Abstract
Keywords
Introduction
Women with breast cancer (BC) show high prevalence rates of cancer-related fatigue (CRF) with 75% to 94% during adjuvant chemotherapy and radiotherapy. 1 In 16.5% to 34% of BC patients, 2 the fatigue still persists 5 to 10 years after initial diagnosis. 3 A study by Groenvold et al 4 found that lower fatigue predicted longer recurrence-free survival in BC survivors, even after adjusting for prognostic biological risk factors. Given the high prevalence, severity, and chronicity, CRF is considered a major public health concern with high costs for the health care systems, for example, Germany. 5
According to the National Cancer Institute, 6 aerobic training (AT) is the only level 1–recommended therapy to treat CRF with small to moderate effect sizes.7,8 Growing evidence, however, emphasizes the relevance of other evidence-based therapies to lower CRF complaints such as cognitive behavioral therapy (CBT), 9 mindfulness-based stress reduction, 10 Qigong, acupuncture, and multimodal approaches. 11 By combining elements of evidence-based therapies, multimodal approaches have the capacity to achieve stronger effect sizes than AT alone addressing the multidimensional character of CRF alongside with coexisting medical 12 and fatigue-related comorbidities, such as anxiety and depression. 13 Two randomized controlled trials using differently composed 10-week multimodal approaches including home-based practice found mixed results in improving CRF in BC survivors. A mono-centric study by Spahn et al 14 comparing a mind-body multimodal therapy (MT) to a walking exercise (AT) revealed no superiority of multimodal approaches versus AT in reducing CRF, after 10 weeks treatment, and 3 months later. A 3-armed comprehensive cohort CRF study 15 compared MT (psychoeducation, sleep education, art therapy, and eurythmy therapy) with control AT and combination therapy (CT; MT and add-on AT) using a closed testing procedure and propensity scores of a composite fatigue and sleep quality score to avoid α-error inflation. The global null hypothesis that stated superiority of CT and noninferiority of MT versus AT was rejected (degrees of freedom [df] = 2; F = 4.68; P = .0115) and explorative tests found sustainably improved sleep quality/fatigue scores after 10 weeks for the MT group (ΔPC = −0.0369, 95% confidence interval [CI] = −0.0705 to −0.0034; p = 0.0314) and 6 months later for CT and MT versus AT (CT vs AT: ΔPC = −0.0436, 95% CI = −0.0781 to −0.0091; P = .0137; MT vs AT: ΔPC = −0.0538, 95% CI = −0.0910 to −0.0166; P = .0050) with strong standardized effect sizes with Cohen’s d ranging between 0.72 and 1.18 for the MT approaches. In the framework of this study, we confirmatively tested secondary pre hoc hypotheses on the identical cohort regarding the impact of the same MT approaches (MT and CT vs AT) on salutogenetic variables.
Though previous epidemiological studies point to the importance of salutogenetic (eg, sense of coherence, self-regulation) and hygiogenetic factors (eg, autonomic regulation) as predictors for successful cancer and CRF management and improved treatment responsiveness,16-18 there is no study to explore the relevance of salutogenetic variables in the context of multimodal CRF treatment. Traditionally, salutogenetic factors have been studied in association with resilience examining an individual’s reaction to stressors and the ability to cope. 19 Well researched is Antonovsky’s sense of coherence (SOC), 20 a health-engendering personality trait explaining how a person comprehends (“comprehensibility”), manages (“manageability”), and makes sense (“meaningfulness”) of stressful life events (“stressors”) using internal and external resources. 21 Besides SOC, 22 accumulating evidence has found other variables such as perceived self-efficacy, 23 benefit finding, 24 positive affect, 25 and potentially internal coherence 26 to be mediators or prognostic resilience factors for CRF management. An improved understanding of how resilience variables operate independently or in concordance with each other may help refine CRF treatment strategies. 23 The importance of salutogenetic variables (internal coherence and self-regulation) in the context of resilience has not been sufficiently researched.
