Abstract
Background
MicroRNA (miR)-99a-5p acts as a tumor suppressor in several tumors, including bladder cancer and breast cancer, but its biological function in oral squamous cell carcinoma (OSCC) is poorly understood.
Methods
miR-99a-5p expression was determined in OSCC tissues and cell lines using quantitative reverse transcription polymerase chain reaction (RT-qPCR). Cell proliferation was assessed by the Cell Counting Kit-8 assay and colony formation assay. Wound healing and Transwell assays were used to analyze migration and invasion abilities, respectively, in OSCC cells. The luciferase reporter assay, RT-qPCR, and western blotting were used to determine the relationship between miR-99a-5p and isoprenylcysteine carboxylmethyltransferase (ICMT).
Results
miR-99a-5p expression in OSCC tissues and cell lines was significantly decreased compared with corresponding controls, and was significantly associated with clinical stage and lymph node metastasis in OSCC. Functional assays revealed that miR-99a-5p overexpression significantly inhibited the proliferation, migration, and invasion abilities of CAL-27 and TCA-8113 OSCC cells. miR-99a-5p was found to directly target ICMT, while ICMT restoration reversed the role of miR-99a-5p in OSCC cells.
Conclusions
Our results indicate that miR-99a-5p-mediates the down-regulation of ICMT, which could be used as a novel potential therapeutic target for OSCC treatment.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) is one of the most common malignant head and neck cancers, with approximately 0.3 million new cases diagnosed each year.1,2 Some of the treatments available for OSCC, including surgical resection, chemotherapy, and radiotherapy, have been improved, but patient outcomes remain unsatisfactory with only a 50% 5-year survival rate.3,4 Additionally, most patients with OSCC are first diagnosed at an advanced stage because of the lack of reliable and specific biomarkers, and thus face uncontrolled disease progression. 5 Therefore, elucidation of the molecular mechanism underlying OSCC progression is urgently needed to improve the prognosis for OSCC.
MicroRNAs (miRNAs/miRs) are a class of short non-coding RNA molecules of 18 to 25 nucleotides, which directly bind the 3′-untranslated region (3′-UTR) of target genes to induce mRNA degradation.6,7 Emerging evidence has indicated that miRNAs play an essential role in a diverse range of biological processes, such as proliferation, apoptosis, differentiation and invasion.
8
In recent years, aberrant miRNAs have been demonstrated to be critical modulators in OSCC progression by acting as oncogenes or tumor suppressors. For instance, Li et al.
9
found that miR-182-5p was up-regulated and promoted the growth and proliferation of OSCC cells by down-regulating CAMK2N1, while Peng et al.
10
revealed that miR-140-5p suppressed OSCC tumorigenesis by targeting p21-activated kinase 4
Chen et al. 11 used bioinformatic analysis to show that markedly lower miR‐99a‐5p expression was associated with poor survival in patients with head and neck squamous cell carcinoma (HNSCC). In fact, miR‐99a‐5p, as the major member of the miR‐99a family, is reported to be down-regulated in several types of cancers, including esophageal squamous cell carcinoma, 12 hepatocellular carcinoma, 13 and bladder cancer. 14 Functionally, miR-99a-5p markedly inhibits the growth of bladder cancer cells via dual inhibition of the mammalian target of rapamycin (mTOR) complex 1 (mTORC1) and mTORC2. 15 Additionally, benzyl isothiocyanate induced the up-regulation of miR-99a-5p decreased insulin-like growth factor 1 receptor (IGF1R), mTOR, and fibroblast growth factor receptor (FGFR)3, which are predicted targets of miR-99a-5p to suppress bladder cancer cell survival. 16 Moreover, miR-99a-5p was found to inhibit breast cancer progression and the cell cycle pathway by down-regulating cell division cycle 25A. 17
Isoprenylcysteine carboxylmethyltransferase (ICMT), a polytopic membrane protein restricted to the endoplasmic reticulum, was shown to contribute to multiple biological functions of tumor cells as its inhibition results in anti-cancer activities in pancreatic cancer, 18 leukemia, 19 and cervical cancer. 20 Several studies have reported an oncogenic role for ICMT in tumor cells, and it was found to be associated with microRNA (miR)-99a-5p by bioinformatics analysis. 21 We therefore speculated that miR-99a-5p might play an important role in OSCC cell function by regulating ICMT expression.
