105 KYSE140 and KYSE510 cells in 200 μL of serum-free and different concentrations of sporoderm-removed Ganoderma lucidum spore powder medium were added to the upper chamber of the device, and the lower chamber was filled with 600 μL conditioned media (with 20% FBS added) of control cells. After 24 hours of incubation at 37°C, the non-migrated cells were removed from the upper surface of the membrane with a cotton swab. The filters were then fixed in methanol for 10 minutes, stained with crystal violet solution for 1 hour and counted. Five random microscopic fields (×100) were counted per well and the mean was determined.
Cell Invasion Assay
A Matrigel invasion assay was carried out to examine cancer cell invasion. At first, Transwell upper chambers were loaded with 0.1 mL Matrigel (Becton Dickinson, Bedford, MA) at 37°C for 1 hour. After this, cells were treated with different concentrations of sporoderm-removed Ganoderma lucidum spore powder medium for 24 hour, and cells were trypsinized and suspended at a final concentration of 5 × 105 cells/mL in serum free RPMI 1640 medium. Cell suspensions were then placed in the upper chamber and medium with 20% fetal bovine serum was added in the lower chamber. Following incubation at 37°C with 5% CO2 for 24 hour, non-invading cells on the upper surface were removed with a cotton swab. All the cells were stained using crystal violet staining and counted under an inverted microscope. We selected ten random views to count the cells and all the independent experiments were repeated 3 times.
Western Blotting
Protein extraction and western blotting were performed as described previously. 17 Briefly, proteins were isolated by RIPA lysis buffer (Beyotime, China). Then 24 μg protein was loaded and separated by 12% SDS-polyacrylamide gel electrophoresis and transferred onto a polyvinylidene difluoride membrane and incubated with antibodies: bax (#2774), bcl-2 (#3498), cleaved caspase-3 (#9661), cleaved caspase-8 (#8592), cleaved-PARP (#5625), p21 (#2947), p27 (#3686s), CDK1 (#4539), E-cadherin (#14472), PI3K (#17366), AKT (#4691), Erk (#4370), JNK1 (#4668), mTOR (#2983), and GAPDH (#51332). These antibodies were purchased from Cell Signaling Technology. MMP2 (sc-13595) and MMP9 (sc-21736) were purchased from Santa and MCP-1 (66272-1-lg) was purchased from Proteintech. The chemiluminescence signals were detected with an Amersham Imager 600 (GE, USA).
Statistical Analysis
All the experiments were performed in duplicate and repeated 3 times. Student’s t-test and 1-way ANOVA were used for statistical analysis of the in vitro data. Comparisons of the different groups were performed with the Student’s t-test. P < .05 was considered to be statistically significant for all data.
Results
MCP-1 Is Upregulated in Esophageal Squamous Cell Carcinoma KYSE70, KYSE140, KYSE180, KYSE410, KYSE450, KYSE510 and KYSE680 Cells
In our previous work, we found that the expression of MCP-1 in non-small cell lung cancer was closely related to the sporoderm-removed Ganoderma lucidum spore powder. In addition, MCP-1 has been shown to correlate with head and neck cancer (HNC) metastases through regulating PI3K/AKT/mTOR and MAPK/ERK pathways. 13 Esophageal squamous cell carcinoma is a primary squamous cell carcinoma of the head and neck, which often metastasizes to the neck and forms HNC. Studies have found that the expression of MCP-1 is significantly increased in ESCC compared with normal esophageal mucosal tissue. 14 Therefore, we hypothesize that sporoderm-removed Ganoderma lucidum spore powder might exert an anti-esophageal squamous cell carcinoma effect by regulating the expression of MCP-1. In order to investigate the effect of sporoderm-removed Ganoderma lucidum spore powder in ESCC, we first checked the expression pattern of MCP-1 in esophageal squamous cell lines by western blotting. We found that expression level of MCP-1 was increased in KYSE70, KYSE140, KYSE410, KYSE450, and KYSE510 cells (Figure 1).

