Abstract
Periampullary carcinoma refers to a malignant tumor within 2 cm of the duodenal ampulla. Primary ampullary carcinoma is very rare, accounting for only 0.2% of malignant gastrointestinal tumors. The small intestine accounts for 75% of the length of the gastrointestinal tract, and primary tumors in the small intestine account for only 2% of all gastrointestinal tumors. Here, we report the case of a duodenal ampullary tumor with malignant transformation of parapapillary polyps. The patient had both a primary ampullary tumor and high-grade intraepithelial neoplasia of juxtapapillary adenomatous duodenal polyps.
Keywords
Introduction
Periampullary cancer is a malignant tumor that occurs within 2 cm of the duodenum. The lesion in periampullary cancer is mainly located in the lower part of the common bile duct, ampulla of Vater, opening of the pancreatic duct, duodenal papilla, and the nearby duodenal mucosa, and the incidence accounts for approximately 5% of all gastrointestinal malignancies. 1 Ampullary cancer belongs to a subset of tumors around the ampulla, and specifically refers to malignant tumors that occur in the distal ampulla of Vater where the common bile duct and pancreatic duct merge. Primary ampullary cancer is very rare, accounting for only 0.2% of gastrointestinal malignancies.2,3 The small intestine accounts for 75% of the length of the gastrointestinal tract, but tumors that originate in the small intestine account for only 2% of all gastrointestinal tumors. 4 The main nipple is in the descending section of the duodenum. Adenomas that occur in this area account for only 10% of all duodenal adenomas, but the rate of malignant transformation is high. 5 The occurrence of small intestinal adenocarcinoma is similar to the adenoma-adenocarcinoma progression pattern of colon cancer. The patient in this report had both a primary ampullary tumor and high-grade intraepithelial neoplasia of duodenal parapapillary adenomatous polyps, which has rarely been reported.
Case presentation
A middle-aged male patient presented with upper abdominal pain of 2 weeks’ duration. Physical examination showed mild tenderness in the upper abdomen, and no other specific symptoms. Outpatient examination showed positive occult blood in stools, and no obvious abnormalities were found on abdominal ultrasonography. Gastroscopy showed a polypoid bulge next to the nipple in the descending duodenum. After admission, further examination revealed that total bilirubin concentration was 29.8 μmol/L, alkaline phosphatase concentration was 256 U/L, and gamma-glutamyl transferase concentration was 339 U/L. Abdominal enhanced computed tomography (CT) demonstrated a nodular shadow at the lower end of the common bile duct near the duodenal papilla, and both the common bile duct and the proximal main pancreatic duct were dilated (Figure 1). Duodenoscopy (Figure 2) revealed a polypoid bulge approximately 1 cm below the main nipple in the descending duodenum. The texture was brittle, and the nipple bled easily when touched. A biopsy was performed, and pathology revealed a villous adenoma. Endoscopic ultrasonography (Figure 3) demonstrated a hypoechoic area in the ampulla, which was enlarged, and the boundary was blurred. Doppler ultrasonography revealed a more obvious blood flow signal, and elastography revealed that the texture of the lesion was hard. The main pancreatic duct and common bile duct were dilated. Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) biopsy was performed, and three ampullary biopsies were obtained. Pathology revealed adenocarcinoma.