Internal Coherence operationalized as a reliable and valid self-report measure (Internal Coherence Scale [ICS]) shows moderate correlations to SOC (Trapp 2014). It captures an attitude referring to the ability to activate internal resources (eg, emotional adaption) to cope with the disease and achieve physical comfort (thermo-coherence). The ICS proved to be specifically relevant for patients with cancer and other chronic conditions. 27 Self-regulation Scale (SRS) as a self-report measure describes the ability to actively achieve well-being by adapting own behavior(s) to reach intended goals. 28 Both variables characterize the adaptive capacity of a person to engage in health promoting attitudes and behaviors. SRS and ICS have not been sufficiently explored in the context of multimodal CRF treatment; thus, there is evidence they may be relevant for successful fatigue management. A pilot study found that high baseline measures of SRS and ICS in metastasis-free BC survivors were positively correlated with the responsiveness to a multimodal CRF therapy after treatment. 26 Additionally, high SRS was found to predict less depression in epidemiological studies in BC patients.16,28
The aim of this comprehensive cohort design study 15 was to investigate secondary pre hoc hypotheses and test whether MT and CT (compared with AT) are more effective to improve salutogenetic variables (SRS and ICS) in BC patients with CRF. Additionally, the relevance of both salutogenetic variables in association with CRF management was examined. Patients’ satisfaction and qualitative feedback regarding the therapies were evaluated to further refine CRF treatment strategies.
Methods
Ethics and Framework of the Study
We conducted a mixed-method, 3-center, prospective, open-label pragmatic trial for metastasis-free BC patients with CRF, from June 2011 to December 2013. After a detailed study explanation, patients received information about the study and all 3 study interventions and signed informed consent. The patients were allocated by either preference or block randomization using balanced randomization lists (eg, group proportion of 1:1:1) with varying permutation block sizes for each center. Randomized allocation was conducted by a central randomization service at the Institute for Clinical Research, Berlin.
The study was conducted according to the Declaration of Helsinki Guidelines and approved by the local ethics committees (Ärztekammer Berlin: Eth-06/11) of the 3 centers. The study was subject to GCP-conform on-site monitoring and registered in the German Clinical trial register (DRKS-ID: DRKS00003736).
Participants, Inclusion, and Exclusion Criteria
A total of 126 patients with metastasis-free BC and CRF were included in the study, 105 started the intervention, 84 finished treatment, and 81 completed follow-up assessments (see Figure 1). Sociodemographic description for patients is outlined in Table 1. Inclusion and exclusion criteria are summarized in Table 2.
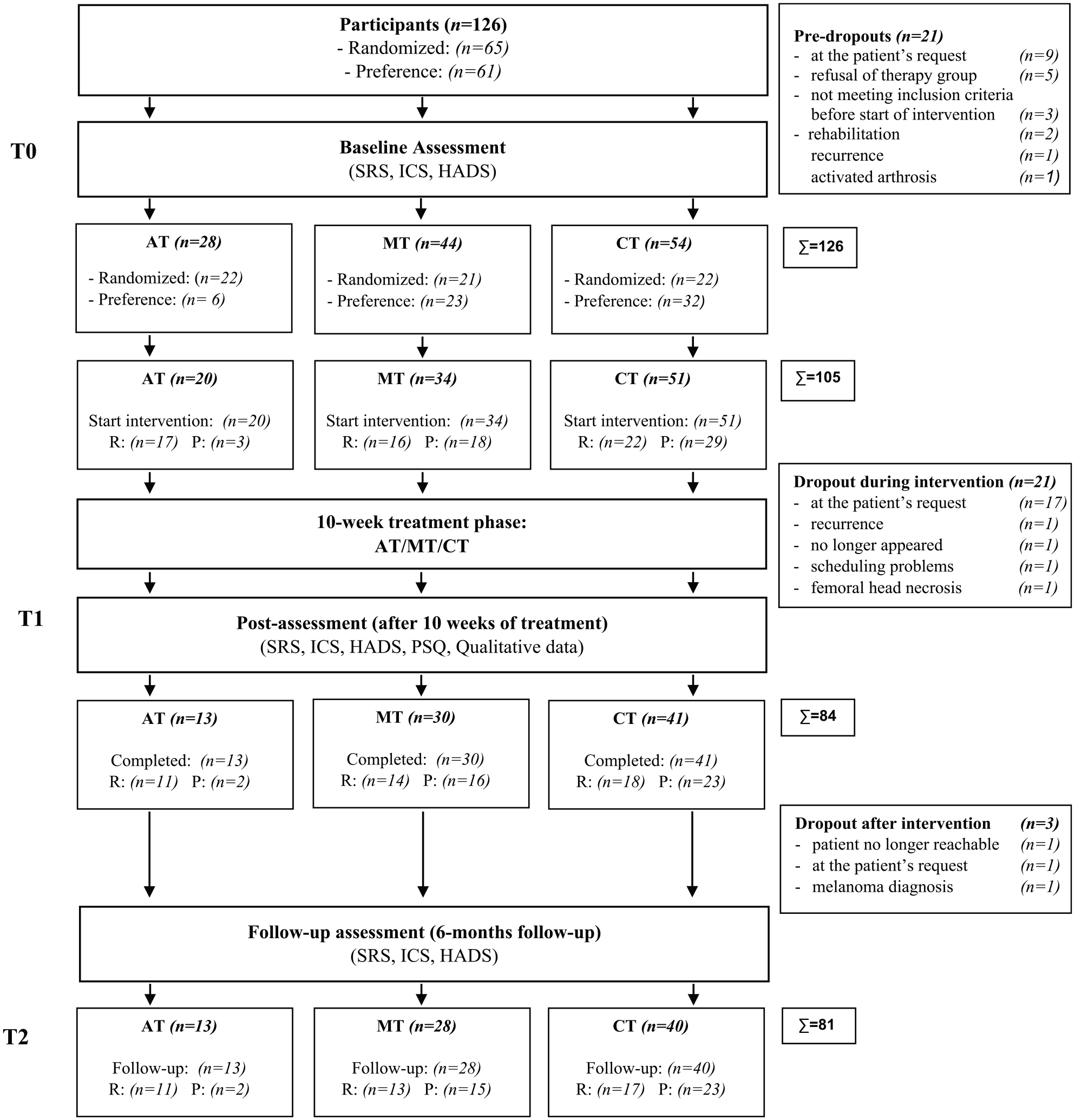
Flow chart for recruitment.
Study Group Characteristics at Baseline (T0).

Inclusion and Exclusion Criteria.

Abbreviations: CRF, cancer-related fatigue; NYHA, the New York Heart Association; COPD, chronic obstructive pulmonary disease; TSH, thyroid stimulating hormone.
Patient-Reported Questionnaires
Patients of the study completed questionnaires at baseline, after 10 weeks of treatment (T1) and 6 months later (T2). Patients’ satisfaction with the treatment was assessed alongside qualitative feedback for T1.