High expression of MCP-1 in human esophageal squamous cell carcinoma cell lines.
Sporoderm-Removed Ganoderma lucidum Spore Powder Inhibits ESCC Cell Proliferation
We treated ten ESCC cell lines (YES2, KYSE30, KYSE70, KYSE140, KYSE150, KYSE180, KYSE410, KYSE450, KYSE510, and KYSE680) with sporoderm-removed Ganoderma lucidum spore powder at concentrations ranging from 50 to 1000 μg/mL for 24, 48, and 72 hour, respectively. And then we checked cell viability with the MTS assay. Among all the tested cell lines, KYSE140 and KYSE510 showed higher sensitivity under sporoderm-removed Ganoderma lucidum spore powder treatment; the inhibitory effect was dose and time-dependence (Figure 2A and B). At 72 hour, the inhibitory concentration of 50% of cells (IC50) was 945 and 771 µg/mL in KYSE140 and KYSE510 cells, respectively. Combined with the high expression of MCP-1 in esophageal squamous cell carcinoma cells KYSE70, KYSE140, KYSE410, KYSE450, and KYSE510 cells (Figure 1), we selected KYSE140 and KYSE510 cells for further experiments.
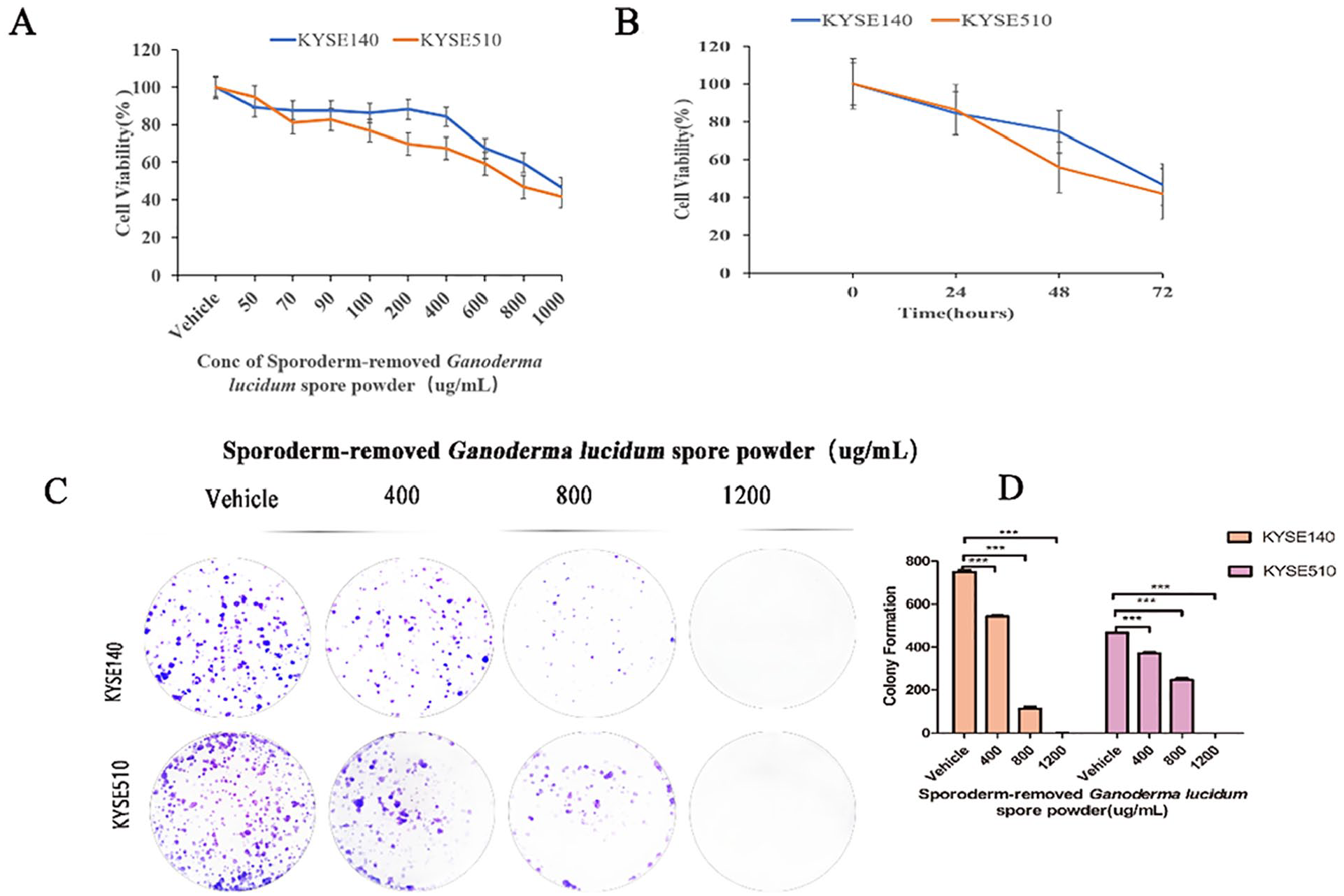
Inhibition of proliferation of human esophageal squamous cell carcinoma cell lines KYSE140 and KYSE510 treated by sporoderm-removed Ganoderma lucidum spore powder. (A) KYSE140 and KYSE510 cells were treated with the indicated concentrations of sporoderm-removed Ganoderma lucidum spore powder for 72 h, cell viability was assessed using MTS assay. IC50 values were calculated using the GraphPad Prism 5.0 software. Data are presented as mean ± SD. (B) KYSE140 and KYSE510 were treated with 1000 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 24, 48, and 72 hour, respectively, cell