To validate our hypothesis, we first analyzed the expression of miR-99a-5p in OSCC tissues and cell lines. We next carried out a series of functional experiments to determine the role of miR-99a-5p in OSCC cell proliferation, migration, and invasion. Finally, we explored the association between miR-99a-5p and ICMT in OSCC cells to determine whether ICMT is a functional regulator in the control of cell proliferation, migration, and invasion by miR-99a-5p.
Materials and methods
Clinical samples
Thirty-six tumor specimens and matched adjacent tissues were collected from patients with OSCC undergoing surgical resection at The Yan’an Hospital of Traditional Chinese Medicine (Shaanxi, China) between March 2015 and October 2017. All enrolled patients received no radiotherapy or chemotherapy, and all provided written informed consent. Patient characteristics, including sex, age, and clinical stage are presented in Table 1. Tissue samples were immediately frozen in liquid nitrogen and stored at −80°C until analysis. This study was performed in accordance with the Declaration of Helsinki and was approved by the Yan’an Hospital of Traditional Chinese Medicine (Shaanxi, China; approval number YHM-87DA).
The relationship between microRNA-99a-5p expression and clinicopathological characteristics of patients with oral squamous cell carcinoma.

*p < 0.05.
Cell lines and transfection
The human normal oral keratinocyte hHOK cell line and OSCC cell lines (SCC-9, CAL-27, and TCA-8113) were purchased from American Type Culture Collection (Manassas, VA, USA). All cell lines were cultured in Dulbecco’s modified Eagle medium (DMEM) with 10% fetal bovine serum (FBS) (Gibco, Carlsbad, CA, USA) in a humidified incubator containing 5% CO2 at 37°C.
miR-99a-5p mimics and the corresponding negative control (miR-NC) were purchased from GenePharma Co., Ltd. (Shanghai, China). The pcDNA3.1 vector with a full-length ICMT open reading frame (pcDNA3.1-ICMT) was purchased from GenScript (Nanjing, China). Empty pcDNA3.1 vector was used as a control. For miR-99a-5p overexpression, CAL-27 or TCA-8113 cells were plated in six-well plates (2 × 105 cells/well) and transfected with miR-99a-5p mimics or miR-NC at a final concentration of 50 nM. For the rescue experiments, CAL-27 or TCA-8113 cells were co-transfected with miR-99a-5p mimics and pcDNA3.1-ICMT or pcDNA3.1. All transfections were performed using Lipofectamine 2000 (Invitrogen Corp., Carlsbad, CA, USA) according to the manufacturer’s instructions and were incubated for 48 hours.
Quantitative reverse transcription polymerase chain reaction (RT-qPCR)
Total RNA was extracted from tissues or cells using Trizol RNA reagent (Invitrogen Corp.). First-strand cDNA was reverse-transcribed from 2 µg total RNA using PrimeScript RT reagent (TaKaRa Biotech Corporation, Dalian, China). Quantitative real-time PCR was performed using SYBR Green qPCR SuperMix (Invitrogen) with the following PCR amplification conditions: 95°C for 3 minutes, followed by 40 cycles of 94°C for 10 s and 60°C for 30 s. Primer sequences were as follows: miR-99a-5p forward, 5′-
Cell proliferation assay
Transfected OSCC cells were seeded in 96-well plates at a density of 3000 cells per well and incubated with 10 µL cell counting kit (CCK)-8 solution (Dojin do Molecular Technologies, Inc., Rockville, MD, USA) per well at 37°C. Subsequently, the optical density value was read at a wavelength of 450 nm at the specified time points (0, 24, 48, and 72 hours) using a microplate reader (BioTek, Winooski, VT, USA).
Colony formation assay
Transfected OSCC cells were seeded in six-well plates at a density of 500 cells/well and cultured for 2 weeks to form colonies. Colonies were fixed in methanol for 15 minutes and stained with 0.5% crystal violet (Sigma, St. Louis, MO, USA) for 30 minutes at room temperature. They were then washed with phosphate-buffered saline, air dried, and photographed and counted under a light microscope in five random fields of view.
