Abstract
Introduction:
Chemotherapy-induced peripheral neuropathy (CIPN) is one of the prevalent and disabling side effects of cancer treatment. However, management strategies for CIPN currently remain elusive, with treatment restricted to neuropathic pain medications, supportive care, and chemotherapy dosing adjustments. This overview explores evidence on the potential benefits and safety of nonpharmacological interventions in preventing and treating CIPN in cancer patients.
Methods:
Seven databases were searched for systematic reviews of randomized controlled trials (RCTs). The methodological quality of the selected reviews was assessed by AMSTAR 2, and the quality of evidence was judged by GRADE. Twenty-eight systematic reviews were considered eligible for this review.
Results:
It was found that nonpharmacological interventions (acupuncture, exercise, herbal medicine, nutritional supplements) provided potential benefits for patients with CIPN. Furthermore, Chinese herbal medicine, administered orally or externally, significantly prevented and/or relieved the incidence and severity of CIPN in comparison to control groups (no additional treatment, placebo, and conventional western medicine). However, the quality of evidence and strength of recommendations were compromised by the inconsistencies and imprecision of included studies. The main concerns regarding the quality of systematic reviews included the lack of sufficiently rigorous a priori protocols, and the lack of protocol registration adopted in the included studies.
Conclusions:
Though looking across reviews, Chinese herbal medicine appear generally effective in CIPN, uncertainty remains about the effects of many other nonpharmacological interventions. The evidence on what works was particularly compromised by reporting and methodological limitations, which requires further investigation to be more certain of their effects.
Keywords
Introduction
Chemotherapy-induced peripheral neuropathy (CIPN) is one of the prevalent and disabling side effects of cancer treatment regimens including neurotoxic chemotherapeutic agents (eg, taxanes, platinum compounds, vinca alkaloids, proteasome inhibitors, immunomodulatory agents). 1 The prevalence of CIPN varies from 30% to 80%, and many patients have chronic symptoms during treatment. CIPN manifestations include certain variation of numbness, tingling, shooting pain, stabbing pain, burning, and increased thermal sensitivity, which may lead to day-to-day functional comorbidity.2,3 Published reviews for the prevention and treatment of CIPN were dedicated to evaluating pharmacologic therapies.4-6 However, only duloxetine was recommended by the American Society of Clinical Oncology (ASCO) with limited effectiveness. Most pharmacologic medications, including tricyclic antidepressants and anticonvulsants, either present limited efficacy in CIPN or pose intolerable risk of adverse events to patients.7-10
Nonpharmacological therapies comprise a broad range of physical therapies, mind and body practices, natural products, and supplements. There is increasing interest in the effect of nonpharmacological therapies in integrative oncology.11,12 However, evidence remains incomplete for many of these therapies.13,14 To date, a number of systematic reviews (SRs) have been published broadly on the use of nutraceuticals, complementary and integrative remedies, whereas no reviews have comprehensively assessed the studies of these therapies to manage CIPN for cancer care. This was based on a preliminary search for existing overview of reviews on the topic conducted on the databases (ie, Cochrane Library, CINAHL, PubMed, and PROSPERO) searched on September 21, 2019. Furthermore, SRs may demonstrate varied scope, quality, population size, reporting of outcomes, and heterogeneous effects, making interpretation of the evidence on overall treatment efficacy difficult. Hence, this overview explores evidence on the potential benefits and safety of nonpharmacological interventions in preventing and treating CIPN in cancer patients.
Methods
Protocol and Registration
The protocol of this overview was registered on PROSPERO (CRD42019129145). The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 15 and the Cochrane Collaboration Handbook 16 were followed to undertake this overview of reviews.
Literature Search
A comprehensive literature search of SRs and meta-analysis of randomized controlled trials (RCTs) was performed in the MEDLINE, EMBASE, Cochrane Library, PROSPERO, CNKI, VIP, and Wanfang databases from inception to October 13, 2019. The literature search was composed of the Medical Subject Headings (MeSH) and free-text words for “CIPN,” “systematic review,” and “meta-analysis,” which were implemented for different databases. MEDLINE, EMBASE, and Cochrane Library search strategies are shown in the appendix (supplementary file, available online). The reference lists of all the appraised articles were screened for relevant citations that might have been missed from the electronic searches. There were language restrictions on SRs published with a title and abstract in English or Chinese.
Study Selection
Initially, all duplicates were removed from the references. Two independent researchers (JH and XZ) selected the relevant reviews by screening the titles and abstracts of the identified articles. The full texts of these were then retrieved for further assessment of their potential eligibility. Any disagreements about inclusion were resolved by discussion or consultation with a third assessor if a consensus was not reached (AB).
The inclusion criteria were as follows: (1) SRs and meta/analyses of clinical studies in which at least 1 RCT was included; (2) target population was any type of cancer participants with CIPN (any type of chemotherapy) where the nonpharmacological management was the primary focus of the review; (3) the interventions included were nonpharmacological approaches such as lifestyle interventions, physical therapy, nutritional supplements, and complementary medicine therapies (eg, acupuncture, herbal medicine); (4) comparator(s)/control were pharmacological control or any other forms of control (eg, placebo, no intervention); and (5) the main outcomes reported were neurotoxicity incidence and/or severity measured by standardized and validated clinical assessment tools, including, but not limited to, patient-reported outcomes, clinician-rated neuropathy assessments, and physical/functional measures. The additional outcomes were safety outcomes (eg, adverse events).