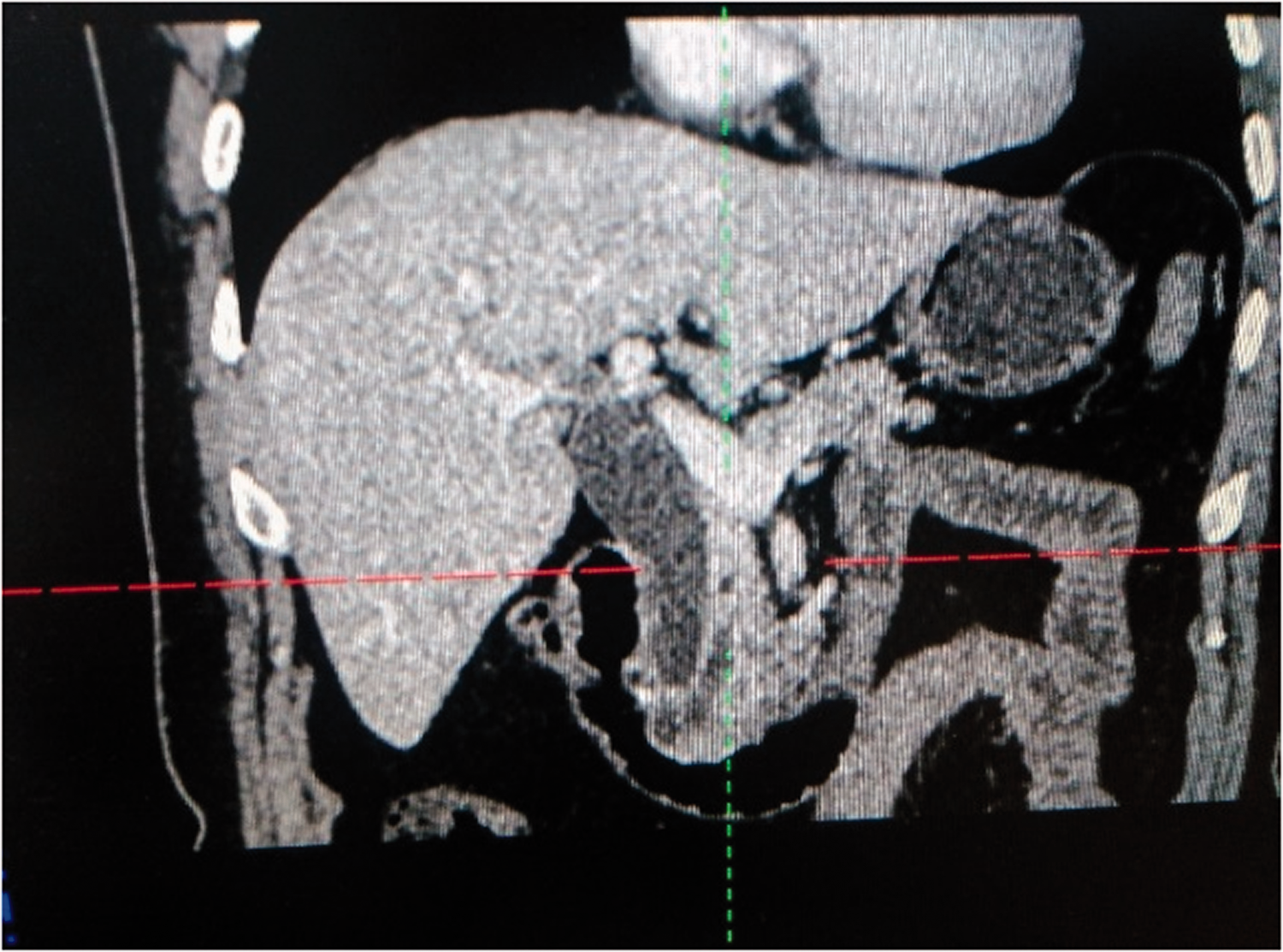
Enhanced computed tomography (CT) images of the upper abdomen. A nodular shadow is seen at the lower end of the common bile duct near the duodenal papilla, and the common bile duct and the proximal main pancreatic duct are dilated.

Polypoid bulge (red arrow) seen approximately 1 cm below the main nipple in the descending duodenum.

Endoscopic ultrasonography. The low-echo area of the ampulla is enlarged, and the boundary is blurred. Elastography showed that the lesion was hard. The main pancreatic duct and common bile duct are dilated.
