Abstract
Background:
This systematic review and meta-analysis aimed to determine whether chemotherapy-induced peripheral neuropathy (CIPN) affects the risk of falls and physical function in patients with cancer.
Methods:
A literature search was conducted in the CINAHL, Scopus, and PubMed databases for articles published from January 1950 to April 2022. Seven review authors retrieved studies using predetermined eligibility criteria, extracted the data, and evaluated the quality.
Results:
Nine studies were included in the analysis. Patients with CIPN had a significantly higher risk of falls than those without CIPN (risk ratio = 1.38, 95% confidence interval [CI] =1.18-1.62). Patients with CIPN had lower grip strength (standardized mean difference [SMD] =−0.42, 95% CIs = −0.70 to −0.14, P = .003), longer chair stand time (SMD = 0.56, 95% CIs = −0.01 to 1.17, P = .05), worse timed up and go test time (SMD = 0.79, 95% CIs = 0.41 to 1.17, P < .0001), and lower mean Fullerton Advanced Balance scale score (SMD = −0.81, 95% CIs = −1.27 to −0.36, P = .005) than patients without CIPN. There were no significant differences in gait speed (P = .38) or Activities-specific Balance Confidence Scale score (P = .09) between patients with and without CIPN.
Conclusions:
This systematic review and meta-analysis demonstrated that patients with CIPN are prone to falls and impaired balance function and muscle strength.
Introduction
Chemotherapy-induced peripheral neuropathy (CIPN) is an adverse effect of chemotherapy and is one of the major concerns in patients with cancer.1,2 Patients with CIPN may experience symptoms even after completing chemotherapy and may require several years to recover or may experience permanent neurologic dysfunction. 3 Platinum and taxane-based chemotherapy regimens are likely to lead to CIPN.4,5 The estimated prevalence of CIPN after platinum-based chemotherapy was 58%, 45%, 32%, and 24% at 6, 12, 24, and 36 months, respectively. 6 Furthermore, CIPN persisted in 46.7% of patients 24 months after taxane-based chemotherapy. 7 Risk factors for CIPN include the cumulative dose, increased age, baseline neuropathy, diabetes mellitus, smoking history, decreased creatinine clearance, and obesity.8-12 Various mechanisms underlie the development of CIPN, including impairment of microtubule formation, oxidative stress, mitochondrial dysfunction, neuronal apoptosis, altered ion channel activity, and DNA damage.13,14 Patients with CIPN often present with sensory neuropathy of the hands and feet, including hypesthesia, numbness, and pain.9,15 In addition, patients with CIPN may develop motor and, occasionally, autonomic dysfunction,9,15 which can cause psychological symptoms and affect the quality of life and medical costs.16-18
The sensory and motor nerve changes due to CIPN have been shown to negatively affect physical function, such as muscle strength, 19 gait velocity,20,21 and balance function in patients with cancer.20,22,23 Of major concern is the high risk of falls in patients with CIPN during or after chemotherapy.24-26 Falls among older patients with cancer treated with neurotoxic chemotherapy may have more severe consequences, such as fractures, than falls among older patients without cancer. 27
Previous studies have found that patients with CIPN have an increased risk of falls and deterioration of physical function.19-22,25,26,28 However, other studies have failed to show that CIPN is significantly associated with fall risk.24,29 Similarly, previous studies found no statistically significant association between CIPN and physical dysfunction.30-32 Therefore, these conclusions are controversial, as inconsistent results have been reported regarding the association between CIPN and the risk of falls or physical dysfunction in patients with cancer. Hence, in this systematic review and meta-analysis we aimed to determine whether CIPN affects the risk of falls and physical dysfunction in patients with cancer.
Methods
Protocol
This study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines and used a pre-specified protocol, registered with PROSPERO (CRD: 42022336390).
Search Strategy
We conducted a literature search for articles published between January 1950 and April 2022 in the CINAHL, Scopus, and PubMed databases. The following terms related to “participants” were searched: “cancer,” “tumor,” “neoplasm,” “hematopoietic malignancy,” “lymphoma,” “sarcoma,” “carcinosarcoma,” or “leukemia.” The following terms related to “events” were searched: “chemotherapy induced peripheral neuropathy” or “CIPN.” The following terms related to “outcomes” were searched: “fall,” “gait,” “walk,” “locomotion,” “ambulation,” “muscle,” “balance,” “postural,” “dynamic,” “exercise capacity,” “tolerance,” “physical,” “activity,” “behavior,” “lifestyle,” “quality of life,” “The European Organization for Research and Treatment of Cancer,” or “QLQ-c30.”
