Abstract
Colorectal cancer (CRC) is the third leading cause of cancer-related death in the world. Multiple evidence suggests that there is an association between excess fat consumption and the risk of CRC. The long chain n-3 polyunsaturated fatty acids (LC n-3 PUFA), especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are essential for human health, and both in vitro and in vivo studies have shown that these fatty acids can prevent CRC development through various molecular mechanisms. These include the modulation of arachidonic acid (AA) derived prostaglandin synthesis, alteration of growth signaling pathways, arrest of the cell cycle, induction of cell apoptosis, suppression of angiogenesis and modulation of inflammatory response. Human clinical studies found that LC n-3 PUFA combined with chemotherapeutic agents can improve the efficacy of treatment and reduce the dosage of chemotherapy and associated side effects. In this review, we discuss comprehensively the anti-cancer effects of LC n-3 PUFA on CRC, with a main focus on the underlying molecular mechanisms.
Background
Colorectal cancer (CRC) is the third most common cancer after lung and breast cancer and the fourth leading cause of cancer death worldwide.1,2 CRC is a multifactorial disease caused by the interaction of genetic and environmental factors 3 and it presents in 1 of 3 patterns: sporadic, inherited, and familial. The majority of CRC cases are sporadic and approximately 70% to 80% are derived from somatic mutations without any family history. 3 The inherited and familial causes (about 35%) of CRC are derived from germline mutations. 4 Age, poor diet, and sedentary lifestyles are considered the main environmental contributors to the disease. 5
There is considerable evidence indicating that red and processed meats, and alcoholic beverages are risk factors for CRC. However, grains, vegetables and fruits, dairy products, and fish and other seafood are linked with a decreased risk of CRC.6,7 Studies have also shown that high intakes of energy, saturated fatty acids (SFA) and sucrose are associated with increased risk of CRC; while high intakes of dietary fibers, calcium and long chain n-3 polyunsaturated fatty acids (LC n-3 PUFA) contributed to low incidence of CRC.8 -11
The onset of CRC is a complex process, and, in most cases, CRC starts with polyps occurring on the epithelial layer of the colon or rectum. These polyps may be benign (for example hyperplastic polyp), pre-malignant (eg, tubular adenoma), or malignant (eg, colorectal adenocarcinoma). 12 Several genetic, molecular, cellular, and histological changes were found to be associated with the transformation of normal epithelium to adenoma and invasive metastatic adenocarcinoma.13,14 This usually takes 10 to 15 years due to the incremental accumulation of genetic alterations.3,15 Three main causes of colorectal carcinogenesis have been identified. 16 The first one is the suppressor pathway of chromosomal instability (CIN) involving the accumulation of molecular alterations that influence oncogene activation (KRAS) and inactivation of tumor suppressor genes (DCC, APC, SMAD4, and TP53). 17 The second mechanism is the accumulation of errors in DNA replication as a result of mutations of the genes that are responsible for DNA repair (MSH2, MLH1, MSH6, PMS2, MLH3, MSH3, PISI, and Exol). This is known as the microsatellite instability pathway (MSI). 18 The third mechanism is the CpG island methylator phenotype (CIMP) pathway. This is due to the vast hyper-methylation of promoter CpG island sites that silence the tumor suppressor genes. 19 The progression of CRC is commonly associated with multiple steps such as hyperplasia of colonic crypts, colonic crypt dysplasia, adenoma, adenocarcinoma, invasion, and distant metastasis. 20
Currently available treatments for CRC are surgery, chemotherapy, radiotherapy, and molecular–targeted therapy.3,21 Surgery might be curative only when the disease is diagnosed at its early stages. Treatments for patients diagnosed at advanced stages are chemotherapy, radiotherapy, or a combination of both. However, these therapies are associated with numerous side effects leading to a significant impact on patients’ quality of life. 22 In recent years, a significant focus of cancer therapeutic research has been shifted to marine sources as they serve as a province for a range of bioactive compounds.23,24 Nutraceuticals that have little or no side effects have received considerable attention for the prevention and management of CRC and other invasive metastatic carcinomas.25,26
There is growing evidence showing that LC n-3 PUFA originated from marine oils (eg, fish oil and krill oil) have anti-CRC properties. These fatty acids have been reported to exhibit multiple anti-cancer effects on various stages of CRC from primary to tertiary, including advanced metastasis.26,27 Epidemiological studies have found that populations consuming LC n-3 PUFA-rich diets (such as fish and other seafood) have fewer cases of CRC compared to populations that consume diets containing less LC n-3 PUFA.28,29 A meta-analysis reported that a 50 g increment in the daily consumption of fish was associated with a statistically significant 4% reduction in CRC risk. 30 A recent European Prospective Investigation showed that CRC incidence decreases with increasing proportions of red blood cell membrane n-3 PUFA, particularly EPA. 31 Conversely, a diet containing a relatively high proportion of n-6 PUFA (eg, typical western style) was associated with an increased risk of inflammatory bowel disease (IBD) and colon carcinogenesis.32,33 This review discusses the beneficial effects of LC n-3 PUFA, mainly eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) and their two main marine oil sources, fish oil and krill oil, on the initiation, progression and apoptosis of CRC. A particular focus is on the molecular mechanisms underlying the anti-cancer properties of these fatty acids and their marine sources.
Methods
The databases of PubMed, PubMed Central, MEDLINE, Springer Link, and Wiley Online Library were searched using the key words of n-3 PUFA, EPA, DHA, fish oil, krill oil, colorectal cancer, molecular mechanism, signaling pathway, cell apoptosis, cell cycle, and anti-inflammatory. The inclusion criteria are defined molecular targets of LC-n3 PUFA in colorectal cancer. As there are a large number of studies available, the title and abstract were used for initial screening. Full text screening was then applied for original research and their relevant and key citations. Exclusion criteria include the preprints, conference proceedings, articles with only abstract available, and articles were not written in English.
