Abstract
This is a preliminary study to investigate the feasibility of electronic moxibustion in breast cancer patients with upper limb lymphedema. As current treatment options for lymphedema are unsatisfactory and time consuming, there have been attempts to manage symptoms using integrative treatments. Electronic moxibustion was developed to compensate for the shortcomings of conventional moxibustion and is widely used in clinical practice. However, there have been no studies on using electronic moxibustion in breast cancer-related lymphedema. To investigate the feasibility of electronic moxibustion in treating breast cancer-related lymphedema, this study included subjects who completed primary cancer treatment at least 6 months ago and had more than 10 mm difference in arm circumference of upper limbs. All subjects were assigned to the treatment group. Subjects were treated with 16 sessions (30 minutes/session) of electronic moxibustion for 8 weeks followed by 4 weeks of follow-up. For outcome measures, upper limb circumferences, shoulder range of motion, bioimpedance analysis, and quality of life questionnaire were assessed. All 10 subjects completed the study. The effective index showed 38.21% reduction after treatment (
Introduction
Lymphedema is defined as regional accumulation of protein-rich interstitial fluid due to impaired lymphatic circulation. Breast cancer-related lymphedema (BCRL) is one of the most common causes of secondary lymphedema, and it often occurs after surgical removal of lymph nodes and adjuvant radiation therapy.1,2 Swelling in the upper limb and hand leads to heaviness, pain, decreased range of motion (ROM), and skin thickening. These symptoms can cause deterioration of the quality of life and seriously impact careers.3-5 Overall incidence of BCRL varies widely from 0 to 63.4%, and it mostly occurs during the first 6 to 12 months after surgery.6,7 Although conservative approaches to surgery and radiotherapy have lowered the incidence, a meta-analysis of 30 prospective cohort studies estimated BCRL incidence of 21.4%. 8 The risk of lymphedema is known to peak between 6 and 12 months for patients who underwent axillary lymph node dissection and between 18 and 24 months for patients who underwent both axillary lymph node dissection and regional lymph node radiation. The overall estimated 5-year incidence is 13.7%. 9
Currently, there is no medication to alleviate lymphedema and no gold standard for treatment of BCRL.1,10 The current standard treatment is complete decongestive therapy (CDT), which involves various physiotherapeutic interventions, such as manual lymphatic drainage, multilayer bandaging, skin care, and gentle exercise. Although the treatment effect of CDT varies, studies have shown that a combination of various physical therapies may help to reduce lymphedema volume and improve quality of life for patients with lymphedema.11-13 Since it is difficult to receive regular and frequent treatment from physical therapists, it is important for patients to maintain their symptoms after achieving initial volume reduction. Adherence to long-term use of compression garments is crucial in the maintenance phase but is highly dependent on patient compliance. Surgical treatments such as excisional procedures, liposuction, and microsurgical reconstruction may be effective in some patients who are refractory to CDT. However, due to lack of evidence to suggest these treatments, they are only used as alternatives to the standard treatment. 13
As current treatment options are not satisfactory and are often time consuming, there have been attempts to treat BCRL using integrative treatments, such as acupuncture, moxibustion, cupping, and kinesiology taping.14-18 Based on traditional East Asian medicine, treatment methods to stimulate the meridians have been used to treat edematous conditions by promoting blood circulation and enhancing energy. Acupuncture has been suggested as an effective and safe treatment for BCRL in many studies.19-22 Although a series of studies has shown that acupuncture may be an effective treatment, invasive stimulations are generally avoided in patients with lymphedema. While a recent randomized controlled study showed that acupuncture is safe, it did not result in meaningful reduction of lymphedema compared to usual care. 23 Therefore, other possible treatments for BCRL are needed.
Moxibustion, a non-invasive traditional treatment method widely used in East Asia, involves burning herbal materials on the body surface to stimulate acupoints. Although it shares some common therapeutic mechanisms with acupuncture, possible mechanisms of moxibustion also involve thermal stimulation and biophysical effects.24,25 In South Korea, moxibustion is the third most used treatment method among cancer patients after herbal medicine and acupuncture. 26 However, to date, only a preliminary study has explored the effects of moxibustion for treating BCRL. 15
Despite the possible benefits of moxibustion, there have been barriers for clinical utilization. It can be difficult to control the intensity of heat stimulation, and burns are possible. 27 Smoke produced from moxibustion may cause discomfort to patients and practitioners, such as coughing, headache, and dry throat, and there have been concerns about harmful substances in moxa smoke.27,28 Electronic moxibustion (EM) was developed to reduce the discomfort caused by smoke and to reduce risk of burns while stimulating the acupoint with a constant temperature.29,30 Due to convenience, EM has been widely used in clinics in South Korea, but there are not yet many clinical trials using EM. Thus, we designed a preliminary study to investigate the feasibility of a clinical study of EM for treatment of BCRL.