Selection Criteria
The inclusion criteria were as follows: (1) observational and cross-sectional studies that included (2) patients of all ages and genders who were (3) diagnosed with any type of cancer, (4) received chemotherapy, and were (5) diagnosed with CIPN, that (6) assessed falls or physical function (i.e., muscle strength, gait ability, balance) and (7) compared outcomes among cancer patients with and without CIPN. The exclusion criteria were as follows: studies with an (1) ineligible design, (2) ineligible patients, (3) ineligible outcomes, or (4) other reasons (e.g., different language, not an original paper). Regarding the study design, systematic reviews, editorials, randomized controlled trials, case reports, and case series were excluded. Regarding the systematic reviews, we checked one by one for inclusion of eligible articles. Regarding languages, we included all languages in the initial search, although we excluded articles in other languages later in the screening process. The different stages of the study selection process are shown in Figure 1. The titles and abstracts of all retrieved articles were screened by 7 independent reviewers to ensure their eligibility. Full-text articles were retrieved for review when there was an indication that they met the inclusion criteria or when there was insufficient information in the abstract and title to make a decision. The corresponding authors were to be contacted to obtain data if necessary; however, this was not required as all the included studies contained complete data. To perform a meta-analysis, detailed data were examined. Final inclusion of eligible observational and cross-sectional studies was determined in consensus meetings, which were attended by all authors.
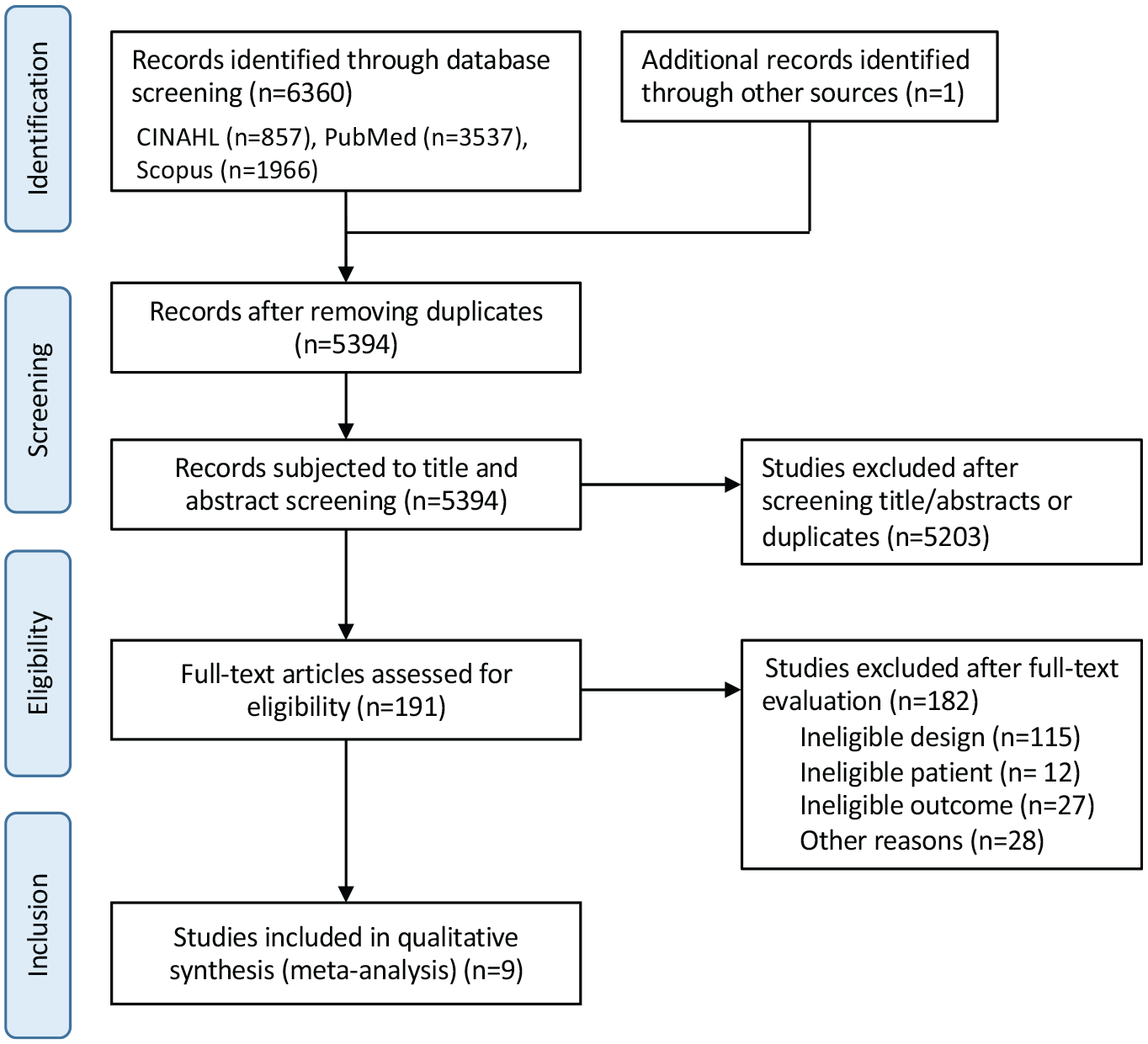
Flow chart of the article selection process.
Data Extraction
Three reviewers were responsible for data extraction. When the data in the full text of an article were deemed insufficient, the authors of the article were contacted by email for additional information. The following data were extracted from each included study: first author’s last name, publication year, nationality, sample size, sex distribution, mean age of the sample, cancer type, and outcomes.
Quality Assessment
The quality assessment of studies, including their risk of bias, was performed using the Newcastle–Ottawa scale. 33 This tool includes the following 8 domains: (1) case definition (the study received 1 point if the case definition was appropriate); (2) representativeness of the cases (the study received 1 point if all eligible cases had appropriately defined outcomes); (3) selection of controls (the study received 1 point if the controls were from the same community); (4) definition of controls (the study received 1 point if the controls had