Abstract
Purpose:
Cancer fatigue guidelines recommend routine fatigue screening, with further assessment for people reporting moderate to severe fatigue. There is neither a gold-standard, nor a broadly accepted screening method, and knowledge about the impact of screening on care processes is limited. This study aimed to explore the feasibility of 2 fatigue screening methods and current clinical practice in cancer outpatient clinics.
Methods:
Hospital outpatients attending cancer clinics during 1 week completed a five-item survey: a numeric scale for current tiredness, 2 categorical pictorial scales rating tiredness last week and the impact of fatigue (Fatigue Pictogram), screening tool preference and help needed for survey completion. Participant demographics and fatigue documentation by clinical staff for that appointment were extracted from medical records. Analyses used descriptive statistics. Groups were compared using appropriate statistical tests.
Results:
Over 75% of participants rated their fatigue consistently as mild or significant on both screening tools. Of 1709 eligible outpatients, 533 (31%) completed the survey. Records were audited for 430 (81%) identifiable participants. Over half of the participants reported moderate or severe tiredness either “now” (237, 57%) and/or “last week” (226, 53%). Clinician documentation of fatigue seldom matched self-reports. Fatigue was rated as severe by 103 participants (24%), yet was noted in only 21 (20%) of these individuals’ clinical notes. Both screening tools were equally preferred.
Conclusion:
The numeric rating scale and Fatigue Pictogram are equally applicable for screening fatigue in cancer outpatient care. There is a high prevalence of clinically significant fatigue in a hospital outpatient setting that is not documented. Adequate care pathways for further management should be established alongside fatigue screening.
Keywords
Introduction
Fatigue is a significant and distressing problem for many people with cancer. 1 Several conceptual definitions of cancer-related fatigue (CRF) exist, 2 with that of the National Comprehensive Cancer Network 3 commonly used by guideline development groups.4-6 CRF must coincide with cancer diagnosis or treatment, it is distressing, persistent, out of proportion with recent activity, interferes with usual functioning 3 and is not relieved by rest.3,7 However, it can be difficult to separate CRF from “everyday” fatigue without a focused evaluation. 8
Evidence-based guidelines for managing CRF advocate routine screening at a range of time points, and further evaluation and management for people with moderate and severe fatigue.3,4 These levels of fatigue interfere with physical and daily activities 4 and are often referred to as “significant” or “clinically significant” fatigue.9,10
There has been limited research into how to implement guideline recommendations for fatigue, including screening, 11 and the implication of routine fatigue screening on resources is unclear. Although “routine screening” of fatigue is recommended, 12 there is scant evidence to guide when, where and how screening should occur. Studies have found that if not directly asked, patients may not mention their fatigue during a clinical contact.13,14 To ensure timely education and management of fatigue, and to enable self-reporting, patients must be asked. 15 However, it is unclear which screening method health practitioners should use. Guidelines recommend routine symptom screening in the clinical setting using brief self-report measures, but there is no “gold standard” method to identify fatigue. The Canadian Association of Psychosocial Oncology (CAPO) fatigue guideline recommends either using a valid quantitative measure with established severity cut points to rate fatigue now, such as 0-10 numeric rating scale (NRS), or a semi-quantitative tool for example, the Fatigue Pictogram.4,16 Other CRF guidelines suggest using NRS over 7 days,3,6 and researchers have advocated short measures such as PROMIS-CAT17,18 or three CRF screening questions.19,20
A second challenge for implementing fatigue screening is lack of clarity or empirical evidence about the appropriate timing and location for screening. Peak body recommendations include “Screen every patient at regular intervals”, 3 “Screen for fatigue at entry to system, periodically throughout treatment, post-treatment follow-up and advanced disease” 4 and “All health care providers should routinely screen for the presence of fatigue from the point of diagnosis onward, including after completion of primary treatment . . . as clinically indicated and at least annually.” 6
Thirdly, the impact of routine fatigue screening on care processes has not been explored, and doubts about capacity to respond appropriately remain.