The exclusion criteria were as follows: (1) CIPN was assessed as a part of a broader topic; (2) interventions were administered intravenously only; and (3) control comparisons were related to nonpharmacological therapy. If there were duplicate publications, we selected the latest complete version.
Assessment of Quality of Included Reviews
Two reviewers (JH and XZ) independently assessed the methodological quality of the included reviews using the AMSTAR 2 (Assessment of Multiple Systematic Reviews) appraisal tool. 17 Any discrepancies were resolved by consultation with a third reviewer (AB). This checklist can be used to appraise SRs that include RCTs of health care interventions and also those that include nonrandomized studies, or both. It includes 16 domain-specific questions, each referring to a relevant methodological aspect of the study. By assessing the potential impact of an inadequate rating for each item, the reviews were rated by the overall confidence (High/Moderate/Low/Critically low) detected on the number of critical and noncritical items of the review. 17 The quality of evidence for each main and additional outcome across studies was individually determined by 2 assessors as per the GRADE (Grading of Recommendations Assessment, Development, and Evaluation) recommendations. 18 We downgraded the level of evidence if there were: risk of bias, unexplained heterogeneity, indirectness of evidence, imprecision of the pooled estimate, and publication bias. The overall quality of evidence was judged as either high, moderate, low, or very low.
Data Extraction and Analysis
Two reviewers independently extracted information from the reviews and cross-checked the other’s extracted data. Discrepancies were resolved via judgement from a third author. The following data were collected from the included SRs: authors, publication year, databases search, number of included clinical studies and patients, target population, type of chemotherapy, interventions, controls, outcomes and outcome measurements, risk of bias tool, statistical model for data pooling, estimates of effect size, heterogeneity, publication bias, and funding source. Descriptive summaries of the included studies and their methodological quality are displayed in Tables 1 and 2. In Table 3, we extracted the estimates of effect size from meta-analyses, and reported these as relative risk (RR), odds ratio (OR) for dichotomous outcomes, and mean difference (MD) or weighted/standardized MD (WMD/SMD) for continuous outcomes, with the 95% confidence intervals (CIs).
Characteristics of Included Systematic Reviews in the Overview.

Abbreviations: 6MWT, 6-minute walk test; ADL, activities of daily living; CBM, Chinese BioMedical Literature Database; CINAHL, Cumulative Index to Nursing and Allied Health Literature; CIPNAT, chemotherapy-induced peripheral neuropathy assessment tool; CMAP, compound motor action potential; CNKI, China National Knowledge Infrastructure; COM, center of mass; CQVIP, VIP Database for Chinese Technical Periodicals; CR, complete remission and the grade of CIPN reduced to 0 grade and all symptoms disappeared; CSPD, Wanfang Database of China Science Periodical Database; DEB-NTC, Neurotoxicity Criteria of Debiopharm; DGI, dynamic gait index; EC, eyes closed; EO, eyes opened; EORTC-QLQ (CIPN20/C30), European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Chemotherapy-Induced Peripheral Neuropathy scale; EPIC, European Prospective Investigation into Cancer; FACT/GOG-NTx, Functional Assessment of Cancer Therapy/Gynecological Oncology Group–Neurotoxicity; FACT-G/O, Functional Assessment of Cancer Therapy–General/Ovarian Cancer-Specific Scale; FES-I, Falls Efficacy Scale International; HADS, Hospital Anxiety and Depression Scale; ICHUSHI, Japanese Database of Scientific Literature and Abstracts of Scientific Meetings; IST, Incremental Step Test; KISS, Korean Information Service System; KISTI, The Korea Institute of Science and Technology Information; KMbase, Korean studies Medical Database; KoreaMed, Korean Association of Medical Journal Editors; KSI, Korean Studies Information; LANSS, Leeds Assessment of Neuropathic Symptoms and Sign; mCTSIB, modified Clinical Test for Sensory Interaction in Balance; MET, metabolic equivalent; mTNS, Modified Total Neuropathy Score; Nanet, National Assembly Library of Korea; NCI-CTCAE, the National Central Cancer Institute Common Terminology Criteria for Adverse Events; NCS, nerve conduction studies; NCV, nerve conduction velocity; NHMRC, the Australian National Health and Medical Research; NPS, Neuropathy Pain Scale; NRS, Neuropathic Symptoms on Numerical Rating Scale; NSS, Neurological Severity Score; OCEBM, the Oxford Center for Evidence Based Medicine; PNQ, Patient Neurotoxicity Questionnaire; PR, partial remission; QOL, quality of life; RECIST, rate of response to chemotherapy; RISS, Research Information Service System; SED, symptom examination daily; SGA, Subjective Global Assessment; SF-12, Short-Form Health Survey–12; SF-36, 36-Item Short-Form Survey; SNAP, sensory nerve action potential; STROBE, Strengthening the Reporting of Observational Studies in Epidemiology; TNS, Total Neurological Score; TNSc, Clinical Total Neuropathy Score; TNSr, Total Neuropathy Score Reduced; TUG, timed up and go; VAS, Visual Analogue Scale; VO2max, maximal oxygen consumption; VPT, vibration perception threshold.
AMSTAR 2 Tool of Quality Assessment of the Included Systematic Reviews.

Abbreviation: AMSTAR, Assessment of Multiple Systematic Reviews.
AMSTAR 2 critical domains.
GRADE Quality of Evidence Score for Significant Outcomes Reported in the Systematic Reviews Included in the Overview.