no history of the outcome); (5) comparability of cases on the basis of the design or analysis (the study received 1 point if the design was comparable and adjusted for one of the main elements as a confounder, and 1 point if it was adjusted for additional elements); (6) assessment of exposure (the study received 1 point if there was a record of exposure); (7) same method of ascertainment used for cases and controls (the study received 1 point if the ascertainment of case and control exposures were the same); and (8) non-response rate (the study received 1 point if the non-response rate was the same for cases and controls). Two trained reviewers scored each item according to the criteria established by Wells et al 33 Potential disagreements were resolved during consensus meetings in the presence of all the authors. The total scores were calculated, with higher scores indicating higher quality studies.
Data Analysis
All statistical analyses were conducted using the Review Manager software, version 5.1 (Cochrane Collaboration, London, UK). The influence of CIPN on falls among patients with cancer was estimated using a forest plot of risk ratio (RR) with 95% confidence intervals (CIs), whereas the influence of CIPN on the physical function (gait speed, chair stand test, grip strength, timed up and go test, Activities-specific Balance Confidence Scale, and Fullerton Advanced Balance scale) in patients with cancer was estimated using a forest plot of the standardized mean difference (SMD) with 95% CIs. The random-effects model was used for pooling. We also assessed statistical heterogeneity using the I2 statistic. Furthermore, we adopted the I2 levels suggested by the Cochrane Handbook for Systematic Reviews of Interventions (0%, 25%, 50%, and 75% representing no, low, moderate, and high heterogeneity, respectively). The threshold for interpreting the I2 value can be misleading. Therefore, we determined the importance of the observed I2 value by assessing the magnitude and direction of the effect as well as the strength of evidence for clinical heterogeneity.
Results
Study Selection
The database search yielded 6360 articles, which were reduced to 5394 articles after excluding the duplicates. These 5394 articles were screened for titles and abstracts, resulting in the exclusion of 5203 studies due to irrelevant study design or discrepancies regarding the population or outcomes. A full-text review of 191 articles identified 115 studies with ineligible designs, 12 with ineligible patients, 27 with ineligible outcomes, and 28 with other reasons eligible for exclusion (e.g., different language, not an original paper); therefore, 182 articles were excluded. Finally, 9 studies were found to meet all the inclusion criteria. The flow diagram of the study selection process is presented in Figure 1.