Polyunsaturated Fatty Acids and Their Effects on Colorectal Cancer
There are two major families of polyunsaturated fatty acids (PUFA) including omega 3 (n-3 PUFA) and omega 6 (n-6 PUFA); and they are not metabolically interchangeable. 34 The members of n-3 and n-6 families are considered essential fatty acids for humans, since they cannot be synthesized de novo in the body and, therefore, must be obtained from dietary sources.27,34 Plant-based α-linolenic acid (ALA, C18:3, n-3) is known as the parent form of the LC n-3 PUFA, eicosapentaenoic acid (EPA, C20:5, n-3), docosapentaenoic acid (DPA, C22:5, n-3), and docosahexaenoic acid (DHA, C22:6, n-3), and it is an essential fatty acid. 35 ALA is commonly found in green leafy vegetables, nuts, especially in walnuts, and oils, such as flaxseed oil, soybean oil, and canola oil.36,37 After consumption, ALA can be utilized to synthesize LC n-3 PUFA by enzymes desaturases (delta-6 and 5) and elongases (2 and 5) mainly in the liver. But this process can also take place in other organs such as brain, kidney, and testicles.38,39 However, the body cannot synthesize EPA and DHA from ALA in sufficient quantities. 40 Only about 5% of the consumed ALA is converted to EPA in the body due to a lack of delta desaturases to catalyze the addition of double bonds.41,42 Therefore, these fatty acids are obtained mainly from fish (especially those from cold-water fatty fish, such as mackerel and salmon), fish products, and other seafood including oysters, mussels, and shrimps, as well as from dietary supplements.43,44 Fish oil and krill oil are the two major commercially available sources of EPA and DHA supplements.
The n-6 PUFA, linoleic acid (C18:2, n-6) is another essential fatty acid and the parental form of arachidonic acid (AA). Linoleic acid is mainly found in plant oils such as sunflower and corn oils, grains, and animal fats. Humans can readily metabolize linoleic acid to obtain AA via desaturation and elongation by the same set of enzymes as in the synthesis of EPA.27,39 In addition, AA can be obtained directly from beef, pork and eggs. 39 The LC n-3 PUFA and AA are biologically vital for the human body, because of their important roles in the phospholipid cell membrane structure, modulation of cellular signaling, membrane fluidity, cellular interaction, and lipid metabolism.27,45 EPA and AA are released from the cell membrane by the action of phospholipase enzymes, especially phospholipase A2 (PLA-2) and C (PLC) and metabolized through three main pathways of cyclo-oxygenase (COX), lipoxygenase (LOX), and cytochrome P450 (CYP450) . 46 The main metabolites are prostaglandins (PGs) and thromboxanes by COX-1 and COX-2, leukotrienes (LTs) and lipoxins by LOX, and hydroxyeicosatetraenoic acids (HETEs) and dihydroxyeicosatetraenoic acids (DHETEs) by cytochrome P450. In general, eicosanoids derived from AA are typically involved directly in the development of inflammation. Moreover, they are also involved in a range of biological process and modulate diverse physiological responses.46 -48 Prostaglandins have received more attention for their roles in modulating inflammation. It has been reported that prostaglandin-E2 (PGE-2), derived from the metabolism of AA, as an important pro-inflammatory agent, is associated with the onset and progression of several cancers through cell proliferation, angiogenesis, migration, and invasion while inhibiting apoptosis.49,50 In addition, it plays a significant role in the early stages of colorectal carcinogenesis. 51 LTB4, another metabolite of AA, apart from its pro-inflammatory action, has also been reported to stimulate cancer cell growth52,53
In contrast, prostaglandin-E3 (PGE-3), derived from EPA, is generally known for its anti-inflammatory and anti-cancer properties54,55. It has been found that PGE-3 can antagonize the effects of PGE-2, although few studies have shown discrepant results that PGE-3 causes the similar deleterious effects as PGE-2 on epithelial barrier function or promotes the proliferation of cancer cell line.56,57
In the cells, there is a competition between n-3 and n-6 PUFA for their metabolic process via COX enzymes. However, these enzymes have a higher affinity for EPA of n-3 PUFA rather than AA of n-6 PUFA, especially COX-2. 58 This leads to the formation of anti-tumorigenic PGE-3 and the reduction in the synthesis of PGE-2. In addition, LTB5 produced by LOX from EPA has shown anti-inflammatory and anti-cancer properties.59 -61 Furthermore, EPA and DHA can also produce a family of pro-resolving anti-inflammatory mediators including resolvins, protectins and maresins.62,63 The food sources, metabolism and functions of major metabolites of PUFA are summarized in Figure 1.