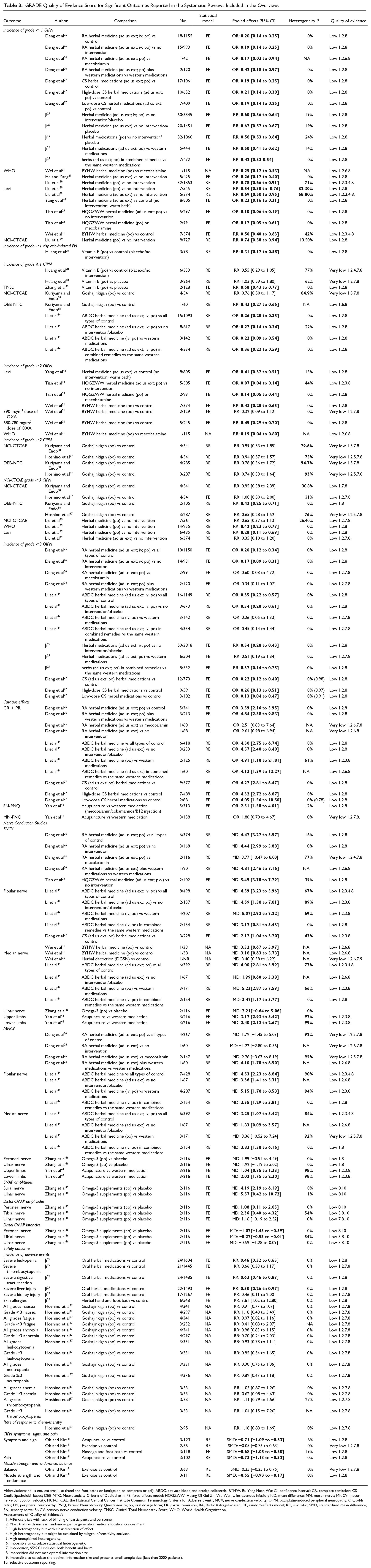
Abbreviations: ad us ext, external use (hand and foot baths or fumigation or compress or gel); ABDC, activate blood and dredge collaterals; BYHW, Bu Yang Huan Wu; CI, confidence interval; CR, complete remission; CS, Caulis Spatholobi–based; DEB-NTC, Neurotoxicity Criteria of Debiopharm; FE, fixed-effects model; HQGZWW, Huang Qi Gui Zhi Wu Wu; iv, intravenous infusion; ND, mean difference; MN, motor nerve; MNCV, motor nerve conduction velocity; NCI-CTCAE, the National Central Cancer Institute Common Terminology Criteria for Adverse Events; NCV, nerve conduction velocity; OIPN, oxaliplatin-induced peripheral neuropathy; OR, odds ratio; PN, peripheral neuropathy; PNQ, Patient Neurotoxicity Questionnaire; po, oral dosage form; PR, partial remission; RA, Radix Astragali–based; RE, random-effects model; RR, risk ratio; SMD, standardized mean difference; SN, sensory nerve; SNCV, sensory nerve conduction velocity; TNSC, Clinical Total Neuropathy Score; WHO, World Health Organization.
Assessments of ‘Quality of Evidence’:
All/most trials with lack of blinding of participants and personnel.
Most trials with unclear random-sequence generation and/or allocation concealment.
High heterogeneity but with clear direction of effect.
High heterogeneity but might be explained by subgroup/sensitivity analyses.
High unexplained heterogeneity.
Impossible to calculate statistical heterogeneity.
Imprecision, 95% CI includes both benefit and harm.
Imprecision did not met optimal information size.
Impossible to calculate the optimal information size and presents small sample size (less than 2000 patients).
Selective outcome reporting.
Results
Literature Search
The literature search and cross-reference search retrieved 378 potentially relevant articles, of which 147 were duplicates. After screening the titles and abstracts, 183 records were excluded. Of the remaining 48 articles that were assessed as full text, 20 were excluded for the following reasons: 1 of them was only a protocol, 6 did not review on CIPN, 6 were administrated intravenously only, 4 were not systematic, and 4 did not include any RCTs as stated in the inclusion criteria. Finally, 28 SRs of RCT on nonpharmacological interventions for CIPN met the inclusion criteria (Figure 1).19-46

Flow chart of the selection of systematic reviews included in the overview.