This study explored fatigue screening and documentation in outpatient cancer clinics, guided by 2 main research questions: which ultra-short screening tool is appropriate for identifying fatigue in cancer outpatient clinics, and how well cancer fatigue levels documented by health professionals’ align with patient-reported fatigue. The main study aims were:
(1) To evaluate the feasibility of 2 ultra-short fatigue screening methods.
(2) To describe health professional fatigue measurement practice in specialist cancer clinics.
(3) To estimate demand for fatigue management among cancer patients attending clinic visits.
Methods
Participants and consent. Over a one-week period in November 2018 volunteers at a cancer center invited adult outpatients in waiting areas to complete a paper survey. Of 1864 patients across all tumour streams attending appointments, 1709 (91.7%) checked in during recruitment times. There were no additional eligibility criteria, however surveys were excluded from analysis if participants did not have a cancer diagnosis or did not provide an identifier because the survey was deliberately short with no details about participants. Participants completed the survey independently, or assisted by a companion or interpreter if available. Participants were asked to provide their hospital identifier and/or name to enable medical record audit and team notification if required. An implied consent approach was used. The survey cover page explained that participation was voluntary and how data would be collected and used (see Supplemental Material 1).
Instrument. The 5-item survey included 2 screening tools and 2 questions, asking participants their preferred method to rate fatigue and whether help was needed for the survey. The screening tools were selected for brevity and psychometric properties:
(1) The Revised Edmonton Symptom Assessment System (ESAS-r) 21 is commonly used to screen symptoms including tiredness in oncology and palliative care in Canada. 22 The original ESAS has shown acceptable reliability and validity, 23 although fewer studies have explored reliability and validity of the ESAS-r.21,22 Valid cut points for moderate and severe fatigue 24 and minimal clinically important differences have been determined. 25 The “tiredness” item is rated on an NRS from 0 (no tiredness) to 10 (worst possible tiredness) and the recall period is “now.”
(2) The Fatigue Pictogram uses icons with descriptors to rate (1) tiredness over the last week and (2) impact/interference. 16 Responses can be recorded in numerical form (ordinal scale) from 0 to 4 (most fatigue) (Supplementary Material 1). The Pictogram demonstrates good validity and test-retest reliability. Its properties are equivalent when administered face-to-face or by phone. 16
ESAS-r fatigue ratings were categorised as none (0), mild (1-3), moderate (4-6) and severe (7-10) according to established cut points. 24 The Pictogram icons have no set interpretations, so research team members aligned them to CAPO category descriptors of mild, moderate and severe fatigue 4 by consensus, shown in Table 1. The second Pictogram impact response item “I can do almost everything I normally do” was difficult to align with CAPO categories, due to conflicting descriptors for mild fatigue in the CAPO algorithm pages 19-20. 4 Since some impact on normal activities is suggested, this item was categorised a priori as “moderate” fatigue (Table 1, Impact A).
Calibration of Fatigue Responses Across Different Fatigue Measures.

ESAS-r: Edmonton Symptom Assessment System Revised 21 . Impact B was used for the analysis
Record audit. Hospital records of identifiable participants were audited 1 week after survey completion. Data extracted included date of birth, gender, language, postcode, cancer diagnosis, treatment phase and documentation of fatigue management in the electronic medical record. The CAPO guideline recommended actions for severe fatigue are “urgent management of contributing factors” and safety. 4 To ensure duty of care, if participants recorded severe fatigue on 1 or more screening tool question, a treating team member was notified by email and/or a note entered in the electronic medical record. The note included a suggested action according to the CAPO guideline (“assess and manage contributing factors” or “review fatigue/safety next visit”). These records were re-audited to examine health professional actions within the week following team notification.
All study procedures were approved by the Human Research Ethics Department of the cancer center (LNR/46671/PMCC-2018).