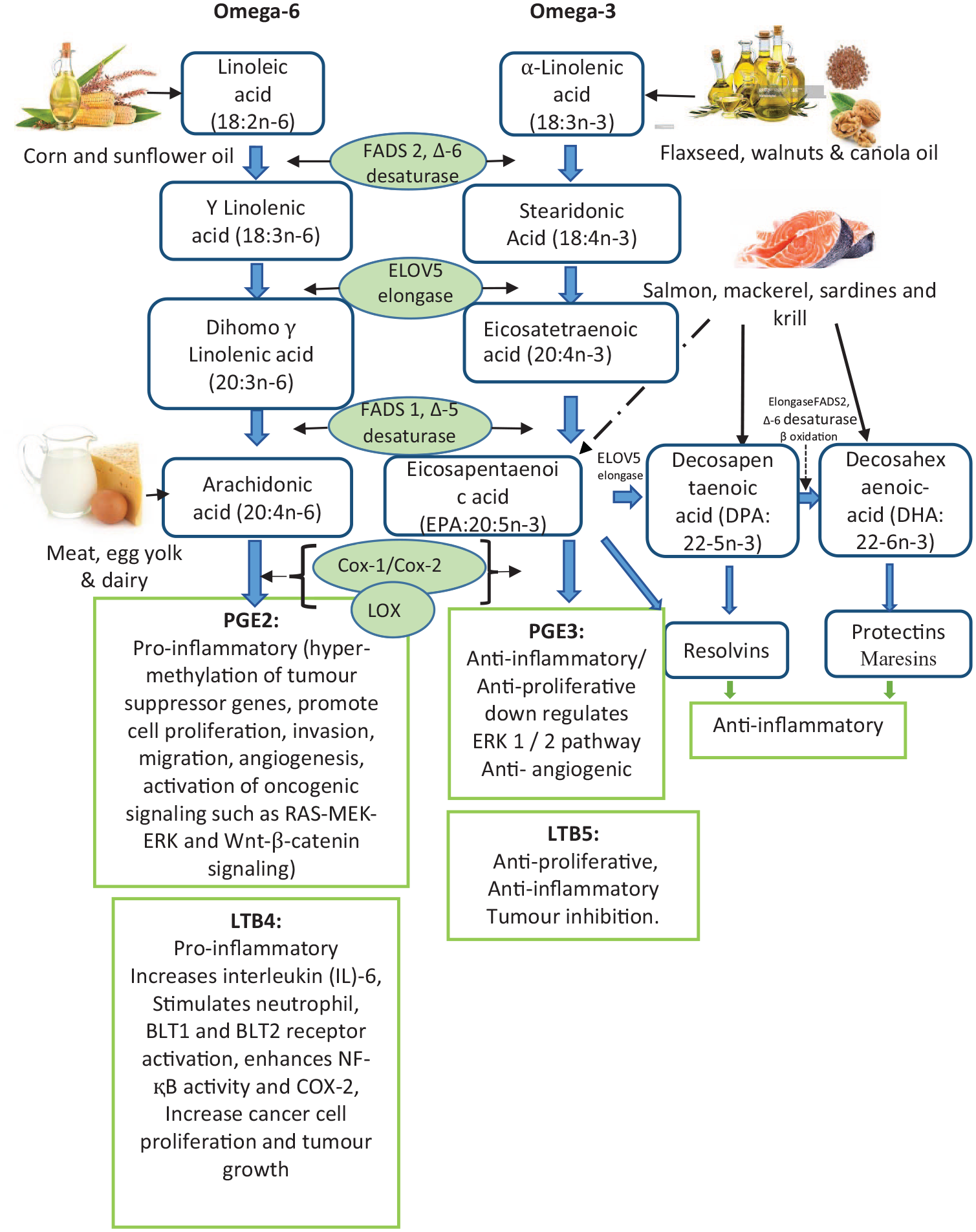
Schematic overview of biosynthesis of long chain PUFA and actions of main metabolites from arachidonic acid verse that from EPA. High metabolism of linoleic acid to arachidonic acid contributes to cancer risk and progression through the synthesis of pro-inflammatory and pro-tumor lipid metabolites. High metabolism of α-linolenic acid to EPA and DHA reduces cancer risk and progression through the synthesis of anti-inflammatory and anti-tumor lipid metabolites.
The main anti-cancer properties of LC n-3 PUFA involve the modulation of COX-2 enzymatic activity, alteration of the functions of cell surface receptors and membrane characteristics, enhancement of cellular oxidative stress, and production of anti-inflammatory mediators including resolvins, protectins, and maresins.27,62 It has been reported that LC n-3 PUFA can inhibit cancer cell proliferation and reduce tumor growth through various mechanisms, including the alteration of signaling pathways involved in carcinogenesis such as angiogenesis and cell metastasis64 -67; regulation of cell cycle; as well as induction of cell apoptosis.68 -70 The characteristic functions of those proposed molecular mechanisms are summarized in Figure 2.

Targeted signaling pathways by LC n-3 PUFA in relation to the death of cancer cells. (A) represents the inhibition of EGFR activation and the inhibition of its downstream signaling pathways. (B) represents the suppression of phosphorylation of the RAS/MEK pathway. (C) shows the inhibition of the PI3K/AKT/mTOR signaling cascade. Phosphorylated AKT activates mTOR, which further activates the transcription factors necessary for the transcription of genes essential for cell proliferation, metastasis, angiogenesis. (D) shows an intrinsic apoptosis pathway that activates through the up-regulation of pro-apoptotic Bcl-2 protein and down-regulation of anti-apoptotic Bcl-2 protein that leads to alteration of MMP to release cytochrome c. Cytochrome c forms an apoptosome with the binding of Apaf and caspase-9 to further activate caspase-3 to induce apoptosis. (E) represents the intrinsic apoptosis pathway through the endoplasmic reticulum. (F) represents the extrinsic pathway of apoptosis showing Eas/FasL interaction and DISC formation leading to caspase-8 activation. (G) represents the suppression of NF-қB that upregulates the transcription of genes involved in inflammation, angiogenesis, and metastasis. (H) represents all phases of the cell cycle together with its cyclin and CDKs. The LC n-3 PUFA suppress the cell cycle by alteration of cyclin and CDKs. Abbreviations: EGFR, epidermal growth factor receptor; EGF, epidermal growth factor; P, phosphorylation; RAS, retrovirus-associated DNA sequences; RAF, rapidly accelerated fibrosarcoma; MEK, mitogen-activated protein kinase; ERK, extracellular signal-regulated kinase; PI3K, Phosphatidylinositol-3-kinase; AKT, threonine-protein kinase; mTOR, Mammalian target of rapamycin; Bcl-2, B-cell lymphoma-2; BAX, Bcl-2 Associated X-protein; BAK, Bcl-2 Antagonist/Killer; tBid, truncated Bid; Puma, p53 upregulated modulator of apoptosis; MCL-1, myeloid cell leukemia-1; Bcl-xL, B-cell lymphoma-extra-large; BIM, Proapoptotic Bcl-2 homology 3-only protein; FADD, Fas-associated death domain protein; TRAIL-1 /2, Tumor necrosis factor-related apoptosis-inducing ligand receptor 1 /2; FAS, Fas Cell Surface Death Receptor; DISC, death-inducting signaling complex; CDK, cyclin-dependent kinase; G1, gap 1/growth 1; G2, gap-2/growth 2; M, mitosis; S, Synthesis Phase; DNA, deoxyribonucleic acid; JNK, Jun N-terminal kinase; NF-ĸB, nuclear factor kappa-light-chain-enhancer of activated B cells; COX-2, cyclooxygenase-2; PGE-2, prostaglandin E2; iNOS, inducible nitric oxide synthase; NO, nitric oxide; BH3, canonical mitochondrial apoptosis.