Characteristics of Included Systematic Reviews
Detailed characteristics of the included 28 SRs are presented in Table 1. The 28 SRs involved the following therapies: herbal medicine (n = 13), natural products and complementary therapies in general (n = 4), acupuncture (n = 3), physical exercise (n = 3), vitamins (n = 3), and omega-3 oral supplements (n = 1). All studies were published between 2013 and 2019, with 16 published in English and 11 published in Chinese and 1 published in Korean. RCTs and quasi-RCTs only were included by 21 reviews; the other 7 reviews also included controlled clinical trials, case studies, and other types of clinical studies. These SRs included a median of 8 trials (range = 2-75), involving a total of 25 719 participants, and each SR contained a median of 647 participants (range = 78-4286). Three authors (10.7%) did not assess the risk of bias of included studies. In contrast, 13 authors (46.4%) used Cochrane collaboration’s RoB assessment tool; 7 (25%) used improved Jadad scale; and 5 (17.9%) evaluated the quality with one each by Jadad scale, Cochrane Collaboration Back Review Criteria, National Health and Medical Research Council clinical evidence assessment matrix, Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) scale, and OCEBM the Oxford Levels of Evidence. Ten studies (35.7%) did not report a source of funding, and the authors in 1 study (3.6%) received research support from industry. The MEDLINE database was the most used electronic source searched by the authors from 27 SRs (96.4%), followed by the Cochrane Register of Controlled Trials (CENTRAL)/Cochrane Library (23; 82.1%), EMBASE (16; 57.1%), CNKI (15; 53.6%), and Wanfang (12; 42.9%). The majority of authors (25; 89.3%) specified the date of literature searching; only 3 authors did not. Thirteen authors (46.4%) searched reference lists for additional studies as supplementary strategies, and another 2 (7.1%) also searched conference proceedings. Ten SRs (35.7%) were focused on oxaliplatin-induced peripheral neuropathy, 11 (39.3%) were reported on mixed chemotherapy regimens, and 7 (25%) were not detailed with the type of chemotherapy involved. Thirteen authors (46.4%) reported on preventive effects of nonpharmacological interventions, 11 (39.3%) reported on treatment efficacy, and the other 4 (14.3%) reported on the both. A variety of outcomes were reported, but mainly with neurotoxicity incidence and/or severity measured by patient-reported outcomes, clinician-rated assessments, and physical/functional measures. Clinician reported scales, such as Levi grade, World Health Organization (WHO) grade, National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE), Neurotoxicity Criteria of Debiopharm (DEB-NTC), and Clinical version of the Total Neuropathy Score (TNSc) were used for the clinical grading of CIPN. Patient-reported CIPN Scales, such as Patient Neurotoxicity Questionnaire (PNQ), were also used. Clinical effectiveness was assessed by symptom remission completely or partially, in accordance with the grading of CIPN reduced at least over 1 grade or down to 0 grade.47,48 Changes in the parameters of the nerve conduction studies were reported. Adverse events were also extracted. Among all the measure tools, nerve conduction studies (13; 46.4%), Levi scale (11; 39.3%), and WHO Neurotoxicity scale (9; 32.1%) ranked the top 3 outcome instruments being applied to included studies.
Methodological Quality of the Included Systematic Reviews
The methodological quality of the reviews is displayed in Table 2, as determined using AMSTAR2 tool. As overall rating of quality was evaluated, only one study scored “high,” over half (16) of the studies scored “critically low,” and another 11 studies scored “low.” Most studies (27/28) showed shortcomings in reporting on the list of excluded studies (Q7), and the funding information of the included studies (Q10). In terms of individual domains of risk of bias, most authors (27/28) explained their selection of the study design in inclusion criteria (Q3). Twenty-three authors described the components of PICO (population, intervention, control, and outcome; 82.1%) in their research questions and inclusion criteria (Q1), but only 2 studies detailed all aspects of the included studies adequately (Q8). Twelve authors (42.9%) established an “a priori” design of review methods from a written protocol/guide (Q2). All studies executed a comprehensive literature search including at least 2 electronic databases, keywords of the search strategy, and publication restrictions. However, they were all rated “partial yes” because the searching did not include the references lists/bibliographies of included studies, trial registries, gray literature, and content from experts in the field (Q4). Twenty-three review authors (82.1%) stated that 2 independent authors determined the eligibility of studies for inclusion in SRs (Q5), of which 22 authors (78.6%) indicated 2 assessors independently performed data extraction (Q6). Twenty-four reviewers (85.7%) assessed the risk of bias for included individual studies, but largely from unconcealed allocation and lack of blinding of patients and assessors (Q9). Half of the authors properly justified combining the data in the meta-analysis (Q11), but only 8 of them (28.6%) assessed the potential impact of risk of bias in individual studies on the results of meta-analysis (Q12). By comparison, 2 studies did not use appropriate methods to conduct the meta-analysis (Q11). Nine SRs (32.1%) did not conduct a meta-analysis because of the heterogeneity in study design and treatments (Q11 and Q12). Twenty-one (75%) studies discussed the likely impact of risk of bias in individual studies on the results (Q13). Twenty-five (89.3%) authors provided a satisfactory explanation for heterogeneity in the results (Q14). Only 6 (21.4%) carried out an adequate investigation on publication bias (Q15). Most studies (26/28) reported on potential sources of conflict of interest (Q16).
Quality of Evidence in the Included Systematic Reviews Assessed by GRADE
On the basis of pooled data from 28 trials, evidence was graded as “low” or “very low” quality using the GRADE approach. The detailed information regarding the reason for downgrading of each outcome is presented in Table 3.