viability was assessed using MTS assay. (C) Results of colony formation assays for KYSE140 and KYSE510 cells. ESCC cells were treated with the indicated concentrations of sporoderm-removed Ganoderma lucidum spore powder for 14 days. (D) The quantification of the cell colonies in (C) is presented as the mean percentage of viable cells (mean ± SD), averaged from 3 independent experiments, each with 3 replicates per condition.
Next, we performed the cell colony formation assay using KYSE140 and KYSE510 cells to assess cell population dependence and proliferation capacity. After being seeded in 6-well plates and colony formatted for 2 weeks, ESCC cells displayed a decreased number of colonies with increased sporoderm-removed Ganoderma lucidum spore powder concentration. Figure 2C and D show results of each colony formation assay. Overall, according to the results of the MTS and cell colony formation assays, these findings suggest that sporoderm-removed Ganoderma lucidum spore powder treatment, in a dose-dependent manner, significantly inhibits ESCC cell proliferation and colony formation.
Sporoderm-Removed Ganoderma lucidum Spore Powder Decreases MCP-1 Expression
Based on our previous findings, the expression of MCP-1 in non-small cell lung cancer was closely related to sporoderm-removed Ganoderma lucidum spore powder. We were further interested in investigating the activation of MCP-1 in esophageal cancer cells KYSE140 and KYSE510 after sporoderm-removed Ganoderma lucidum spore powder treatment. The results demonstrated that sporoderm-removed Ganoderma lucidum spore powder reduced the expression of MCP-1 in KYSE140 and KYSE510 (Figure 3).