The Self-Regulation Scale
The Self-regulation Scale (SRS) by Büssing et al 29 is a short 16-item adaption of Grossarth-Maticek’s Self-Regulation Scale. 30 It measures self-regulation on a 6-point Likert-type scale via 2 subscales: (1) Change of Behavior to achieve goal and (2) Achieve Satisfaction and Well-being that form the sum score. The SRS is a valid and reliable self-report instrument with robust internal consistency (Cronbach’s α of r = 0.95) and satisfying to good retest reliability (rtt = 0.82) assessed in BC survivors with CRF. 29
The Internal Coherence Scale
The ICS is a self-report questionnaire with 10 items based on a 5-point Likert-type scale. 27 It contains 2 subscales: (1) Inner Coherence and Resilience with 8 items and (2) Thermal Coherence with 2 items. Higher scores indicated higher ICS with values between 10 and 50. The ICS is a highly reliable with Cronbach’s α of r = 0.91 and sound test-retest reliability (after 4-8 weeks) with rtt = 0.80. 27 It displays sufficient validity and correlates moderately with SOC31,32 and aspects of self-regulation. 27
The Cancer Fatigue Scale (CFS-D)
The Cancer Fatigue Scale is a 15-item scale to assess cancer fatigue via 3 subscales (physical, cognitive, and affective fatigue) on a 5-point Likert-type scale (0-4). Its global score ranges from 0 (no fatigue) to 60 (maximum fatigue). The CFS-D classifies values ≥30 as severe fatigue, CFS-D-scores ≥24 as moderate, and values ≤23 as minor fatigue. The CFS-D is a highly reliable and valid self-report measures with robust internal consistency (Cronbach’s α of r = 0.94) and test-retest reliability (rtt = 0.82). 33
The Hospital Anxiety and Depression Scale (HADS)
The German version of the HADS is a widely used self-report measure assessing psychological distress in medical practice and contains 14-item Likert-type items ranging from 0 to 3. 34 The HADS is a valid and reliable scale with 2 subscales. One for depression with 7 items and one for anxiety with 7 items. Both subscales range from 0 to 21 with cutoff for probable cases of ≥11. The HADS shows good reliability and is a valid instrument used in many studies with oncologic patients. 35
Patient Satisfaction Questionnaire (PSQ)
The Patient Satisfaction Questionnaire is a self-report measure with 8 items. Patients indicate their overall satisfaction with the treatment (1 item) and its treatment components (5 items) on a 5-point Likert-type scale from “very satisfied = 1” to “not satisfied at all = 5.” Two additional open-format items are included, as follows: Item 7—“Would you recommend our treatment to others. If yes/no why?” and Item 8—“Please share your ideas of how to improve our treatment?” 36
Treatment
MT comprised psychoeducation, sleep education, eurythmy therapy, and anthroposophic painting therapy. CT additionally included AT (group-based walking exercise). Stand-alone AT was used as control intervention. All study interventions were designed as manualized treatments over a period of 10 weeks, varying between 360 minutes for trainer-guided AT including home-based AT (average 223 per week), 1450 minutes for the MT group, and 1810 minutes for the CT group. Debriefing and homework were included in the time frame of the study. Psychoeducation (behavioral alteration) comprised 8 sessions to improve patients’ self-competence to cope with CRF-related symptoms including stress management, social competence, communication, relaxation, cognitive restructuring, emotional coping, and in vivo exercises to improve self-responsibility. Sleep