Incidence of Grade ≥1 CIPN/OIPN
A total of 12 studies summarized evidence on this outcome.21,23,26-29,31,38,39,43,44,46 Four studies reported on peripheral neuropathy (PN) caused by various types of chemotherapy, including one on cisplatin-induced neurotoxicity. Eight other studies reported evidence on overall incidence of oxaliplatin-induced peripheral neuropathy (OIPN). Regarding the incidence of grade ≥1 CIPN, Kuriyama and Endo 38 found that it was significantly lower in oral goshajinkigan group when measured by DEB-NTC (RR = 0.43 [0.27-0.66]), but there was no statistical difference in NCI-CTCAE grade. Li et al 44 concluded that this outcome was significantly decreased in all forms (externally:ad us ext; orally: po) of herbal medicine groups (OR = 0.26 [0.20-0.35]). These results were consistent with those of the subgroup analyses when the herbs were compared with no intervention/placebo (OR = 0.22 [0.14-0.34]) or western medications alone (OR = 0.22 [0.09-0.54]) or herbs in combined remedies compared with the same western medications (OR = 0.36 [0.22-0.59]). Huang et al 28 found oral vitamin E supplements resulted in superior effects to placebo/no intervention group (RR = 0.31 [0.17-0.58]) in subgroup grade ≥1 cisplatin-induced PN, but not in CIPN. In contrast, Zhang et al 46 reported that oral vitamin E supplements significantly reduced grade ≥1 TNSc, compared with placebo (RR = 0.58 [0.43-0.77]). In terms of incidence of grade ≥1 OIPN, 3 studies examined the pool effects between all forms (ad us ext; po) of herbal medicine and control. Deng et al 26 investigated evidence on both Radix Astragali–based herbs and Caulis Spatholobi–based herbs for this outcome. Compared with all types of control, they reported a reduced incidence of grade ≥1 neurotoxicity in the Radix Astragali–based herbs group (OR = 0.20 [0.14-0.25]). In the relevant subgroup analyses, these results were consistent when compared with no intervention or mecobalamin alone. Another SR found the neurotoxicity incidence in the Caulis Spatholobi–based herbal group was lower than that of all the control groups (OR = 0.19 [0.14-0.25]), regardless of a low dose (15 g) or high dose (20 g-45 g) of Caulis Spatholobi included in the herbal medicine. 27 Ji 29 reported this outcome in favor of all forms of herbal medicine (ad us ext; po) compared with no intervention or placebo (RR = 0.60 [0.56-0.64]) or western medications (RR = 0.50 [0.41-0.62]); and herbs plus western medications to the same western medications (RR = 0.42 [0.32-0.54]). Two SRs pooled effects in oral and external herbs versus control.23,43 Both studies reported a reduction in the neurotoxicity incidence by the herbal medicine group (Yang et al 43 : OR = 0.23 [0.16-0.31]; Tian et al 23 : OR = 0.10 [0.06-0.19]). Orally applied herbal medicine was described in 3 studies (Ji 29 : RR = 0.58 [0.53-0.64]; Wei et al 31 : RR = 0.25 [0.12-0.53]; Liu et al 39 : RR = 0.78 [0.66-0.91], I2 = 71%; RR = 0.54 [0.38-0.76], I2 = 82.3%; RR = 0.74 [0.58-0.94]) with superior effects with this outcome compared with control groups. Three additional SRs summarized evidence of external use of herbal medicine versus no intervention/placebo control.29,31,39 External herbs had significant benefits (He and Yang 21 : OR = 0.26 [0.17-0.40]; Ji 29 : RR = 0.62 [0.57-0.67]; Liu et al 39 : RR = 0.69 [0.50-0.95], I2 = 68.8%). It should be noted that substantial heterogeneities were found in the meta-analyses from Liu et al’s study, 39 but in the sensitivity analyses, the pooled result of studies remained significant without heterogeneity (I2 = 0%) after omitting 4 studies.49-52
Incidence of Grade ≥2 CIPN/OIPN
Five studies reported evidence on incidence of grade ≥2 neurotoxicity.23,31,37,38,43 Two studies reported on CIPN (evidence of very low quality). Another 3 studies focused on oxaliplatin (evidence of low quality). In terms of the incidence of grade ≥2 OIPN, Tian et al 23 found that this outcome was in favor of Huang Qi Gui Zhi Wu Wu (HQGZWW) herbal medicine when compared with no intervention (OR = 0.07 [0.04-0.14]) or mecobalamine (OR = 0.14 [0.05-0.44]). Yet, there was moderate heterogeneity between HQGZWW (ad us ext; po) and no intervention control (I2 = 44%). Wei et al 31 reported orally used Bu Yang Huan Wu (BYHW) herbal medicine was superior to control by different measures (Levi grade: RR = 0.43 [0.28-0.65]); WHO grade: RR = 0.19 [0.04-0.80]). But these results were only consistent with those of the subgroup analyses for oxaliplatin dose at 680 to 780 mg/m2 group, not for dose at 390 mg/m2 group. Yang et al 43 also reported that the grade ≥2 neurotoxicity was significantly decreased in external herbal medicine group (OR = 0.41 [0.32-0.51]). Compared with control, goshajinkigan had no statistically significant difference in decreasing grade ≥2 CIPN irrespective of using NCI-CTCAE or DEB-NTC grade.37,38
Incidence of Grade ≥3 CIPN/OIPN
A total of 7 studies summarized evidence on this outcome (evidence of low quality).26,27,29,37,38,44 Three studies included 8 comparisons on CIPN. But only 3 comparsions reported a reduction in the neurotoxicity ≥3 grade of herbal medicine group. 38 Kuriyama and Endo 38 found that oral goshajinkigan decreased the incidence of DEB-NTC grade ≥3 CIPN (RR = 0.42 [0.25-0.71]). Liu et al 39 reported that oral herbal medicine was superior to no intervention in grade ≥3 CIPN with WHO (RR = 0.42 [0.23-0.77]) and Levi scale (RR = 0.28 [0.11-0.69]). Four other studies were dedicated to oxaliplatin regimens. Three of the 4 studies reported that this outcome in favor of different types of herbal medicine compared with no intervention (Deng 26 : OR = 0.17 [0.09-0.31]; Ji 29 : RR = 0.34 [0.28-0.43]; Li et al 44 : OR = 0.34 [0.20-0.61]), but not to western medications. Only Ji 29 reported add-on benefit to western medications (RR = 0.32 [0.14-0.75]). Additionally, the grade ≥3 neurotoxicity was significantly decreased in the oral and external use of Caulis Spatholobi–based herbal group (Deng et al: OR = 0.22 [0.12-0.40]), irrespective of high or low dose of Caulis Spatholobi. 39
Curative Effects (Ratio of Complete and Partial Remission)
Four studies reported on curative effects, referring to the integral of complete remission plus partial remission.26,27,42,44 Three herbal studies assessed this outcome with the grading of CIPN and another one acupuncture study with PNQ. Herbal medicines were described in these 3 studies with superior effects on this outcome to the control group (Deng et al: OR = 3.59 [2.16-5.95]; Li et al: OR = 4.30 [2.75-6.74]; Deng et al: OR = 4.27 [2.81-6.47]).26,27,44 In subgroup analyses, compared with no intervention/placebo group, this benefit was only consistently reported in externally applied activating blood and dredging collaterals herbs group (Li et al 44 : OR = 4.57 [2.48-8.40]). Compared with western medication, curative effects were reported significantly improved in Radix Astragali–based herbs (ad us ext; po) plus western medication groups (Deng et al 26 : OR = 4.84 [2.38-9.83]), activating blood and dredging collaterals herbs group (po; OR = 4.91 [1.10-21.81], I2 = 61%) and activating blood and dredging collaterals herbs plus western medications groups (Li et al 44 : OR = 4.13 [1.39-12.27]). Also compared with western medicine, Yan et al 42 reported that acupuncture significantly enhanced the curative effects by PNQ sensory scale (OR = 2.51 [1.58-4.01]).