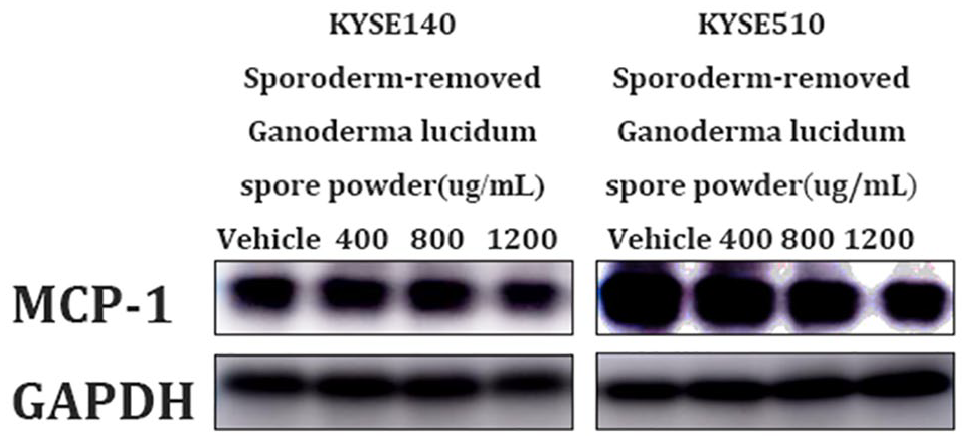
MCP-1 was inhibited in KYSE140 and KYSE510 treated by sporoderm-removed Ganoderma lucidum spore powder. Western blotting analysis of MCP-1 was performed when KYSE140 and KYSE510 were treated with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 72 hours.
Sporoderm-Removed Ganoderma lucidum Spore Powder Induces Cell Cycle Arrest and Apoptosis in ESCC Cells
After getting confirmation of the sensitivity of KYSE140 and KYSE510 cell lines under sporoderm-removed Ganoderma lucidum spore powder treatment, we were further interested in determining the underlying molecular mechanism of sporoderm-removed Ganoderma lucidum spore powder inhibition of the proliferation of ESCC cells KYSE140 and KYSE510. To this end, we detected the cell cycle and apoptosis after sporoderm-removed Ganoderma lucidum spore powder treatment. We found that sporoderm-removed Ganoderma lucidum spore powder induced cell cycle arrest in the S phase in KYSE140 and G1 phase in KYSE510 (Figure 4A and B), the data were analyzed by Mod Fit 5.0 (Figure 4C). At the same time, we also found that the expression of CDK1, CDK7, p21, and p27 were suppressed by sporoderm-removed Ganoderma lucidum spore powder in a concentration-dependent manner. However, sporoderm-removed Ganoderma lucidum spore powder did not affect the expression of CDK4 (Figure 4D).

Sporoderm-removed Ganoderma lucidum spore powder arrested cell cycle in KYSE140 and KYSE510 cells. (A) KYSE140 and KYSE510 cells were treated with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 72 hours, and then stained with DAPI and subjected by FACS. (B and C) cell cycle was analyzed by Mod Fit 5.0. All data are presented as the mean ± SD. A 1-way analysis of variance, followed by a Tukey’s post-hoc test, was used to compare the different groups. *P < .05, **P < .01, ***P < .001 versus vehicle. (D) The expression of CDK1, CDK4, CDK7, p21, and p27 were examined by Western blotting. Lysates from KYSE140 and KYSE510 cell were probed with antibodies after treatment with sporoderm-removed Ganoderma lucidum spore powder for 72 hours.
In addition, the apoptosis rate increased as we enhanced the dose of sporoderm-removed Ganoderma lucidum spore powder (Figure 5A and B). Next, we examined the expression level of apoptosis markers by Western Blot. We found that sporoderm-removed Ganoderma lucidum spore powder increased the expression level of Fas (Figure 5C).

Sporoderm-removed Ganoderma lucidum spore powder induces apoptosis in KYSE140 and KYSE510 cells. (A) KYSE140 and KYSE510 cells were treated with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 72 hours. Flow cytometry analyzed apoptotic cells stained by Annexin V and PI. (B) data are presented as the mean ± SD. A 1-way analysis of variance, followed by a Tukey’s post-hoc test, was used to compare the different groups. *P < .05, **P < .01, ***P < .001 versus vehicle. (C) Expression of the apoptosis-associated protein Fas was determined by western blotting.