Materials and Methods
Study Design
This was a single-arm pilot clinical trial to assess the feasibility of the study design and determine adequate sample size for further research. Study subjects who had voluntarily signed informed consent visited the hospital to review study eligibility and blood analysis. Subjects who met the eligibility criteria visited for baseline evaluation within 2 weeks after the screening visit and were started on intervention for 8 consecutive weeks followed by 4 weeks of follow-up. The outcome measures were performed at the 5th, 9th, and 13th week, and blood tests were performed twice, once at the screening visit and once at the 9th week. Detailed schedule of enrolment, interventions and assessments is shown in Supplementary Table 1. The study was conducted in Dunsan Korean Medicine Hospital of Daejeon University (Daejeon, Republic of Korea) from 8th June 2018 to 8th October 2019. The institutional review board at Dunsan Korean Medicine Hospital of Daejeon University approved the study protocol (approval no. DJDSKH-17-BM-30), and the protocol was registered at the Clinical Research Information Service (CRIS registration number: KCT0003423, https://cris.nih.go.kr/cris/search/search_result_st01_en.jsp?seq=14035<ype=&rtype=).
Participants
As this is a preliminary pilot study, there was no prior research to determine required sample size. Ten study participants were recruited through advertisements in local newspapers, subway billboard ads, and posters in a hospital bulletin board. Those who voluntarily signed informed consent underwent eligibility screening. Inclusion criteria were as follows: (1) woman over 19-years-old diagnosed with breast cancer, (2) subjects who completed primary cancer treatment at least 6 months ago (hormone therapy in progress was allowed), (3) subjects with lymphedema in the upper limb of the treated area after breast cancer surgery or radiotherapy, (4) subjects with lymphedema for at least 6 months and who still have lymphedema, (5) subjects with more than 10 mm difference in arm circumferences between upper limbs, (6) subjects with Eastern Cooperative Oncology Group (ECOG) performance status below 2 points, and (7) subjects who signed a written agreement. Only those who met all of the inclusion criteria above were included in the study.
Subjects were excluded if any of the exclusion criteria below were applicable: (1) subjects already had lymphedema before breast cancer treatment, (2) Breast cancer recurrence or metastasis, (3) Subjects with other diseases that affect edema, such as severe heart failure, kidney failure, or cirrhosis, (4) Abnormal hepatic, or renal function tests (AST, ALT more than twice the upper limit of normal range, or creatinine 2.0 mg/dL or more), (5) Lymphedema accompanied by deformed bones or vascular malformations, (6) HbA1c more than 10%, (7) Platelet count below 60 000/μL, hemoglobin below 8 g/dL, or absolute neutrophil count (ANC) below 1000, (8) White blood cell (WBC) above 10 000/μL, (9) Subjects with life expectancy less than 6 months, (10) Subjects planning to undergo surgery, chemotherapy, and radiation therapy during the trial, (11) Subjects who have been actively treated to improve lymphatic edema within the last 4 weeks (compression stockings, compression bandages etc. were allowed), (12) Subjects who participated in other clinical trials within the last 4 weeks, (13) Subjects who experienced hypersensitivity reactions or serious adverse reactions after moxibustion treatment.
Intervention
All participants were treated with EM therapy for 8 consecutive weeks and received 30-minute treatments twice per week for 8 weeks. The EM device (Cettum, K-medical Co., Republic of Korea) is a cube-shaped apparatus with a base embedded in an electrical board controlled by a heat sensor (Supplementary Figure 1). The EM device can be stably attached on acupoints by double-sided medical tape. When the top button is pushed, the electrical board emits heat and the plateau temperature rises up to 45 ± 1°C, which is similar to the peak temperature of conventional moxibustion. EM treatment was conducted by certified Korean Medicine doctors who completed a 6-year regular medical course. Participants received treatment on both affected and unaffected upper limbs. An expert group selected 6 acupoints, bilateral LI14 (
Follow-up
After an 8-week treatment period, we continued to record changes in outcome measure and adverse events for a 4-week follow-up period. During the follow-up period, participants were prohibited to undergo any treatments to attenuate lymphedema, except for the use of compression garments, compression stockings, and self-massage.
Outcome measures
To assess the degree of lymphedema, both affected and unaffected arm circumferences were measured with a tapeline at 4 points: wrist crease, 10 cm proximal to wrist crease, elbow crease, and 10 cm proximal to elbow crease. 14 To exclude changes in the circumference of the unaffected arm caused by unexpected factors, such as weight change, the difference in circumferences between affected and unaffected arms was obtained. The effective index was calculated using the point with the largest circumference difference. Effective index is the percent change in lymphedema calculated according to the following formula:14,20 Effective Index (%) = (Pre-post difference of affected arm circumference)/(Pre-treatment difference between affected and unaffected arm circumference)*100.
To obtain single frequency bioelectrical impedance analysis (SFBIA) values, body composition analysis was performed using an Inbody 720 (Bispace Co., Republic of Korea). Bioelectrical impedance analysis can be used to measure the ratio of extracellular fluid volume.34-36 SFBIA values were noted for both affected and unaffected arms at 1 kHz and 5 kHz.
Based on the physical condition of the upper extremities, staging of lymphedema was performed with a 0-3 staging system established by The International Society of Lymphology (ISL). 37 Stage 0 refers to no edema but presence of lymphatic impairment. Stage I represents mild edema that is reversible with appropriate limb position. Stage II refers to moderate edema that is not reversible with limb elevation. Stage III represents lymphostatic elephantiasis with alterations in skin characteristics and thickness.
The ROM of both affected and unaffected shoulder joints was evaluated using an electrogoniometer to examine mobility limitations due to lymphedema. The maximum angles of flexion, extension, abduction, adduction, external rotation, and internal rotation that subjects could reach without pain or discomfort were measured to evaluate the degree of altered arm use due to lymphedema.
