Abstract
Hypothese:
Cancer-related fatigue (CRF) and sleep disorders are some of the most wearing and common symptoms in disease-free breast cancer patients (BC). Aerobic training (AT) is the treatment with the best available evidence, even though it seems to be insufficient with regards to improvements in cognitive fatigue. We introduced a new multimodal therapy concept (MM) consisting of psycho-, sleep-education and new approaches based on anthroposophic medicine such as eurythmy and painting therapy.
Study design:
This pilot study will test the implementation of MM and yield first results of the MM and AE in our centres.
Methods:
31 out of 34 patients suffering from BC and CRF were fully assessed in a ten-week intervention study. 21 patients chose MM and 10 decided on AT. CRF was measured with the help of the Cancer Fatigue Scale (CFS-D), and the global quality of sleep was measured with the Pittsburgh Sleep Quality Index (PSQI). We also captured autonomic regulation (aR) and patients’ satisfaction with questionnaires. Statistical analysis was done with SAS 9.1.3 for windows.
Results:
The new MM therapy can be implemented with high satisfaction among patients. Significant improvements were found in the MM group with regards to CFS-D, global quality of sleep, sleep efficiency (PSQI), aR and rest/activity regulation compared to baseline (all p<0.05). In the AT group aR orthostatic-circulatory and rest/activity regulation improved significantly (p<0.05), too. However, no improvement in cognitive fatigue was seen in either group.
Conclusion:
The multimodal therapy concept was feasible and improved cancer fatigue, sleep quality, autonomic and rest-/activity regulation in breast cancer patients. It may therefore constitute a valuable treatment option in addition to aerobic training for BC patients with CRF. A further study with larger sample size needs to be carried out to assess the efficacy of combined multimodal-aerobic therapy.
Keywords
Introduction
Within the past decade, there has been an increasing amount of research into cancer-related fatigue (CRF) because it is one of the most wearying and insufficiently treated symptoms among oncological patients. 1 In breast cancer patients in particular it is of outstanding importance as it is among the most pronounced symptoms affecting health-related quality of life in patients with metastasized 2 and primary tumours. 3 Apart from insomnia, CRF is the most common symptom in recurrence-free breast cancer patients 1 to 3 years after initial diagnosis, with a mean symptom score for CRF of 37.5% and for insomnia of 45.9% in the EORTC QLQ (European Organization for Research and Treatment quality of life questionnaires) symptom scales, with 80% and 72%, respectively, of all patients reporting such symptoms. 4 CRF is viewed as a complex phenomenon that includes physical, affective, and cognitive or mental fatigue and is adequately measured by multidimensional questionnaires. 5 Particularly, cognitive impairments—often following adjuvant chemotherapy—which have been called “chemobrain” on the basis of cross-sectional studies are detectable as neurocognitive deficits in learning, memory, concentration, slower processing speed, and slower executive functions, 6 that is, with a posttreatment frequency of 9% to 75%.7,8 Prospective studies rendered evidence that these cognitive dysfunctions exist partially prior to chemotherapy and are influenced, in addition to chemotherapy, radiotherapy, hormone therapy, and operation, by fatigue, anxiety, depression, predisposition, 9 and sleep disturbances. 6 They should therefore be renamed as “cancer- or cancer treatment–associated cognitive changes.” 9 Even if there are at best weak correlations between physiological neurocognitive tests and self-assessed cognitive fatigue and CRF, there is a clear need for “more practical and viable” adequate self-report measures that capture the underlying distinct dysfunctions such as in the form of a multidimensional cancer-related fatigue questionnaire. 6
