Abstract
Introduction:
Cancer-related fatigue (CRF) occurs frequently in breast cancer patients. The aim of this real-world study was to analyze the longitudinal changes of CRF in breast cancer patients receiving an integrative medicine program, which includes the application of non-pharmacological interventions (NPIs) and Viscum album L. (VA) extracts.
Methods:
All data were collected from the clinical register of the Network Oncology of a German certified breast cancer center of the Gemeinschaftskrankenhaus Havelhöhe (GKH). Primary breast cancer patients, treated upon initial diagnosis with integrated NPIs, comprising art and exercise therapy, nursing interventions, and educational components, during their hospital stay, and who had answered the German Cancer-Fatigue Scale (CFS-D) questionnaire at first diagnosis and 12 months later, were included. The associations between NPIs and CFS-D changes were analyzed with adjusted multivariable regression analyses, considering received treatment regimens and demographic variables, using the software R.
Results:
231 female breast cancer patients of all tumor stages were evaluated. While chemotherapy exhibited significant severe deterioration, add-on VA applications seem to partially mitigate this impairment on CRF. 36 separate multivariable regression analyses for all NPIs showed that in particular significant associations between CFS-D improvements and the interventions nursing compresses (6 point change; P = .0002; R² = 28%) or elaborate consultations and life review (ECLR) (4 point change; P = .0002; R² = 25%) were observed.
Conclusions:
Breast cancer patients benefit from a hospital-based integrative medicine program. To alleviate fatigue symptoms during oncological therapy, an expansion of this concept should be developed in the future.
Keywords
Introduction
Cancer-related fatigue (CRF), accompanied by psychological disorders, sleep disturbances, and cognitive impairments, is one of the most frequent symptoms in cancer patients, and this manifold symptom complex has a profound negative impact on daily and social activities, as well as on health-related quality of life (HRQL).1,2 Breast cancer still is the most common cancer in women worldwide and due to the growing effectiveness of oncological therapies the number of cancer survivors is increasing. 3 About 33% of breast cancer patients experience moderate to severe fatigue symptoms, not infrequently even persistently many years after oncological treatment.4-6 For supportive care, breast cancer patients commonly wish to make use of complementary and integrative medicine methods.7,8 However, clinical evidence regarding supportive interventions in oncology is limited. Educational treatments were often described as a component of psychosocial interventions to ameliorate CRF during cancer treatment 9 and currently it is accepted that moderate physical activity and psychological/educational interventions are beneficial therapeutic strategies to prevent and counteract fatigue symptoms. 10 From meta-analyses it was concluded that educational interventions may have reducing effects on fatigue. 11 In a systematic literature review it was presumed that especially mind-body therapies such as meditation and yoga are effective supportive strategies during care of breast cancer patients. 12 Other systematic reviews and meta-analyses revealed that significant fatigue benefits were achieved with supervised, combined aerobic-resistance exercises and also with some other non-pharmaceutical interventions.13-15
In the breast cancer center at the Gemeinschaftskrankenhaus Havelhöhe (GKH) which has been certified by the German Cancer Society since 2012, integrative strategies are implemented in the routine care of cancer patients.16,17 Based on scientific standards and guidelines of conventional medicine, the integrative concept of Anthroposophic Medicine includes additional pharmacological and non-pharmacological interventions (NPIs) such as nursing applications (eg, compresses, embrocation, and therapeutic bathes), art therapy (eg, music and painting), and mindfulness-oriented exercise therapy (eg, eurythmy therapy), and also educational components. 18 Among pharmacological treatments, extracts of mistletoe, Viscum album L (VA) are used in integrative medicine in breast cancer patients during chemotherapy, to reduce adverse side effects and to enhance quality of life.19-21 Furthermore, conducting elaborate consultations and life review (ECLR) has always been part of Anthroposophic concepts. 22 We previously reported that ECLR was associated with relevant improvements in global health/ quality of life and emotional, social, and cognitive functioning of breast cancer patients. 23 In addition, recently it was shown that a multimodal treatment concept with NPIs can lead to an enhancement of the internal coherence and resilience in breast cancer patients. 24
The present real-world study evaluated longitudinal changes in self-reported fatigue in breast cancer patients, receiving a hospital-based integrative medicine program. CRF was assessed by the German Cancer Fatigue Scale (CFS-D),25,26 which enables to distinguish between affective, physical, and cognitive characteristics of fatigue. The CFS-D questionnaires were assessed at first diagnosis and 12 months thereafter.
Methods
Study Design and Patients
We conducted a non-controlled, non-randomized longitudinal real-world study by analyzing patient registry data (Network Oncology, NO). The NO is a conjoint clinical register of hospitals, practitioners and out-patient centers for the evaluation of integrative oncological therapy concepts in health services research oncology. 27 Oncological patients from whom written informed consent has been obtained were included in the NO. Demographic data as well as information on diagnosis, histology, and treatment regimens were documented and surveys of questionnaires on quality of life at different time points were conducted. For the present study, patients with a histologically proven primary diagnosis of breast cancer with all tumor stages were included.