A self-reported questionnaire, the European Organization for Research and Treatment of Cancer QLQ-BR23 questionnaire (EORTC QLQ-BR23), was used to assess quality of life related to breast cancer.38,39 The QLQ-BR23 questionnaire consists of 23 questions recorded on a 1-4 Likert scale. Lower scores indicate lighter symptoms and higher quality of life.
Measurement of arm circumferences, ISL stage determination, ROM measurements, and questionnaire results were evaluated at baseline and at the 5th, 9th and 13th weeks. Body composition analysis was performed at the 1st, 9th, and 13th weeks.
To assess safety, blood pressure and body temperature were evaluated at every visit, and adverse events were continuously monitored throughout the study. Laboratory tests were performed at the screening visit and at week 9. Laboratory tests included complete blood count with differential, absolute neutrophil count (ANC), aspartate transaminase, alanine transaminase, blood urea nitrogen, creatinine, and hemoglobin A1c. To investigate the reliability of and satisfaction with EM treatment, a credibility/expectancy questionnaire was administered before and after the intervention (weeks 1 and 9). 40 Since there was no validated Korean version of credibility/expectancy questionnaire, we used the translated questionnaire from the previous study. 41
Statistical Analyses
SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA) was used for all statistical analyses. Due to the small sample size, nonparametric analysis, such as Wilcoxon signed rank test was performed for the pre-post comparison. All values are presented as mean ± standard error (SE) or median [interquartile range]. For categorical data, data are shown as mean and percentage, and Fisher’s exact test was performed for the analysis.
Results
Baseline Characteristics
Eleven volunteers were screened for eligibility criteria, and 10 subjects were enrolled in this study (Figure 1). All 10 subjects were allocated to the treatment group, and all subjects completed the study without drop-out. Baseline characteristics of study participants are shown in Table 1. Median age at consent was 53.0 years, and all subjects were non-smokers and non-alcoholic drinkers. The majority of subjects (80%) exercised regularly. Median height and weight were 154.40 cm and 56.95 kg, respectively. The median duration of BCRL symptoms was 82.5 months. All 10 subjects underwent surgery and radiation therapy for cancer treatment.

Study flow diagram.
Baseline Characteristics.
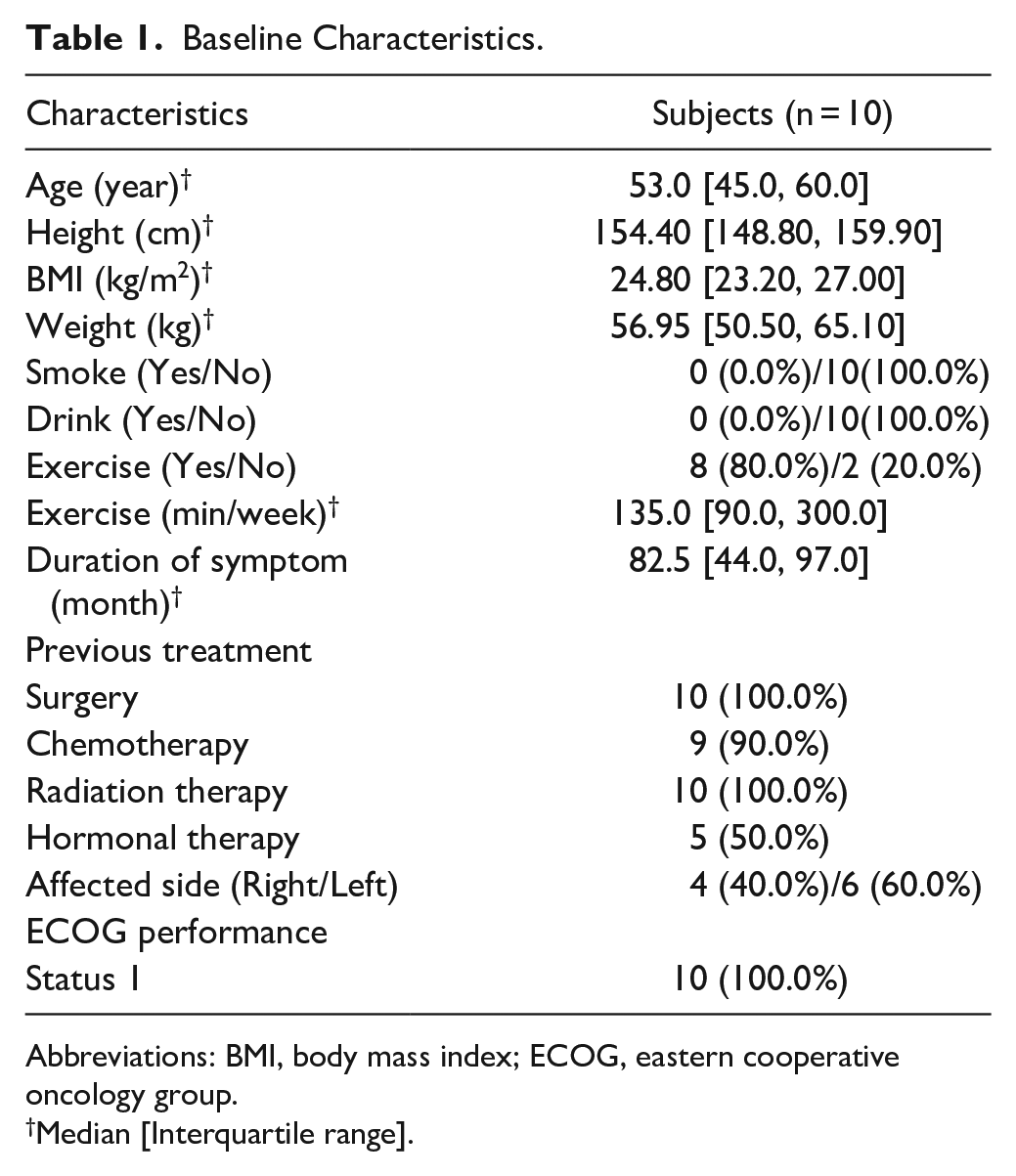
Abbreviations: BMI, body mass index; ECOG, eastern cooperative oncology group.