The best available evidence for therapeutic effects on CRF in breast cancer patients exists for aerobic training, followed by sleep hygiene, sleep restriction, stimulus control, behaviour treatment,10,11 and other modes of cognitive behaviour therapy. 12 Pharmacological therapies (apart from transfusion in the case of anemia) such as the use of erythropoietin are controversial because of a 17% elevated mortality risk. 13 This also applies to the psychostimulant methylphenidate showing a lack of improvement in a randomized controlled trial (RCT) 14 as well as Modafinil improving CRF in one open label study only. 15 Surprisingly, concomitant therapy of mistletoe extracts with chemotherapy appears to render growing evidence for control or reduction of CRF, particularly in the case of breast cancer patients, which is supported by 4 RCT studies, although none of these studies specifically had used CRF as primary outcome instrument. 16 It therefore has to be stated that all currently accepted therapeutic approaches are ineffective in influencing cognitive dysfunctions and cognitive fatigue, including aerobic training, sleep restriction, and stimulus control. 6 A substantial component of classical psychophysiological insomnia therapy is “relaxation.” 17 For this very reason innovative approaches focusing on reduction of cancer fatigue applying relaxation 18 or meditative methods inducing a reduction of distress and an improvement in coping strategies with possible controlling influence on sleep disturbances are being considered. This includes the method of mindfulness based stress reduction which improves the relaxation response the Carlson and Garland 19 which could even “play a mediating role in improving subjective cognitive functions” although there is presently no actual evidence based on RCT. 20 In the integrative approach of anthroposophical medicine, founded by Rudolf Steiner and Ita Wegman, 21 the traditional care of breast cancer patients, next to standard and mistletoe therapy, includes treatment approaches to potentially harmonize and balance relaxation responses and which focus on fine motor skills and concentration exercises such as eurythmy therapy and painting therapy. 22 An RCT study on eurythmy therapy yielded a significant stress reduction in subjects suffering from burnout. 23 In case of eurythmy and painting therapy, there are at present no confirmatory studies available to affect CRF or cognitive dysfunctions, but the preliminary results of pilot studies support their validity justifying further research.24,25
In this article, we present a pilot study investigating the feasibility and first results of a new multimodal therapy, including behavioral therapy (sleep and psychoeducation), eurythmy, and painting therapy in breast cancer patients with chronic CRF compared with the current standard therapy, namely aerobic training.
Methods
Sample
The pilot study was conducted from February 2009 to December 2009. Participants were recruited from February to September 2009 at 2 centers during oncological, internal medicine, and physical medicine consultations as well as via local newspaper advertisements.
Inclusion criteria: Breast cancer patients with relevant chronic cancer fatigue (>6 months and with a Fatigue Numerical Scale ≥4), 26 either recurrence-free or with low tumor burden (bone metastases or visceral metastases with or without antihormonal and/or bisphosphonate treatment).
Exclusion criteria: Chemotherapy or radiotherapy in the past 6 months, hemoglobin <10 g/dL, other severe internal conditions (ie, cardiac insufficiency New York Heart Association [NYHA] 2, unstable angina pectoris, peripheral arterial disease stage ≥2, chronic obstructive pulmonary disease stage ≥2, renal insufficiency (creatinine >2 mg/dL), apparent, untreated hypothyroidism (thyroid-stimulating hormone basal >4 µU/mL, fT4 < 9 pmol/L) or hyperthyroidism (thyroid-stimulating hormone <4 µU/mL, fT4 > 24 pmol/L), and severe limitations of the musculoskeletal system. Furthermore, we excluded patients with severe psychiatric conditions, such as apparent major depression or psychosis, or patients with untreated sleep apnea, restless legs syndrome, and narcolepsy. Finally, we excluded patients already undergoing intensive (trainer-controlled optimized, twice a week, or more often) aerobic training, and those receiving erythropoietin or transfusions.