Education (rhythm alteration) with 2 sessions included individualized information on sleep and chronobiologic regulation. Eurythmy as mindfulness therapy in 10 sessions included speech articulation and movement exercises, and Painting Therapy comprised 10 sessions including spontaneous paintings in sessions 1 and 10. In session 2, 9 patients were instructed to create paintings with day and night motifs using the dynamic form drawing technique, progressing from the dark to the bright color spectrum. Both treatments were integrated as mindfulness components to improve patients’ relaxation response 37 with nonverbal ways of self-expressions (relaxation alteration). Aerobic Therapy comprised 8 sessions up to 45 minutes of trainer-guided group-based walking to improve physical endurance and performance. Aerobic heart rates (70% to 80% of maximum heart rate) were monitored with a pulse watch.
Statistical Analyses
Statistical Procedure
Baseline characteristics were compared between treatment arms with the Cochran-Mantel-Haenszel χ2-test or van Elteren test for categorical or ordinal/continuous data. A general linear model with propensity score (PS) tested for differences in SRS, ICS, or HADS between MT and CT versus AT after 10 weeks of treatment (T1) and 6 months later (T2). Group differences of changes from pretreatment to posttreatment (T1-Baseline) or 6 months later (T2-Baseline) were displayed as means and standard deviations with associated P values. Posttreatment changes from baseline were calculated for all parameters. All analyses were based on the intention-to-treat principle including all patients with valid screening/baseline measurements. Missing items were substituted according to their respective manuals. For the primary effectiveness analysis missing data at T1 were imputed by last-value-carried-forward as provided for in the protocol. Sensitivity analyses were done using worst case and multiple imputations of missing data, omitting PS from the model, and using PS values derived from various models. All tests were carried out 2-sided at an α error level of α = 5% and with corresponding 95% CIs. Randomization lists, sample size estimates, and statistical analyses were produced using SAS version 9.1 (SAS Institute). A Pearson’s correlation analysis was calculated for all variables at baseline, after 10 weeks and 6 months later. For detailed information on adverse events, please see Kröz et al. 15 Qualitative feedback was assessed by 2 open-format items of a patient satisfaction questionnaire, considered and analyzed in relative and absolute frequencies.
Qualitative Analyses
We used the “open coding” method from “Grounded Theory” for our content analysis. 38 The original data of 2 open-format items (“Would you recommend our treatment to others. If yes/no why?”; “Please share your ideas of how to improve our treatment”) of the Patient Satisfaction Questionnaire was considered inductively with an open coding procedure. The first researcher (psychologist) inspected the written feedback of all patients and used “open coding” to group words and phrases into labeled categories. Memos were kept to assess and facilitate associations between categories. In the second step, a group of researchers (1 statistician and 2 medical doctors) reassessed each written statements and assigned them to one of the categories. Unclear statements were rediscussed. Interrater reliability between raters was not examined. In the last step, absolute and relative frequencies were calculated for each category.