Sporoderm-Removed Ganoderma lucidum Spore Powder Inhibited the Metastasis and Invasion of KYSE140 and KYSE510 Cells
After confirming that sporoderm-removed Ganoderma lucidum spore powder treatment played a crucial role in regulating cell cycle arrest and apoptosis of ESCC cells, we were further interested in investigating the inhibitory effect of sporoderm-removed Ganoderma lucidum spore powder on the migration of KYSE140 and KYSE510 cells. In this line, the confluent monolayer was scraped by Scale bar, 200 μm, to create a scratch wound, and the cells migrated to the denuded zone. The levels of the wound closure area were analyzed after 0, 12, and 24 hours incubation with sporoderm-removed Ganoderma lucidum spore powder. As shown in Figure 6A, the results indicated that treatment with sporoderm-removed Ganoderma lucidum spore powder significantly reduced migration of KYSE140 and KYSE510 cells to the denuded zone as compared to control cells in a concentration-dependence, indicating that sporoderm-removed Ganoderma lucidum spore powder effectively inhibited the motility of KYSE140 and KYSE510 cells.

Sporoderm-removed Ganoderma lucidum spore powder inhibited migration and invasion in KYSE140 and KYSE510 cells. (A) KYSE140 and KYSE510 cells were treated with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 72 hours. Cell migration was evaluated by the wound healing assay. Scale bar, 200 µm. (B and C) Gap Width data were presented as the mean ± SD. A 1-way analysis of variance, followed by a Tukey’s post-hoc test, was used to compare the different groups. *P < .05, **P < .01, ***P < .001 versus vehicle. (D) KYSE140 and KYSE510 cells, pretreated with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 24 hours, were plated onto the apical side of filters in serum free medium containing either vehicle or sporoderm-removed Ganoderma lucidum spore powder, medium containing 20% FBS was placed in the basolateral chamber to act as a chemoattractant for 24 hour. Cells on the bottom of the filter were stained by 0.5% crystal violet and then counted. (E) Quantification of the migrated cells in (D) was displayed on the right. The results were displayed as the mean ± SD. A 1-way analysis of variance, followed by a Tukey’s post-hoc test, was used to compare the different groups. *P < .05, **P < .01, ***P < .001 versus vehicle. (F) Before experiment, transwell chamber was covered with matrix glue. KYSE140 and KYSE510 cells, pretreated with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 24 hours, were plated onto the apical side of filters in serum free medium containing either vehicle or sporoderm-removed Ganoderma lucidum spore powder, medium containing 20% FBS was placed in the basolateral chamber to act as a chemoattractant for 24 hours. Cells on the bottom of the filter were stained by 0.5% crystal violet and then counted. (G) Quantification of the invasive cells in (F) was displayed on the right. The results were displayed as the mean ± SD. A 1-way analysis of variance, followed by a Tukey’s post-hoc test, was used to compare the different groups. *P < .05, **P < .01, ***P < .001 versus vehicle. (H) The expression of MMP2 and MMP9 that reflect cell migration and invasion was examined by Western blotting. Lysates from KYSE140 and KYSE510 cells were probed with antibodies after treatment with vehicle, 400, 800, and 1200 µg/mL of sporoderm-removed Ganoderma lucidum spore powder for 72 hour.
In addition, the inhibitory effect of sporoderm-removed Ganoderma lucidum spore powder on cell migration was also determined by the Transwell assay. As shown in Figure 6D, compared with the vehicle, migration of KYSE140 and KYSE510 cells treated with sporoderm-removed Ganoderma lucidum spore powder was inhibited. This inhibition occurred in a dose-dependent manner. The results were analyzed by ImageJ and GraphPad prism 5.0 (Figure 6E).
