Abstract
Purpose:
Examine the feasibility and preliminary effects of a lifestyle intervention of rice bran plus navy bean supplementation, and physical activity (PA) education on intake of fiber and whole grains, and PA levels.
Design:
Randomized-controlled, single-blinded.
Setting:
Academic institution and free-living.
Subjects:
Adults >18 years, with ≥1 adenomatous polyp removed within 3 years.
Intervention:
Participants received powder and pre-prepared meals and snacks that contained either rice bran (30 g/day) plus navy bean (30 g/day), or Fibersol-2® (10 g/day), for 12-weeks. All participants received a 1-hour (PA) education session.
Measures:
Feasibility was assessed by recruitment and retention rates, and compliance to the study foods and procedures. Three-day food logs were analyzed using Nutritionist Pro™ to estimate fiber intake, and the Automated Self-Administered 24-hour (ASA24®) Dietary Assessment Tool calculated Healthy Eating Index (HEI) whole grain and total scores. PA was measured using an ActivPAL™ accelerometer.
Analysis:
Continuous data were summarized as median, range, and percent change from baseline to post-intervention.
Results:
N = 20 (86.9%) completed the intervention. Compliance was 92% in the rice bran plus navy bean versus 89% in Fibersol-2®. Navy bean consumption increased from 2 g/day to 30 g/day, and rice bran from 0 g/day to 30 g/day. Fiber intake (g/day) increased by 73% versus 82%, HEI whole grain improved by 270% versus 37%, and HEI total improved by 10% versus 9.1% in rice bran plus navy bean and Fibersol-2®, respectively. Total PA (MET-hours/day) showed minimal change for intervention (+0.04%) and control (+4%).
Conclusion:
Findings merit a larger trial of rice bran plus navy bean and PA to evaluate efficacy for dietary and cancer prevention-related outcomes.
Introduction
Colorectal cancer (CRC) is the third most common cancer in both men and women in the United States, and it is estimated that by 2024, 1.6 million Americans will be diagnosed.1,2 The World Cancer Research Fund has estimated that 30% to 40% of all cancers can be prevented by appropriate diet and body weight, and this risk reduction is nearly 70% for CRC. 3 Furthermore, consuming whole grains and legumes as a source of dietary fiber is associated with reduced risk for CRC.4-6 Evidence also suggests that brown rice consumption at least one to two times per week, 7 or daily cooked dry bean intake (1.5 cups/week) reduces risk for pre-cancerous adenomatous polyps. 8 Rice bran and navy bean consumption each separately have shown dietary efficacy for reducing aberrant colonic polyp formation in animals. We and others have demonstrated observation and intervention feasibility of increased rice bran or navy bean intake among healthy adults and CRC survivors.9-13 The combination of rice bran/navy bean foods was palatable in a study of at-risk children with elevated cholesterol. 14
Another important lifestyle factor with convincing evidence for decreased incidence of CRC is physical activity (PA). 15 Previous studies have observed associations between PA and colon cancer, with risk reduction ranging from 16% to 24%.16-18 Adherence to dietary and PA recommendations, such as those outlined by the American Cancer Society (ACS), World Cancer Research Fund, and American Institute for Cancer Research (AICR) is associated with reduced odds of having 3 or more adenomas colorectal adenomas.19-21
Thus, the objective of this pilot study was to evaluate a 12-week rice bran/navy bean intervention among individuals at high risk for CRC, while measuring and educating participants on PA. We assessed the feasibility of recruiting and retaining participants, compliance to study foods, compliance to, and acceptability of study procedures, and finally, evaluated the effects of the intervention on fiber intake, whole grain intake, Healthy Eating Index (HEI), and PA. Our primary hypothesis was that the rice bran/navy bean intervention via study foods and as powder supplements would be feasible to improve healthy eating indices in all categories of fiber intake when compared to a fiber powder.