Endpoints
The aim of this study was to monitor the longitudinal changes of the CFS-D scales of breast cancer patients from first diagnosis (T0) and subsequent 12 months (T1) thereafter, considering their applied oncological treatments, and NPIs.
Data Collection
Primary NO breast cancer patients who were seen at the certified Breast Cancer Centre GKH, Berlin, Germany between June 2012 and April 2019, were screened as previously reported.23,28,29 According to the German National Cancer Plan social services and psycho-oncological support were offered to all patients. At first diagnosis during the hospital stay, the patient received standard therapies in conjunction with integrative ones and appointments with NPI therapists were arranged in a personalized manner as described in detail previously.17,24 The following 9 different NPIs were the most frequently used by the breast cancer patients, and were the subject of present study: nursing interventions (embrocation, therapeutic bath, and compresses), rhythmic massages, exercise therapy (eurythmy therapy and physiotherapy), art therapies (music and painting), and elaborate consultations and life review (ECLR) treatment. The patients visited the surveillance and study center at different time points. After receipt of their written consent to be included in the NO-study, the patients received and answered the questionnaires. For the present evaluation the CFS-D questionnaires at T0 and T1 were assessed.
All data reported here are based on retrievable data from the NO registry at cut-off date of April 15, 2019. Patients, from which assessable data-sets at least for T0 and T1 were available, were enrolled in the present study. Furthermore, only female primary breast cancer patients >18 years were included. In addition, demographic and medical data (diagnosis, histology, pre-treatment and treatment, received medications and NPIs) of the enrolled patients were retrieved from the NO.
Ethics Approval and Consent to Participate
The study complies with the principles laid down in the Declaration of Helsinki. The NO study has been approved by the ethics committee of the Medical Association Berlin (Berlin – Ethik-Kommission der Ärztekammer Berlin). The reference number is Eth-27/10. This study had been retrospectively registered at the WHO approved register German Register for Clinical Trials (Deutsches Register Klinischer Studien, DRKS), trial registration number DRKS00013335 on 27/11/2017. Written informed consent has been obtained from all patients prior study enrollment.
Analyses of Cancer-Related Fatigue
For exploratory evaluation of longitudinal effects on fatigue the German version of the Cancer Fatigue Scale (CFS-D) was used and analyzed. The questionnaires were assessed at T0 and T1. The Cancer Fatigue Scale was originally developed in Japan 26 and after transcultural adaption was validated in German. 25 The CFS-D consists of a 15-item questionnaire on 3 subscales (physical, cognitive and affective fatigue), based on a 5-point Likert scale with a possible range of 0 (no fatigue) to 60 (maximum fatigue). 25 A decline of scores indicates an improvement of fatigue while higher scores represent a higher degree of fatigue symptoms respectively. The CFS-D is highly reliable with a robust validity and classifies values ≥30 points as clear symptoms of fatigue, ≥24 points as suspected moderate fatigue, and ≤23 points as no or only minor fatigue symptoms. 30
Statistical Analysis
All statistical analyses were performed using the software R (R Version 3.1.2 [2014]). 31 Continuous variables were described as median with interquartile range (IQR); categorical variables were summarized as frequencies and percentages. Student’s t-tests were applied, to detect differences; P-values <.05 were considered to be significant. All analyses were descriptive, therefore no p-value adjustment for multiple comparisons were made. For characterization of group differences, Pearson’s Chi-squared tests with Yates’ continuity correction were performed. To identify influencing factors and to address potential sources of bias, adjusted multivariable linear regression analyses were performed and potential confounders were addressed. In order to yield reliable model results, stepwise regression selections were performed and models with high adjusted R2 were chosen. According to Cohen’s interpretation 32 R2 values between 13% to 25% indicate medium and R2 values 26% or above indicate high effect sizes. Predicting or mediating variables were age (in years), body mass index BMI (in classes: underweight (BMI < 18.5), normal (18.5 ≤ BMI < 25), overweight (25 ≤ BMI ≤ 30), obese (BMI ≥ 30)), Union for International Cancer Control (UICC) tumor stage (numerical, 0 - IV), hormonal status (categorical, post-, peri-, pre-menopausal), and the respective CFS-D values at T0. Received treatments with chemotherapy (Ctx) and mistletoe (VA) (categorical, no treatment, Ctx only, VA only, Ctx+VA) and endocrine treatment (yes/no). For the 9 different NPIs, rank-ordered variables were assigned. Zero was allocated for no treatment, 1 was allocated when treatment occurred between T0 and T1, and when patients filled in the T0 questionnaire after the application period of NPIs, in those cases 0.5 was assigned. To analyze synergism of different NPIs a sum parameter for the different NPIs were formed.