Median [Interquartile range].
Among the permitted concurrent treatments, all 10 subjects reported using the compression garments only on days when the affected side of upper limb was heavily used.
Lymphedema Symptoms
Differences in arm circumferences between affected and unaffected upper limbs are shown in Table 2. The mean difference in circumferences between affected and unaffected upper limbs at 10 cm above the elbow crease decreased significantly at week 5 (mean difference of −4.6 mm,
Differences in Circumferences between Affected Arm and Unaffected Arm.

Abbreviations: SE, standard error; IQR, interquartile range.
During 8 weeks of EM treatment, the physical condition of the upper extremities assessed by ISL stage classification improved, although the changes were not statistically significant (Supplementary Figure 2). Some participants with stage II lymphedema became stage I, while some participants with stage I lymphedema became stage 0. Although physical changes were observed during the treatment period, the changes did not last during the 4-week follow-up period.
Differences in ROM between Affected and Unaffected Limbs
After the 8-week intervention, only flexion and internal rotation showed significant improvements (Table 3). The differences in ROM between affected and unaffected upper extremities improved an average of 8.3° for flexion (
Differences in Range of Motion between Affected Arm and Unaffected Arm.
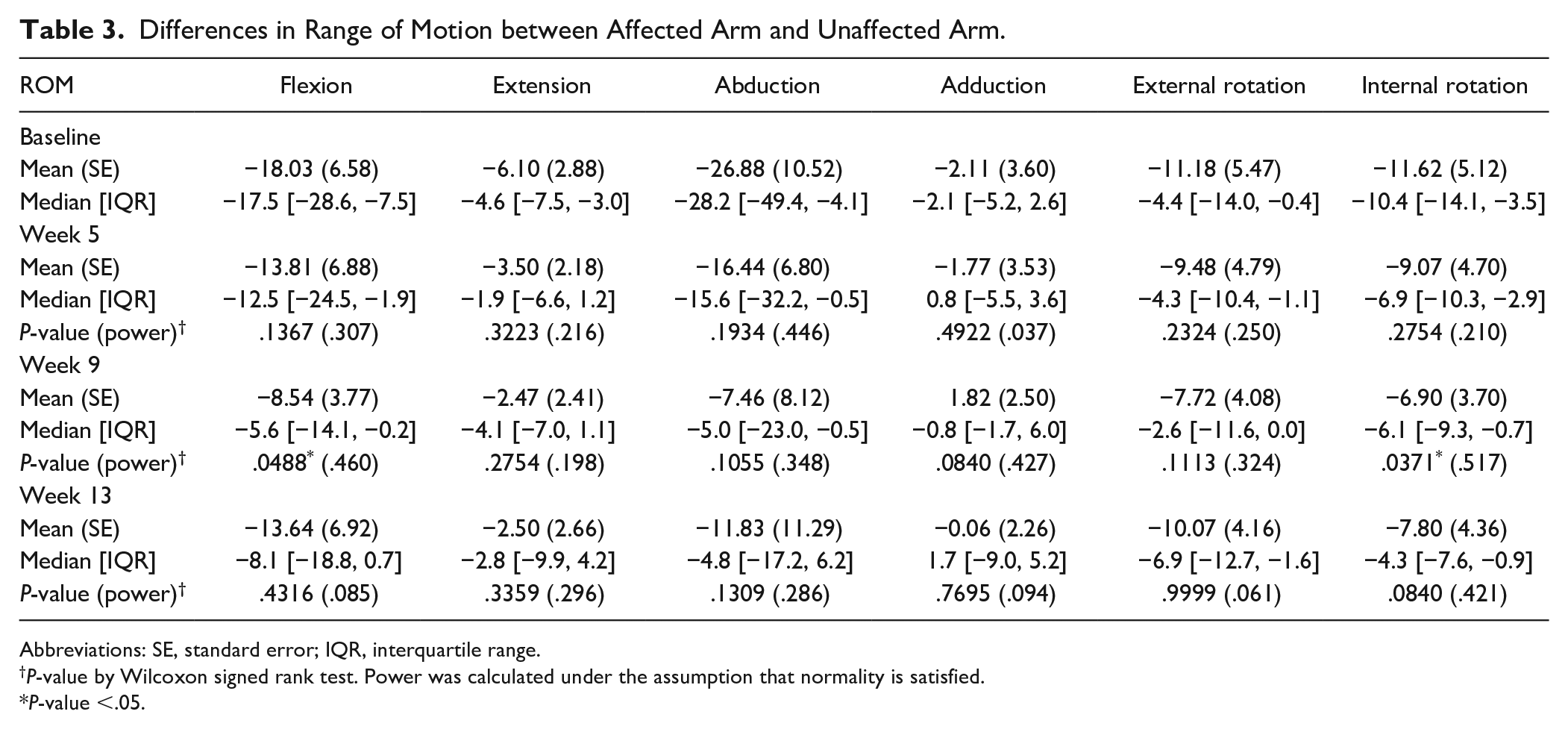
Abbreviations: SE, standard error; IQR, interquartile range.
