Abstract
Patients with precancerous gastric conditions are at a high risk for gastric carcinoma. The Chinese medicine Weifuchun (WFC) is used in treating chronic superficial gastritis and in postoperative adjuvant treatment of gastric cancer. Both monotherapy and combination therapy of WFC with other drugs can result in a favorable therapeutic outcome. WFC can dramatically improve clinical outcomes in patients with gastric precancerous lesions by targeting multiple pathways including pathways involved in the pharmacological action of Radix Ginseng Rubra (red ginseng), Rabdosia amethystoides, and fried Fructus Aurantii, including regulation of NF-κB, RUNX3/TGF-beta/Smad, Hedgehog (Hh) and Wnt signaling pathways, modulation of the expression of oncogenes and tumor suppressor genes, and indirect inhibition of Helicobacter pylori (Hp) by maintaining gastric microbial ecosystem. In this review, we will discuss the clinical efficacy and therapeutic regimen of WFC for gastric precancerous lesions and the molecular mechanisms involved. This review will highlight WFC-based therapeutic strategies in disrupting progress to gastric cancer and provide more information on the pharmacological mechanisms of WFC and its clinical application for the treatment of precancerous gastric lesions.
Introduction
Gastric cancer is currently one of the most common cancers and has a high mortality rate worldwide. 1 Development of gastric cancer requires a cascade of multistep processes to lead to carcinogenesis, including multifocal atrophic gastritis; intestinal metaplasia; dysplasia and intestinal-type carcinoma (carcinoma in situ); and invasive carcinoma.2,3 Atrophic gastritis and intestinal metaplasia are precancerous lesions that supply the proper conditions for the progress of gastric carcinoma. 3 Thus, early intervention of precancerous conditions can play a critical role in disrupting progression to gastric carcinoma. Atrophic gastritis is a chronic gastric disease characterized by immune damage to the structures of the gastric gland that leads to a lack of production of gastric acid and intrinsic factor.4-6 Hp-induced immunosuppression is a major cause of atrophic gastritis. 4 During Hp infection, the bacteria stimulate IL-1b expression, which inhibits gastric acid secretion and leads to chronic and progressive gastric mucosal damage.7,8 To date, the ideal treatment for Hp infection is still lacking. 8 Intestinal metaplasia is classified as complete or incomplete conditions. Complete intestinal metaplasia exhibits reduced expression levels of gastric mucins such as MUC1, MUC5AC, and MUC6 in absorptive and goblet cells. In incomplete metaplasia, gastric and intestinal mucins are co-expressed in the goblet and non-absorptive cells.9–11 Gutiérrez-González et al. revealed the existence of spasmolytic polypeptide-expressing metaplasia with expression of trefoil factor 2 (TFF2) polypeptide associated with oxyntic atrophy. 12 Anti-Hp treatment and endoscopic monitoring and management are currently the main strategies for gastric precancerous lesions.13,14 In the early stages, anti-Hp treatment can effectively improve gastric mucosal inflammation and reverse gastric mucosal atrophy, but whether it can reverse intestinal metaplasia and progression to cancer is still controversial. 14 Although studies have shown that cyclooxygenase-2 (COX-2) inhibitors,15–18 the antioxidant vitamins A, C, E, and β-carotene,19–21 and retinoic acid 22 might reverse intestinal metaplasia, the current evidence is still insufficient to support intervention using these agents as approaches to reduce the risk of gastric precancerous lesions. 13
Weifuchun (WFC) was developed for the treatment of gastric diseases and postoperative adjuvant treatment for gastric cancer in China. There have been multiple clinical studies on the effect of WFC on gastric precancerous conditions and lesions.23–30 The results of those studies showed that WFC is effective for improving atrophic gastritis and intestinal metaplasia. Therefore, an updated review of the molecular mechanisms and efficacy of WFC is urgently required for usage of WFC in treating gastric precancerous lesions.