Methods
Participants and Study Procedures
Participants who met eligibility criteria and consented to participate were randomized by body mass index (BMI) and sex to one of the 2 study groups. Participants completed 3 scheduled study visits at baseline (week 0), midpoint (week 6), and post-intervention (week 12). Fasted blood samples were collected at 3 scheduled visits by venipuncture. Blood was processed for serum lipid panel. Study ID-labeled containers were given to participants for self-collection of stool (week 0 and week 12) and urine (week 0, week 6, and week 12). Blood, stool and urine samples were analyzed for non-targeted and targeted metabolomics (data not reported here). Participants visited the study site bi-weekly to pick up study foods/powders, return empty containers from completed study foods, and check compliance. The CONSORT diagram is shown in Figure 1.

CONSORT flow diagram.
Study Foods
Sixteen different meal and snack food items were developed for each of the study arms (control group and rice bran/navy bean intervention group). The control study foods were isocaloric-matched to the intervention foods without the inclusion of rice bran or navy bean. Intervention foods had 10 g of heat stabilized rice bran and 10 g of pre-cooked navy bean powder using a similar design to our published study food protocols.9,12 Nutritionist Pro™, a diet analysis software, was used for a complete nutritional analysis on each meal/snack recipe (Axxya Systems, Redmond, WA). Pre-cooked navy bean powder was purchased from Archer Daniels Midland (ADM) Edible Bean Specialties, Inc (Decatur IL). Heat-stabilized rice bran (RBT300) was purchased from Rice Bran Technologies (Sacramento, CA). Fibersol-2®, digestion-resistant maltodextrin soluble corn fiber was purchased online. Western Innovations (Denver, CO) packaged the study powders into blinded sachets for the control group (10 g Fibersol-2®) and intervention group (10 g of heat-stabilized rice bran and 10 g of pre-cooked navy bean powder). Table 1 shows calorie (kcal) and fiber (g) for study foods and study powders used for each group.
Caloric and Fiber Content of Study Foods a .

Two foods and 1 study powder packet were consumed by all participants. Intervention group received 10 g rice bran/10 g navy beans per meal/snack. Control study foods were macronutrient matched.
Study powder packet contains 10 g Fibersol-2® (soluble corn fiber) or 10 g rice bran/10 g cooked navy bean powder. Study powders packets were self-incorporated by participants into daily diet.
Dietary Intervention
Every other week, both intervention and control participants were provided with a 2-week supply of study foods and study powders along with storing, thawing and reheating instructions. 12 To ensure participants remained blinded, study-provided foods and powders were coded so that only the study coordinator was aware of group assignment. Participants in the control group received one 10 g/day Fibersol-2® powder, and 2 study foods that contained small amounts of fiber (see Table 1). Participants in the intervention group began with 1 study food (or powder) item per day for the first 2-weeks. There were 2 study food (or powder) items consumed per day by week 4, and by week 6, the participants were consuming the full dose of 3 study food items daily for the remainder 6-week duration of the study for a total of 30 g rice bran and 30 g navy bean powder). Participants were free-living for the daily dietary foods consumed in addition to the intervention. The study provided meals, snacks and powders supplied ~35% of the daily caloric intakes.
Physical Activity Education
Participants in both control and intervention arms met individually with a health and exercise science-trained graduate research assistant who provided a 1-hour PA education session specific to CRC prevention. The topics included the benefits of PA for CRC control and prevention, 18 summarizing the PA guidelines for cancer prevention. 22 As part of this session, participants were also offered behavioral support for PA using strategies based on social cognitive theory, 23 such as goal setting, PA barrier identification, and problem-solving PA barriers, and environmental resource (ie, local PA facilities and programs).
Outcome Measures
Participants completed a 3-day food log at baseline, 6-weeks, and post-intervention (12-weeks), which included recording all food and drink consumed on 2 weekdays (Monday-Thursday) and 1 weekend day (Friday-Sunday), as previously described.9,12 The participant food logs were analyzed using Nutritionist Pro™ (Axxya Systems, Redmond, WA) to estimate and caloric and fiber intake, and macronutrient and micronutrient consumption. The Automated Self-Administered 24-hour (ASA24®) Dietary Assessment Tool was used to calculate the Healthy Eating Index (HEI) for each participant. ASA24® is a web-based tool that enables multiple, automatically coded, self-administered 24-hour recalls and/or single or multi-day food records, also known as food diaries. It is based on the 2015 Dietary Guidelines for Americans. A SAS macro (SAS Institute Inc., Cary, NC) which incorporated nutritional composition of rice bran and navy beans was utilized to calculate HEI in the current study.