Credibility/Expectancy Questionnaire
Overall, patient credibility and expectancy increased after EM treatment compared to the baseline. Credibility/expectancy scores significantly increased, except for the effectiveness score (Figure 2 and Supplementary Table 2). Subjects with moderate credibility at the start about EM treatment rated the effect more positively after treatment (
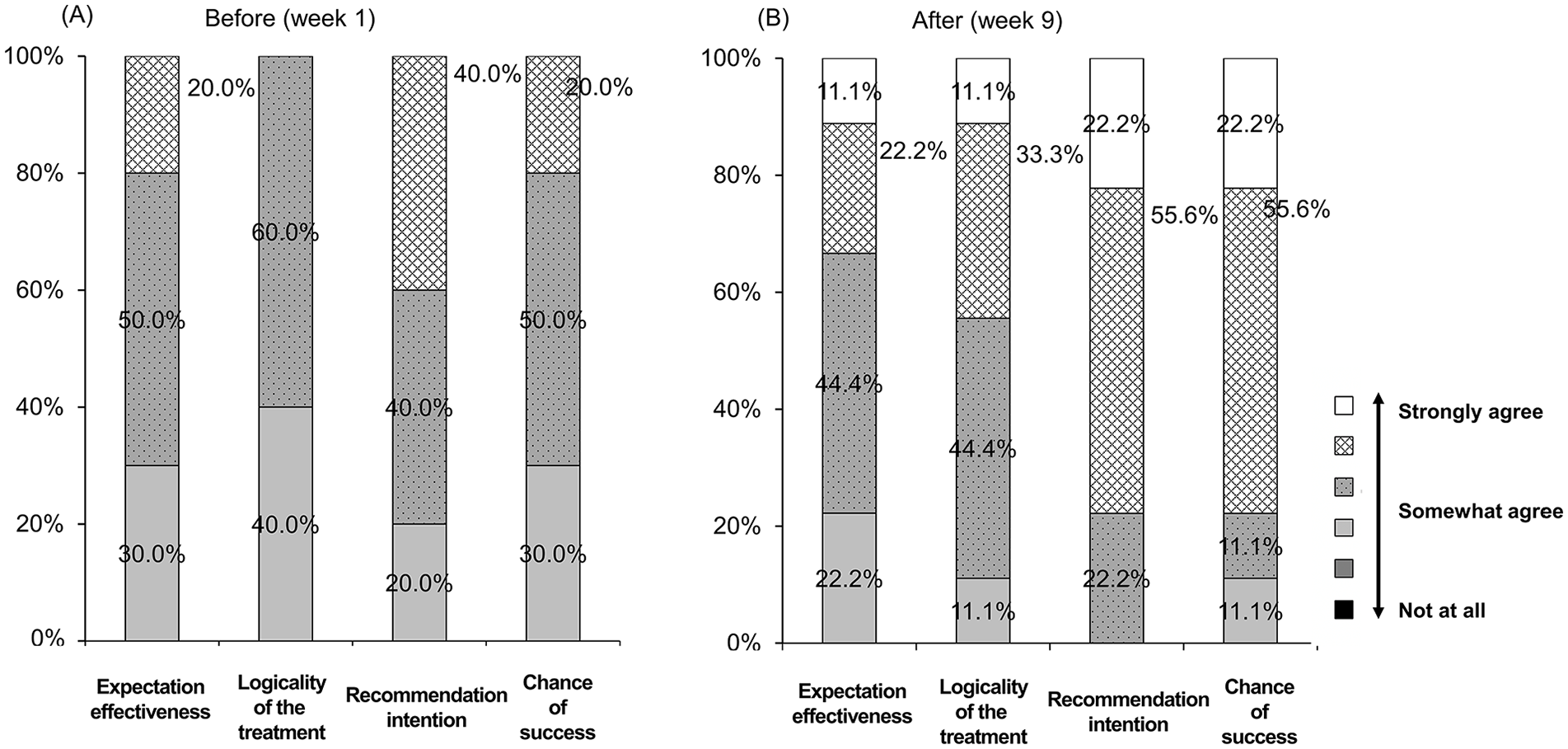
Changes in perception of the reliability of electronic moxibustion treatment using Credibility/Expectancy questionnaire. (A) Before. (B) After the study.
Quality of Life
Overall quality of life of participants was not significantly different throughout the study (Table 4). Among categories of the EORTC QLQ-BR23 questionnaire, improvement of arm symptoms was significant only at week 5 (
Changes in Quality of Life Scores Using EORTC QLQ-BR23 Questionnaire.