Results
Patients’ Characteristics
In total, 425 breast cancer patients, treated between 2012 and 2019 at the certified German Breast Cancer Centre, GKH received CFS-D questionnaires at different time points. A total of 194 patients were not yet available for analyses, of which 92 patients were still under follow-up at cutoff time and for 102 patients no completed questionnaires for both time points T0 and T1 were available for any reasons. For 231 patients, assessable data-sets at least for T0 and T1 were available and were included in this analysis (Figure 1). In Table 1 the main characteristics of the 231 analyzed patients are given. At T0, the median age of the entire study cohort was 59 years and the median body mass index (BMI) was 24. All different UICC tumor stages 0-IV were represented, 56% had early stage (tumor stage 0 or I) cancers (Table 1). The hormonal status of the majority (70%) was postmenopausal. 13 patients (6%) were triple negative. The oncological interventions which were applied to the patients after surgery were listed (Table 1). 81% of the patients received radiotherapy, 67% endocrine therapy, 37% mistletoe treatments, 61 patients (26%) received chemotherapy, and 21 of them in addition immunological therapy. For 11 patients the hormonal status, BMI, or tumor stage respectively was not known (Table 1); accordingly, data from 220 patients were available for multivariable analyses (Figure 1).

Flow chart of the study population.
Demographic Data and Characteristics of Primary Breast Cancer Patients at Baseline.

Abbreviations: BMI, body mass index; ECLR, elaborate consultations and life review; IQR, interquartile range; UICC, union for international cancer control stages at first diagnosis according to the 7th edition of the ‘TNM Classification of Malignant Tumors’; n, number of patients and portion (%).
CFS-D Evaluation
Completed CFS-D questionnaires were evaluated and analyzed for the entire study cohort at T0 and T1. The CFS-D total scale and all the subscales were determined for T0 and T1 (Table 2). Statistical analysis revealed no clinically relevant but significant longitudinal increase of total fatigue and in particular a significant increase for the cognitive component at T1 compared to the T0 scales (a mean increment of 0.9 score-points for cognitive fatigue with P = .0001) was observed. However, regarding physical and affective fatigue components respectively, no remarkable changes were detected (Table 2). The longitudinal CFS-D changes from T0 to T1 were calculated and multivariable regression analyses were carried out as outlined in the methods. Except for age as being a continuous variable, all other explanatory variables were of a categorical nature. Of the 61 patients receiving chemotherapy (Ctx) the majority (40 patients) in addition received VA extracts, therefore for Ctx and VA treatment a grouped variable was assigned (no treatment, Ctx only, VA only, Ctx+VA). On multivariable analysis, Ctx was significantly associated with a deterioration of the CFS-D scales (Table 3). The deterioration of the total CFS-D changes was above 5 points if no VA was received (Ctx only; estimate β = 5.44 point change; P = 0.026) and about 3.5 points if Ctx and VA were received (Ctx+VA; estimate β = 3.5 point change; P = 0.064) (Table 3). In particular, Ctx was significantly associated with a deterioration of the physical CFS-D (Ctx only; estimate β = 3.1 point change; P = 0.016), but less pronounced if VA extracts were additionally received (Ctx+VA; estimate β = 1.8 point change; P = 0.013) (Table 3). Also, endocrine treatment was significantly associated with a deterioration of the physical CFS-D (estimate β = 1.55 point change; P = 0.039) (Table 3). However, no significant associations between receiving VA extracts and CFS-D changes were observed (Table 3).
CFS-D Questionnaires of the Entire Study Cohort at First Diagnosis (T0) and 12 months Thereafter (T1).
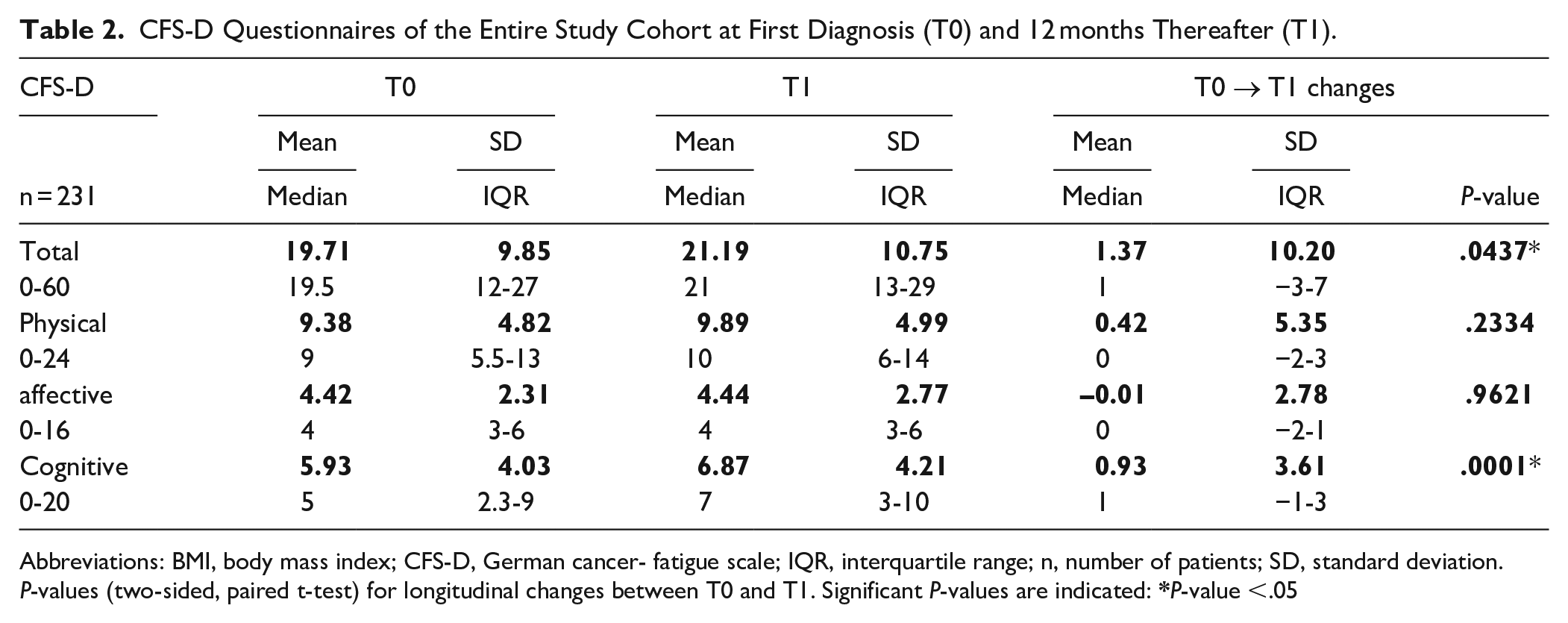
Abbreviations: BMI, body mass index; CFS-D, German cancer- fatigue scale; IQR, interquartile range; n, number of patients; SD, standard deviation.
P-values (two-sided, paired t-test) for longitudinal changes between T0 and T1. Significant P-values are indicated:
Association Factors for Longitudinal Changes of CFS-D Changes and Pharmacological Treatments.

Abbreviation: CFS-D, German cancer-fatigue scale.