PA was measured using an activPAL accelerometer (PAL Technologies, Glasgow, Scotland). The activPAL quantifies free-living sedentary and ambulatory activities. It has been validated as one of the most accurate wearable activity monitors and has previously been used in healthy adults as well as cancer survivors.24-26 The activPAL activity monitor is a small device worn on the anterior thigh that uses information about static and dynamic acceleration to (1) distinguish body posture as sitting/lying, standing and stepping and (2) estimate energy expenditure (EE) (expressed as metabolic equivalents (METs)). 26 The activPAL was wrapped in a nitrile sleeve for waterproofing and attached to the anterior aspect of the right thigh with Tegaderm dressing. Participants wore the activPAL for 7 consecutive days, 24 hours per day, for the week immediately following the baseline study visit. Participants were asked to record times of activPAL removal and sleep/wake-up times. Data was downloaded using the activPAL software (v. 7.2.38) and summarized using the activPAL Processing package in R. 27 Days of data collection required at least 600 minutes (10 hours) of wear time to be considered valid. Non-wear periods were excluded from analyses; these were defined using participant logs and verified via visual inspection of the accelerometer data. A cadence based linear regression was used to estimate total MET hours and time spent in sedentary (METs <1.5), light (METs 1.5-2.99), and moderate-to-vigorous (METs ≥3) intensity activity per day. 26 Each variable was summed between all valid days and divided by valid days to calculate daily values.
Twelve-hour fasted blood samples were collected between 7:00 am and 11:00 am. Blood was drawn through venipuncture into EDTA tubes (Greiner bio-one, Monroe, NC) and immediately stored on ice. Total cholesterol, HDL, and LDL were measured with PiccoloÒ Lipid Panel Plus (Abaxis, Inc, Union City, CA). Urine and stool samples were self-collected within 24 hours of their scheduled appointment and stored as previously described for metabolite profiling analysis (data not included).10,28
Randomization and Statistical Analyses
Participants were randomized in blocks, 1:1 to the intervention and control arm. Randomization was stratified by sex, cancer stage (0 or 1), and BMI at time of consent. The project statistician generated the randomization schedule, and was concealed from the study coordinator until a participant was enrolled and ready to be randomized.
The primary outcomes of this feasibility study were (1) participant recruitment and retention, (2) compliance to the study foods, (3) compliance to, and acceptability of study procedures, and (4) collection of preliminary data examining changes in fiber intake, whole grains, total HEI score, and physical activity. Participant recruitment and retention was based on accrual and study attrition. Compliance to the dietary intervention was determined by (1) recording the study meals and snacks that participants consumed each day divided by the total number provided to calculate percent compliance, and (2) the percent of daily caloric intake due to rice bran/navy bean consumption. To calculate the percent of daily caloric intake due to rice bran/navy bean consumption the following equation was used:
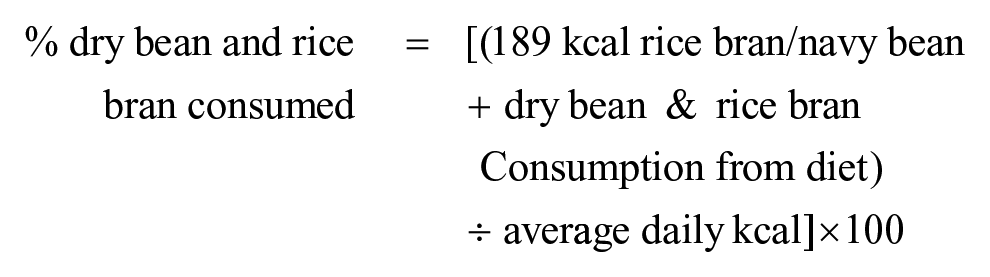
Compliance to study procedures was based on completion of blood draws, urine and stool sample collections. For the activPAL accelerometer, compliance was defined as ≥10 hours of wear time per day, for ≥4 days, with at least on weekend day. Acceptability of study foods was based on responses to a participant satisfaction questionnaire, administered at the end of intervention. Continuous data were summarized and presented as median, range, and median percent change from baseline to post-intervention within the intervention and control group.