Wound healing assay
Transfected OSCC cells were seeded in six-well plates at a density of 4 × 105 cells per well and cultured in DMEM. When they reached 90% confluency, a scratch was artificially created using a 200-µL aseptic pipette tip on the OSCC cell monolayer. At 0 and 24 hours incubation, the scratch area was photographed using a light microscope. The relative migration distance was then calculated as follows: (area of original wound at 0 hours – area of wound at 24 hours)/area of original wound at 0 hours ×100%.
Transwell assay
A Transwell invasion assay was conducted to analyze the invasion ability of transfected OSCC cells. Approximately 5 × 104 cells re-suspended in serum-free medium were plated into the upper chambers of Matrigel-coated Transwell inserts (BD Biosciences, Franklin Lakes, NJ, USA). Complete medium with FBS was added to the bottom chambers as a chemoattractant. After 24 hours of incubation, cells that had invaded the bottom chambers were fixed with 20% methanol at 4°C for 30 minutes and stained with 0.1% crystal violet (Sigma-Aldrich, St. Louis, MO, USA). They were then counted under a fluorescence microscope (Olympus, Tokyo, Japan) in five randomly selected fields of view. The migration assay procedure was identical except that the Transwell insert was not coated with Matrigel.
Luciferase reporter assay
The association between miR-99a-5p and ICMT was predicted by TargetScan 7.1 (http://www.targetscan.org/vert_71) and confirmed by luciferase reporter assay. Wild-type and mutated sequences of the ICMT 3′UTR were cloned into the pmiRGLO vector (Promega, Madison, WI, USA) and named WT ICMT and MUT ICMT, respectively. Subsequently, OSCC cells were co-transfected with synthetic miR-99a-5p mimics or miR-NC and WT ICMT or MUT ICMT using Lipofectamine 2000 (InvitrogenCorp., Carlsbad, CA, USA). After 48 hours, luciferase activity was measured using the Dual-Luciferase Reporter Assay System (Promega). Relative luciferase activities were shown as the ratio of Renilla luciferase activity to firefly luciferase activity.
Western blot
Proteins were extracted with radioimmunoprecipitation assay lysis buffer containing a protease inhibitor (Sigma-Aldrich). After protein quantification by a bicinchoninic acid assay (Beyotime Biotech. Inc., Jiangsu, China), 30 µg protein was separated by 10% sodium dodecyl sulphate–polyacrylamide gel electrophoresis, transferred to a nitrocellulose membrane, and blocked with 5% non-fat milk for 1 hour. The membrane was incubated with primary antibodies against ICMT and GAPDH (Abcam, Cambridge, UK) at 4°C overnight. Following incubation with horseradish peroxidase-conjugated secondary antibodies (Abcam) for 2 hours at 37°C, protein signals were detected using a BeyoECL kit (Beyotime Biotech. Inc.) with GAPDH as the internal control.
Statistical analysis
All experiments were independently repeated at least three times and data are expressed as mean ± standard deviation. Statistical analysis was performed using SPSS 21.0 software (IBM Corp, Armonk, NY, USA). The chi-square test was used to evaluate the relationship between miR-99a-5p expression and clinical pathological parameters. The Student’s t-test or two-way analysis of variance followed by post-hoc Tukey’s test were used to assess differences among groups, with values of p < 0.05 representing statistical significance.
Results
miR-99a-5p was down-regulated in human OSCC tissues and cell lines
miR-99a-5p expression was determined using RT-qPCR in 36 OSCC tissues and matched adjacent healthy tissues. As shown in Figure 1a, miR-99a-5p expression was significantly down-regulated in OSCC tissues compared with matched adjacent tissues (p < 0.001). Patients with OSCC were classified into high expression (n = 18) and low expression groups (n = 18) based on the median value of miR-99a-5p in OSCC tissue samples (0.813). Chi-square analysis showed that miR-99a-5p expression was significantly associated with clinical stage and lymph node metastasis (p = 0.0023 and p = 0.0442, respectively; Table 1). Similarly, we observed that miR-99a-5p expression was significantly reduced in all three OSCC cell lines compared with the human normal oral keratinocyte hHOK cell line (p < 0.001; Figure 1b).