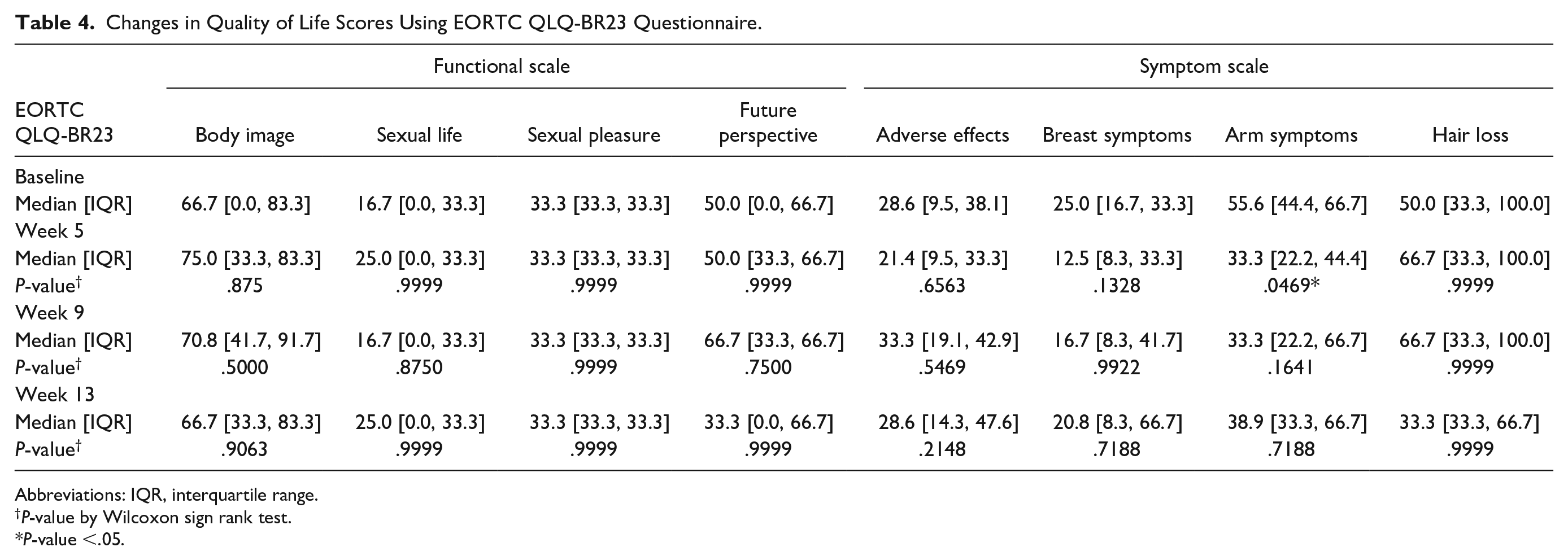
Abbreviations: IQR, interquartile range.
Safety Assessments
After 8 weeks of intervention, there were no significant changes in blood analysis and vital signs (Table 5). One serious adverse event (SAE) and 6 adverse events were reported during the trial, but most were irrelevant to the intervention. The participant with a SAE was hospitalized to treat back pain due to a traffic accident but completely recovered within the follow-up period. There was a case of mild local burn on an acupoint area that recovered completely. One patient experienced an inflammatory response of redness and swelling of the affected axillary area. A specialist was consulted and was uncertain about the relationship with moxibustion. The patient recovered after antibiotics and anti-inflammatory drugs. Other adverse events included finger abrasion, sinusitis, and 2 cases of upper respiratory infection.
Comparison of Safety Assessments Before and After 8-week Electronic Moxibustion Treatment.