Follow-up
After surgical consultation, the patient chose pancreaticoduodenectomy (PD). Postoperative pathology (Figure 4) demonstrated an adenocarcinoma of the ampulla, measuring 2.7 × 2.7 × 0.8 cm, which infiltrated the muscle layer, with suspicious intravascular tumor thrombus, and no clear lymph node metastasis or nerve invasion (Figure 5a). Immunohistochemical findings indicated that the tumor was cytokeratin (CK) 20-positive (+) (Figure 5b). A polypoid bulge was seen approximately 1 cm from the nipple. The tumor measured 0.8 × 0.7 × 0.5 cm and was identified as a tubular adenoma with focal high-grade intraepithelial neoplasia, pathologically (Figure 5c).
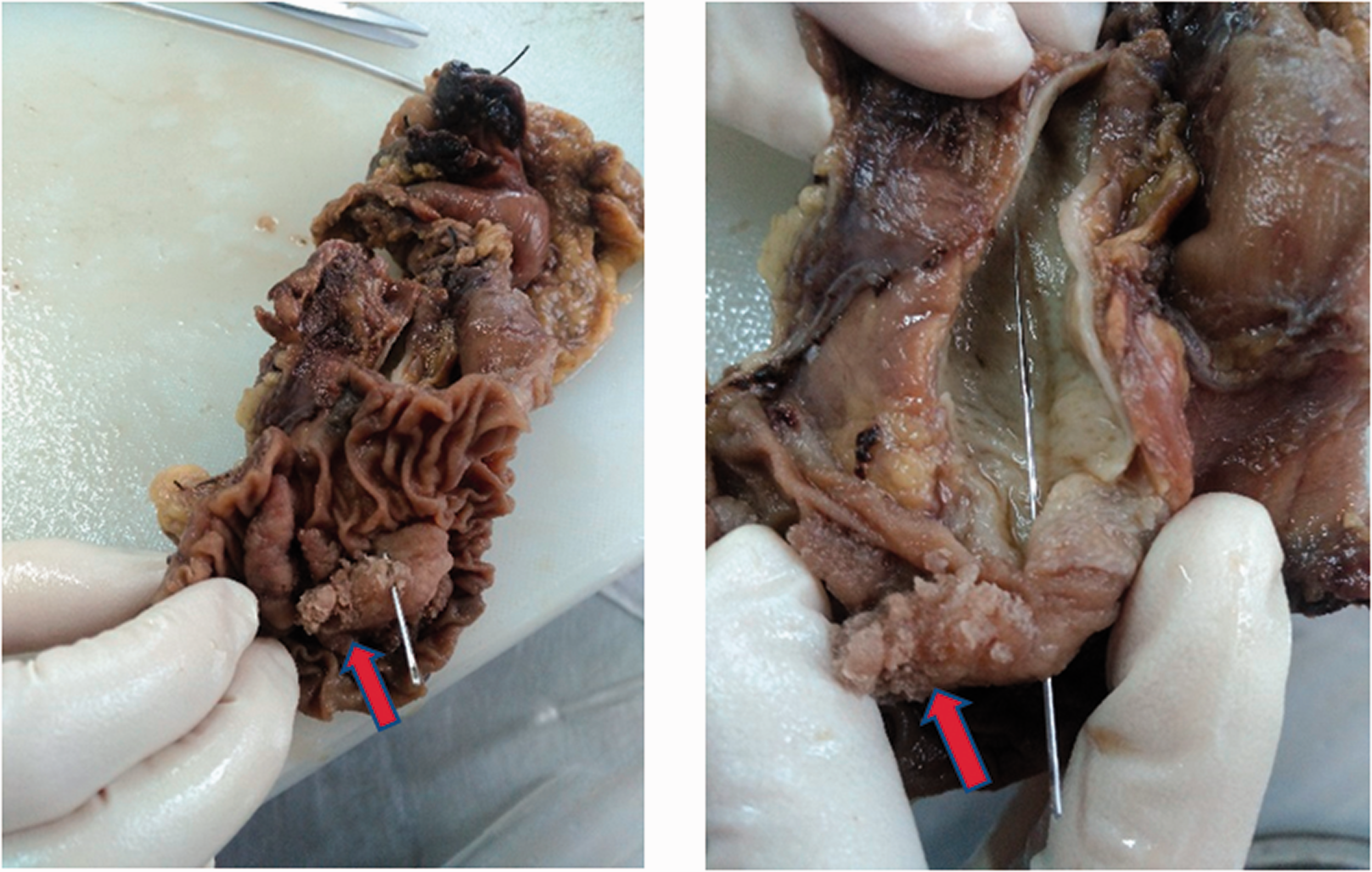
Pathology findings. The image shows an ampullary tumor (blue arrow) and polyps with high-grade intraepithelial neoplasia near the nipple (red arrow).