Sensory Nerve Conduction Velocity (SNCV)
Six studies summarized evidence on this outcome.23,26,27,31,42,44 Three herbal studies reported on SNCV of both median nerve and fibular nerve and 1 acupuncture study of upper limbs and lower limbs. Other 2 herbal studies did not specify either the nerve or the body area for SNCV testing. With herbal medicine, compared with control, both forms of activate blood and dredge collaterals herbal medicine and oral BYHW herbal decoction showed a significant improvement in SNCV of both fibular nerve (Li et al: MD = 4.59 [3.23-5.96]; Wei et al: MD = 3.32 [0.67-5.97]) and median nerve (Li et al: MD = 4.00 [2.81-5.99]; Wei et al: MD = 3.18 [0.63-5.73]).31,44 Caulis Spatholobi–based herbal medicine administered orally or externally had beneficial influences on improving the SNCV of the fibula nerve (Deng et al: MD = 2.12 [1.04-3.20]). 27 However, heterogeneity was significant or not reported in these meta-analysis. Besides that, 2 authors (Tian et al: MD = 5.49 [3.70-7.29]; Deng et al: MD = 4.42 [3.27-5.57]) reported an increase of SNCV in Radix Astragali–based herbal medicine and HQGZWW herbal decoction, administered orally or externally.23,26 One study found that acupuncture enhanced the SNCV of upper limbs (MD = 3.17 [2.9- 3.42]) and lower limbs (MD = 2.40 [2.12-2.67]), but these results were represented with extremely high heterogeneity. 42
Motor Nerve Conduction Velocity (MNCV)
Three studies summarized evidence on this outcome.26,42,44 One herbal study reported on MNCV of both median nerve and fibular nerve and 1 acupuncture study of upper limbs and lower limbs. Another one herbal study did not specify either the nerve or the body area for MNCV testing. Compared with control, all forms of activate blood and dredge collaterals herbal medicine were found with a significant improvement in MNCV of both fibular nerve (MD = 4.53 [2.23-6.84]) and median nerve (MD = 3.25 [1.07-5.42]). 44 With Radix Astragali–based herbal medicine comparisons, only with western medications, RA plus western medications was superior to improving the MNCV (Deng et al: MD = 4.10 [1.70-6.50]). 26 Additionally, Yan et al 42 reported that acupuncture increased MNCV of upper limbs (MD = 1.04 [0.75-1.33]) and lower limbs (MD = 2.02 [1.75-2.30]), compared with western medication. However, heterogeneity was significant or not reported in all these meta-analyses.
Sensory Nerve Action Potential (SNAP) Amplitudes
Zhang et al 46 reported the superior effect of omega-3 polyunsaturated fatty acid (PUFA) oral supplements compared with placebo on SNAP amplitudes of the ural nerve (MD = 4.19 [2.19-6.19]) and ulnar nerve (MD = 5.57 [0.42-10.72]).
Distal Compound Motor Action Potential (CMAP) Amplitudes and Latencies
Zhang et al 46 reported significant differences in distal CMAP amplitudes, favoring the omega-3 group over placebo, in both the peroneal nerve (MD = 1.08 [0.11-2.05]) and tibial nerve (MD = 2.36 [0.40-4.32]). Similarly, omega-3 PUFA oral supplements have shown to better preserve CMAP latencies of the peroneal nerve (MD = −1.02 [−1.45 to −0.59]) and tibial nerve (MD = −0.27 [−0.53 to −0.01]). However, heterogeneities were evident (I2 = 54%) in CMAP amplitudes and latencies of tibial nerve. 46
Safety Outcome
Two studies reported on safety outcomes with regard to incidence of adverse events and hematological toxicities.29,37 One also reported on the rate of response to chemotherapy. Hoshino et al 37 reported that goshajinkigan did not influence the risk of grade ≥1 and ≥3 nausea, fatigue, anorexia, leukocytopenia, neutropenia, anemia, thrombocytopenia, or rate of response to chemotherapy. Ji 29 found that oral herbal medications lowered the incidence of severe leukopenia (RR = 0.46 [0.32-0.65]), severe digestive tract reaction (RR = 0.63 [0.46-0.87]), and severe liver injury (RR = 0.50 [0.26-0.97]), but there was no statistically significant difference in severe thrombocytopenia and severe kidney injury. As well, no significant differences were observed on incidence of skin allergies between herbal hand and foot bath and control. However, the evidence reported was of low-grade quality.