Multivariable linear regression analyses using R-statistics were performed for the CFS-D changes (T0->T1). Adjusted were for age and the hormonal status (categorical, post-, peri-, pre-menopausal). Received treatments with chemotherapy (Ctx) and mistletoe (VA) were categorized (no treatment, Ctx only, VA only, Ctx+VA) and endocrine treatment (yes/no). Negative estimate β-values indicate an association with an improvement while positive β-values indicate an association with a worsening of fatigue.
Significant P-values are indicated:
Assessment of Association Factors between NPIs and CFS-D Changes
All patients were offered various different NPIs, and the percentages of those 9 NPIs received and documented for the breast cancer patients are given in Table 1. The most frequent applied NPIs were nursing interventions (96% embrocation, 84% therapeutic bath, 31% nursing compresses), and exercise therapies (85% eurythmy therapy, 71% physiotherapy), and 70% rhythmic massage. Furthermore, art therapies (74% music, 32% painting) were applied and 54 patients (23%) received ECLR treatment. Mean number of the 9 different NPIs applied to 1 patient was 5.66 ± 1.58 NPIs, median: 6, ICR: 5–7 NPIs. One patient received none of these 9 NPIs and 2 patients received all 9 different NPIs. Embrocations can be obtained several times a day, while some other NPIs are more extensive and can only be applied once during the stay in the clinic. As an example, Figure 2 illustrates for 3 patients of the study cohort the NPIs received within a 7-10-day hospital stay. In addition, their individual demographics, time frames, and oncological treatments received over the course of 12 months are also indicated (Figure 2).

NPI treatment regimen course.
Since the CFS-D scales at T0 varied highly (Table 2) they were used as a predictive continuous variable for the following multivariable regression analyses. In Table 4, the multivariable analyses for longitudinal changes of the total CFS-D (T0->T1) and ECLR-treatment and nursing compresses respectively are shown. Significant associations between the respective T0-values and the longitudinal CFS-D changes were observed (estimate β = –0.4 point change; P < .00001; Table 4). Significant association between ECLR treatment and an improvement in fatigue was observed (β = –3.97; P = .02) and also if nursing compresses were received (β = –5.96; P = .0002). Multiple R² of the regression analyses if ECLR therapy was received revealed a medium effect size (R² = 25%) and receiving nursing compresses (R² = 28%) even a strong effect size (Table 4). As in Table 3 a significant association between Ctx and a deterioration of total CFS-D changes (β = 5.8; P = .01) were observed and not significant but elevated fatigue (β = 2; P = .09) was associated if endocrine therapy was received. Furthermore, significant associations between overweight and increased fatigue levels were observed (Table 4; β = 2.7; P = .05). Multivariable linear regression analyses as in Table 4 were also performed for the other 7 NPIs separately, and also for all the CFS-D subscales, thus a total of 36 separate analyses. A summary of these analyses is listed in Table 5. For total CFS-D and all 9 different NPIs the estimate β-values were negative, indicating that between received NPIs and fatigue changes no worsening associations were observed (Table 5). The largest significant effect sizes were observed with nursing compresses, in particular for physical fatigue (β = –2.685; P = 0.0002; R² = 35%). Similarly, significant associations between receiving ECLR therapy and improvements of physical (β = –1.865; P = .026; R² = 33%) and affective fatigue (β = –1.227; P = .009; R² = 25%) were observed. For the CFS-D subscale cognitive fatigue significant associations were observed only if nursing compresses were received (β = –2; P = 0.0004; R² = 25%). In addition, although not significant, receiving eurythmy (β = –1.863; P = .28; R² = 23%) or music therapy (β = 1.415; P = .36; R² = 23%) were associated with improved CFS-D changes (Table 5). However, for therapeutic baths, painting therapy, physiotherapy, rhythmic massages, or embrocation no clinical relevant differences were found (Table 5). To investigate whether the association factors of the NPIs are additive, multivariable analyses with the total number of applied NPIs were performed. The sum of the variables for the different NPIs received varied between 0 and 9 (median = 4, IQR: 3-6). Multivariable linear regression analyses with the sum of received NPIs (Table 5) revealed associations with an improvement of about 0.6 point changes per received NPI for the total CFS-D (β = –0.56; P = .09; R² = 24%), improvements for the physical (β = –0.27; P = .09; R² = 33%), and the affective fatigue (β = –0.15; P = .09; R² = 24%) components were also associated.