Data analysis. Survey and audit data of identifiable participants were entered manually into a RedCap® database. All available data was used. Analyses were performed in Excel and R™. 26 Descriptive statistics were used for the participant sample and to identify prevalence of any level of fatigue, because hypotheses were not being tested in this feasibility study. 27 Clinical (treatment phase) and demographic (age, gender) differences between respondents who reported none/mild, moderate and severe fatigue for each of the 3 items separately were assessed using two-sided ANOVA for numerical variables and chi-squared tests for categorical variables. 28 No adjustments were made for the number of outcomes assessed. 29 Alpha was set at 0.05. If the chi-squared test showed evidence of association, the individual cells that had different values to those expected were identified by examining the Pearson residual, where a value of 2 or greater was considered evidence of lack of fit. 28
Individuals’ responses to NRS fatigue severity (now) and Pictogram fatigue severity (last week), and Pictogram fatigue severity last week and impact last week were cross-tabulated to examine consistency of fatigue categories.
On descriptive analysis of fatigue impact (Pictogram), participant distribution was excessively skewed to the moderate fatigue category. Of 132 participants who endorsed the impact response choice I can do almost everything I normally do, 66% indicated mild or no fatigue last week, suggesting its interpretation as “mild fatigue impact” is reasonable for screening purposes. After re-categorising this response option to the mild category (Table 1, Impact B), responses were more evenly distributed. Impact B categories were used for analysis.
Indicators of feasibility were instrument ability to classify clinically significant fatigue, participant preference, help needed to complete the survey and missing fatigue data.
Results
Of 1709 patients presenting for a clinic appointment during the survey period, 533 (31%) returned surveys and 430 (25%) were eligible for analysis. Due to this volume of patients attending appointments it was not possible to approach all patients. Figure 1 illustrates the flow of survey completion and inclusion for analysis.

Survey eligibility for analysis.
Table 2 shows demographic and clinical characteristics of 430 identifiable participants with a malignancy. Ages ranged from 18 to 93 years, three-quarters of participants lived in a major city and 398 (93%) spoke English as their primary language.
Participant Demographic and Clinical Characteristics (n = 430).

Accessibility and Remoteness Index of Australia https://www.abs.gov.au/websitedbs/D3310114.nsf/home/remoteness+structure.
Responses to the fatigue items on the survey overall, and by age, gender and treatment phase, are shown in Table 3. Using the established categories for the ESAS-r and our categorization of the Fatigue Pictogram (Table 1), 237 (57%) participants reported moderate-severe levels of fatigue “now” and 226 (53%) “last week.” Over half of respondents reported moderate-severe fatigue impact. The overall results of the chi-squared tests indicated treatment phase was significantly associated with both pictogram items—fatigue last week and fatigue