Ingredients of WFC
WFC mainly contains Radix Ginseng Rubra (red ginseng), Rabdosia amethystoides H. Hara, and fried Fructus Aurantii.31,32
Red Ginseng
Red ginseng contains ginsenosides that have a wide range of pharmacological activities such as anti-tumor, neuroprotective, and antioxidant effects, as well as promotion of gastrointestinal motility and immune regulation. 33 Studies have shown that the intake of red ginseng was closely correlated with a reduced risk of colorectal, lung, stomach, and liver cancers, and that this effect was not organ-specific. 34 The anti-cancer mechanisms of red ginseng are believed to act through cell cycle arrest, induction of apoptosis, and inhibition of angiogenesis. 35 As reported by Oyagi A et al. red ginseng dramatically alleviated gastric ulcers induced by exposure to ethanol or indomethacin in mouse models by improving local blood supply to the gastric mucosa and suppressing cellular damage caused by oxidative stress. 36 Jim et al. and Bae et al. showed that red ginseng effectively prevented inflammation induced by Hp infection in gastric mucosa.37,38 Red ginseng inhibited the upregulation of inflammatory cytokines, lactoperoxidase (LPO) activity and myeloperoxidase (MPO) activity in gastric mucosa. Although red ginseng did not affect the colonization of Hp, it reduced neutrophil infiltration and intestinal progression to intestinal metaplasia. 38
Rabdosia Amethystoides
R. amethystoides has been found to possess antitumor, antioxidant, anticoagulant, antibacterial, anticomplement, and antipyretic activities, and its main components, glaucocalyxin A, glaucocalyxin B, and glaucocalyxin D, are diterpenoids similar to the antitumor drug paclitaxel.39,40 R. amethystoides can effectively improve the microcirculation of the gastric mucosa, eliminate inflammation, repair the damaged gastric mucosa, and promote mucosal regeneration in gastric precancerous lesions. 41
Fructus Aurantii
Fructus Aurantii is a dried, unripe citrus fruit that contains a variety of chemical components including volatile oil, coumarin, alkaloids, and flavonoids such as naringin and neohesperidin. Fructus Aurantii flavonoids were proven to effectively enhance gastrointestinal motility by regulating tryptophan metabolism, regenerate the glands of the gastric mucosa, and influence corticosterone and sphingolipid metabolism. 42
WFC in Treating Gastric Precancerous Lesions and Its Clinical Efficacy
WFC Single Therapy
Accumulated clinical evidence has proven that WFC is effective in improving gastric premalignant conditions. A clinical trial in 76 patients with gastric precancerous lesions showed that administration of WFC dramatically decreased the degree of glandular atrophy, intestinal metaplasia, and Hp infection in these patients. 23 In this study, WFC was taken by patients 3 times per day for 24 weeks, with gastroscopy and pathological examination performed before and after WFC treatment for evidence of Hp infection, inflammation in the gastric mucosa, and degree of precancerous conditions. The results indicated that the effective rates of WFC for attenuating glandular atrophy and intestinal metaplasia were 76.47% and 62.5%, respectively. Furthermore, the elimination rate of Hp in the WFC patients was 61.71%. Thus, WFC can effectively alleviate the clinical symptoms of gastric precancerous lesions. 23
In a clinical study conducted by Lu L et al., the efficacy of WFC in 96 patients diagnosed with dysplasia and intestinal metaplasia was evaluated. The results showed that the effectiveness of WFC on dysplasia and intestinal metaplasia reached 66.0%, which was significantly more effective than that in a control group receiving basic treatment with omeprazole and sucralfate (40.8%), indicating that WFC exerts obvious therapeutic effects on gastric precancerous lesions. 24 The indicators of blood rheology such as whole blood viscosity, plasma viscosity, and hematocrit were reduced in patients who received WFC treatment, suggesting that the hypercoagulability was improved by WFC treatment. Moreover, plasma endothelin-1 was decreased and the levels of calcitonin gene-related peptide (CGRP) were increased in response to WFC treatment, suggesting that blood flow in gastric mucosa and circulation disorders in these patients were dramatically improved. 24
WFC-Based Combinatory Therapeutic Strategies
More and more studies have developed therapeutic strategies using WFC in combination with other drugs to halt progression of gastric precancerous conditions. Min S et al. carried out a clinical study in 126 patients with chronic inflammation of the gastric mucosa with or without gastric epithelial metaplasia confirmed by endoscopic biopsy and pathology examination. 25 The results showed the patients benefited more from combined treatment of WFC with retinoic acid compared to retinoic acid or WFC alone and that combination treatment significantly upregulated expression of the tumor suppressor Rb while downregulating the expression of the oncogene C-erbB2. Thus, this study concluded that WFC in combination with retinoic acid was a promising therapeutic strategy for gastric precancerous lesions. 25