Association Factors for Longitudinal Changes of Total CFS-D and ECLR Treatment and Application of Nursing Compresses.

Abbreviations: BMI: body mass index (categorical, in classes), the hormonal status (categorical, post-, peri-, pre-menopausal), and received treatments with chemotherapy (Ctx) and mistletoe (VA) (categorical, no treatment, Ctx only, VA only, Ctx + VA) and endocrine treatment (yes/no); CFS-D, German cancer- fatigue scale; ECLR, elaborate consultations and life review; NPI, non-pharmacological intervention.
Multivariable linear regression analyses using R-statistics were performed for the CFS-D changes (T0->T1). Adjusted were for age, the respective total CFS-D values at T0, the Union for International Cancer Control (UICC) tumor stages (numerical, 0-IV), Negative estimate β-values indicate an association with an improvement while positive β-values indicate an association with a worsening of fatigue.
Significant P-values are indicated:
Association Factors for Longitudinal CFS-D Changes and NPI Treatment.
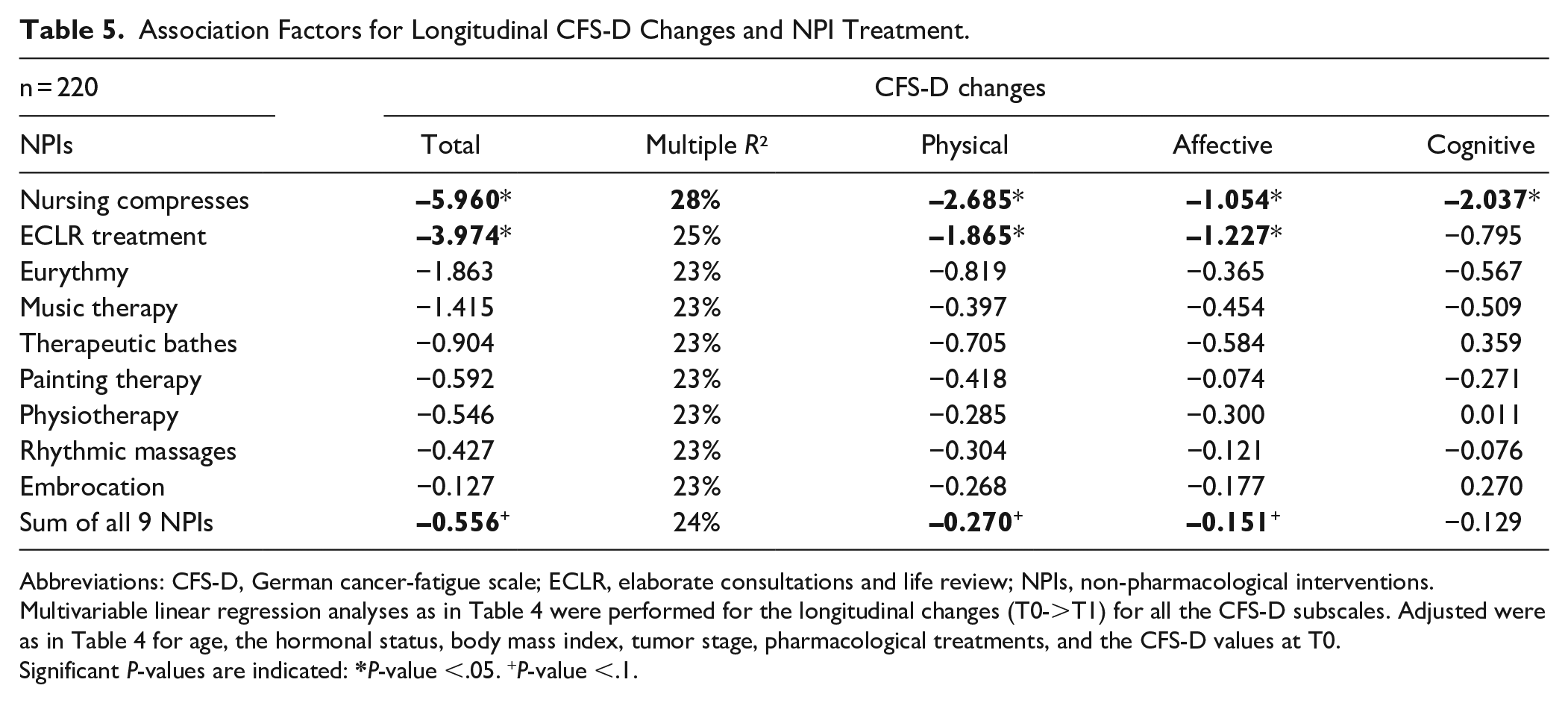
Abbreviations: CFS-D, German cancer-fatigue scale; ECLR, elaborate consultations and life review; NPIs, non-pharmacological interventions.