impact over the last week. 30 Significantly more participants in the “managing recurrent, residual and metastatic disease” phase reported severe tiredness in the last week, with fewer reporting mild tiredness and mild fatigue impact than in other phases (P < .05). More participants in the “diagnosis, staging and treatment planning” phase reported severe fatigue impact during the last week than expected. The association between gender and fatigue during the last week was also statistically significant, with fewer female participants reporting mild fatigue and more reporting severe fatigue, with the opposite found for male participants (P < .05). See Table 3.
Responses to Fatigue Patient Reported Measures by Participant Characteristics.
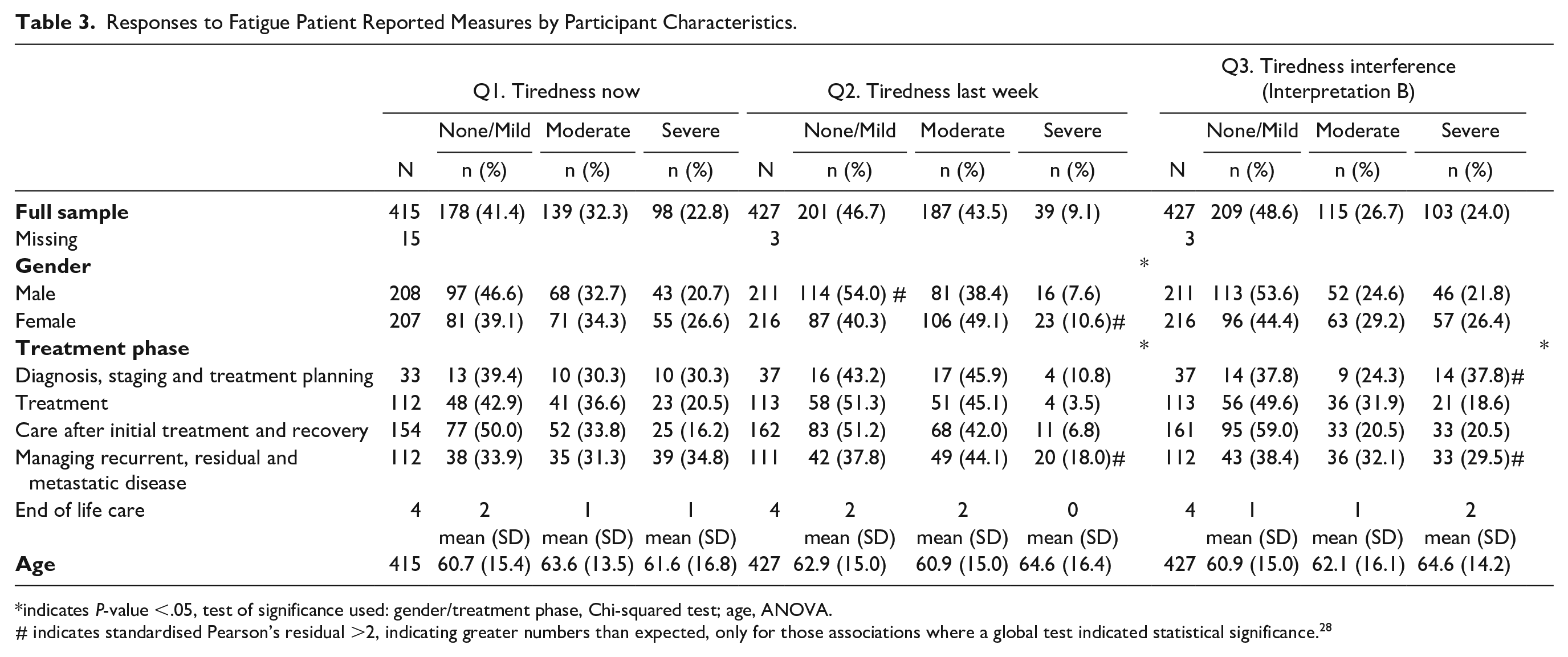
indicates P-value <.05, test of significance used: gender/treatment phase, Chi-squared test; age, ANOVA.
# indicates standardised Pearson’s residual >2, indicating greater numbers than expected, only for those associations where a global test indicated statistical significance. 28
Figure 2 shows a cross-tabulation of individual participants’ fatigue severity levels over 2 recall periods, and severity and impact levels (Pictogram). Shown within heavy borders in the figure, most participants consistently rated their fatigue severity both “now” on the NRS and “last week” on the Fatigue Pictogram as either none-mild (33.8%) or moderate-to-severe (45.2%). 16 Greater fluctuation was observed between the finer categories of none, mild, moderate and severe, with 62% of respondents consistently reporting fatigue severity for both recall periods (colored fields). For the 2 Pictogram items (fatigue severity and impact last week), again shown within the heavy borders in the figure, 36.3% of participants consistently rated none-mild and 40.7% moderate-severe.

Cross-tabulation of individuals’ fatigue levels on 3 questions (numbers presented are percentages).
Despite its apparent simplicity, 38 (9%) participants needed help to complete the survey. Of these participants, 15 spoke a primary language other than English, another 10 had advanced disease and 1 person was visually impaired. Preference to rate fatigue using numbers and pictures was evenly endorsed, with 30% and 28% of participants endorsing the Pictogram and NRS respectively, while 41% had no preference. Fifteen surveys had missing data for the NRS, and 3 participants omitted the 2 Pictogram items.
Record audit. Three hundred participants (70%) indicated moderate or severe (“clinically significant”) fatigue in at least one survey question. Fatigue was mentioned in just 54 (17%) of these clinic notes. When noted, health professionals’ appraisal of fatigue severity was often inconsistent with self-rated fatigue, with only 1 in 3 notes indicating clinically significant fatigue (Table 4). Many clinic notes only listed fatigue as a possible treatment side effect. In most reports, descriptive language was used for fatigue, with just 9/54 (17%) using a fatigue grade (NRS, CTCAE 31 or Brief Fatigue Inventory 32 ) or CAPO category. 4 Health professionals’ descriptions of fatigue included: no/mild fatigue, fatigue not a huge issue; lethargy; fatigued/tired; and significant/troublesome/persistent/moderate/notable fatigue.