CIPN Symptoms and Signs
One study reported that this outcome measured by a mix of multiple scales. 41 Compared with control group, CIPN symptoms and signs were relieved significantly with acupuncture (SMD = −0.71 [−1.09 to −0.33]) and massage and foot bath (SMD = −0.68 [−1.05 to −0.30]). Acupuncture was also statistically effective in reducing CIPN pain (SMD = −0.73 [−1.13 to −0.32]).
Muscle Strength, Endurance, and Balance
One study reported that exercises were effective in improving muscle strength and endurance (SMD = −0.55 [−0.93 to −0.17]). 41
Discussion
Key Findings From the Overview
Twenty-eight SRs of varied methodological quality including nonpharmacological treatment modalities for the clinical management of CIPN were identified. The strengths and weaknesses of each study were evaluated, and the level of evidence was summarized. We did not set any restrictions on the CIPN diagnoses or cancer types of included SRs. This approach reflects real-world practice and improves the external validity of this overview.
There was some evidence to suggest the superior effects of Chinese herbal medicine on preventing the development of CIPN, but evidence was mainly limited to low-quality trials. Radix Astragali, Caulis Spatholobi–based herbal combination, and the additional use of herbal medicine with active components promoting blood circulation presented superiority in reducing the incidence of grade ≥1 OIPN/CIPN. BYHW and HQGZWW herbal decoction was effective in both grade ≥1 and ≥2 OIPN. In SRs synthesizing evidence on decreasing grade ≥3 OIPN, Caulis Spatholobi–based herbal combination played an active role. Regarding treatment effects, evidence indicated that Radix Astragali–based herbal combination plus western medication, Caulis Spatholobi–based herbal combination and herbs promoting blood circulation action presented curative effects in OIPN. Radix Astragali–based herbal combination, Radix Astragali–based herbal combination plus western medication, Caulis Spatholobi–based herbal combination, BYHW, and HQGZWW herbal decoction had the potential of being more effective in improving sensory nerve conduction velocity. With regard to MNCV, positive results were observed on Radix Astragali–based herbs plus western medications and herbs with promoting blood circulation. In term of safety outcomes, 2 meta-analyses that reported on the incidence of adverse events agreed that herbal medications did not increase this risk.
There was insufficient evidence to make any judgements on the efficacy of vitamin E, omega-3 supplementation, exercise, massage, and foot baths in the treatment of CIPN with low certainty from single meta-analysis. Although evidence from 2 meta-analyses reported that acupuncture has the potential to alleviate CIPN symptoms and enhance nerve conduction velocity, the confidence of it remains low due to poor reporting quality, little details on acupuncture procedures, and how outcomes were measured. In addition, available evidence cannot demonstrate clear/consistent add-on benefits of vitamin E administration for CIPN. Goshajinkigan also did not appear to reduce the risk of CIPN nor did it reduce the severity of CIPN.
In general, results from the identified SRs demonstrated add-on protection and benefit potentials from acupuncture, natural products (including vitamins, omega-3 PUFAs), herbal medicine, and physical exercise for CIPN symptoms control. Seventeen (60.7%) of the 28 reviews in this overview reported a meta-analysis with the topic. A significant proportion of meta-analyses indicated that herbal medicine, regardless of being administered orally or externally, reduced the overall OIPN incidence significantly compared with no intervention, placebo, or pharmaceuticals, thus increased adherence to chemotherapy. Acupuncture, omega-3 supplementation, vitamin E supplementation, massage and foot baths, and exercise showed some positive effects, but their effects were taken with caution as low certainty or less consistent overall and so need further study to be more certain of their effects.