Study Characteristics
The characteristics of the 9 included studies, involving a total of 1834 individuals, are shown in Table 1.29-32,34-38 The publication years ranged from 2014 to 2022. Eight studies were conducted in the USA29-32,34-36,38, one in Germany. 37 The study with the largest sample size of 623 was conducted by Miaskowski et al, 36 whereas the study with the smallest sample size had 17 participants. 31 Two studies included only females,34,38 while the others included patients of both genders.29-32,35-37 Of the 1843 participants, 1684 were females (91.4%), and 159 were males (8.6%). The mean participant age range was 50.75 to 71.1 years. Although pediatric patients with cancer were also included in this study, studies of pediatric patients were excluded because of ineligible study designs and outcomes. All of the studies that ultimately met the inclusion criteria only involved adult patients with cancer. Studies involving multiple cancer types were commonly reported (n = 6),29-31,36-38 followed by breast cancer (n = 1), 34 breast and colorectal cancer (n = 1), 32 and breast and ovarian cancer (n = 1). 35 CIPN was diagnosed using the Numerical Rating Scale in 3 studies,34-36 Common Terminology Criteria for Adverse Events in 2 studies,30,37 Functional Assessment of Cancer Therapy/Gynecologic Oncology Group–Neurotoxicity in 2 studies,31,38 European Organization for Research and Treatment of Cancer Quality of Life Questionnaire-CIPN 20-item scale in one study, 32 and by clinical symptoms documented in the electronic medical record in one study. 29 Six studies controlled for other causes of neuropathy by establishing exclusion criteria such as diabetes mellitus, vitamin deficiency, human immunodeficiency virus, and stroke.29,30,32,35-37 Seven studies reported physical function outcomes30-32,35-38, 3 described falls.29,34,38 Physical function outcomes were evaluated using gait speed (n = 3),31,32,38 chair stand test (n = 2),37,38 leg press test (n = 1), 38 grip strength (n = 3),30,35,36 short physical performance battery (SPPB) score (n = 1), 38 timed up and go test (TUG) (n = 3),35-37 Activities-specific Balance Confidence Scale score (n = 2),31,37 Fullerton Advanced Balance scale score (n = 3),35-37 Chemotherapy-Induced Peripheral Neuropathy Assessment Tool (frequency balance trouble scores) (n = 1),35 six-min walk test (6 MWT) (n = 1), 37 and physical activity (n = 1). 38
Characteristics of the Studies Included in the Systematic Review.

Abbreviations: CIPN, chemotherapy-induced peripheral neuropathy; NR, not reported.
Study Quality
The risk of bias assessment of the selected studies is shown in Table 2. According to the Newcastle–Ottawa scale, 2 studies were considered to be of high quality (8 or 9 points),34,38, 7 studies were deemed to be of moderate quality (6 or 7 points).29-32,35-37
Quality Assessment of the Case Control Studies Included in the Systematic Review Using the Modified Newcastle–Ottawa Scale.

Influence of CIPN on Falls
Three studies involving 889 participants were included in a random-effects meta-analysis of falls. Patients with CIPN were found to have a significantly higher risk of falls than those without CIPN (RR = 1.38, 95% CIs = 1.18 to 1.62, I2 = 0%) (Figure 2).

Risk ratio for falls associated with chemotherapy-induced peripheral neuropathy in patients with cancer.
Influence of CIPN on Physical Function
Six outcomes of physical function (gait speed, chair stand, grip strength, TUG, Activities-specific Balance Confidence Scale, and Fullerton Advanced Balance scale) from 7 studies were included in a random-effects meta-analysis.
Gait
Three studies involving 543 participants compared the gait speeds of CIPN and non-CIPN groups. The meta-analysis showed no statistically significant difference in this parameter between the 2 groups (SMD = −0.25, 95% CIs = −0.81 to 0.31, P = .38, I2 = 48%) (Figure 3A).