In a clinical study of combination therapy of WFC with rebamipide, an amino acid analog used for treating gastroduodenal ulcers, patients diagnosed with chronic atrophic gastritis receiving combination treatment for 12 weeks showed greater improvement compared to either treatment alone. WFC in combination with rebamipide effectively alleviated the carcinogenesis process of gastric mucosa. 26
In a clinical study conducted by Chen WJ et al., the efficacy of 12-week combination therapy of WFC and Lizhu Weisanlian (bismuth potassium citrate/tinidazole/clarithromycin tablets) was evaluated in 82 patients with Hp-positive atrophic gastritis. 27 The total effective rate of single treatment, Hp eradication rate, pathological histological improvement and adverse reaction rates of the control group were 73.17%, 68.29, 41.46%, and 21.96%, respectively, which were significantly inferior to those of combination therapy (97.56%, 95.12%, 53.66%, and 7.32%, respectively). The data showed that the combination treatment for Hp-positive atrophic gastritis enhanced therapeutic responses more impressively than Lizhu Weisanlian alone did. 27
Zhang YQ et al. reported that WFC in combination with folic acid and Hp eradication treatments could dramatically alleviate precancerous lesions of gastric cancer. 28 In this study, a total of 130 patients with precancerous lesions of gastric cancer were enrolled and gastroscopy and pathology examination were undertaken after 6 months of treatment. For those patients who failed in Hp eradication there was no dramatic improvement of intestinal metaplasia, but a combination of WFC and folic acid still significantly improved intestinal metaplasia and low-grade intraepithelial neoplasia. 28
In a clinical study on 126 patients with Hp-negative atrophic gastritis treated with combination of WFC and marzulene-S or single agent for 36 weeks, the effective rate in the combination group was dramatically higher than that of single therapy control groups and the combination of WFC and marzulene-S dramatically inhibited the carcinogenic effect of N-nitroso compounds on gastric mucosa. 29
Teprenone is one of the commonly used protective agents for gastric mucosa. In combination with teprenone, WPC significantly improved the clinical outcomes and pathological grading of patients with chronic atrophic gastritis compared with WFC or teprenone alone. 30 The combination treatment of WFC and teprenone dramatically increased blood flow in gastric mucosa and enhanced the reconstruction of local microcirculation and gastric mucosa repair in comparison to the single treatments. 30
Molecular Mechanisms Underlying the Therapeutic Effects of WFC on Gastric Precancerous Lesions
Molecular Mechanism of Action of Red Ginseng in WFC
Being a major component of WFC, red ginseng has been shown to reduce cytotoxicity caused by Hp infection in the stomach and DNA mutations induced by oxidative stress. 37 The mechanism of the action is mediated through the following pathways: (1) Red ginseng blocks ERK1/2 signal transduction thereby reducing the expression and release of the inflammatory mediator IL-8; (2) Red ginseng suppresses IL-8-induced inflammation by inactivating the c-jun pathway, which leads to decreased 5-LOX activity and 5-LOX expression; (3) Red ginseng improves atrophic gastritis by inhibiting the activation of caspase-3 in the apoptosis pathway. 37
Molecular Mechanism of Action of R. Amethystoides in WFC
As another important ingredient of WFC, R.a amethystoides can inhibit the proliferation of gastric cancer cells by inducing cell cycle arrest in G0/G1, thereby preventing progression through the cell cycle. 43 R. amethystoides can also induce apoptosis by activating Caspase3 and inhibiting PARP. Finally, R. amethystoides plays an inhibitory role in regulating the migration and invasion of gastric carcinoma cells by promoting the expression of miR-212-3p, a micro RNA that targets and inhibits Sox6. 43
Molecular Mechanism of Action of Fructus Aurantii in WFC
Studies have shown that the flavonoid compound neohesperidin in Fructus Aurantii can significantly inhibit the formation of hydrochloric acid/ethanol-induced gastric lesions in rats, increase the amount of mucus in gastric mucosa, and reduce the secretion of gastric acid, resulting in protection of the gastric mucosa. 44 In addition, neohesperidin exhibits antioxidant activity in the 1,1-diphenyl-2-pyridylpyrazolyl (DPPH) free radical scavenging test, indicating that it can block the gastric precancerous process by reducing oxidative stress. 44 Naringin, another critical flavonoid contained in Fructus Aurantii, was proven to induce autophagy by activating the MAPK pathway and inhibiting the PI3K/Akt/mTOR cascade, thereby suppressing proliferation of gastric cancer cells. 45
Deactivation of NF-κB Signaling Pathways by WFC
The transcription factor NF-κB is an important protein in controlling inflammation and immunity. Deregulated activation of NF-κB signaling pathways has been implicated in a wide variety of pathological inflammatory conditions. 31 WFC exerts an immune modulatory effect and alleviates Hp-induced chronic gastritis by deactivating NF-κB pathways, thus attenuating IL-8 and IL-4 production by gastric epithelial cells. In the presence of WFC, the Hp-induced increase in the expression of intranuclear P65 and decrease in IκBα were abrogated. WFC may act by preventing the translocation of P65 into the nucleus and subsequent degradation of IκBα. 31