Multivariable linear regression analyses as in Table 4 were performed for the longitudinal changes (T0->T1) for all the CFS-D subscales. Adjusted were as in Table 4 for age, the hormonal status, body mass index, tumor stage, pharmacological treatments, and the CFS-D values at T0.
Significant P-values are indicated:
Missing Data
A total of 194 patients were not yet available for analyses (Figure 1). For 92 patients their first diagnosis was after December 2017, thus their T1 questionnaires could not yet collected at cutoff time. For the residual 102 patients no completed CFS-D questionnaires for both time points T0 and T1 were available for different reasons. Table 6 shows the base line characteristics of the 102 patients with incomplete data sets compared to the 231 patients of the entire study cohort. The patients with incomplete data were a little younger, however, no significant differences with regard to demographic characteristics were seen (Table 6). For 28 patients no completed CFS-D questionnaires at first diagnosis were available. For the residual 74 patients for which no completed questionnaires at T1 were available, the CFS-D scales were determined at T0 and compared with the entire study cohort (Table 6). T-test analyses (two-sided, unpaired) revealed significant differences between the total CFS-D values at T0 between patients with incomplete data (P = .04) and the entire study group (Table 6). Minor or no fatigue at T0 was self-reported by 64% of the entire study cohort, while only 51% of patients, who had not completed the questionnaires at T1, reported it (Table 6).
Baseline Characteristics of Patients with Incomplete Data Sets.
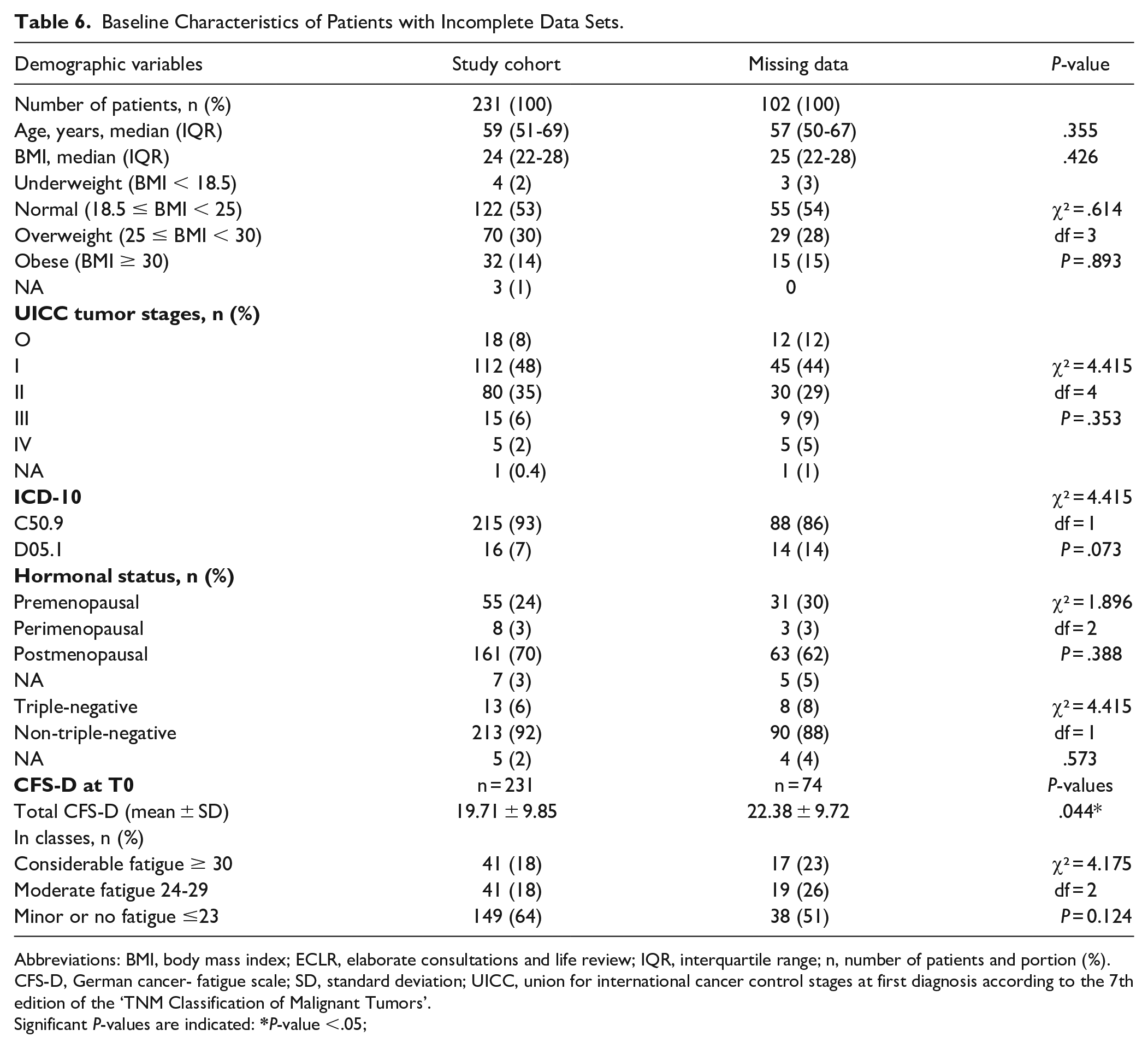
Abbreviations: BMI, body mass index; ECLR, elaborate consultations and life review; IQR, interquartile range; n, number of patients and portion (%). CFS-D, German cancer- fatigue scale; SD, standard deviation; UICC, union for international cancer control stages at first diagnosis according to the 7th edition of the ‘TNM Classification of Malignant Tumors’.
Significant P-values are indicated:
Discussion
In the present longitudinal real-world analysis of primary breast cancer patients, association factors for received NPIs and pharmacological treatments including VA extracts were assessed for CFS-D changes from first diagnosis until 12 months thereafter. Clear associations between an improvement of fatigue and 2 NPIs of an integrative medicine program were observed. Adjusted multivariable linear regression analyses revealed in 220 patients that receiving nursing compresses or ECLR therapy were significantly associated with improvements of fatigue. Furthermore, VA therapy concomitant to Ctx seemed to alleviate the increase of fatigue.