microRNA-99a-5p expression was reduced in oral squamous cell carcinoma (OSCC) tissues and cell lines. (a) The expression level of microRNA-99a-5p was significantly lower in 36 OSCC tissues compared with adjacent healthy tissues (***p < 0.001). (b) The expression of microRNA-99a-5p was significantly lower in OSCC cell lines compared with the normal oral keratinocyte cell line hHOK. Data are expressed as mean ± standard deviation. ***p < 0.001, compared with hHOK.
Overexpression of miR-99a-5p suppressed proliferation of OSCC cells
CAL-27 and TCA-8113 OSCC cell lines, demonstrating the lowest expression of miR-99a-5p, were selected for subsequent gain-of-function assays. RT-qPCR first confirmed that miR-99a-5p expression was significantly increased in the miR-99a-5p mimics-transfected group compared with the miR-NC-transfected group in both CAL-27 and TCA-8113 cells (p < 0.001; Figure 2a). Then, cell proliferation was assessed using the CCK-8 assay and colony formation assay. As depicted in Figure 2b, CAL-27 and TCA-8113 cells transfected with miR-99a-5p mimics had significantly lower cell proliferation than corresponding miR-NC groups (p < 0.001). Similarly, the colony formation ability was significantly impaired in both CAL-27 and TCA-8113 cells after miR-99a-5p overexpression (p < 0.001; Figure 2c). These data indicate that miR-99a-5p inhibits OSCC cell proliferation.
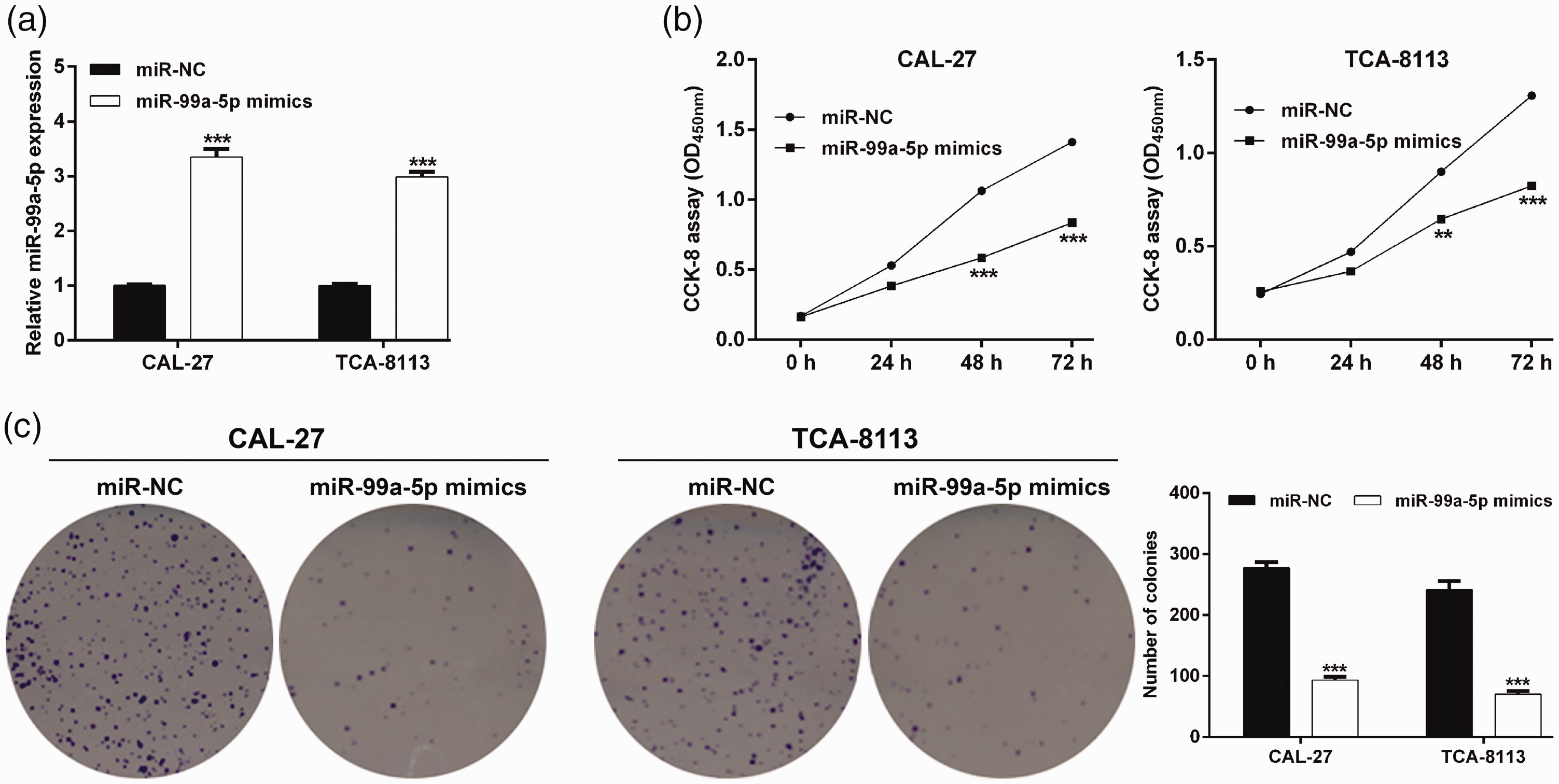
Overexpression of microRNA-99a-5p suppressed the proliferation of oral squamous cell carcinoma (OSCC) cells. (a) Expression of microRNA-99a-5p in CAL-27 and TCA-8113 cells was determined after transfection with microRNA-99a-5p mimics or microRNA-NC (negative control). The effect of microRNA-99a-5p on CAL-27 and TCA-8113 cell proliferation was evaluated by the cell counting kit‐8 assay (b) and colony formation assay (c). Data are expressed as mean ± standard deviation. **p < 0.01, ***p < 0.001, compared with microRNA-NC.
Overexpression of miR-99a-5p inhibited the migration and invasion ability of OSCC cells