Restoration of RUNX3/TGF-beta/Smad Signaling Pathway by WFC
In a study conducted by Ma et al., WFC was found to regulate atrophic gastritis through the RUNX3/TGF-beta/Smad signaling pathway. 46 This study demonstrated a drastic increase in methylation of RUN3 and inhibition in RUNX3/TGF-beta/Smad signaling in intestinal atrophic gastritis and dysplasia atrophic gastritis compared to non-atrophic gastritis. Administration of WFC decreased the methylation of RUN3 and enhanced the activation of RUNX3/TGF-beta/Smad signaling, suggesting that this pathway could be targeted by WFC in the treatment of intestinal atrophic gastritis and dysplasia atrophic gastritis. 46
Regulation of Hh and Wnt Signaling Pathways by WFC
Studies have shown that the aberrant expression of Sonic hedgehog (Shh) protein, a ligand of the Hh signaling pathway, and Wnt 3A, a critical factor in the Wnt signaling pathway, are closely associated with the pathogenesis of gastric intestinal metaplasia.47–49 Both Hh and Wnt signaling are hot topics in the research of gastric cancer and precancerous pathogenesis. In the gastrointestinal gland, Hh and Wnt signals interact with each other to form an inhibitory circuit, like the yin and yang opposites of Chinese medicine. Shh is essential for gastric differentiation and Wnt3A plays an important role in intestinal differentiation. In gastric intestinal metaplasia, Shh is aberrantly downregulated and Wnt3A is upregulated, which keeps the Wnt signaling pathway activated.47–49 WFC administration significantly promoted the expression of Shh and decreased the expression of Wnt3A, suggesting that restoration of the balance of Hh and Wnt signaling pathways could contribute to the alleviation of gastric intestinal metaplasia. 48
Regulation of the Expression of Oncogenes and Tumor Suppressor Genes by WFC
WFC exerts regulatory effects on the expression of oncogenes and tumor suppressors. Min S et al. and Cui SJ et al. reported that WFC treatment downregulated the expression of the oncogene C-erBb-2 and upregulated the expression of the tumor suppressor gene Rb in human gastric precancerous lesions including mild, moderate and severe intestinal metaplasia.25,50 Zheng B et al. found that WFC combined with colloidal pectin bismuth capsules reduced the expression of oncogenic Notch-1 and β-catenin in gastric precancerous lesions and enhanced the expression of the tumor suppressor gene PTEN. 51 These results suggest that modulation of the expression of oncogenes and tumor suppressor genes by WFC could be the basis of its therapeutic effects on gastric precancerous lesions and the protective effect on gastric mucosa.
Indirect Inhibition of Hp and Maintenance of the Balance of Gastric Microbial Ecosystem by WFC
The balance of the microbial ecosystem is a key factor to maintain the healthy microenvironment in stomach, characterized by the diversity of bacteria, balanced bacterial density and dominance of probiotics.52,53 Under normal circumstances, the microbial ecosystem is in a dynamic equilibrium state, and disruption of equilibrium determines the occurrence of gastric and intestinal diseases including gastric precancerous conditions.52,53 Thus, for the unbalanced microbial ecosystem caused by Hp infection, the ideal treatment is not direct Hp killing, but the restoration of the balance of microbial ecosystem that inhibits adhesion and colonization of Hp, and promotes the activity and growth of probiotics, which is a new strategy against Hp infection. 53 Studies have proven that the bacteriostatic and bactericidal effect of WFC does not mainly rely on its Hp-killing activity, but its capability to restore balance of gastric microbial ecosystem through repairing damaged gastric mucosa caused by Hp infection, regulating the production of inflammatory factors and improving gastric microenvironment, which leads to the suppression of adhesion and colonization, and thus the inhibition of Hp.53,54
Perspectives and Limitations
As a traditional Chinese medicine, WFC is one of the potent drugs against gastric precancerous lesions through multiple mechanisms (Figure 1). Combination therapies using WFC and other agents have exhibited dramatically improved therapeutic results compared with single agents. The activity of WFC to restore the balance of gastric microbial ecosystem makes it also a strong candidate for treatment of resistance to Hp eradication therapies and this mechanism of WFC lays a foundation for the development of new therapeutic strategies based on maintenance of balance of gastric microbial ecosystem. However, most of the studies focus on the improvement in clinical outcomes in short-term treatment, and data regarding long-term treatment plans and follow-ups, evaluation of the rate of relapse and severity of relapse, and long-term improvement of patient quality of life are still lacking. More studies are needed to explore the full scope of the therapeutic mechanisms of WFC and to develop new strategies in combination of WFC with other targeted therapies, based on the molecular mechanisms.