Audit: Consistency of HP Documented and Self-Rated Fatigue.

The delayed audit included 167 (39%) participants who reported severe fatigue or impact to any survey question. Of these, 29 (17%) had a clinic note that mentioned fatigue. The clinical note included fatigue for 29 of 55 (24%) participants recording severe fatigue on more than 1 question. A researcher entered a medical record note for 91% of these participants and notified a team member by email in 85% of cases. Reasons for not notifying or recording fatigue included the participant being acutely ill or admitted, already referred for fatigue management or unknown to the multidisciplinary team (attending annual medical review or second opinion, cancer treatment elsewhere). During the week after notification, 16 (10%) participants had further fatigue management documented by the healthcare team including symptom management, phone calls, education or referrals to allied health.
Discussion
Study results indicate that about half of the study participants in an ambulatory cancer care setting experienced clinically significant fatigue that was rarely documented by a health professional. The 2 screening tools were equally preferred by participants, but the Pictogram had less missing data, suggesting ease of use. In this discussion we will use our findings to further examine the feasibility of the 2 fatigue screening methods, and finally suggest resources needed for optimal implementation of screening and evidence-based management of CRF in ambulatory care.
Feasibility of ESAS-r Tiredness Item and Fatigue Pictogram
Clinical utility is a concept incorporating feasibility and effectiveness. An ideal clinical screening tool is efficient, and easy to administer and interpret.10,33 Questions must be easily understood and response options clear. Screening results must provide information of value to both consumer and clinician. 33 Personal factors such as beliefs, experience, physical and mental state may affect responses. 34
Fatigue screening in clinical practice aims to identify individuals with fatigue requiring deeper assessment and management. 16 Since the CAPO fatigue guideline recommended first step for moderate and severe fatigue is the same, 4 a minimum requirement of screening is to identify clinically significant fatigue. Then, further assessment can stratify to moderate and severe fatigue levels to indicate care pathways for different CRF levels.4,9
Established NRS cut points enabled interpretation of clinically significant fatigue “now”. 24 The Fatigue Pictogram had no previously identified cut points.16,35 Our study results suggest that for fatigue screening, the Pictogram categories assigned a priori for fatigue severity icons are appropriate and that fatigue impact items interpreted as impact B (Table 1) can indicate clinically significant fatigue. More research is needed to validate this finding.
Use and interpretation. The NRS can be used in written form or verbally without a physical copy of the scale. 36 In contrast, although some participants found the Fatigue Pictogram icons more tangible than numbers, a physical or electronic copy is required, and respondents need adequate vision. Nine percent of study participants had help to complete the survey, a reminder that self-completed questionnaires do not suit everyone. Lack of overall consensus among participants about a preferred screening tool, with 60% stating a preference suggests that either screening method could be successfully implemented into routine care.
To meet CRF criteria, that is, persistent tiredness affecting function,3,4 at least moderate fatigue in the past week is implied. Fatigue only rated as significant “now” risks over-or under-management of CRF, although acute fatigue still needs investigation. Twelve percent of our participants rated “tiredness now” moderate to severe and “not at all” or “a little bit” tired in the last week, (ie, likely acute fatigue) while 9% were not currently tired but had significant tiredness in the last week, and would not be identified for further management if only current fatigue were rated (Figure 2).
Current Practice Indicators
We used fatigue documentation to gauge current clinical practice. Several factors may explain the limited recording and management of fatigue revealed in this audit. First, fatigue was not discussed. Reasons patients and professionals avoid discussing fatigue include lack of knowledge of what to do next, lack of time, perception that fatigue is inevitable, a focus on cancer management and fear of treatment reduction.15,37 Next, fatigue was mentioned but not recorded. Medical appointments are scheduled every 10-15 minutes with insufficient time to address all issues, let alone for detailed clinical notes. Finally, the commonly used progress report form lacked fields or prompts, resulting in variable language used to describe fatigue.