Procedures
The study was approved by the local ethics committees. After giving informed consent, participants selected their preferred treatment group. The 2-group intervention study was conducted over 10 weeks. The multimodal concept has been developed by a group of experts (oncologists, internists, sleep specialists, psycho-oncologists, specialists in anthroposophical medicine, physiotherapists and sports therapists, art therapists, and eurythmy therapists). The multimodal experimental group consisted of 4 modules of 225 minutes total, which was carried out once a week over 10 weeks:
The psychoeducative fatigue group focused on CRF comprehension and management, stress management, including exercises for improving mindfulness, relaxation, and handling of emotions in an interactive manner from a psychotherapeutic and anthroposophical point of view.10,27
Sleep education, apart from a lesson on the basics of chronobiology and sleep medicine (circadian, rest/activity, basic rest/activity rhythm, and sleep regulation) included a sleep diary based on sleep hygiene, sleep restriction, and stimulus control consultation with individual recommendations.28-30
Eurythmy therapy is a specific, mind–body orientated active exercise therapy, which was developed in anthroposophical medicine by Rudolf and Marie Steiner. 31 Eurythmy therapy or “harmonious rhythm” can be described as an active exercise therapy integrating cognitive, emotional, and volitional elements as well as sounds of language to express them in movements of the whole body. The inherent images of the sounds are performed by the therapist and recreated by the patient’s activity. 32 The purpose of eurythmy therapy in our setting was to treat CRF-associated dysrhythmia, with the following active exercises: starting with the vowels I, A, and O, clenching and spreading, followed by reciting a hexameter together with the corresponding movements, vowel “Ei”, consonants L, M, and R and preparing the series of O E M L EI B D, which we called the “cancer series.” 33 Patients were also required to exercise these movements at home.
Anthroposophical medicine–orientated painting therapy is an art therapy first developed by Ita Wegman and Margarethe Hauschka. 34 In our intervention we started each session with form drawing at increasing degrees of difficulty to stimulate concentration and fine motor skills. After this we continued with the creation of a series of rhythmic paintings (day–night), beginning with a dark atmosphere in blue, and adding progressively more light with yellow and red, respectively. The outcome of this was a sunrise, and then a tree in a landscape setting. 35 In the first and last sessions, patients were asked to produce a painting of their choice.
The standard therapy group was required to carry out home-based aerobic training, with three to five 45-minute trainer-controlled interventions over 10 weeks. On the first day, we ascertained a physiological exposure of 70% to 80% threshold by an ergometer. We started with 25 watts and increased by 25 watts every 2 minutes, with the heart rate being measured every 2 minutes. The test was repeated and watts further elevated to the point of the patients’ exhaustion. 18 Participants were asked to carry out 30-minute home-based training sessions 3 to 5 times a week.
Every intervention was conducted by a specialist in his or her own field.
Measures
In the context of this study, the German version of the Cancer Fatigue Scale (CFS-D) and the Pittsburgh Sleep Quality Index (PSQI) were administered during 3 weeks before the start of the intervention, and all questionnaires again on day 1 as well as on the last day of the intervention. Furthermore, 48-hour actigraphy and 24-hour long-term electocardiographic measurements were carried out during the 3 weeks prior to and following the intervention (presented elsewhere).
The following questionnaires were used:
Patients’ questionnaire on subjective expectations of rehabilitation with regards to intervention by the Clinic for Physical Medicine and Rehabilitation at the Hannover Medical School. 36
The CFS-D is a short and compact 15-item questionnaire with 3 subscales dealing with physical (6 items), affective (4 items), and cognitive fatigue (5 items). The items of the cognitive subscale capture memory (“become forgetful”), concentration (“lack of concentration,” “become forgetful”), language difficulties (“make errors while speaking”), and slower processing speed (“thinking has become slower”). The scale consists of a 5-point Likert-type scale (0-4) with a range between 0 (no fatigue) and 60 (maximum fatigue). The CFS was originally developed in Japan 26 and was then transculturally adapted and revalidated in Germany, yielding robust reliability (sum-scale: Cronbach’s α: rα = .94, test–retest reliability rrt = .82), and validity. 37 The CFS-D classifies values ≥30 points as CRF, ≥23 to 29 points strongly as CRF cases, and ≤22 points as no cases. 38
The PSQI captures sleep attitudes and sleep quality in the past 4 weeks. It is widely used in sleep medicine research. The questionnaire consists of 18 quantitative items split into 7 components, all scored between 0 and 3. The sum scale detects global sleep quality between 0 (no sleep disturbance) and 21 (maximum disturbed sleep) with a cutoff of 5 (good/bad sleeper). 39
The autonomic regulation (aR) scale captures the self-perceived state of regulation of different autonomic functions such as orthostatic, circulatory functions, as cold hands, sleep quality, being rested in the morning, “pull yourself together” to come to work, circadian well-being, thermoregulation, and digestion. These questions had already been proposed in the early 1920s by Steiner as constitutional and health parameters. 40 The validated scale consists of an 18-item 3-point Likert-type scale with 3 subscales—orthostatic-circulatory (7 items), rest/activity (8 items), and digestive regulation (3 items). The questionnaire ranges from 18 (minimal aR) to 54 (maximum aR), has satisfying to good reliability (Cronbach’s α: rα = .75, test–retest reliability rrt = .85), and good validity. 41 In breast cancer patients, different studies have reported reduced aR and particularly loss of rest/activity regulation in recurrence-free breast cancer patients.41,42 We applied the aR scale for capturing possible improvements in the regulation of autonomic functions under intervention in a more holistic view.