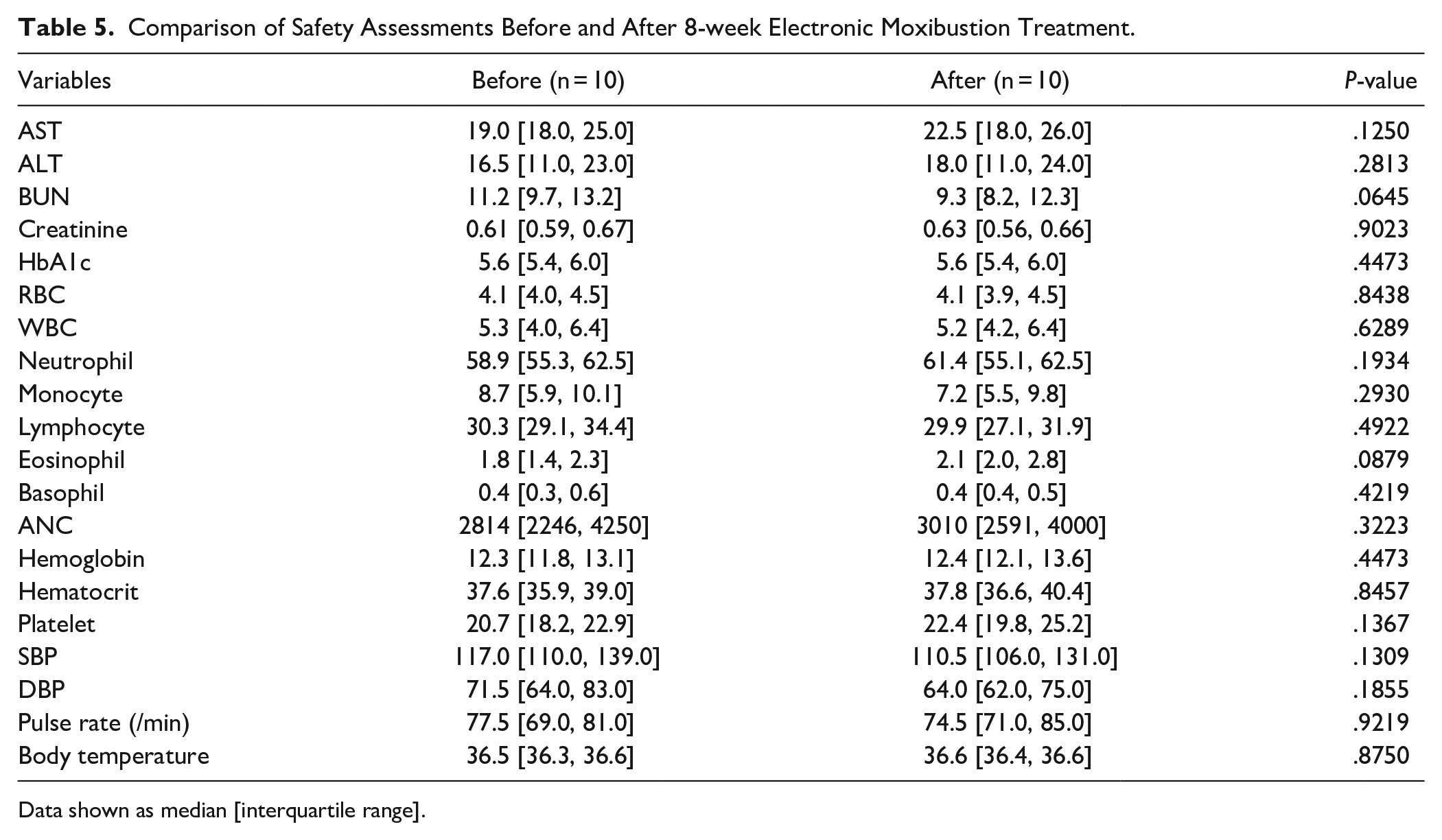
Data shown as median [interquartile range].
Discussion
The results of this pilot study showed that EM treatment reduced the difference in arm circumference, confirming the feasibility of this study. The study had successful 90.91% recruitment rate and 100% adherence and completion rates. However, patient recruitment was not successful with our previous protocol, which had more rigorous inclusion criteria. Our previous inclusion criteria were subjects with more than 20 mm difference in arm circumferences who had BCRL between 6 months and 5 years. Because BCRL is a long-term complication that can cause serious discomfort in daily life even with mild symptoms, we designed a pilot study including patients with 10 mm difference in arm circumferences and included patients who had been diagnosed with lymphedema more than 5 years ago.
In the present study, we confirmed that EM can significantly reduce the difference in circumferences between affected and unaffected arms. The reduction was significant after 8 weeks of intervention and maintained 4 weeks after treatment. A 4-week pilot study using conventional moxibustion showed more improvement than daily compression garment use. 15 Our results suggest that EM treatment can also be used as a heat stimulation strategy on acupoints for BCRL treatment. Another study showed that moxibustion combined with acupuncture treatment has a better effect on relieving edematous conditions than acupuncture alone, and this therapeutic effect was related to increased skin temperature and blood flow measured by laser Doppler analysis. 42 A similar result was shown in a warm acupuncture study, a combined method of acupuncture and moxibustion, in which circumferential reduction was also prominent in the upper arm with an effective index of 50%. 14 The authors inferred that the treatment effect was more pronounced on the upper arm where heat stimulation was provided. 14 Like the previous study, improvement was more pronounced in the upper arm than in the lower arm in our study. However, there is still a lack of evidence to explain that the decrease in arm circumference is more significant near the acupoints. Since each person has different locations of swelling, further research is needed on selection of acupoints for treatment of BCRL. The acupoints in our study were selected within local points around the upper limb based on references and consensus between experienced Korean medicine doctors. According to the ancient theory that disease on one side can be treated through the acupuncture points on the opposite side, bilateral acupuncture points are commonly used in clinics to increase the therapeutic effect. Several studies have shown that the contralateral treatment is as effective as the ipsilateral treatment, but the mechanism has not been fully elucidated and there have been no studies on lymphedema.43-45 Although bilateral acupoints were selected in this study, future studies to clarify which treatment is most effective in BCRL patients among contralateral, ipsilateral and bilateral acupoints are needed.
EM is a type of modern moxibustion that provides a constant amount of thermal stimulation during treatment. The temperature of EM is set between 40 and 45ºC for therapeutic effect, and the amount of heat transferred to the body surface is the same as in conventional moxibustion.29,30 Studies have demonstrated that burns could occur at 47°C but prolonged exposure at 43°C can be safe up to 8 hours. 46 Therefore, EM can be considered safe from burns and it is more efficient because the temperature rises rapidly and has a long-lasting effect compared to conventional moxibustion, which cools down quickly after combustion. 29 In a study comparing conventional moxibustion and EM in knee osteoarthritis patients, both conventional moxibustion and EM showed significant improvement in symptoms compared to the usual care group, but the adverse events related to the moxibustion occurred less in the EM group. 47 It is noteworthy that the conventional moxibustion group reported adverse event such as blister, tingling, redness and crust, whereas the EM group had only milder symptoms such as redness and crust. 47 Although EM maintains a safe temperature, one patient in our study experienced a mild burn. Cautious observation is required during treatment because people have different sensitivity to temperature.