LC n-3 PUFA Alter CRC Growth Signaling Pathways
The cellular behavior in healthy individuals is tightly controlled by a complex network of signaling pathways involving growth factors, which ensure the proliferation of cells only when required. Dysregulation of these growth factor-dependent cell proliferation signaling pathways is one of the hallmarks of cancer development and progression. One of the identified dysregulations of the cell signaling pathway in CRC is the overexpression and activation of the epidermal growth factor receptor (EGFR). The EGFR is a multifunctional member of the ErbB family of tyrosine kinase receptors that transmits a growth-inducing signal to the cell.71,72 The higher expression of EGFR has been recognized as an important player in CRC initiation and progression. The EGFR is activated through interaction with its ligands, epidermal growth factor (EGF), and transforming growth factor-alpha (TGF-α). Overexpression of the EGFR-ligand complex plays a crucial role in cell proliferation, differentiation, survival, adhesion, migration, tumorigenesis, as well as resistance to cancer therapy.72,73 The binding of EGFR with its corresponding ligand triggers the catalytic activity of its intrinsic kinase that leads to the activation of several downstream intracellular signaling pathways, including Ras/Raf/Mitogen Erk1/2, the Phosphatidylinositol 3-Kinase (PI3K)/A Serine/Threonine-Protein Kinase (Akt)/mammalian target of rapamycin (mTOR) and signal transducer and activator of transcription (STAT-3), PLC-gamma-1, and c-Jun N-terminal kinase (JNK). 74 The overexpression of these signaling pathways causes the up-regulation of cell growth and carcinogenesis. 75 The inhibition or inactivation of the EGFR complex involves signaling pathways that are associated with an anti-cancer mechanism.65,66,70,76 Studies have shown that LC n-3 PUFA treatment can inhibit the activation of EGFR and its downstream intracellular signaling pathways Ras/Erk and AKT.66,72 Free fatty acid extract of krill oil treatment was also found to reduce the expression of EGFR/pEGFR and their downstream signaling, pERK1/2 and pAKT along with the downregulation of programed death-ligand 1 (PD-L1). 77
It has also been found that increased expression of vascular endothelial growth factor (VEGF) is associated with the progression of CRC; and the enhancement of VEGF signaling pathways could increase cancer cell proliferation, metastasis, and influences survival rates of CRC patients. 78 Activation of the VEGF receptor could also lead to transphosphorylation, an increase in intrinsic catalytic activity, and the creation of receptor binding sites on tyrosine kinases (RTK) to recruit cytoplasmic signaling proteins that activate mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase 1/2 (ERK 1/2) and mTOR/AKT pathways. 79 Continuous activation of MAPK and mTOR due to VEGF signaling stimulates the inhibition of cellular apoptosis and increases cell survival through the up-regulation of anti-apoptotic B cell lymphoma (Bcl) regulatory proteins, Bcl-2 and Bcl-xL.80,81 Furthermore, COX-2 is also involved in the expression of VEGF, because COX-2 and PGE-2 are well-established upstream regulators of VEGF during angiogenesis.82,83 Some studies have observed that COX-2-derived PGE-2 signaling through the prostaglandin (EP) 4 receptor stimulates cell proliferation, angiogenesis, and resistance to anti-tumor immune response and apoptosis.84,85 Furthermore, experimental results have shown that nitric oxide (NO) can promote cell survival, proliferation, inhibiting apoptosis,86,87 and regulate VEGF-mediated angiogenesis. 88 Studies found that LC n-3 PUFA treatment can reduce the expression of COX-2 and the synthesis of PGE-2.82,89 Furthermore, LC n-3 PUFA induce apoptosis through the modulation of Bcl-2 family proteins90,91 and decrease of VEGF signaling pathways65,92 involved in cell migration, blood vessel formation, and NO production.93,94
The peroxisome proliferator-activated-receptors (PPARs) are a group of the nuclear receptor proteins. 95 The LC n-3 PUFA are thought to play a crucial role in upregulating the transcription factor PPAR-alpha. This regulation is important to reduce the activity of the transcription factor NF-ĸB,96 -98 which is essential to mitigate inflammatory responses. 99 It has been reported that these beneficial effects are associated withthe mediators of LC n-3 PUFA including resolvins, protectins and maresins 99 In addition, the enhanced expression of PPARs was reported to be associated with the suppression of cellular proliferation64,100 and angiogenesis through the downregulation of VEGF. 65 It was found that EPA modulates PPARs to reduce cell viability by inducing cellular apoptosis. 64 Table 1 summarizes the in vitro studies that examined the effects of LC n-3 PUFA on the modulation of different survival signaling pathways involved in colorectal carcinogenesis.
Summary of Studies Investigating the Effects of LC n-3 PUFA on the Modulation of Survival Signaling Pathways in CRC Cells.