Moreover, we attempted to determine whether the inhibitory effects of sporoderm-removed Ganoderma lucidum spore powder on cell migration were associated with decreases of KYSE140 and KYSE510 cell invasion using a Matrigel invasion assay. We found that results were consistent with those of the wound-healing assay and Transwell assay, and demonstrated that sporoderm-removed Ganoderma lucidum spore powder significantly inhibited the invasion of KYSE140 and KYSE510 cells with dose-dependence (Figure 6F).
Furthermore, we observed protein expression levels of MMP2 and MMP9, which are associated with invasion and metastasis. The results showed that sporoderm-removed Ganoderma lucidum spore powder treatment led to decreased expression level of both MMP2 and MMP9 (Figure 6H).
Sporoderm-Removed Ganoderma lucidum Spore Powder Affects MCP-1 and Possibly Has an Anti-Esophageal Cancer Role Through PI3K/AKT and Erk Signaling Pathways
MCP-1 has been found to stimulate the PI3K/AKT and Erk pathways. 13 To elucidate the mechanism by which sporoderm-removed Ganoderma lucidum spore powder affects MCP-1 and has an anti-esophageal cancer role through PI3K/AKT/mTOR and Erk signaling pathways, we investigated the effect of sporoderm-removed Ganoderma lucidum spore powder on PI3K/AKT and Erk signaling pathways. We found that sporoderm-removed Ganoderma lucidum spore powder reduced the expression of PI3K/AKT/mTOR and Erk signaling pathways in a dose-dependence in ESCC cells (Figure 7).

The effect of sporoderm-removed Ganoderma lucidum spore powder on PI3K/AKT/mTOR and Erk signaling pathways. The expression of PI3K, AKT, p-AKT, Erk, p-Erk, JNK1, and mTOR were assessed using the corresponding antibodies via Western blotting. The results were repeated with at least three independent experiments.
Discussion
Despite huge progress in surgery, radiotherapy and chemotherapy, esophageal squamous cell carcinoma remains a deadly malignancy with less than 30% of patients surviving for 5 years.18,19 Therefore, there is an urgent need to identify more effective therapeutic targets to enhance the treatment outcome of ESCC. Emerging evidence suggests that sporoderm-removed Ganoderma lucidum spore powder has great potential in the treatment of human cancers. 8 In this study, we found that sporoderm-removed Ganoderma lucidum spore powder inhibited the proliferation of esophageal squamous cell carcinoma KYSE140 and KYSE510 cells in a dose-dependence and time-dependence.
In terms of the anti-esophageal squamous cell carcinoma mechanism of action of sporoderm-removed Ganoderma lucidum spore powder, we found that MCP-1 was highly expressed in the esophageal squamous cell carcinoma cells KYSE70, KYSE140, KYSE410, KYSE450, KYSE510, and KYSE680 (Figure 1). This confirmed that MCP-1 was highly expressed in many tumors, 20 which would be consistent with previous published reports. In this study, we also found that sporoderm-removed Ganoderma lucidum spore powder inhibited the expression of MCP-1 in esophageal squamous cell carcinoma KYSE140 and KYSE510 cells and it played the same role in non-small cell lung cancer, which was found in our previous study. We speculated that sporoderm-removed Ganoderma lucidum spore powder may exert its anti-esophageal squamous cell carcinoma role by affecting MCP-1. Thus, we would further analyze the cell cycle, apoptosis, metastasis and invasion to verify the anti-esophageal squamous cell carcinoma effect and molecular mechanism of sporoderm-removed Ganoderma lucidum spore powder.