On conclusion of the intervention, individual therapy modules were evaluated with the help of a newly developed questionnaire on patient satisfaction. Possible answers were rated with a 5-point Likert-type scale from 0 (not at all satisfied) to 4 (very satisfied).
Statistics
Sample size
The intention of this pilot study was to obtain first hints regarding possible treatment effects of our experimental multimodal (MM) therapy. Thus, emphasis was put on a small beta error (indicative of a missing treatment effect) of 5% (ie, a power of 95%) while relaxing the alpha error (deducing a nonexistent improvement from the data) to 50%. As no knowledge regarding the expected possible improvement was available, a rather small standardized effect size of 0.33 was chosen. A sample size estimation based on a paired t test for corresponding changes in the patients’ condition from their baseline levels resulted in 25 patients who were to be included in the MM therapy. Because of recruitment problems and patients’ preferences for MM treatment, only half of this sample size could be allocated to the controls of aerobic treatment (AT).
Statistical analysis
Data analysis was performed with SAS version 9.1.3 for Windows. We performed a complete case analysis, which is a valid approach especially in nonrandomized experimental and in therapies at early stages of development where immediate therapeutic effect rather than effectiveness is the focus of research. Group-wise descriptive statistics 43 included absolute/relative frequency, mean, standard deviation, standard error of mean, minimum, maximum, median, and first and third quartiles. Efficacy parameters were analyzed at the end of trial with respect to their differences from baseline levels. For changes from baseline, additionally parametric 95% confidence limits for the mean and nonparametric 95% confidence limits for the median were calculated. We controlled divergence from a symmetric distribution by comparing arithmetic mean and standard deviation with median and quartiles, respectively, and did not observe significant discrepancies. Differences between therapy groups were tested by Fisher’s exact test and the Mantel–Haenszel test for sociodemographic characteristics and by t test for baseline values of the efficacy parameters. Additional sensitivity analyses using propensity scores for adjusting the comparability of the therapy groups with regard to all baseline parameters generally supported the outcome of the primary analysis.
Results
In total, 73 female breast cancer patients and 1 male breast cancer patient who were either known from previous consultations or who responded to newspaper advertisements were initially seen. After clarification, 65 female patients presented for the study consultations, of whom 36 fulfilled the inclusion and exclusion criteria and agreed to participate in the study. Twenty patients opted to participate in the MM therapy and 13 in the AT group. Of these, 2 participants in the MM group (one because of family reasons and one because of exhaustion) and 3 participants in the AT group dropped out of the study (2 because of disease and one because of an operation), resulting in 21 women in the MM group and 10 women in the AT group completing the study (Figure 1).

Recruitment flowchart.
The mean age of the participating breast cancer patients was 57.0 years in the MM group and 56.9 years in the AT group, with a time span since first diagnosis of 3.9 years (MM) and 3.0 years (AT; see Table 1).
Sociodemographic Characteristics, Cancer Stages, and Therapy of Participants With Breast Cancer.