Implication of Results on CRF Guideline Implementation and Care Processes
Study results indicate both the ESAS-r tiredness item (NRS) 21 and the Fatigue Pictogram 16 are feasible and acceptable for identifying clinically significant fatigue. Patients may prefer one method, and a choice can be offered. Re-coloring the Pictogram icons in “traffic-light” green-amber-red may assist with interpretation. Other researchers have suggested more stringent screening methods to identify CRF “cases,” thereby reducing demand on resources. 38 To minimise unnecessary resource use and patient inconvenience, asking about fatigue levels both now and last week is advised, with a step-wise approach to further assessment of fatigue.
The location of fatigue screening must be considered. This and other studies in ambulatory cancer clinics1,39 indicate that 30-66% of people in waiting areas may have clinically significant fatigue—requiring further assessment according to CAPO guidelines. 4 In an outpatient clinic with about 1800 individuals attending weekly, at least 600 would require further fatigue assessment. Capacity to respond is a concern. Our audit suggested that cancer doctors and specialist nurses rarely dealt with fatigue within the allotted clinic time, or documented follow-up within a week after being notified of severe fatigue.
Although our participation rate was low (25%), the high rates of clinically significant fatigue we report in the diagnosis and staging and after initial treatment and recovery phases supports fatigue screening both before and after anticancer treatment. With smaller numbers in some subgroups, these findings should be cautiously interpreted. However, based on fatigue prevalence data in ambulatory services, we do not advocate fatigue screening in outpatient clinics without systems for timely response. To ensure adequate CRF management, screening and patient education within established clinical routines is suggested. Pre-treatment symptom checking in chemotherapy and radiotherapy units should include fatigue, with patients educated to prevent and report fatigue affecting function. Fatigue and related symptoms should be screened via intake questionnaires and initial assessments.
Due to the high occurrence of fatigue in cancer, specialist centers must share responsibility for fatigue management with patients and their local health services. These community practitioners need evidence-based education and resources to manage CRF.
This study has several limitations. Although the sample size was large, the reasons 75% of 1700 eligible patients did not participate are unknown, and our data must be interpreted cautiously. The original plan to invite consecutive clinic attendees to participate when checking in for their appointment could not be implemented. Many patients used self-check-in kiosks and clinic receptionists lacked time to distribute and explain the survey. The resultant convenience sampling method using volunteers is a study limitation. Selection bias and misinterpretation could have been reduced by systematic invitation of consecutive patients and verbal administration. Additionally, it has been reported that research results for fatigue levels may be higher than those reported in clinical practice, due to under-reporting of CRF to the health team, to ensure focus on disease management and maintain cancer treatment choices. 13
Conclusion
The high prevalence of clinically significant fatigue in ambulatory cancer clinics suggests innovative approaches are needed to adequately manage cancer fatigue. Cancer services can educate patients to prevent, self-monitor and report fatigue. Local health services need access to practice resources to assist with cancer fatigue management. More research into fatigue screening participation and implementation is needed.
Supplemental Material
sj-pdf-1-ict-10.1177_1534735420983443 – Supplemental material for Exploring the Use of Two Brief Fatigue Screening Tools in Cancer Outpatient Clinics
Supplemental material, sj-pdf-1-ict-10.1177_1534735420983443 for Exploring the Use of Two Brief Fatigue Screening Tools in Cancer Outpatient Clinics by Elizabeth J. Pearson, Allison Drosdowsky, Lara Edbrooke and Linda Denehy in Integrative Cancer Therapies
Footnotes
Acknowledgements
The study team thanks Ms Trista Koproski for data management, Peter Mac volunteers for survey distribution, and research assistants Ms Amanda Appathurai, Ms Jamie Waterland for data entry and medical record audit.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Elizabeth Pearson, research assistants and Allison Drosdowsky. The first draft of the manuscript was written by Elizabeth Pearson and all authors contributed to subsequent versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Victorian Cancer Agency Fellowship to Dr Pearson.
Ethical Approval
The Human Research Ethics Department of Peter MacCallum Cancer Centre approved the project methods (Approval number LNR/46671/PMCC-2018).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