LC n-3 PUFA Induce CRC Death via Apoptosis
The onset of CRC associates with uncontrolled cell proliferation and a reduction in cell apoptosis. Apoptosis is a programed process of cell death and caspases are fundamental for this mechanism. There are three distinct pathways involved in the apoptotic mechanism, including the intrinsic (mitochondrial), the extrinsic (through the death receptors), and intrinsic endoplasmic reticulum (ER) pathways.111 -113
The intrinsic apoptosis pathway responds to diverse stress signals such as growth factor deprivation, DNA damage, and reactive oxygen species (ROS). Mitochondria are central to this pathway, which involves pro- and anti-apoptotic members of Bcl-2 family proteins.114,115 The pro-apoptotic Bcl-2 proteins include Bax, Bak, Diva, Bcl-Xs, Bik, Bim, Bad, and cBid; and the anti-apoptotic Bcl-2 proteins are Bcl-2, Bcl-XL, Mcl-1, CED-9, A1, Bfl-1. These proteins collectively regulate the mitochondrial membrane potential (MMP). An imbalance in pro- and anti-apoptotic proteins triggers -apoptotic events.116 -118 and results in the release of pro-apoptotic regulators, cytochrome c, Smac/Diablo, endonuclease G, and apoptosis-inducing factor (AIF) from mitochondria into the cytosol.112,119 Cytochrome c then triggers the formation of apoptosome complex via interaction with apoptotic protease activating factor 1 (Apaf1). This complex is involved in the activation of the initiator caspase, caspase-9, then caspases 3 and 7, leading to cell apoptosis.112,120 Furthermore, Smac/Diablo promotes apoptosis through the inhibition of anti-apoptotic protein, endonuclease G, and translocation of AIF in the cell nucleus. This causes a large amount of DNA damage via a caspase-independent apoptotic pathway.121,122
Cell apoptosis through the extrinsic pathway involves death domain receptors, such as tumor necrosis factor (TNF)-α, CD95 (Fas or apoptosis antigen 1), and TNF-related apoptosis-inducing ligand (TRAIL) receptors, after interaction with their corresponding ligands. The activation of these receptor-ligand complexes causes the formation of death-inducing signaling complexes (DISCs), including TNF receptor-associated death domain (TRADD), Fas-associated death domain (FADD), and procaspase-8/FLICE receptor-interacting protein kinase 1 (RIPK1). 112 These complexes activate caspases 8 and 10, and then caspase-7 leading to apoptotic cell death.112,123
Apart from those two caspase pathways, the intrinsic endoplasmic reticulum (ER) pathway is also related to apoptosis. The ER is a site for the synthesis, folding, modification of the cell surface proteins, and intracellular calcium storage compartment. The stress of ER could be induced by the accumulation of unfolded and/or misfolded proteins in the ER lumen. This could then activate the unfolded protein response (UPR) signaling pathway associated with a wide variety of human diseases. Furthermore, the unfolded protein response and continuous ER stress activate the apoptosis through caspases 3 and 7, or via a p53-dependent pathway.124,125
Numerous studies in the last two decades suggest that LC-n-3 PUFA treatment prompts colorectal cancer cell apoptosis through intrinsic or extrinsic pathways, as summarized in Table 2. LC n-3 PUFA elevate pro-apoptotic proteins, and suppress anti-apoptotic proteins, in the Bcl-2 family.126 -132 Furthermore, studies also showed that LC n-3 PUFA induce apoptosis via the activation of extrinsic pathways involving caspases 9 and 8.133,134 Giros et al found that LC n-3 PUFA induce apoptosis through both intrinsic and extrinsic pathways, via the release of Smac/Diablo and cytochrome c into the cytosol and the activation of caspase-8. Their results demonstrated that extrinsic apoptosis is independent of death receptor activation but influenced by LC n-3 PUFA on FLICE-like inhibitory proteins. 127
Summary of Studies Investigating the Effects of LC n-3 PUFA on Apoptosis of CRC Cells.

LC n-3 PUFA Alter CRC Cell Cycle
The cell cycle orchestrates precise molecular events, ensuring the generation of identical cell copies. Healthy cells regulate growth signals activated by factors such as growth factors, cell-to-cell interaction molecules, and extracellular matrix components that affect cell growth and maintain the cell cycle. This helps to control the total number of cells, and the structure and function of normal tissues. 116 However, some cells are progressively transferred into a neoplastic state (cancer) when they escape from the normal cell cycle and produce their own growth factors independently to proliferate infinitely. 116
The cell cycle consists of four sequential phases: G1 (gap 1), S (DNA synthesis), G2 (gap 2), and M (mitosis). Cyclin-dependent kinase (CDK) plays a pivotal role in regulating the progression of cell cycle and preventing transitions between phases.142,143 CDK activation occurs through the interaction with cyclins, forming a cyclin-CDK complex143,144 In response to mitogen signals, CDK4 and CDK6 activate the D-type cyclins, facilitating G1 progression and activation of the transcription factors.144,145 CDK2 is subsequently activated by cyclin E and cyclin A, initiating DNA replication. The activation of cyclin B-CDK1 complex leads to mitosis, while cyclin-dependent kinase inhibitors (CKI) including INK4 and Cip/Kip families monitor and deactivate CDK-cyclin complexes that arrest the cell cycle. The INK family has four members, p15, p16, p18, and p19; and the Cip/Kip family comprises p21, p27, p57, p107, and p130.144,146,147
Several studies reported that LC n-3 PUFA inhibit cancer cell proliferation through the alteration of cell cycle progression. Table 3 summarizes the results of in vitro studies elucidating the impacts of LC n-3 PUFA on the CRC cell cycle. Treatment with DHA and/or EPA arrests the cell cycle progression in the G1 phase in CRC cells.69,126,131 Molecular components crucial for CRC cell cycle progression, such as Cdc25c, Cdc25b, Cdc20, CDK1, CDK2, and cyclin D, A, and B are downregulated by LC n-3 PUFA.69,126,131,136,148 Studies found that DHA treatment increases the level of p21 and reduces the level of cyclin D1, inducing cell cycle arrest.126,131 LC n-3 PUFA also activate the p53 pathway, causing DNA damage and modulating the p21 signaling pathway. 149
Summary of Studies Investigating the Effects of LC n-3 PUFA on the Cell Cycle of CRC.

Overall, a large number of in vitro studies have confirmed the positive impacts of LC n-3 PUFA on CRC cells, and several molecular signaling pathways have been suggested. However, the comparative roles of EPA versus DHA versus total LC n-3 PUFA in the modulation of these molecular pathways are not clear, given that different doses, treatment durations as well as cell lines were used in different studies.