Although breast cancer patients often make use of complementary integrative medicine programs, the contribution and quantification of individual components of multimodal concepts on self-reported outcomes has not yet been specified in detail. Here, using health services research data, we quantify the association factors between distinct treatment components and changes of fatigue burden. Components of the integrative medicine program at the GKH seem to have a favorable outcome on self-reported fatigue of breast cancer patients (Table 5). In line with our previous analyses that ECLR was associated with relevant improvements of HRQL, 23 here we observed that receiving ECLR was also associated with significant improvements of physical and affective fatigue symptoms. In our approach demographic variables were considered and we monitored that the body mass index has a significant influence on the CFS-D scales. Overweight was significantly associated with impaired total CFS-D changes (Table 4). In line with this observation, a longitudinal study with 261 breast cancer patients concluded that increased BMI is a key predictor of aggravated fatigue. 33
Currently, in the clinical practice guidelines on the use of integrative therapies for breast cancer patients, add-on VA applications received Grade C for improving quality of life, in particular CRF during Ctx. 12 In line with this, we found that Ctx showed a strong significant deterioration, which appeared to be partly mitigated by add-on VA applications (Table 3). However, this mitigating effect of VA applications seems to be unrelated to the cognitive fatigue component (Table 3). 34 Nevertheless, Ctx is associated with a severe increase in cognitive fatigue (Table 3), 34 therefore it is important to find any interventions for the cognitive fatigue component. Hence, if chemotherapy has been prescribed, the application of compresses might be effective to alleviate the development of fatigue symptoms.
Strengths of our study are the presentation of real-world data and the prospective, longitudinal data collection. It is noteworthy that in our analyses with the sum of applied NPIs a relevant association was found between the total number of different NPIs received and changes in fatigue, especially for the physical fatigue component with a high effect size (R² = 33%). This may indicate that the favorable outcomes on fatigue could be synergistic with different NPIs, and that this applied multimodal treatment concept may lead to greater benefits on fatigue than a single exercise type. CRF is a very prevalent and distressing long-term side effect among breast cancer patients,4-6 thus it seems unlikely that the evaluated associations with improvements in CFS-D in our study can be fully explained by natural history or regression to the mean. There were various reasons for incomplete records, which were not always registered and clearly identifiable. Some patients dropped out for unknown reasons or could not respond in due time. The proportion of high or moderate fatigue levels at baseline was higher for the patients with incomplete data (Table 6; 49% vs 36%). That is, seemingly the willingness to re-complete the questionnaire 12 months after first diagnosis is better in patients with less fatigue. The potential risk of attrition bias is limited for the results of the present study, as no significant differences in demographic characteristics were found for patients with incomplete data (Table 6) and in the multivariable analyses (Tables 4 and 5) the CFS-D scales at T0 were considered as predictive variable.