Influence of chemotherapy-induced peripheral neuropathy on physical function. (A) Gait speed. (B) Chair stand. (C) Grip strength. (D) Timed up and go test. (E) Activities specific balance confidence scale. (F) Fullerton advanced balance scale.
Muscle strength
Two studies involving 553 participants reported the results of the chair stand test. The results of the meta-analysis suggested that the chair stand time was longer with borderline statistical significance in the CIPN group than in the non-CIPN group (SMD = 0.56, 95% CIs = −0.01 to 1.17, P = .05, I2 = 65%) (Figure 3B). In addition, 2 studies involving 863 participants revealed that the grip strength in the CIPN group was significantly lower than that in the non-CIPN group (SMD = −0.42, 95% CIs = −0.70 to −0.14, P = .003, I2 = 52%) (Figure 3C).
Balance function
Three studies involving 875 participants used the TUG test. The TUG test is simple and can be easily performed to assess a person’s mobility and fall risk, and evaluates both static and dynamic balance. 39 The meta-analysis results suggested that the TUG time was significantly longer in the CIPN group than in the non-CIPN group (SMD = 0.79, 95% CIs = 0.41 to 1.17, P < .0001, I2 = 73%) (Figure 3D). Two studies involving 58 participants demonstrated that the Activities-specific Balance Confidence Scale score was not significantly different between the CIPN and non-CIPN groups (SMD = 0.79, 95% CIs = 0.41 to 1.17, P = .09, I2 = 73%) (Figure 3E). Three studies involving 875 participants used the Fullerton Advanced Balance scale to evaluate participants. The meta-analysis results revealed that the Fullerton Advanced Balance scale score was significantly lower in the CIPN group than in the non-CIPN group (SMD = −0.81, 95% CIs = −1.27 to −0.36, P = .005, I2 = 81%) (Figure 3F).
Other physical function outcomes
Five outcomes of physical function (leg press test, SPPB, frequency balance trouble, 6MWT, physical activity) had insufficient data for a quantitative meta-analysis. A summary of the results showed that the CIPN group had lower SPPB scores, higher frequency balance trouble scores, shorter 6MWT, and lower physical activity, than the non-CIPN group. The leg press test scores were not significantly different between patients with and without CIPN.
Discussion
The present systematic review and meta-analysis showed that CIPN can affect the risk of falls and physical function in patients with cancer. Patients with CIPN had a significantly higher risk of falls than those without CIPN. In addition, the group with CIPN had worse chair stand test results and significantly lower grip strength. Regarding balance function, patients with CIPN had a significantly longer TUG and significantly lower Fullerton Advanced Balance scale score than those without CIPN. However, the Activities-specific Balance Confidence scale score was not significantly different between the 2 groups. Furthermore, gait speed was not significantly different between the CIPN and non-CIPN groups.
Three studies have investigated the relationship between CIPN and falls in patients with cancer. CIPN was significantly associated with the risk of falls in patients with cancer in our meta-analysis. Previous studies have reported that patients with diabetic peripheral neuropathy have a higher risk of falling than those without diabetic peripheral neuropathy. 40 Our meta-analysis yielded similar results, and CIPN may be one of the major factors involved in falls. Falls among patients with CIPN have been attributed to sensory and motor impairments,25,26 muscle weakness, 41 reduced gait ability, 41 and balance dysfunction. 41 Sensory and motor impairments due to CIPN may cause gait and balance problems, ultimately increasing the risk of falls in patients with cancer.
CIPN was also found to affect muscle strength in patients with cancer in this meta-analysis. The chair stand test score reflects the lower extremity muscle strength, 42 while grip strength reflects the overall muscle strength, not only the upper extremity muscle strength. 43 Patients with CIPN may have muscle atrophy in the lower extremities and the entire body. A previous study reported that patients with diabetic polyneuropathy have decreased lower extremity muscle strength compared to patients without diabetic polyneuropathy. 44 Other studies have shown that patients with chronic inflammatory demyelinating polyneuropathy also have decreased upper and lower extremity muscle strength compared to age- and sex-matched healthy controls. 45 Similar findings may be observed in patients with CIPN. However, no significant difference was observed between patients with and without CIPN with respect to the leg press test scores, a measure of lower limb muscle strength. This finding suggests that CIPN tends to occur distally. Therefore, the effect of CIPN on proximal lower extremity muscles is considered less substantial than that on distal muscles. Indeed, the effect size for grip strength was larger than that for the chair stand test in the present study, confirming that CIPN has a greater influence on distal than on proximal muscles. These findings suggest that grip strength (i.e., distal muscle strength) is a better indicator of muscle strength in patients with CIPN than the chair stand test score.