The Anti-inflammatory Effects of LC n-3 PUFA on CRC Cells
The relationship between chronic inflammation and cancer initiation and progression has been well recognized and documented for several types of cancer, including CRC.153 -155 The risk of CRC could be increased by 10 times in patients with a history of inflammatory bowel disease. 156 The cyclic auto-activation process of the inflammatory signaling pathways increases the production of immune cells, and release of several pro-inflammatory cytokines (IL-1, IL-2, IL-4, IL-6, IL-12, IFNɤ, and TNFα), chemokines (IL-8, monocyte chemo-attraction protein-1 (MCP-1/CCL2)), growth factors, reactive oxygen and nitrogen species, and lipid molecules (saturated fatty acids)157,158 These mediators stimulate signaling processes within the cells that play an important role in the growth and development of cancer. 157
Furthermore, the soluble mediators generated by cancer cells also contribute to the recruitment and activation of immune cells and the excessive production of pro-inflammatory mediators. The inflammatory reaction generated by immune cells activates and maintains several signaling processes. These processes continue to stimulate proliferative signaling, the survival of cancer cells, and extracellular matrix (ECM), which facilitate tumor growth by modifying the enzymes that enable angiogenesis and metastasis.116,159 -163 In addition, some tumor and myeloid cells, including macrophages, polymorphonuclear neutrophils (PMN), and myeloid-derived suppressor cells (MDSCs) express mediators of the immunosuppressive checkpoint, such as programed death-ligand 1 (PD-L1) and ligand 2 (PD-L2) resulting in cellular interactions that suppress T-cell proliferation and function.164,165 Studies have found that LC n-3 PUFA have an ability to down-regulate inflammation and inflammatory cell infiltration of tumors. 166 Furthermore, LC n-3 PUFA have pro-resolving effects on both innate and adaptive immunity through multiple mechanisms, including influence on various cellular phenotypes that coordinate the host response against tumors. Resolvins that are the metabolites from LC n-3 PUFA have an endogenic pro-resolution activity that protects against abnormal/uncontrolled innate inflammatory responses. 165 Altogether the inflammatory responses play a crucial role in tumor development at different stages including initiation, promotion, malignant transformation, invasion, and metastasis. 161 Recently, the proliferative effects of pro-resolvin mediators on Caco-2 cell line have been reported. 167 The proliferation of intestinal epithelial cells stimulated by pro-resolvin mediators was related to wound closure. More studies using other cell lines are warranted to elucidate the effect of pro-resolvin mediators on CRC growth. LC n-3 PUFA have shown useful anti-inflammatory properties, due to the immunomodulatory effects of substances derived from these fatty acids through eicosanoid metabolism. 168 The main anti-tumor effect of LC n-3 PUFA can be mediated through the downregulation of the synthesis of pro-inflammatory eicosanoids from n-6 PUFA of AA. 169
The phospholipid membrane normally contains more AA than other 20-carbon PUFA. 169 AA is the molecular substrate causing the over-activation of some enzymatic pathways in CRC, such as COX and LOX.170,171 The PGE-2 produced by COX from the metabolism of AA is typically pro-inflammatory,27,172 and excess PGE-2 has been found to be linked with the onset and progression of colorectal carcinogenesis through increased cell proliferation, angiogenesis, cell migration and invasion, as well as the inhibition of apoptosis.173,174
Dietary supplementation with LC n-3 PUFA significantly decrease the concentration of AA in the cell membrane, as well as its ability to displace AA as a molecular substrate in the COX and LOX pathways.27,175 The actions of metabolized derivatives of LC n-3 PUFA, such as prostaglandin E3 (PGE-3), on the substrate of COX and LOX are typically anti-inflammatory. 176 Therefore, it has been found to reduce angiogenesis,177,178 inflammation,27,179 and exert anti-cancer properties.27,180 The studies on the anti-CRC effects of LC n-3 PUFA via modulation of the inflammatory response are summarized in Table 4.
Summary of Studies on Anti-Inflammatory Effects of LC n-3 PUFA in CRC Cells.

LC n-3 PUFA can downregulate the Bcl-2 and inhibit the Nuclear factor-kB (NF-κB) pathway. 181 A similar effect has been observed in other studies, showing that LC n-3 PUFA can cause the suppression of genes involved in the NF-ĸB pathway.94,174,180 NF-ĸB is a transcription factor that regulates the expression of many genes involved with the upregulation of COX, production of inflammatory cytokines, the progression of the tumor cell cycle, and adhesion molecules. These activities play an important role in tumor invasiveness and provide tumors with the inflammatory microenvironment that supports tumor progression, invasion of surrounding tissue, angiogenesis, and metastasis.183 -186 In addition, the experimental evidence shows that LC n-3 PUFA can decrease the gene expression of inflammatory molecules such as the inflammatory cytokines interleukin IL-1β, IL-6 and TNF-α.187,188 The anti-inflammatory properties of LC n-3 PUFA include the reduction of anti-apoptotic genes, Bcl-2 and survivin and the increase of pro-apoptotic factors.172,181 More studies are warranted to further identify the specific interactions of LC n-3 PUFA and gut microbiota, and their impacts on the immune response in CRC.
The Anti-Cancer Effects of LC n-3 PUFA in Vivo
Animal studies have demonstrated the beneficial effects of LC n-3 PUFA on CRC at the early stages of carcinogenesis. EPA and DHA can reduce tumor growth, progression, and invasion by inhibiting cell proliferation and inducing cell apoptosis.109,189-193 The LC n-3 PUFA supplementation in immunosuppressed mice with human CRC cell xenografts significantly reduced tumor size compared to the control group.150,189,190,194,195 Several studies have also reported positive outcomes following supplementation with LC n-3 PUFA-rich marine oils including fish oil and krill oil.178,193,195,196 In addition, these studies have reported several molecular actions of LC n-3 PUFA in CRC tumor inhibition (Table 5). For example, modulation of COX metabolism and reduction of PGE-2 production in tumors, 180 inhibition of EGFR and VEGF signal transduction through the alteration of lipid raft composition and fluidity, 197 and downregulation of pathways associated with CRC promoting signals such as AKT90,195,198 and Wnt/β-catenin. 180
Summary of In Vivo Studies Investigating the Effects of LC n-3 PUFA and Their Marine Oil Sources on CRC and Associated Signaling Pathways.