We found that the sporoderm-removed Ganoderma lucidum spore powder blocked the cell cycle of KYSE140 in the S phase and KYSE510 in the G1 phase, respectively. The different results between in KYSE140 and KYSE510 might be associated with cellular characteristics, which indicated that the sporoderm-removed Ganoderma lucidum spore powder acted on KYSE140 and KYSE510 cells through different pathways. However, we found that the sporoderm-removed Ganoderma lucidum spore powder consistently inhibited the expression of CDK1, CDK7, p21, and p27 in esophageal squamous cell carcinoma cells KYSE140 and KYSE510, but the effect on CDK4 expression was not significant. Previously, most researchers found that cancer cells would be arrested in the G2 phase subsequent to the induction of CDK1 loss, 21 whereas some reports described that meclofenamic acid down-regulated the expression of CDK1 through regulating the cellular m6A level, which caused cell cycle arrest in the G1/S transition and decreased of cell proliferation. 22 Sporoderm-removed Ganoderma lucidum spore powder may arrest the cell cycle of KYSE140 in the G1 phase and KYSE510 in the S phase by regulating the cellular m6A level, but this should be further explored. As for CDK7, our research found that sporoderm-removed Ganoderma lucidum spore powder, as an inhibitor of CDK7, inhibits the expression of CDK7 in KYSE140 and KYSE510, which blocks the cell cycle in the G1 or S phase. CDK7 has been known as a component of the CDK activating kinase (CAK) complex that contributes to cell cycle progression by phosphorylating other CDKs. 23 In terms of p21 and p27, though p21 and p27 have been shown to inhibit the growth of many cancers such as colon cancer, leukemia, brain tumors and breast-cancer cells.24,25 Sporoderm-removed Ganoderma lucidum spore powder downregulates the expression of p21 and p27 in KYSE140 and KYSE510 cells. The difference might be explained as p21 and p27 play crucial roles when cyclins bind to CDK (4, 6) because it also activates cyclin D/ CDKs (4 or 6) complex or cyclin E/ CDK2 complex and promotes the cell cycle. 26 When p21 and p27 are inhibited, it may prevent the formation of cyclin D/ CDKs (4 or 6) complex, and block the cell cycle in G1 or S phase.
A decrease in tumor growth and cell viability has been attributed to the induction of cell cycle arrest and apoptosis. 27 In this line, we investigated the impact of sporoderm-removed Ganoderma lucidum spore powder treatment in regulating expression of apoptosis markers and found that Fas was greatly active in both KYSE140 and KYSE510. Fas is an important promoter for apoptotic pathways, and the molecular mechanism of sporoderm-removed Ganoderma lucidum spore powder in regulating cell apoptosis may be the Fas signaling pathway. We would further explore the Fas signaling pathway and its downstream protein expression.
Tumor metastasis is closely related to prognosis and is quite vital in tumor treatment. Here, for the first time we revealed that sporoderm-removed Ganoderma lucidum spore powder inhibited the metastasis and invasion of esophageal squamous cell carcinoma KYSE140 and KYSE510 cells. Meanwhile, we also found that matrix metalloproteinase 2 (MMP2) and matrix metalloproteinase 9 (MMP9) were significantly inhibited in KYSE140 and KYSE510 that had been treated with sporoderm-removed Ganoderma lucidum spore powder. MMPs play a crucial role in cancer progression, including in metastasis and invasion, by degrading extracellular matrix. 28 MMP2 and MMP9 are key members of the MMP family. MMP2 allows tumor cells to metastasize through hydrolysis of extracellular matrix components, invading surrounding tissue. Research has demonstrated that increased expression of MMP9 in cervical cancer, renal cell carcinoma, and ESCC promotes the invasion and metastasis of tumor cells through the degradation of extracellular mechanisms. 29 Thus, we speculate that suppression of MMP2 and MMP9 may contribute to the inhibition of metastasis and invasion of KYSE140 and KYSE510 cells via proteolytic activities of the MMPs.
Various signaling pathways are strongly associated with cell proliferation, cell cycle arrest, apoptosis, cell metastasis and invasion of cancers. Here, we found that sporoderm-removed Ganoderma lucidum spore powder treatment led to downregulation of MCP-1, PI3K/AKT/mTOR, and Erk pathways in a dose-dependence. Previously, it was reported that MCP-1 functioned via PI3K/AKT and MAPK/Erk pathway in breast cancer. 30 Thus, we assumed that the anti-esophageal cancer mechanisms of sporoderm-removed Ganoderma lucidum spore powder may be affected by MCP-1 and regulated by PI3K/AKT/mTOR and Erk signaling pathways.