Results
Study Group Characteristics at Baseline
No significant differences at baseline were found between the 3 groups concerning mean age, sociodemographic variables, time since first diagnosis, tumor biology, stage, and treatment (see Table 1). No significant group differences between AT, MT, and CT were found for all parameters except for the HADS anxiety subscale: MT: mean = 7.3 (SD = 3.1); CT: mean = 9.3 (SD = 3.5); AT: mean = 9.4 (SD = 4.3), with P = .024. All baseline means and standard deviations and P values are displayed in Table 3.
P Values for Test for Superiority of MT and CT Versus AT a .

Abbreviations: MT, multimodal therapy; CT, combined therapy; AT, aerobic training; SRS, Self-Regulation Scale; ICS, Internal Coherence Scale; HADS, Hospital Anxiety and Depression Scale.
Baseline mean (SD), T1-Baseline, T2-Baseline.
P < .05. **P < .01; significant P values in boldface.
Treatment Effects for Intervention Groups at T1 and T2
Group differences from pretreatment to posttreatment (T1-Baseline) and 6 months later (T2-Baseline) for SRS, ICS, HADS (all with subscales), and patients’ satisfaction only for T1 are displayed in P values compared with AT, with associated means and standard deviations (Table 3). ICS and SRS (total scores) are displayed in Figures 2 and 3.

Means and standard errors for the Internal Coherence Scale (sum score) for all groups (aerobic training [AT], multimodal therapy [MT], combined therapy [CT]) at baseline (T0), after 10 weeks (T1) and after 6 months (T2),

Means and standard errors for the Self-Regulation Scale (sum score) for all groups (aerobic training [AT], multimodal therapy [MT], combined therapy [CT]) at baseline (T0), after 10 weeks (T1) and after 6 months (T2),
Correlations at Baseline, T1 and T2
Significant positive moderate to strong Pearson’s correlations (r = 0.50 to r = 0.93; mean r = 0.68) between the SRS, the ICS, and the HADS (all total scores and subscales) were detected. At baseline, the strongest correlation was found between the SRS total score and the ICS total score with r = 0.75. Significant negative moderate to strong correlations between SRS (total score) and CFS-D (total score) were found, ranging from r = −0.36 at baseline to r = −0.62 at T1 and r = 0.63 for T2. Moderate to strong correlations were detected between the ICS total score and CFS-D total score, with r = −0.47 at baseline, r = −0.59 at T1 and the strongest correlation with r = −0.70 for T2 with a mean correlation coefficient of r = −0.60.
Qualitative Treatment Effects at T1
Seven categories were extracted by content analysis: well-being, self-regulation, resources, inner peace, sense of coherence, physical effects, and social support. Sixty-eight patients reported positive effects in the 7 categories: well-being (64.8%), self-regulation (42.6%), resources (42.6%), inner peace (40.7%), sense of coherence (33.3%), physical effects (29.6%), and social support (3.7%) of n = 139 responses. Descriptive results reveal n = 68 (81.9%) of patients recommended their therapy. Eurythmy therapy was specified most frequently with 76.6% of n = 197 responses.
Discussion
Our comprehensive mixed-method cohort study for non-metastasized BC survivors with CRF is, to our knowledge, the first to examine the effectiveness of MT and CT versus control AT on 2 salutogenetic variables. Study results indicate that MT was superior to AT to improve SRS after treatment and ICS 6 months later. CT was superior to AT to improve SRS at both endpoints. Patients’ satisfaction was superior for patients in the MT and CT arms. Additionally, qualitative patients’ feedback underpins the positive effects of the multimodal therapies. Distress did not improve significantly for the MT and CT compared with AT. Patients with a major depressive episode (Visual Analogue Scale >60) 39 were excluded from the study to clearly separate fatigue from major depression and prevent confounding effects that may lower the treatment outcome.
Several implications can be drawn from our study results. First, our multimodal therapies were designed as alternatives to stand-alone AT to improve CRF, but have also been found to additionally affect salutogenetic variables and increase treatment responsiveness for this patient group. Our treatment components seem to be well suited to affect CRF symptom scores and salutogenetic variables relevant for fatigue management. Second, we found that both variables were not static, but manipulable variables responding differently to the treatments. Whereas SRS can be influenced more promptly, ICS showed a delayed treatment response. Screening for SRS and ICS variables at baseline may help refine treatment strategies for specific subgroups (eg, high vs low scores), bearing in mind that previous research points to the relevance of both variables as prognostic factors for fatigue management. 26 We did not test the predictive capacity of both variables for CRF management, but Pearson’s correlations validated CRF associations and found strong increasing correlations for both endpoints between SRS, ICS, and CRF.