Kato et al reported that DHA inhibits tumor growth more efficiently than EPA in athymic mice with COLO 205 subcutaneous xenografts. The DHA treatment reduced tumorigenesis by inhibiting genes responsible for tumor angiogenesis, as well as inducing cellular apoptosis via p53 dependent and independent pathways. These positive effects were found to be correlated with the increased level of LC n-3 PUFA in tumor tissues and the reduction of phosphocholine (PC) in xenografts. 150 Zou et al reported that the molecular mechanisms associated with the anti-cancer properties of LC n-3 PUFA involve the inhibition of several genes, such as COX-2, HIF-1α, VEGF-A, COMP, MMP-1, MMP-9, SCP2, SDC3 in nude mice with HCT-15 subcutaneous xenografts. 194 Kansal et al showed that dietary supplementation with LC n-3 PUFA-rich fish oil significantly suppress CRC development and increased apoptosis by reducing the expression of the Ras-induced Raf/MEK/ Erk 1/2 and Akt signaling pathways in rats treated with a carcinogen 1,2-dimethylhydrazine(DMH). 191 Furthermore, Huang et al found that n-3 PUFA attenuated MNU-induced CRC in rats by inhibiting CRC cell proliferation and inducing CRC cell apoptosis via blocking PI3K/AKT and Bcl-2 signaling pathways. 90 The role of LC n-3 PUFA in decreasing cell proliferation in tumor tissues was also found to be associated with the modulation of inflammatory pathways. Rosa et al discovered that a fish oil-enriched diet reduced interleukin-8 (IL-8) expression and enhanced TGF-β expression in Wister rats treated with DMH. 200 In addition, fish oil was found to suppress the initiation of aberrant crypt foci (ACF) development in male Wister rats, 202 reduce TNF-α production in HCT-116 xenograft tumor-bearing nude mice, 182 reduce the expression of phosphorylated ERK 1/2, lower the PGE-2 and increase the PGE-3 levels in Balb/c mice. 106 Koppelmann et al found that n-3 PUFA inhibit NF-қB/COX-2 induced production of pro-inflammatory cytokines and inhibited cell apoptosis to prevent MTX-induced intestinal damage in male Sprague-Dawley rats. 205 Fish oil diet has also been shown to upregulate p21, induce cell cycle arrest, and promote apoptosis of cancer cells in rats with azoxymethane (AOM)-induced colon cancer. 204 LC n-3 PUFA rich diets were found to dramatically decrease cell proliferation and polyp formation and increase apoptosis of cancer cells through the downregulation of COX-2 and β catenin nuclear translocation in ApcMin/+ mice. 203 Recently, we reported that dietary supplementation with krill oil reduces CRC tumor growth and induces cancer cell death. 195 We also observed that the anti-cancer effects of krill oil are comparable with that of the clinical therapeutic agent, Oxaliplatin. These positive effects are associated with the downregulation of EGFR signaling pathways.195,196 and the activation of the intrinsic mitochondrial death pathway. 196 Available animal studies have confirmed some of reported molecular actions of LC n-3 PUFA in vitro. However, there are still gaps between in vitro and in vivo studies. Future in vivo studies focusing on those yet to be proven molecular signaling pathways are highly recommended.
LC n-3 PUFA as an Adjuvant Therapy for CRC
Several studies have investigated the action of LC n-3 PUFA as a single agent or in combination with chemotherapeutic agents in the treatment of cancer. 206 A recent review of current clinical evidence showed that LC n-3 PUFA supplementation delays cancer progression, maintains body weight, and improves appetite and overall quality of life in CRC patients. In the advanced cancer patients, supplementation also decreases pro-inflammatory cytokines and serious adverse events of chemotherapy. 207 The increased level of LC n-3 PUFA in tumor cell membrane and production of lipid mediators were found to be correlated with their anti-cancer properties. High levels of LC n-3 PUFA resulted in an alteration of lipid rafts leading to a range of beneficial events in tumor cells.105,208 The combined treatments of LC n-3 PUFA with chemotherapy have resulted in reducing the dose of chemotherapeutic agents and decreasing the side effects associated with higher doses of chemotherapy. The lower toxicity profile and nutritional benefits of LC n-3 PUFA provide a useful adjuvant therapy for CRC. 209 Fish oil emulsion rich in LC n-3 PUFA has shown anti-cancer effects in combined treatments with standard chemotherapeutic agents, 5-fluorouracil (5-FU), oxaliplatin (OX), or irinotecan (IRI) in CRC cell lines. 128 Fish oil emulsion improves the efficacy of chemotherapeutic agents through a Bax-dependent mitochondrial pathway. 128 Vasudevan et al have demonstrated that treatment with EPA in combination with 5-FU plus oxaliplatin (FuOx) reduces HT-29 and HCT-116 cancer cell growth. It also induces apoptosis through PARP cleavage, and downregulation of pAKT, β-catenin, and Wnt signaling pathways. 210 5-FU combined with fish oil emulsion inhibits the proliferation of Caco-2 CRC cells lines more significantly compared to cells treated by either agent alone. 152 Similar findings by Calviello et al showed that DHA combined with 5-FU resulted in a decrease of the expression of anti-apoptotic proteins Bcl-2 and Bcl-Xl and excessive expression of pro-apoptotic c-MYC in human CRC cells, with low toxicity. 173 De Carlo et al also observed that EPA combined with standard chemotherapies, 5-fluorouracil and oxaliplatin, synergistically reduces COLO 320 CRC cell proliferation and increases their sensitivity to chemotherapeutic drugs. 211
Similar results have also been observed in several animal models of CRC. Rani et al demonstrated that the administration of 5-FU combined with fish oil to mice with colon cancer induced by DMH and dextran sodium sulfate (DSS) enhances both DNA damage and an apoptotic index through the activation of cellular extrinsic and intrinsic apoptotic pathways while reducing the side effects associated with 5-FU treatment. 212 It was also reported that 5-FU combined with fish oil treatment inhibits tumor growth and arrests the cell cycle in mice with DMH/DSS-induced colon cancer. 213 Other studies have also highlighted the beneficial effect of LC n-3 PUFA as adjuvant therapy. Xue et al have found that LC n-3 PUFA supplementation combined with irinotecan (CPT-11) and a 5-FU cyclical regimen could synergistically increase chemo-sensitivity and reduce body weight loss, anorexia, and muscle wasting in rats bearing Ward colon tumor compared to the control animals. 214 Vasudevan et al have also observed that treatment with EPA combined with FuOx (5-FU + oxaliplatin) reduces the tumor size and pro-inflammatory mediators in SCID mice with HT-29 or HCT-116 cell-induced subcutaneous xenografts. 210 Jeong et al found that the oxaliplatin and DHA combination can further reduce oxaliplatin-induced cell viability and autophagy cell death both in vitro and in vivo. Moreover, the combination of oxaliplatin and DHA increased the expression of the stress-sensitive gene of SESN2 and increased ER stress. 215 Our recent study has demonstrated that krill oil combined with ½ dose of oxaliplatin can reduce tumor growth to a similar extent as oxaliplatin, without side effects. 196