Representative hematoxylin and eosin (HE)-stained images (a, c) and immunohistochemistry (b, cytokeratin (CK)) sections under digital microscopy (original magnification: ×200). (a) adenocarcinoma (b) cytokeratin (CK) (c) adenoma.
Discussion
Histopathological classification of ampullary cancer comprises adenocarcinoma, papillary adenocarcinoma, mucinous adenocarcinoma, and undifferentiated carcinoma, of which adenocarcinoma and papillary adenocarcinoma are more common. 6 From a pathological point of view, these tumors appear in the transition zone between two different epithelia, and can be divided into an intestinal type and a pancreaticobiliary type according to their origin. The former is mostly exogenous or has a bulging growth pattern, and the latter shows mainly infiltrative growth.7,8 Therefore, the prognosis differs between the types. Clinically, most tumors in the area around the ampulla involve not only the ampulla, but also the pancreas, distal bile duct, and the duodenum, and it is often difficult to distinguish the preoperative diagnosis. 3 Perysinakis et al. 9 found that CK20+ status identifies intestinal ampullary carcinoma, and MUC1+ status identifies pancreaticobiliary ampullary carcinoma. Following the pathology results in our case, the patient was diagnosed with intestinal ampullary cancer.
The anatomical structure around the ampulla is complex because it is adjacent to important tissues and blood vessels, combined with the influence of cavity organs, such as the gastrointestinal tract; thus, early diagnosis of ampulla cancer is often difficult. The diagnosis of ampullary lesions should include localization and characterization. Localized diagnosis comprises the duodenal nipple, ampulla, lower part of the common bile duct, pancreatic head (including the pancreatic head uncinate process), and the area within 2 cm of the ampulla of Vater. Qualitative diagnosis includes inflammation, calculus, tumor, and others. Common diagnostic methods are abdominal CT, magnetic resonance cholangiopancreatography (MRCP), duodenoscopy, endoscopic retrograde cholangiopancreatography (ERCP), and EUS. Studies have shown10,11 that the specificity and sensitivity of MRCP in the diagnosis of various bile duct stenoses is slightly lower than that of ERCP, but there is no significant difference between the two modalities. Owing to its advantages of non-invasiveness, high diagnostic rate, and cost-effectiveness, MRCP is beneficial in the diagnosis of biliary stenosis compared with transabdominal ultrasonography, abdominal CT, and ERCP. For malignant bile duct obstruction, such as cholangiocarcinoma, MRCP has a sensitivity of 77% to 86% and a specificity of 63% to 98%. 12 ERCP can be used to make real-time and dynamic observations of the pancreaticobiliary duct, and this method has high diagnostic value for obstructions in which the cause is difficult to determine.13,14 The sensitivity of cytological diagnosis during ERCP is less than 30%, and the positivity rate of biopsy under X-ray guidance can be increased to 40% to 70%.15,16 With the clinical application of choledochoscopy (SpyGlass Direct Visualization System; Boston Scientific, Natick, MA, USA), the diagnostic rate of biliary system tumors can be increased to more than 90%. 17 As ERCP is an invasive examination, the incidence of related complications, such as bacterial cholangitis, pancreatitis, biliary tract perforation, and bleeding is 7% to 20%, and the postoperative hospitalization rate is as high as 10%. 18 Therefore, for many doctors, ERCP is often not the first choice for diagnosis.