A limitation of this study was the monocentric design and the lack of a control group, which did not receive NPIs. Unwanted biases may have been introduced in the analysis, since this study was performed in a non-randomized, non-controlled and un-blinded fashion. Not all initially participating patients filled in all questionnaires completely, representing a major limitation. Furthermore, individual constitutions, comorbidities, outpatient therapies, or received additional routine medications were not considered for analyses. A further limitation bias is that the number or duration of the NPIs received has not yet been taken into account. An expansion of these analyses and quantification of the effects as a function of the number or time of the NPIs received will be evaluated in the future.
A broad variety of psychosocial interventions have been developed for breast cancer patients but their respective effect sizes vary considerably across studies. 35 According to systematic reviews individual and group psychotherapy, as well as psychoeducation interventions improved emotional distress, HRQL, and also fatigue in cancer patients.9,36 A systematic review and meta-analysis of 9 studies involving a total of 1156 breast cancer patients revealed that supervised aerobic exercise in particular was statistically more effective than conventional care in improving fatigue. 15 Currently, multimodal supportive strategies such as mind-body therapies including relaxation techniques, stress management, and mediation, physical activity, 37 and also physical self-management interventions 38 have been established to counteract adversely effects of oncological therapeutics, and seem to generate beneficial effects on their HRQL including fatigue.12,14 An educational program consisting of 6 sessions of 90 minutes was effective in reducing perceived fatigue as well as HRQL, was observed in a German randomized multi-center study with 261 CRF patients. 39 Likewise, in a pragmatic comprehensive cohort study with 126 CRF breast cancer patients it was shown that a multimodal concept where eurythmy therapy and psycho-education were part of the program, HRQL was improved and fatigue significantly reduced.40,41 Exercise and physical activity is proposed as a first-line intervention for CRF. 42 From a detailed systematic review with comparative meta-analyses of 245 studies for exercises and other NPIs it was shown that during cancer treatment relaxation exercises, massage, cognitive-behavioral therapy combined with physical activity, aerobic and resistance training and yoga were able to reduce CRF, showing moderate to large effect sizes. 13
In the present study, the largest effect sizes and significant associations were observed between nursing compresses and improvements of fatigue symptoms, even for the cognitive fatigue component. The application of hot moist compresses on the upper abdomen is used in Japan as a traditional medicine, 43 also in nursing care in Germany, 44 and is commonly used in Anthroposophic Medicine. 18 Currently, there is only little scientific evidence about effects of compresses. In a randomized cross-over study with healthy subjects, all volunteers reported that abdominal hot compresses had been pleasant, most of them fell asleep during the course of hot compresses, and some effects on the liver metabolism were detected. 45 Since metabolic reactions are sensitive to circadian rhythms 46 nursing compresses might influence autoregulation and biorhythm. In another study with heat- and steam-generating sheets to women, a feeling of comfort in the abdomen and improvement of thermal sensation at the fingertips evolved. 47 Since local cold and warm stimuli influence the cerebral hemodynamics and metabolism, 48 it was suggested that the warm sheets may improve the peripheral hemodynamics and autonomic regulation. CRF is a multidimensional syndrome, frequently occurring with other neuropsychological symptoms including depression and cognitive impairments, driven by inflammatory mechanisms, and also acting at sleep/wake centers in the brain. 49 Likewise, subjective mood is influenced by complex interactions between circadian and sleep-wake cycles. 50 As reviewed in detail recently, CRF frequently occurs alongside and is interrelated with sleep disturbances and further neurophysiological symptoms, homeostatic processes under control of the master circadian clock in the subcortical brain. 51 Interventions that affect the biorhythm may therefore also influence mood and well-being, as well as might modulate fatigue symptoms.
The patients of the study cohort examined here are comparable to patients of other German breast cancer centers, 17 suggesting these results as possibly transferable to other cancer centers. Integrative concepts are currently also evaluated under criteria of costs and effectiveness52,53 and this should also be considered for the future.
Conclusion
This longitudinal real-world study of breast cancer patients shows significant associations between improvements of self-reported fatigue and nursing compresses or elaborate consultations and life review. Synergistic effects of multimodal applied NPIs on the improvement of fatigue symptoms need further evaluation. Thus, to improve quality of care for cancer patients, an expansion of this integrative treatment concept and continuation of such a program for supportive follow-up care should be developed in the future.
Footnotes
Acknowledgements
We would like to thank all medical documentation officers at the GKH and the FIH involved in the present work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FS reports grants from Helixor Heilmittel GmbH (travel costs and honoraria for speaking), grants from AstraZeneca (travel costs and honoraria for speaking), grants from Abnoba GmbH, and grants from Iscador AG, outside the submitted work. The other authors have declared that no competing interests exist. No payment was received for any other aspects of the submitted work. There are no patents, products in development or marketed products to declare. There are no other relationships/conditions/circumstances that present a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Network Oncology was partially funded by Iscador AG Arlesheim, Switzerland, Abnoba GmbH Pforzheim, Germany, and Helixor Heilmittel GmbH Rosenfels, Germany. By contract, researchers were independent from the funder.