Results
Recruitment and Retention
We aimed to recruit 20 participants to complete the 12-week intervention within the course of approximately 1 year. Recruitment and study participation occurred between September 2017 to December 2018 and consisted primarily of announcements to the University’s faculty/staff list serve via emails, and flyers distributed to gastroenterology centers and the regional cancer center. A total of 68 individuals were assessed for eligibility, 23 enrolled, for a 33.8% accrual rate. Of the 23 participants randomized to one of the 2 study groups, 20 completed the intervention between September 2017 and December 2018, for a retention rate of 86.9%. Three participants withdrew from the trial due to constipation (N = 1), preferred to join weight watchers (N = 1), or didn’t like the food (N = 1). No adverse events were reported. Participant flow through the study is shown in Figure 2, and baseline participant characteristics are shown in Table 2.

Study design.
Baseline Study Participant Characteristics Across Study Groups a .
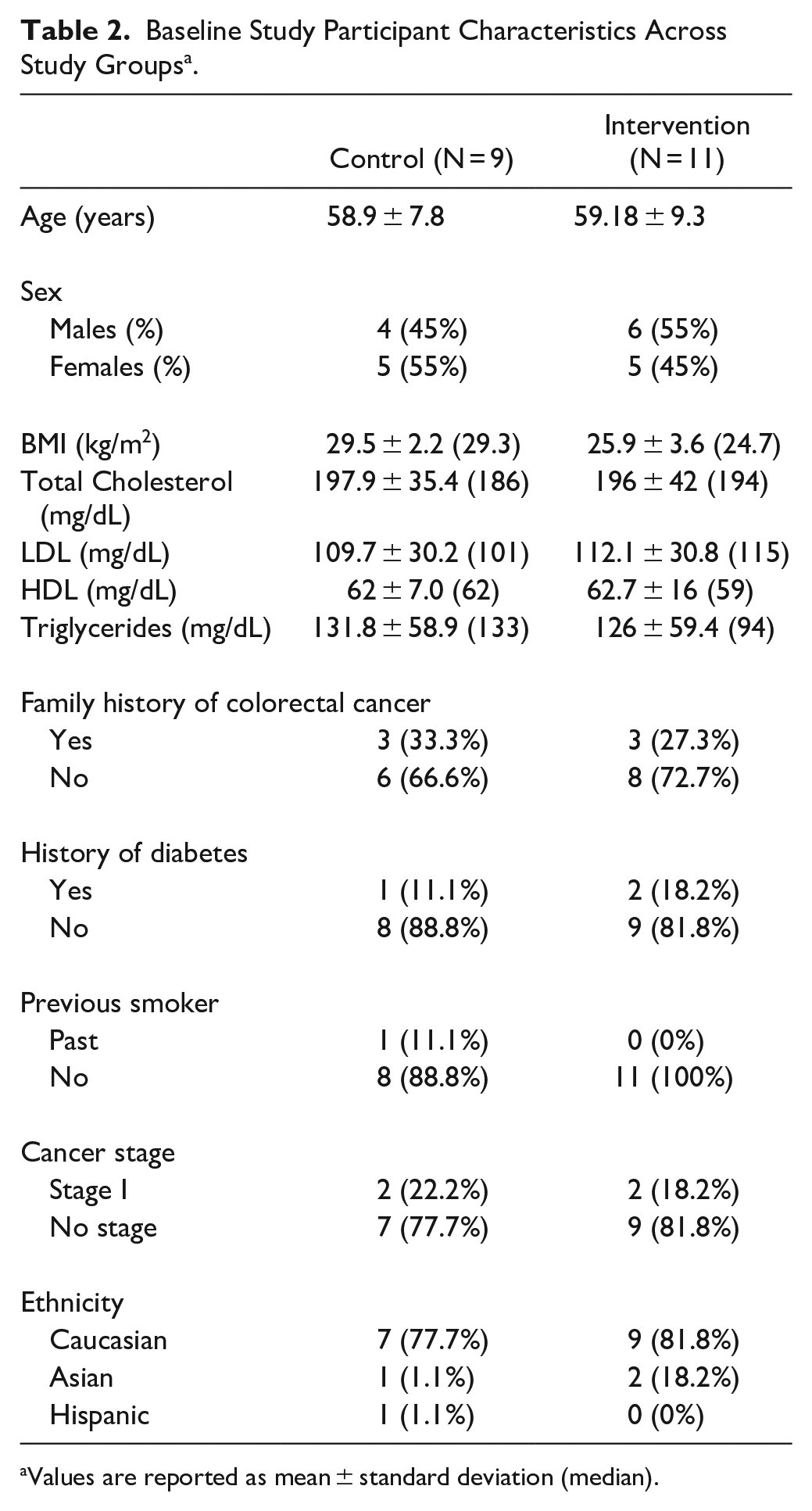
Values are reported as mean ± standard deviation (median).