Some human studies have also demonstrated the anti-cancer effects of LC n-3 PUFA combined with chemotherapy on CRC. It is found that fish oil supplementation positively modulates the nutritional status and reduces pro-inflammatory mediates in CRC patients undertaking chemotherapy. 207 Similarly, Mocellin et al observed that fish oil supplementation during chemotherapy improves C-reactive protein (CRP) values, CRP/albumin status, and prevents weight loss. 216 A study of patients receiving a combination of fish oil and chemotherapy (5-FU and leucovorin) showed that fish oil can prevent the loss of blood polymorphonuclear cells (PMNC), mainly neutrophils, and increase their phagocytosis and production of hydrogen peroxide as well as prevent body weight loss related to chemotherapy. 217 Read et al observed that EPA supplementation maintains the nutritional and inflammatory status in patients having chemotherapy at an advanced stage of CRC. 218
A more recent study by Koppelmann et al has also demonstrated the beneficial role of fish oil as an effective adjuvant therapy for colon cancer. 205 Their data showed that LC n-3 PUFA can prevent intestinal damage and stimulate intestinal recovery. The prospective study by Song et al has found that a higher intake of LC n 3-PUFA may be associated with better survival of patients with stage 3 colon cancer. 219
Taken together, these studies demonstrate that LC n-3 PUFA, either alone or in combination with currently used clinical chemotherapy, may be a useful therapy for CRC. This is attributed to the specific roles of these fatty acids in suppressing tumor growth and development via various molecular signaling pathways discussed in this review. The synergistic impact of LC n-3 PUFA and chemotherapies on CRC are beneficial to CRC patients. Two major sources of LC n-3 PUFA are fish oil and krill oil. The effects of fish oil on CRC have been reported. Preliminary studies have also shown the potential role of krill oil in CRC treatment. More in vivo studies and clinical trials are required to validate the therapeutic efficacy of krill oil.
Conclusion
The data presented in this review have shown the beneficial effects of LC n-3 PUFA on colorectal cancer. The results from several experimental studies using CRC cell lines and animal models provide strong evidence that LC n-3 PUFA supress CRC by modulating different molecular pathways associated with cancer development and progression. These include the action of LC n-3 PUFA on intracellular and extracellular receptors in various signaling pathways involved in cell proliferation, metastasis and apoptosis, as well as angiogenesis and inflammation. Clinical studies have also demonstrated that LC n-3 PUFA can enhance the efficacy and tolerability of chemotherapy by reducing the side effects and toxicity associated with conventional anti-cancer therapies.
Footnotes
Abbreviations
AKT, threonine-protein kinase; BAK, Bcl-2 antagonist killer; BAX, Bcl-2-associated X protein; Bax/Bak, nuclear encoded proteins; Bcl-2, B cell lymphoma protein-2; Bcl-xL, B cell lymphoma protein-xL; CDC25, cell division cycle 25 homolog; CDHA, conjugated docosahexaenoic acid; CDK-1, cyclin-dependent kinase 1; CDK-2, cyclin dependent kinases 2/4; c-fos, proto-oncogene; C/EBP, enhancer-binding proteins; CHOP, C/EBP homologous protein; CiPi/waf1, cell cycle inhibitor proteins; CO, corn oil; COMP, cartilage oligomeric matrix protein; COX-2, cyclooxygenase-2; cyclic GMP, cyclic guanosine monophosphate; DHA, docosahexaenoic acid; DMH, 1,2-dimetilhidrazine; DNA, deoxyribonucleic acid; EGFR, epidermal growth factor receptor; eIF2α, eukaryotic translation initiation factor 2-α; EMT, epithelial-to-mesenchymal transition; EP4, G protein–coupled prostaglandin receptor; EPA, eicosapentaenoic acid; EPA-EE, ethyl ester eicosapentaenoic acid; EPA-FFA, eicosapentaenoic free fatty acids; ER, endoplasmic reticulum; ERK 1/2, extracellular signal-regulated kinase; FFA, free fatty acid; FLIP, FLICE-inhibitory protein; GrB, granzyme B; GRP78, glucose related protein of 78 kDa; GSK3β, glycogen synthase kinase 3β; HIF-1α, hypoxia-inducible factor 1-alpha; IFN, interferon; IL-1-β, interleukin 1 beta; IL-6, interleukin 6; IL-8, interleukin 8; iNOS, inducible nitric oxide synthase; FO, fish oil; LTB4, leukotrienes B4; MAG-EPA, monoglyceride eicosapentaenoic acid; MAPK, mitogen-activated protein kinase; MEK 1/2, mitogen-activated protein kinase 1/2; mir-21, microRNA-21; MMP, mitochondrial membrane potential; MMP-1/9, matrix metalloproteinase-1/9; mPGE-2, messenger function for prostaglandin E2; mRNA, messenger ribonucleic acid; mTOR, mammalian target of rapamycin; NF-ĸB, nuclear factor kappa-light-chian-enhancer of activated B cells; n-SREBP1 nuclear sterol regulatory element-binding protein 1; P21/27, cyclin-dependent kinase inhibitor 21/27; PERK, PER-like ER kinase; PG, prostaglandin; PGE-2, prostaglandin E2; PGE-2/E-3, prostaglandin E2/E3; PGE-3, prostaglandin E3; PPAR-γ, peroxisome proliferator-activated receptor-γ; PUFA, polyunsaturated fatty acids; Raf, rapidly accelerated fibrosarcoma; Ras, retrovirus-associated DNA sequences; ROS, reactive oxygen species; SCD3, syndecan 3; SCP2, sterol carrier protein 2; TCF, T-cell factor; TNF-α, tumor necrosis factor; TRAIL, tumor necrosis factor-related apoptosis inducing ligand; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor; Wnt, Wingless-related integration site; Wtp53, wild type p53; XIAP, X-chromosome-linked inhibitor of apoptosis protein.
Author Contributions
AJ, XS, KN conceived the idea for this article. AJ performed literature search, data analysis and drafted the manuscript. XS, KN and RL critically reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