In our systematic review, 4 of the included studies investigated the relationship between CIPN and balance function in patients with cancer. In the meta-analysis, the TUG time was significantly longer in the group with CIPN than in the group without CIPN. In addition, the Fullerton Advanced Balance scale score was also significantly lower in the group with CIPN. However, the Activities-specific Balance Confidence scale score showed no significant difference. Among balance functions, the TUG test assesses mobility, balance, and fall risk, 39 while the Fullerton Advanced Balance scale measures mixed balance ability, including rotation and one-legged standing.46,47 The Activities-specific Balance Confidence Scale measures balance confidence while performing various activities, such as walking around the house, sweeping the floor, or walking through a crowded shopping mall. 48 Thus, although CIPN decreases balance function, it may not affect the balance confidence of patients with cancer.
Given that patients with CIPN demonstrated a higher risk of falls, which was influenced by the gait velocity, we hypothesized that patients with CIPN have a slower gait speed. It has been reported that patients with diabetic peripheral neuropathy have a slower gait speed than those without diabetic peripheral neuropathy. 49 Another study showed that chronic inflammatory demyelinating polyneuropathy patients have a slower gait speed than age- and sex-matched healthy controls. 44 Contrary to our hypothesis, there was no significant difference in the gait speed between the groups with and without CIPN in our study. Although CIPN affects muscle strength and balance function in patients with cancer, CIPN may not affect the gait speed in patients with cancer. Furthermore, in the extracted studies, gait speed was assessed over a short distance. Previous studies have claimed that the calculated gait speed differs depending on the distance of the walking test. 50 Therefore, a short-distance walking test may not be able to reflect the exact influence of CIPN.
This review has several limitations that should be noted. First, the number of included studies was small. Several analyses revealed high heterogeneity. However, owing to the small number of studies that investigated physical function in patients with CIPN, determining the source of heterogeneity was difficult. Furthermore, as we could not perform a stratified analysis by cancer type, treatment type (e.g., taxane, platinum), or evaluation time (i.e., during or after treatment), we cannot conclude that these results are applicable to patients in various treatment settings. Second, the number of databases used in this study was possibly insufficient. Our review and search criteria may have been inadequate, and relevant studies may have been missed. Third, since only one article each reported on the SPPB score, 6MWT, and physical activity, statistical analysis could not be performed when integrating these articles. In patients with cancer, the SPPB score, 6MWT, and physical activity are important outcomes associated with treatment-related complications and mortality.51,52 Therefore, further studies are required to verify the influence of CIPN on SPPB scores, 6MWT, and physical activity. Finally, we conducted a meta-analysis of patients with and without CIPN and the risk of falls and deterioration of physical function in these groups; however, the patient groups in all the studies were different. Therefore, a direct reference cannot be made to the association between falls and physical function in patients with CIPN. However, this study revealed that patients with CIPN are prone to falls and to decreased muscle strength and balance function compared to patients without CIPN. We believe that these findings may be useful for planning rehabilitation programs for patients with cancer after chemotherapy and for instructing them on how to independently exercise at home.
Conclusions
This systematic review and meta-analysis demonstrated that patients with CIPN are prone to falls and impaired balance function and muscle strength. The impaired physical functions may increase the risk of falls in patients with cancer. However, only one article each reported the leg press test, SPPB, frequency balance trouble, 6MWT, and physical activity scores; consequently, statistical analysis could not be performed by integrating the articles. Further research is needed to identify and understand the influence of CIPN on physical function in patients with cancer.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation and data collection and analysis were performed by Katsuyoshi Suzuki, Shinichiro Morishita, Jiro Nakano, Taro Okayama, Junichiro Inoue, Takashi Tanaka, and Takuya Fukushima. The first draft of the manuscript was written by Katsuyoshi Suzuki, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
All data generated or analyzed during this study are included in this published article. The authors have retained complete records of the extracted data in a database and will make this database available to the journal if requested.