Third, we hypothesize on certain characteristics of both variables. In contrast to SOC (theorized to be a more or less static trait across the life span, naturally improving with age for young healthy adults 22 and important for long-term survival and morbidity prognosis 18 ), ICS may be a more pragmatic resilience marker than SOC, to examine inner coherence and resilience in clinical routine care. Additionally, ICS has a more coherent and replicable factor structure than SOC and reveals high sensitivity to change. 27 A recent study found that BC patients with an indication for chemotherapy who display higher ICS scores were more compliant in following recommended treatment strategies compared with patients with low ICS scores who reject recommended chemotherapy. 40 Additionally, SRS and ICS may be markers for adaptability, whereas high baseline scores indicate more effective symptom management. SRS refers to the adaptive capacity to change behavioral to achieve intended goals, ICS describes a more general life attitude similar to SOC but with more relevance for clinical practice. Other studies confirmed that improvements of SRS had a prospective impact on morbidity and life extension in cancer patients. 41 Both variables were improved in patients by the multimodal therapies and have stabilized over time.
Clinical Implications
There are several clinical implications deriving from the study results. By design, multimodal therapies are eclectic and comprise elements of various evidence-based body-mind interventions and add a group setting to the treatment portfolio. As mentioned multimodal therapies exhibit the capacity to affect potential salutogenetic variables (self-regulation and ICS) and patients’ satisfaction that separately contributes to improved fatigue management. 42 Given the strong effect sizes for the multimodal therapies in our previously published article 15 and their positive impact on self-regulation and internal coherence, they may be a considerable alternative to stand-alone AT.
Our qualitative data underscore the positive effects of a group-based eclectic treatment model for CRF. Similar to CBT group therapy 43 and social skills training 44 for other patient groups (eg, depression, social anxiety), studies revealed that patients with CRF seem to experience beneficial effects from a group-based setting too.45,46 We found that to “interconnect patients” who go through the same difficulties and share similar experiences helped build social support structures, reduces feelings of loneliness, and engage patients in authentic communication including self-disclosure statements (“I have benefited from the group support strongly”; “The group helped me to learn that others go through the same thing and I am not alone”; “I could open up and speak my truth”).
Published literature also suggests that it is crucial “to give patients a voice” by allowing critical feedback regarding the therapy and involving them in the decision-making process on which treatment they would receive (randomization vs self-preference). 47 We found to “offer patients a toolbox” is important to build on patients’ skill set to manage CRF by cognitive, emotional, and behavioral alterations (CBT-based psychoeducation), rhythm alteration (sleep education and stimulus control), and practicing at home. Our patients additionally reported they were able to “reconnect to own resources” (“My life improved generally by treatment”; “I discovered new possibilities to manage the fatigue”; “Treatment helped to regain my daily structure”; “I have more self-esteem”; “I have started gardening again”), which improved the general well-being and life-quality.
Two mind-body interventions (eurythmy and painting therapy) were included to improve “relaxation response” with promising effects, as patients stated: “My fatigue improved and I have more energy”; “I feel more alert and can concentrate better”; “I feel more relaxed”; “I feel inner peace.” Additionally, eurythmy and painting therapy were rated as most beneficial by our patients. Their positive effects on fatigue levels have been proven in other studies 48,49.
Study Limitations
There are limitations to our study. Due to the small sample size including only metastasis-free BC survivors with CRF, the scope of generalization is limited. A control group of metastasized patients with CRF would have elucidated whether positive treatment effects on salutogenetic variables are mainly a result of a better overall prognosis for this patient group. We used a mixture of allocation and self-preference assignment (randomized participations were equally distributed across treatment groups; see Figure 1), and ensured data quality with propensity scores to reduce confounding effects in our observational data. 50
Missing data were last-value-carried forward replacements that could contain biases. Another limitation is the high dropout rate in the AT arm. However, overall dropout rate with 20% was comparable to other studies using AT. Another limitation is that treatment arms varied strongly in length with AT being the shortest treatment. This could have resulted in lower experienced social support that has found to be a prognostic factor for improved fatigue. 51 Other limitations concern time and cost intensity of multimodal therapies compared with AT and the qualitative data analysis. Interrater reliability was not examined between raters and categories were formed without a special content-analysis software.