Although there was little difference in patient quality of life, patients were somewhat satisfied, as demonstrated by the high adherence rate and credibility/expectancy results.
Both objective arm volume and perception of arm volume are important in patients with BCRL and may contribute to decreased quality of life. 4 Therefore, symptom distress and arm circumference or volume should be considered important indicators in future trials. Because many results are collected through self-reported outcomes, it is important to select appropriate outcome measures for target symptoms. In our study, subjects commonly mentioned that symptoms of stiffness in the affected arm improved. Because subjects in this study were mainly people with mild symptoms, they reported more symptoms of stiffness and discomfort than pain or limited shoulder ROM. The EORTC QLQ-BR23 questionnaire includes questions regarding pain, swelling, and ROM for arm symptoms, but does not include questions on symptoms such as stiffness and discomfort, so the results did not fully reflect the improvement. The authors of a conventional moxibustion study also mentioned that subjective symptoms improved although there was no measurable reduction in circumference. 15 As EORTC QLQ-BR23 is the first disease-specific questionnaire and it is the most widely used questionnaire, it may not be appropriate for patients with mild symptoms like our study. In future study, inclusion of additional outcome measures that may effectively reflect such symptoms should be considered. For example, a quality of life scale in upper limb lymphoedema questionnaire developed by the French Society of Lymphology can be considered because it consists of detailed questions regarding physical symptoms which can be better indicators of quality of life in BCRL patients. 48 A recently updated version of EORTC QLQ-BR23 supplemented with a target symptom scale or Lymphedema Symptom and Intensity Survey-Arm (LSIDS-A) can also be considered in future studies.49,50
According to the theory of traditional Chinese or Korean medicine, moxibustion helps circulation of meridians, detoxification, and promotes circulation of
Our study is limited by lack of a comparison group, such as a usual care group or sham group, and inclusion of a specific group of subjects among patients with BCRL. Recruitment focused on chronic BCRL and excluded patients with recurrent breast cancer and metastatic cancer or patients currently undergoing cancer treatments. Although we attempted to evaluate objective outcome measures other than arm circumference, it was difficult to observe changes using the SFBIA. In addition, our study has limitations in that use of compression garments or self-massage were allowed. In order to evaluate the effect of EM treatment alone, studies that allows only EM treatment or that compares EM treatment with usual care are needed. Nonetheless, this is a novel study using EM for patients with BCRL that included patients with mild BCRL symptoms. Ongoing studies have highlighted the importance of subclinical lymphedema, but there are not yet ideal detection tools for subclinical BCRL.57,58 One way to detect subclinical BCRL is through self-reported outcomes reflecting various arm symptoms. Further studies to optimize study protocol are needed.
Conclusion
This pilot study suggested that EM treatment is feasible for patients with BCRL. After 8 weeks of EM treatment, patients showed reduced difference in circumferences of both arms and increased ROM in flexion and internal rotation. Our results provide preliminary data for a future randomized controlled trial to investigate the efficacy and safety of EM for patients with BCRL.
Supplemental Material
Supplementary_figure_1 – Supplemental material for Electronic Moxibustion for Breast Cancer-Related Lymphedema: A Pilot Clinical Trial
Supplemental material, Supplementary_figure_1 for Electronic Moxibustion for Breast Cancer-Related Lymphedema: A Pilot Clinical Trial by Kyungsun Han, Ojin Kwon, Hyo-Ju Park, Ae-Ran Kim, Boram Lee, Mikyung Kim, Joo-Hee Kim, Chang-sup Yang and Hwa-Seung Yoo in Integrative Cancer Therapies
Supplemental Material
Supplementary_tables – Supplemental material for Electronic Moxibustion for Breast Cancer-Related Lymphedema: A Pilot Clinical Trial
Supplemental material, Supplementary_tables for Electronic Moxibustion for Breast Cancer-Related Lymphedema: A Pilot Clinical Trial by Kyungsun Han, Ojin Kwon, Hyo-Ju Park, Ae-Ran Kim, Boram Lee, Mikyung Kim, Joo-Hee Kim, Chang-sup Yang and Hwa-Seung Yoo in Integrative Cancer Therapies
Supplemental Material
Supplmentary_figure_2 – Supplemental material for Electronic Moxibustion for Breast Cancer-Related Lymphedema: A Pilot Clinical Trial
Supplemental material, Supplmentary_figure_2 for Electronic Moxibustion for Breast Cancer-Related Lymphedema: A Pilot Clinical Trial by Kyungsun Han, Ojin Kwon, Hyo-Ju Park, Ae-Ran Kim, Boram Lee, Mikyung Kim, Joo-Hee Kim, Chang-sup Yang and Hwa-Seung Yoo in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Korea Institute of Oriental Medicine (grant number: K18121 and KSN1522120).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