Potentials of Nonpharmacological Interventions
The underlying mechanisms of these nonpharmacological interventions on CIPN have not yet been fully understood. Acupuncture may relieve neuropathic pain by improving central neurotransmission of GABA-ergic, serotoninergic, and adrenergic.53-57 It may also downregulate nerve growth factor signaling with a parallel decrease in sensory neurons hypersensitization. 58 Balance exercises, such as sensorimotor training or whole-body vibration, have shown the highest effect on the crucial side effects of nonmetabolic peripheral neuropathic disorders. 24 It may work through inducing neural adaptations and nerve fibers regeneration, 59 activating deafferented neurons, 60 lowering the excitability threshold, 61 or activating supraspinal learning effects.59,62 In vivo, HQGZWW decoction, a classic Chinese herbal formula, may relieve pain and ameliorate sciatic nerve conduction velocity in rats with CIPN. Its extract AC591 reduced oxaliplatin-induced cold hyperalgesia, mechanical allodynia as well as morphological damage of dorsal root ganglion. 63 Hydroalcoholic Astragali Radix extract (50%) was also found to reduce oxaliplatin-induced cold hypersensitivity, effectively block the onset of the pro-allodynia action, and protect pain induced by neuro damage in rat models.64,65 Potential nerve growth–promoting factors in peripheral nerves regeneration were also found in other vitro and vivo studies with Radix Paeoniae alba extract, puerarin from Pueraria lobata and Tanshinone IIA from Salvia miltiorrhiza.66-68 Ginkgo extract EGb761 was observed to promote faster nerve conduction velocity. This was probably due to its neuroprotective effect on pathological changes of decrease in somatic and nuclear size, nucleolar segregation, and multinucleolation. 69 Explanations of the standardized herbal granule goshajinkigan (Pilula renales plantaginis et achyranthis), well known as Niu Che Shen Qi Wan in China, included its antioxidant and C fiber activation properties for inhibiting the development of CIPN.70-72 In animal models of diabetic neuropathy, it is possible that the action of omega-3 PUFAs lead to beneficial effects on enhancing nerve conduction velocity, 72 and attenuating adverse changes in nerve structure and function.73,74 In other vitro and in vivo experiments, omega-3 PUFAs has been found to reduce neuronal cell death, recover peripheral nerve injury, 75 and stimulate neurite growth in sensory neurons. 76
Strength and Limitations
This is the first overview that summarizes evidence on nonpharmacological therapies for one of the most common side effects of conventional cancer care (CIPN). We comprehensively summarized and critically appraised all available evidence on this topic. Using the GRADE tool, we generally judged the efficacy and safety as being supported by low quality of evidence (and some very low). Thus, further studies are required to enhance confidence in the estimate of effect. The presence of high heterogeneity, small sample size, unclear random allocation concealment, inadequate blinding and follow-up, and selective outcome reporting were identified by authors as possible risk factors for downgrading the level of evidence. Additionally, none of the studies discussed the clinical appropriateness of combining the RCTs prior to choosing a fixed- or random-effects model. Although we attempted to include all key outcomes reporting on CIPN, most included studies from these SRs reported on subjective clinician-rated scales, which may become less optimal when blinding was not well executed. The definitions of PN grades differ between the scales, NCI-CTCAE, WHO, and TNSc scales place focus on the severity of a range of objective neurotoxicity, whereas Levi and DEB-NTC scales place emphasis on the duration of neurotoxicity. Only a small number of SRs included objective neurological assessments and patient-reported questionnaires. Furthermore, reporting safety outcomes (adverse events) of included SRs was unsatisfactory, which may be a result of minimal details provided in the primary studies.
This overview has been designed, conducted, and reported rigorously, but presents with its own limitations. First, the retrieval language limited to SRs with an abstract in English and Chinese can result in a potential risk of positive publication bias. 77 Besides, our evaluation depended on what was reported in the SRs. The authors may have designed and conducted their article more rigorously but removed some key details that we were looking for in their reports. In this instance, the reporting quality of the included article may have had an impact on our results. Future SR should be in accordance with the PRISMA statement. 15 Finally, as the majority of results reporting on herbal medicine therapies came from the Chinese population, the generalizability of current findings is limited. It is worth noting that the Chinese herbs included in SRs, even with the same name of category group, were heterogeneous in treatment duration, formula composition, and doses of individual herbs, likely because one of the features in traditional Chinese medicine is to use the different prescriptions accommodating for different individuals.
Research with the aim of proving the benefits of nonpharmacological interventions has not made much advancement in the CIPN area; however, if a review study fails to include such results, nonpharmacological interventions (eg herbal therapies) would not find acceptance and integration in conventional therapies. Reliable studies are essential to make future research valuable. To provide more rigorous evidence on the effectiveness of nonpharmacological interventions, prudent methods and high reporting standards must be complied within future study design.
Conclusions
This overview compiles the clinical evidence of one of the most common side effects from chemotherapy in a systematic and comprehensive way. It provides a coherent summary of the totality of evidence from SRs of nonpharmacological interventions for CIPN management. This may help busy clinicians, policy makers, and patients. However, the evidence is not sufficiently robust because of the unsatisfactory flaws in both reporting and methodology. Based on the identified evidence, published SRs for CIPN described the potential benefits of nonpharmacological interventions as follows: acupuncture, exercise, and herbal medicine, nutritional supplements. As an adjuvant option to prevent and relieve CIPN, herbal medicine showed promise but required further study to be more certain of their effects. The variability in the use of various intervention methods and outcome measures in different trials was a major challenge in assembling this overview and the individual SRs on which it is based. This made it difficult to pool results and derive conclusions. Readers with a particular interest in one specific intervention would probably look up the relevant SRs and refer to the primary articles.
Supplemental Material
References_to_studies_excluded_in_this_review – Supplemental material for Effects of Nonpharmacological Interventions in Chemotherapy-Induced Peripheral Neuropathy: An Overview of Systematic Reviews and Meta-Analyses
Supplemental material, References_to_studies_excluded_in_this_review for Effects of Nonpharmacological Interventions in Chemotherapy-Induced Peripheral Neuropathy: An Overview of Systematic Reviews and Meta-Analyses by Jie Hao, Xiaoshu Zhu and Alan Bensoussan in Integrative Cancer Therapies
Supplemental Material
Search_strategies – Supplemental material for Effects of Nonpharmacological Interventions in Chemotherapy-Induced Peripheral Neuropathy: An Overview of Systematic Reviews and Meta-Analyses
Supplemental material, Search_strategies for Effects of Nonpharmacological Interventions in Chemotherapy-Induced Peripheral Neuropathy: An Overview of Systematic Reviews and Meta-Analyses by Jie Hao, Xiaoshu Zhu and Alan Bensoussan in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Western Sydney University Research Training Scheme (Grant Number UWS18405473); China Scholarship Council Higher Degree (Doctor of Philosophy) Program (Grant Number CSC201506550006).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