We then evaluated the effects of miR-99a-5p overexpression on migration and invasion in OSCC cells. The wound healing assay showed that cell migration was significantly reduced following transfection with miR-99a-5p mimics compared with the control in both CAL-27 (p < 0.01; Figure 3a) and TCA-8113 (p < 0.01; Figure 3b) cells. In Transwell invasion experiments, miR-99a-5p overexpression significantly decreased the invasive ability in CAL-27 (miR-99a-5p mimics

Overexpression of microRNA-99a-5p suppressed the migration and invasion of oral squamous cell carcinoma (OSCC) cells. (a, b) Migration of CAL-27 and TCA-8113 cells following transfection with microRNA-99a-5p mimics or microRNA-NC (negative control) for 48 hours was determined by a wound healing assay (magnification, ×200). (c) Invasion of CAL-27 and TCA-8113 cells following transfection with microRNA-99a-5p mimics or microRNA-NC for 48 hours was analyzed using a Matrigel-based invasion assay (magnification, ×200). Data are expressed as mean ± standard deviation. **p < 0.01, ***p < 0.001, compared with microRNA-NC.
ICMT is the target of miR-99a-5p
We performed a target search using TargetScan 7.1 software which predicted that ICMT was a direct target gene of miR-99a-5p with possible binding sequences in the 3′-UTR of ICMT (Figure 4a). Next, luciferase reporter plasmids containing the WT or MUT 3′-UTR of ICMT were constructed to further confirm the potential interaction between miR-99a-5p and the 3′-UTR of ICMT. We found that miR-99a-5p overexpression significantly reduced luciferase activity in CAL-27 (p < 0.001; Figure 4b) and TCA-8113 (p < 0.01; Figure 4c) cells transfected with WT ICMT, but that luciferase activity was not affected following MUT ICMT transfection. RT-qPCR (p < 0.001; Figure 4d) and western blot assays (Figure 4e) also verified that ICMT expression in CAL-27 and TCA-8113 cells was significantly suppressed in the miR-99a-5p mimics-transfected group compared with the miR-NC-transfected group. Collectively, these data suggest that ICMT is a direct target gene of miR-99a-5p in OSCC.