Compliance to Study Foods
Compliance to the rice bran and navy bean intervention averaged 92% (range 80%-100%) compared to 89% (range 82%-100%) in the Fibersol-2® control. The intervention group increased consumption of dry beans from 2 g/day at baseline to 30 g/day at post-intervention, and increased consumption of rice bran from 0 g/day at baseline to 30 g/day at post-intervention. Figure 3 illustrates percentage of total caloric intake accounted for by rice bran and navy beans at baseline, mid-point, and post-intervention.

Rice bran and navy bean consumption.
Compliance to, and Acceptability of Study Procedures
Of the 20 participants who completed the intervention, N = 11 (55%) completed optional bi-weekly blood draws, and N = 20 (100%) completed the required blood draws (at baseline, midpoint and endpoint). All participants, N = 20 (100%) supplied urine samples for three-time points (baseline, week 6 and week 12). Of the 20 participants who completed the intervention, N = 19 (95%) supplied stool samples at baseline and post-intervention. Of the 40 activPAL data collections (20 participants × two-time points), N = 20 (100%) were compliant with procedures and provided valid data. At the completion of the intervention, 16 (80%) of participants completed the intervention satisfaction questionnaire. Detailed information from this questionnaire is shown in Table 3. Overall, participants were very satisfied with their experience in the study and would encourage others with increased risk of CRC to participate. The majority of comments provided were related to study foods and personal preferences.
Participant Satisfaction Survey.

Changes in Dietary Fiber, Whole Grains, HEI, and PA
Compared to baseline, post-intervention median scores for fiber intake and total HEI score increased by a similar magnitude, whereas HEI whole grain score increased to a much greater extent in the intervention group (Table 4). As expected, there was minimal change in PA from baseline to post-intervention in both the intervention and control group, with the largest magnitude of change in light PA (Table 4).
Change in Fiber, Whole Grains, Overall Diet Quality and Physical Activity.

HEI-2015 total score 100 maximum.
HEI-2015 Whole grain component score, 10 maximum.
A MET is a rate of intensity where 1 MET is approximately equal to 1 kcal/kg/hour and roughly equivalent to the energy cost of sitting quietly. MET-hours are calculated by multiplying the MET value of a given activity by the duration (in hours) it was performed. In this study, MET-hours for all daily activities are then summed to provide an estimate of total daily physical activity.
Discussion
A dietary fiber intervention using a combination of rice bran and navy bean among individuals at high risk for CRC (i.e., previous adenomatous polyps removed) has potential to become a high impact lifestyle intervention for primary and secondary CRC prevention. Notably, no previous studies have examined the feasibility and acceptability of the dietary combination of rice bran and navy bean for increasing dietary fiber and while also addressing other healthy lifestyle factors such as physical activity. The results from this pilot study support the feasibility and acceptability of a rice bran and navy bean intervention that included physical activity education. Recruitment was timely, compliance to study foods and procedures was high, and the intervention was overall well-received, with participants providing constructive feedback regarding study foods and procedures. Results from this study also provided support for practical means to improve daily fiber intake, overall diet quality, and specifically, whole grain intake.