MCP-1 is ubiquitously expressed in adipose tissue, skeletal muscle and various cancers. 31 Overexpression of MCP-1 is associated with tumor cell proliferation and angiogenesis. 32 Based on our data, we are the first to propose that sporoderm-removed Ganoderma lucidum spore powder may involve MCP-1 against ESCC.
The PI3K/AKT/mTOR and Erk signaling pathways are quite crucial in regulating cell proliferation, invasion and other biological behaviors.33,34 PI3K, as a signaling protein with the catalytic activity within cells, will be activated under the action of extracellular cytokines, drugs, stress and other factors; it then phosphorylates PIP2 into PIP3 and promotes AKT activation so as to regulate the expression of a variety of cell proliferation and invasion-related genes.35-37 AKT is a protein kinase involved in multiple cellular processes, including cell survival, proliferation, metabolism, apoptosis and tumorigenesis. 38 In addition, by activating the mTOR complex 1 (mTORC1) and downstream translation factors, 39 AKT activity strongly enhances protein synthesis and cell proliferation. Thus, p-AKT suppression has been reported to inhibit proliferation and induces apoptosis in multiple tumor cells. 40 Except for AKT, Erk/p-Erk have been widely reported to promote tumor development. Erk/p-Erk promote proliferation, survival and the epithelial-mesenchymal transition (EMT), as well as metastasis. 41 Consistently in our research, sporoderm-removed Ganoderma lucidum spore powder affected the MCP-1 expression pattern and inhibited the expression of PI3K/AKT/mTOR and Erk signaling pathways. However, the direct mechanism by which sporoderm-removed Ganoderma lucidum spore powder inhibited these pathways was not determined.
Among the above pathways, the inhibition of PI3K, p-AKT, p-Erk, and mTOR is generally considered beneficial for promoting tumor killing. However, the function of the JNK1 in tumor killing is controversial.42,43 Some findings support the pro-oncogenic function of JNK1, while others suggest that JNK1 is a tumor suppressor, 44 but the result of the present study did not show obvious change.
Although we found novel findings, some limitations cannot be ignored. First, we investigated the potential role of sporoderm-removed Ganoderma lucidum spore powder treatment in regulating ESCC cell proliferation, cell cycle arrest, apoptosis, metastasis and invasion by affecting MCP-1 through PI3K/AKT/mTOR and Erk signaling pathways, but the activation of PI3K/AKT/ mTOR pathway in cells could not be checked due to various reasons. Second, although we identified that sporoderm-removed Ganoderma lucidum spore powder treatment affected MCP-1 expression pattern, we could not study the knockdown effects of MCP-1 using MCP-1 siRNA or anti-MCP-1 antibodies. Nevertheless, these novel findings provide first insights into the profound impact of sporoderm-removed Ganoderma lucidum spore powder treatment in regulating ESCC onset and progression.
Our Data
Our data indicated that sporoderm-removed Ganoderma lucidum spore powder treatment effectively inhibits the metastasis and invasion and also induces cell cycle arrest and apoptosis in KYSE140 and KYSE510 cells, thereby providing a promising therapeutic target. However, further research is needed to explore the molecular mechanism.
Footnotes
Authors’ Contributions
Jingyan Liu performed experiments, analyzed the data, and wrote the manuscript. Tao Zeng analyzed the data and reviewed the manuscript. All authors read and approved the final manuscript.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Start-up Fund for High-level Talents in Affiliated Hospital of Guangdong Medical University (grant No. 51301Z20200007) and Discipline construction project of Guangdong Medical University (grant No. 4SG21266P and grant No. 4SG21276P) to TZ. The funders had no role in study design, data collection and analysis, manuscript preparation, or decision to publish.