Study Strengths and Conclusions
We also want to report on strengths of the study. The study is the first comprehensive mixed-method cohort design study to investigate the explorative effectiveness of 2 multimodal therapies compared with stand-alone AT, to improve salutogenetic variables of adaptability. As mentioned, the original study found an explorative superiority for the multimodal therapies (compared with AT) to improve a composite score of CFS-D/sleep quality, after 10 weeks (MT) and 6 months later (MT + CT). 15 Our eclectic manualized treatments (MT and CT) exhibit high relevance for patients displaying higher patients’ satisfaction scores and more positive qualitative feedback in the multimodal arms. SRS and ICS seem to be important manipulable variables that show strong associations with CRF, improving and stabilizing over time. Our results suggest to examine SRS and ICS in future studies to refine knowledge of the underlying variables contributing to improved CRF management. To broaden understanding of CRF and CRF management by examining how salutogenetic variables operate separately and in concordance with each other can help refine treatment. Qualitative feedback may be included to complement quantitative data analysis and elucidate that treatment elements improve quality of care for this particular patient group. To screen CRF patients at baseline for self-regulation and ICS can provide important information about who responds well to which treatment. Further research should investigate the prognostic relevance of high versus low SRS and ICS for successful CRF management and its long-term relevance for BC survivors with CRF.
Footnotes
Acknowledgements
The authors thank the CRF-2 study group collaborators: Fadmire ten Brink and Anette Zander for study coordination and documentation; Matthias Girke and Christian Bartsch for their contribution in the conceptualization; Annette Weninger for documentation; Andreas Nikolaou, Augustina Glinz, and Nina Klara for recruitment and documentation; Mahmud Naghavi, Peter Klug, Andrea Küssmann, and Petra Wundram for exercise training; Michaele Quetz, Sarah Zastrutzki, Anne Kristin Meyer, Judith Schmotz, Martina Jackmuth, and Suzanne Mehrtens for psychoeducation; Sarah Zastrutzki and Christian Heckmann for sleep-education; Barbara Trapp, Ursula Heusser, Frederike Rettigand, and Elisabeth Rieger for eurythmy therapy; Astrid Didwiszus, Beatrice Gelin-Kröz, Karen Baumhöver-Wegener, Birgit Lindemann, and Sonja Steffens for painting therapy. We thank Ramona Beutke for the monitoring and all other scientists and coworkers of the CRF study group for their dedicated work.
Author’s Note
Matthias Kröz is also affiliated with Arlesheim Hospital, Arlesheim, Switzerland.
Author Contributions
Conception and design of the study: MK, CG, RZ, MR, AB, Bettina B; Development of the intervention program: DF, MK, MR, RZ, CG; Collection and assembly of data: MK, Benno B, RZ, DP; Data analysis and interpretation: MR, MK, AM, RZ, AB, CG, DP; Manuscript writing: AM, MK, MR. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The primary study was funded by Mahle Stiftung, Stuttgart, Germany, Christophorus Stiftung, Stuttgart, Germany, Dr Hauschka Stiftung, Bad Boll/Eckwälden, Germany, Stiftung Helixor, Rosenfeld, Germany, and Gyllenberg Foundation, Helsinki, Finland. The study sponsors had no involvement in the study design, collection, analysis or interpretation of data. MK, AM, and RZ received financial support from Software AG Foundation, Darmstadt, Germany, and Humanus Institute, Berlin, Germany,
Ethical Approval
The study was positively approved by the Ethics Committee of the “Ärztekammer Berlin” (Reference Number: ETH-06/11, May 5, 2011) and confirmed by the other local ethics committees of the Hannover Medical School (Reference Number: 1119-2011, June 27, 2011) and of the University Witten/Herdecke (Reference Number: 125/2011, October 25, 2011). The study follows the guidelines for clinical trials (Declaration of Helsinki, ICH-GCP).
Informed Consent
All included patients signed a written consent.
Trial Registration
The study was registered in the German Clinical Trial Register (DRKS-ID: DRKS00003736, date of registration: June 19, 2012). https://www.drks.de/drks_web/navigate.do?navigationId=trial.HTML&TRIAL_ID=DRKS00003736.