Total HEI score increased in both the intervention and control group, and HEI whole grain score increased to a greater extent in the intervention group. The HEI-2015 scoring system ranges from 0 to 100 and has an overall HEI score that is made up of 13 components that reflect the different food groups and key recommendations in the 2015 to 2020 Dietary Guidelines for Americans. 29 Higher HEI scores have been associated with decreased risk for CRC in men and women. 30 The total HEI-2015 score for Americans is 59 out of 100, indicating that average American diet does not conform to dietary recommendations. 31 Dietary Guidelines for Americans suggests eating 6 ounces of grains each day and half of your grain intake as whole grains (based on a 2000-calorie diet), which would equate to a HEI score of 10 out of 10. 32 Studies have shown that increase of whole grains is associated with decreaded risk of colorectal cancer recurrence and mortality, 33 and insufficient whole grain consumption is a leading dietary factor associated with preventable cancer burden. 34 At baseline, none of the participants in the current study achieved a HEI whole grain score of 10, but at post-intervention N = 11 in the intervention group scored a 10 (compared to N = 0 in the control group at post-intervention). Taken together, our findings support improvement in overall HEI score with fiber supplementation, and improvement in whole grain component score for the intervention group. Contributions of other macronutrients, and total caloric intake are shown in Supplemental file 1.
While both the control and intervention groups received daily fiber, the intervention group maintained higher amounts at week 6 and week 12. There is a general guideline, 14 g of fiber per 1000 calories intake in the diet. 35 Currently the dietary reference intakes (DRI) for adult’s total fiber ranges between 21 and 38 g/day depending on age and sex, with less than 3% of Americans meeting recommended intake. 36 Dietary fiber is a major macronutrient with convincing evidence for decreased risk of CRC. 37 Hayes et al 38 reported that dietary fiber was associated with decreased risk of colon adenoma. The National Institutes of Health-AARP Diet and Health Study showed fiber specifically from whole grains and legumes (namely, beans) was associated with a lower risk of CRC. 39
Although the results of this intervention are promising, several limitations should be considered. There was a small sample size, yet it was sufficient for a pilot trial design and supports feasibility for recruiting larger cohorts to evaluate changes in efficacy outcomes such as colonic inflammation and recurrence of polyps-neoplasia. Also, a majority of participants enrolled were already achieving high levels of PA at baseline, and thus we did not anticipate substantial increases in PA due to the education intervention. Future investigations with this dietary intervention may seek out sedentary or inactive individuals, and include a supervised PA intervention component.
One strength of this study was the novel and practical approach using “real foods” (eg, snacks, casseroles, etc.) that are commonly consumed, yet with inclusions of rice bran/bean powders. This approach allowed for flexibility in participants’ free-living environment, and likely contributed to the high compliance rates. While preliminary, our results demonstrate increased rice bran/navy bean consumption with subsequent improvements in whole grains and overall diet quality. These results are encouraging when integrated with the monitoring of physical activity. Further, the investigation of microbiome and metabolome changes are additional tools that will help us to better understand the metabolic mechanisms in the prevention of neoplastic lesions into colon cancer, and through mitigation of inflammation.10,28,40
In summary, a lifestyle intervention, which includes both a dietary component of rice bran and navy bean and PA promotion, represents a plausible strategy for primary and secondary CRC prevention. A larger randomized controlled trial is needed to evaluate efficacy, and to explore the physiologic mechanisms for cancer prevention (eg, microbiome, metabolomics, inflammation).
Supplemental Material
Supplementary_File_1_macronutrients – Supplemental material for Feasibility of Beans/Bran Enriching Nutritional Eating For Intestinal Health & Cancer Including Activity for Longevity: A Pilot Trial to Improve Healthy Lifestyles among Individuals at High Risk for Colorectal Cancer
Supplemental material, Supplementary_File_1_macronutrients for Feasibility of Beans/Bran Enriching Nutritional Eating For Intestinal Health & Cancer Including Activity for Longevity: A Pilot Trial to Improve Healthy Lifestyles among Individuals at High Risk for Colorectal Cancer by Heather J. Leach, Bridget A. Baxter, Melanie N. Beale, Hillary V. Smith, Sangeeta Rao, Sarah Hibbs-Shipp and Elizabeth P. Ryan in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a pilot grant from the University of Colorado Cancer Center (P30CA046934), and graduate research assistantships from the Colorado School of Public Health at CSU.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