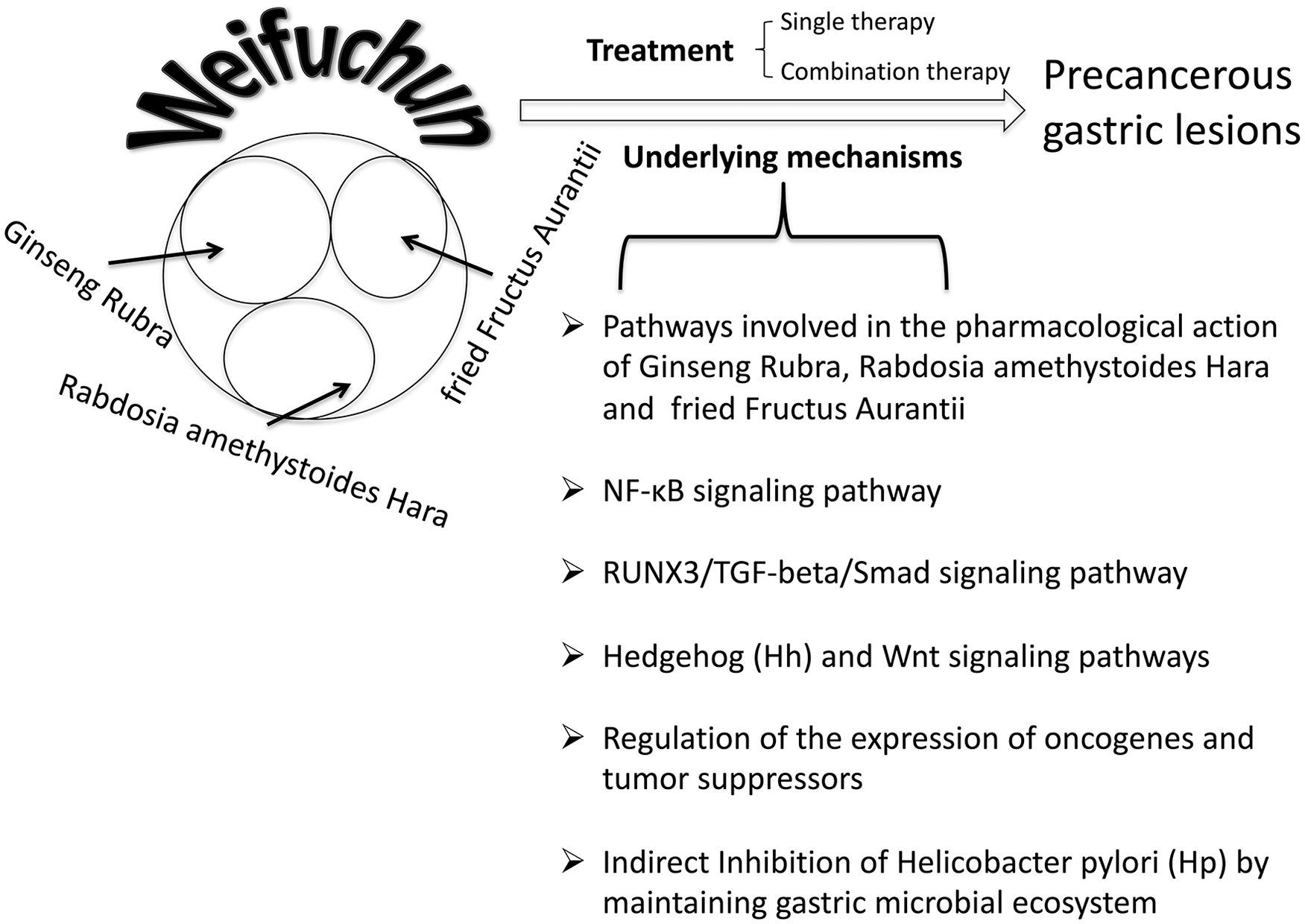
Summary of the therapeutic effects and the pharmacological molecular mechanisms of Chinese medicine Weifuchun in treating precancerous gastric conditions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Evidence-based Traditional Chinese Medicine Foundation of Administration of Traditional Chinese Medicine, China (No. 2019XZZX-XH013), National Natural Science Foundation, China (No. 81774079) and National Foundation for Key Research and Development, China (No. 2018YFC1707300).