Isoprenylcysteine carboxylmethyltransferase (ICMT) is a direct target of microRNA-99a-5p. (a) The putative microRNA-99a-5p binding sequence in the 3′-untranslated region (UTR) of ICMT was predicted using TargetScan Human 7.1. (b, c) Luciferase activity was determined in CAL-27 and TCA-8113 cells co-transfected with luciferase reporter plasmids containing the wildtype (WT) or mutant 3′-UTR of ICMT and microRNA-99a-5p mimics or microRNA-NC (negative control). (d) Quantitative reverse transcription polymerase chain reaction analysis of ICMT mRNA expression after transfection of CAL-27 and TCA-8113 cells with microRNA-99a-5p mimics or microRNA-NC. (e) Western blot analysis of ICMT protein expression after transfection of CAL-27 and TCA-8113 cells with microRNA-99a-5p mimics or microRNA-NC. Data are expressed as mean ± standard deviation. **p < 0.01, ***p < 0.001, compared with microRNA-NC.
Overexpression of ICMT rescued the suppressive effects of miR-99a-5p in OSCC cells
To confirm whether ICMT was a functional target of miR-99a-5p in regulating the proliferation, migration, and invasion of OSCC cells, rescue experiments were performed in CAL-27 cells. Briefly, CAL-27 cells were co-transfected with miR-99a-5p mimics and pcDNA3.1-ICMT/pcDNA3.1 plasmids. Western blotting was used to evaluate the transfection efficiency of pcDNA3.1-ICMT, and ICMT protein expression was found to be clearly up-regulated in the miR-99a-5p mimics + pcDNA3.1-ICMT group compared with the miR-99a-5p mimics + pcDNA3.1 group (Figure 5a). The CCK-8 assay also indicated that the cell proliferation impaired by miR-99a-5p overexpression was significantly attenuated by restoring ICMT expression in miR-99a-5p mimics-transfected CAL-27 cells (Figure 5b). Moreover, the effect of miR-99a-5p mimics in decreasing colony numbers was remarkably reversed by ICMT overexpression (Figure 5c). Subsequently, wound healing assay results demonstrated that miR-99a-5p overexpression markedly suppressed cell migration. However, ICMT up-regulation significantly abolished the miR-99a-5p mimics-mediated suppression of migration in CAL-27 cells (Figure 5d). Furthermore, the inhibitory effect of miR-99a-5p mimics on cell invasion was rescued by pcDNA3.1-ICMT transfection in CAL-27 cells (Figure 5e). We also observed that ICMT overexpression significantly elevated proliferation, migration, and invasion in CAL-27 cells (Figure S1). Similarly, pcDNA3.1-ICMT transfection abolished the suppressive role of miR-99a-5p on TCAA-8113 cell proliferation, migration, and invasion (Figure S2). These results clearly demonstrate that miR-99a-5p inhibits OSCC cell proliferation, migration, and invasion at least partially via down-regulating ICMT.