For ampullary tumors, EUS can eliminate intestinal gas interference, and can more accurately determine the relationship between the lesion and the surrounding tissue structure. A meta-analysis involving 422 patients in 14 clinical studies showed that the diagnostic sensitivity and specificity of EUS for T1 ampullary tumors was 77% and 78%, respectively, and the specificity was 84% and 74%, respectively. 19 In addition, Ogura et al. 20 reported that the diagnostic accuracy of EUS-FNA for ampullary lesions was almost 100%. Studies have shown that EUS is superior to CT, magnetic resonance imaging (MRI), and transabdominal ultrasonography in the diagnosis of smaller ampullary cancers. Chien 21 EUS can assess the relationship between the lesion and the structure surrounding the ampulla and whether the lesion extends into the biliary and pancreatic ducts, and assists in judging the T and N stages of ampullary tumors before surgery.22–25 Owing to abdominal discomfort and positive fecal occult blood, our patient was admitted to the hospital for endoscopic polyp treatment. Laboratory examination then showed that the bilirubin concentration was increased, and CT showed ampullary lesions. Finally, with the help of EUS-FNA, pathological examination confirmed the diagnosis and staging, and follow-up treatment was planned.
The treatment of ampullary cancer involves surgical resection, radiotherapy and chemotherapy, and palliative treatment for patients who cannot be cured by surgery. Surgical resection is the only treatment providing a potential cure for an ampullary tumor. The main surgical methods are pancreatoduodenectomy (PD), or Whipple’s procedure, local resection (transduodenal ampullectomy (TDA)), and endoscopic ampullary resection (EA). Local resection for ampullary tumors has the advantages of less trauma, less bleeding, and quick recovery. Most scholars believe that the indication for local resection of an ampullary tumor is a benign tumor or an early tumor with local malignant transformation.26,27 To date, most studies have shown that endoscopic treatment is most suitable for benign lesions, which can be completely removed.28,29 Thus, higher requirements are needed for diagnostic and endoscopic treatment technologies. Compared with traditional PD, endoscopic papilloma resection and transduodenal local resection are associated with fewer perioperative complications and lower mortality rates. 30 However, the limitation of these interventions is that incomplete resection may occur, and appropriate lymph node dissection cannot be performed. Even in early disease, the possibility of successful resection is higher, but over time, approximately 45% of the treated population will relapse. 31 Therefore, PD remains the gold standard treatment for ampullary tumors. Radical PD should be performed for ampullary lesions highly suspected of malignancy or that are confirmed by endoscopic biopsy. 32 The present patient was diagnosed with a malignant ampullary tumor accompanied by a parapapillary adenoma; therefore, PD was performed. In addition, there is a relationship between ampullary adenoma and familial adenomatous polyposis, which requires screening colonoscopy for follow-up evaluation. 33 There was no obvious abnormality on enteroscopy before discharge, in our patient.
Conclusion
An ampullary tumor is a rare disease, but because of its special location, compared with pancreatic cancer, patients with ampullary tumors often present with symptoms at an earlier stage. 34 Therefore, the disease involves a higher resection rate. 34 The key to improving our understanding of this disease is early localization and qualitative diagnosis by imaging, endoscopy, and pathological biopsy. The best individualized treatment plan for patients comprises surgery, chemotherapy, radiotherapy, targeted therapy, and multidisciplinary combinations.
Footnotes
Ethics statement
The authors obtained approval from the Ethics Committee of Shidong Hospital of Shanghai to publish the case details (approval no.: 2021-17). The patient provided written informed consent for the publication of her images and case details. The reporting of this study conforms to the CARE guidelines. 35
Data sharing statement
The data generated and analyzed during the current case report are not publicly available to protect patient privacy, but can be obtained upon reasonable request from the corresponding author, Dr Xuanfu Xu (email address:
Declaration of conflicting interest
All authors have completed the ICMJE disclosure form. The authors have no conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