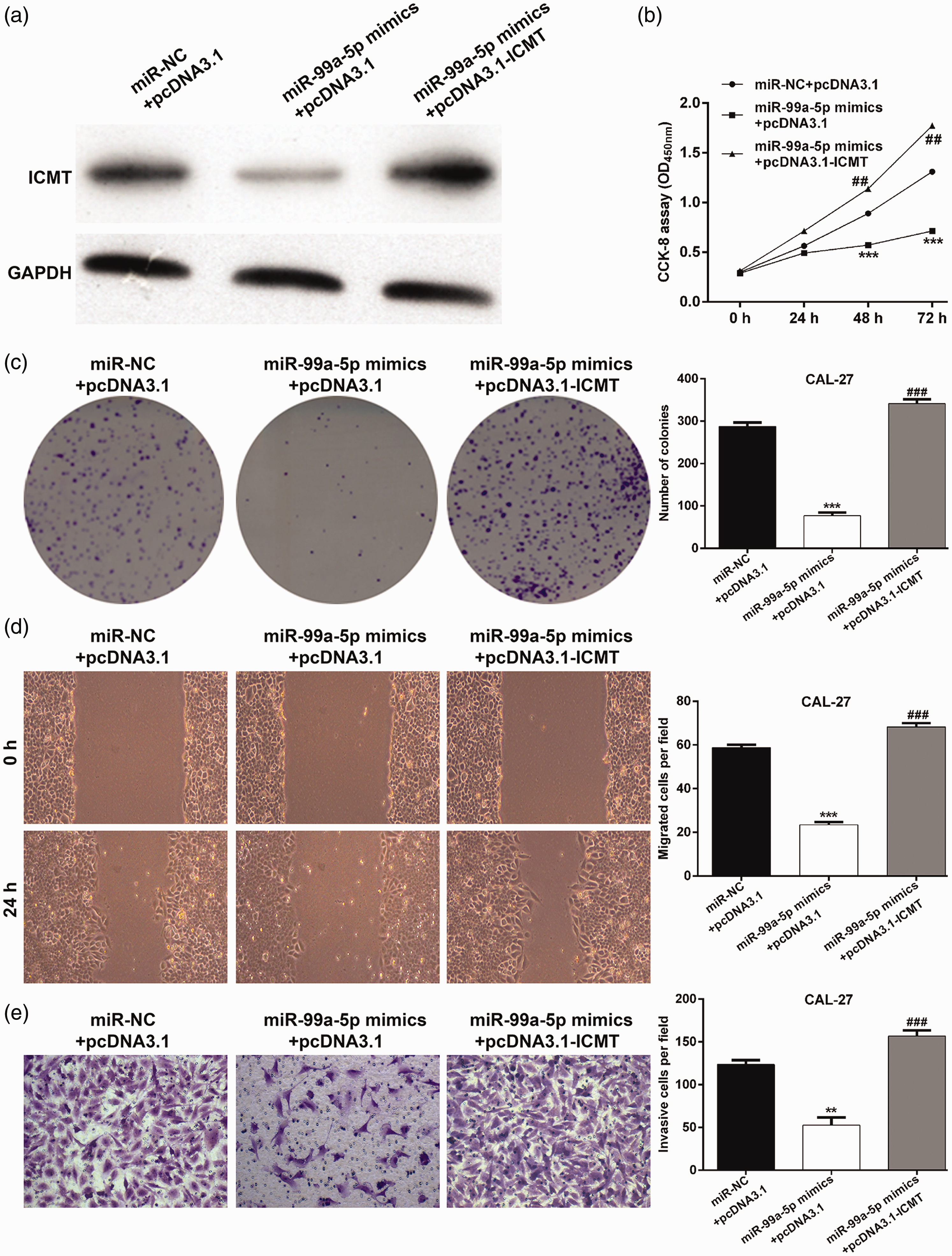
Restoration of isoprenylcysteine carboxylmethyltransferase (ICMT) attenuated the suppressive roles of microRNA-99a-5p in CAL-27 cells. CAL-27 cells were co-transfected with microRNA-99a-5p mimics and pcDNA3.1-ICMT/pcDNA3.1 plasmids. (a) Western blot analysis was performed to detect ICMT protein expression in transected CAL-27 cells. (b) Cell proliferation was analyzed by the cell counting kit-8 assay in transfected CAL-27 cells. (c) Representative images of colonies (left panel) and quantification of colonies (right panel) in transfected CAL-27 cells. (d) Cell migration was assessed by the wound healing assay. (e) Cell invasion was analyzed by the Transwell assay. Data are expressed as mean ± standard deviation. **p < 0.01, ***p < 0.001, compared with microRNA-NC + pcDNA3.1; ##p < 0.01, ###p < 0.001, compared with microRNA-99a-5p mimics + pcDNA3.1.
Discussion
Accumulating studies have suggested that aberrant expression of miRNAs is a common event in human cancers, including OSCC. 22 Here, our preliminary results showed that miR-99a-5p was significantly down-regulated in OSCC tissues and cell lines compared with corresponding adjacent tissues and normal hHOK cells, respectively. Moreover, miR-99a-5p expression was significantly associated with clinical stage and lymph node metastasis in OSCC. Further functional analysis indicated that up-regulation of miR-99a-5p remarkably suppressed the proliferation, migration, and invasion of OSCC cells.
It is accepted that hallmarks of tumor cell growth, proliferation, migration, and invasion play essential roles in the progression of OSCC. 23 Here, the suppressive effects of miR-99a-5p on OSCC cells (CAL-27 and TCA-8113) appear to identify miR-99a-5p as a tumor suppressor in OSCC. Our data agrees with the study performed by Feng et al., 24 who reported that lower expression of miR-99a-5p was correlated with the more aggressive phenotypes of bladder cancer, and that enforced expression of miR-99a-5p inhibited the proliferation of human bladder cancer cells. Qin et al. 17 also showed that overexpression of miR-99a-5p impaired breast cancer cell proliferation and invasion and facilitated apoptosis and cell cycle arrest.
Interestingly, aberrant miR-99a-5p expression has been identified in several studies of human cancers. For example, Hilly et al.
25
revealed that aggressive carcinoma of the oral tongue in young patients showed higher expression of miR-99a-5p than non-aggressive tumors, while Chen et al.
11
found that miR-99a-5p was expressed at higher levels in patients with HNSCC and low histological grade neoplasms than those with high histological grade neoplasms, and that members of the miR-99 family (miR-100 and miR-99a) were down-regulated in HNSCC.
26
These differences in miR-99a-5p expression levels might be ascribed to different tissue sources. Consistent with our
miRNAs can function as either oncogenes or tumor suppressors through binding specific targets. 29 Through target prediction algorithms and luciferase reporter assays, we confirmed ICMT as a potential target of miR-99a-5p. Moreover, miR-99a-5p was shown to negatively regulate the expression of ICMT at both the transcript and protein levels. Previous research demonstrated that miR-99a-5p targets different genes in human oral carcinoma cells, including NADPH oxidase (NOX)4 30 and IGF1R. 28 ICMT is critical for malignant transformation and tumor maintenance by functioning as a substrate for all RAS isoforms. 31 Indeed, RAS-dependent tumor growth requires ICMT, so ICMT is a target for anti-RAS therapy. 32 Currently, several studies have reported the oncogenic role of ICMT in tumor cells. For example, ICMT inhibition induced cell cycle arrest and apoptosis through p21 and p21-regulated BNIP3 induction in pancreatic cancer, 18 while specific ICMT inhibitors have been shown to induce tumor death in prostate cancer 33 and liver cancer.32,34 Furthermore, a recent study by Xu et al. 35 found that ICMT contributes to cell growth, survival, migration, and chemoresistance by regulating RAS/RAF/mitogen-activated protein kinase/extracellular signal-regulated kinase signaling and epithelial–mesenchymal transition in hepatocellular carcinoma.
In line with these reports, our rescue experiments demonstrated that restoration of ICMT attenuated the suppressive effects of miR-99a-5p on OSCC cell proliferation, migration, and invasion, which indicates an oncogenic role for ICMT in OSCC. These data suggest that miR-99a-5p suppresses OSCC cell proliferation, migration, and invasion by targeting ICMT. Interestingly, a recent study by Shi et al. 30 reported that miR-99a-5p overexpression inhibited TSCC1 cell viability, proliferation, migration, and invasion through targeting NOX4, which adds further support to a tumor suppressive role for miR-99a-5p in OSCC. The novelty of the present study included its clinical significance, use of two OSCC cell lines and rescue experiments, and identification of ICMT as a different target gene of miR-99a-5p. We speculate that our work will increase our knowledge of miR-99a-5p as a tumor suppressor in OSCC therapy treatment.
In summary, our study confirms a tumor suppressive role for miR-99a-5p in OSCC
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520939031 - Supplemental material for MicroRNA-99a-5p suppresses cell proliferation, migration, and invasion by targeting isoprenylcysteine carboxylmethyltransferase in oral squamous cell carcinoma
Supplemental material, sj-pdf-1-imr-10.1177_0300060520939031 for MicroRNA-99a-5p suppresses cell proliferation, migration, and invasion by targeting isoprenylcysteine carboxylmethyltransferase in oral squamous cell carcinoma by Xiang Sun and Huixin Yan in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_0300060520939031 - Supplemental material for MicroRNA-99a-5p suppresses cell proliferation, migration, and invasion by targeting isoprenylcysteine carboxylmethyltransferase in oral squamous cell carcinoma
Supplemental material, sj-pdf-2-imr-10.1177_0300060520939031 for MicroRNA-99a-5p suppresses cell proliferation, migration, and invasion by targeting isoprenylcysteine carboxylmethyltransferase in oral squamous cell carcinoma by Xiang Sun and Huixin Yan in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Authors’ contributions
YHX designed and supervised the study. SX mainly performed the experiments, analyzed the data, and wrote the paper. YHX revised the manuscript. Both authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