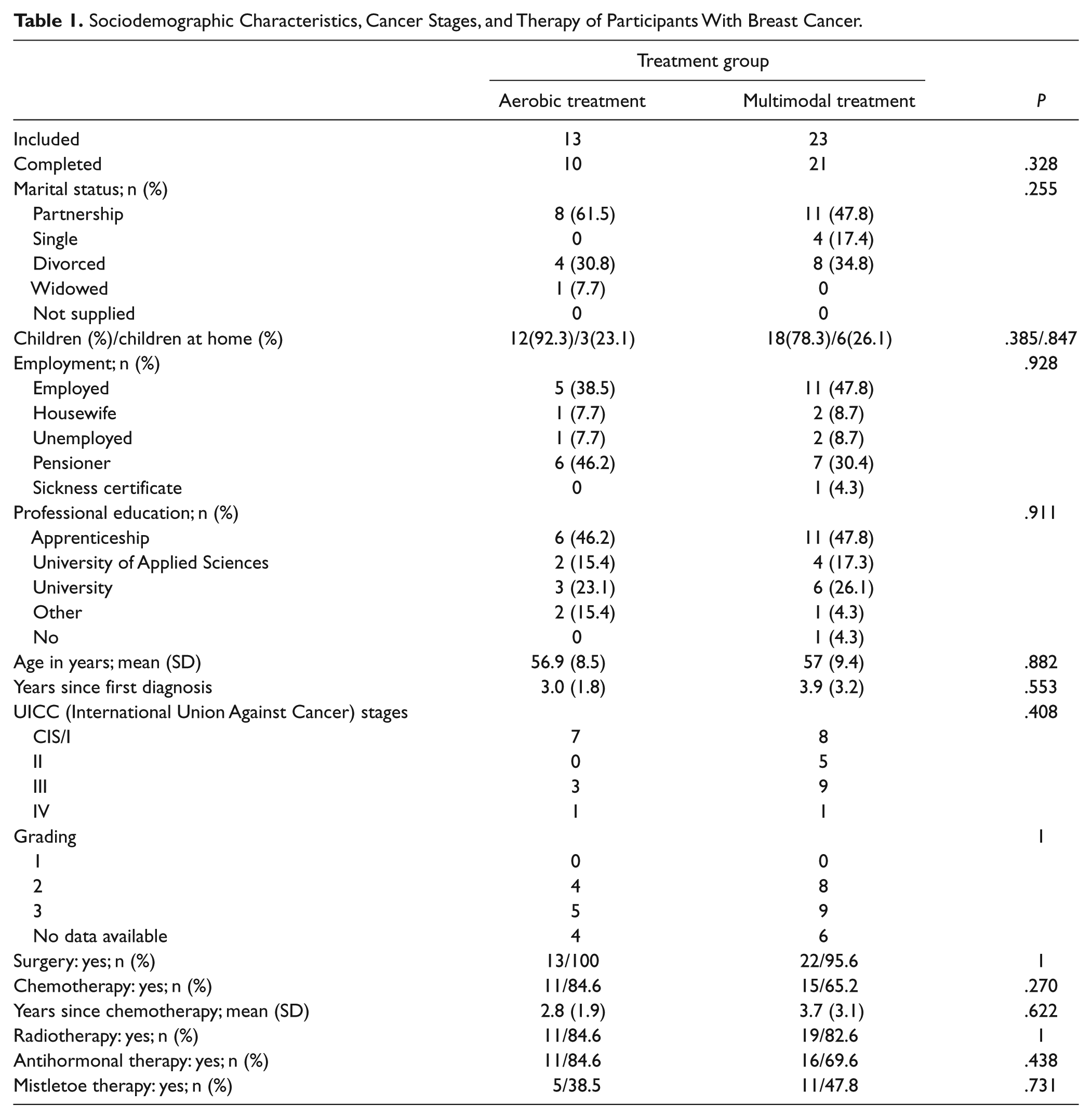
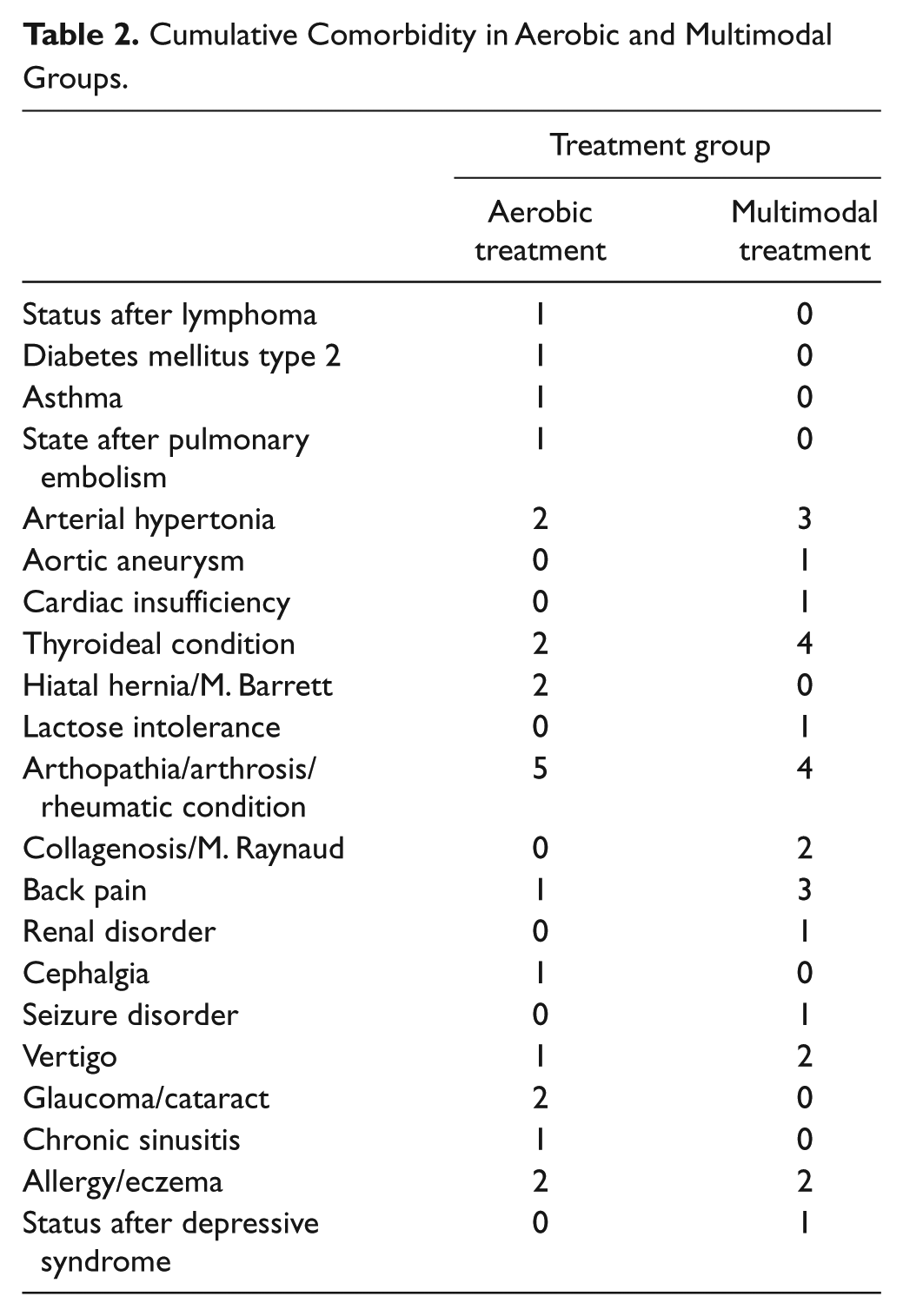
Both groups consisted of recurrence-free breast cancer patients, with one exception in each group showing low-level metastases (Table 1). The patients had received the following standard treatment before inclusion in the study: surgery (MM = 22, AT = 13), chemotherapy (MM = 15, AT = 11), radiotherapy (MM = 19, AT = 11), antihormonal treatment (MM = 16, AT = 11), and mistletoe treatment (MM = 11, AT = 5; see Table 1). Comorbidities in the 2 groups are presented in Table 2.
Cumulative Comorbidity in Aerobic and Multimodal Groups.
Patients’ expectations did not differ significantly between both groups, with one exception, that is, that the aerobic training would be more likely to reduce physical complaints (MM, mean ± SD = 2.0 ± 0.95; AT, mean ± SD =1.33 ± 0.49; P = .035) and there was a tendency in the MM group to achieve better emotional recovery (MM, 1.68 ± 0.84; AT, 2.09 ± 0.94; P = .19; see Figure 2).
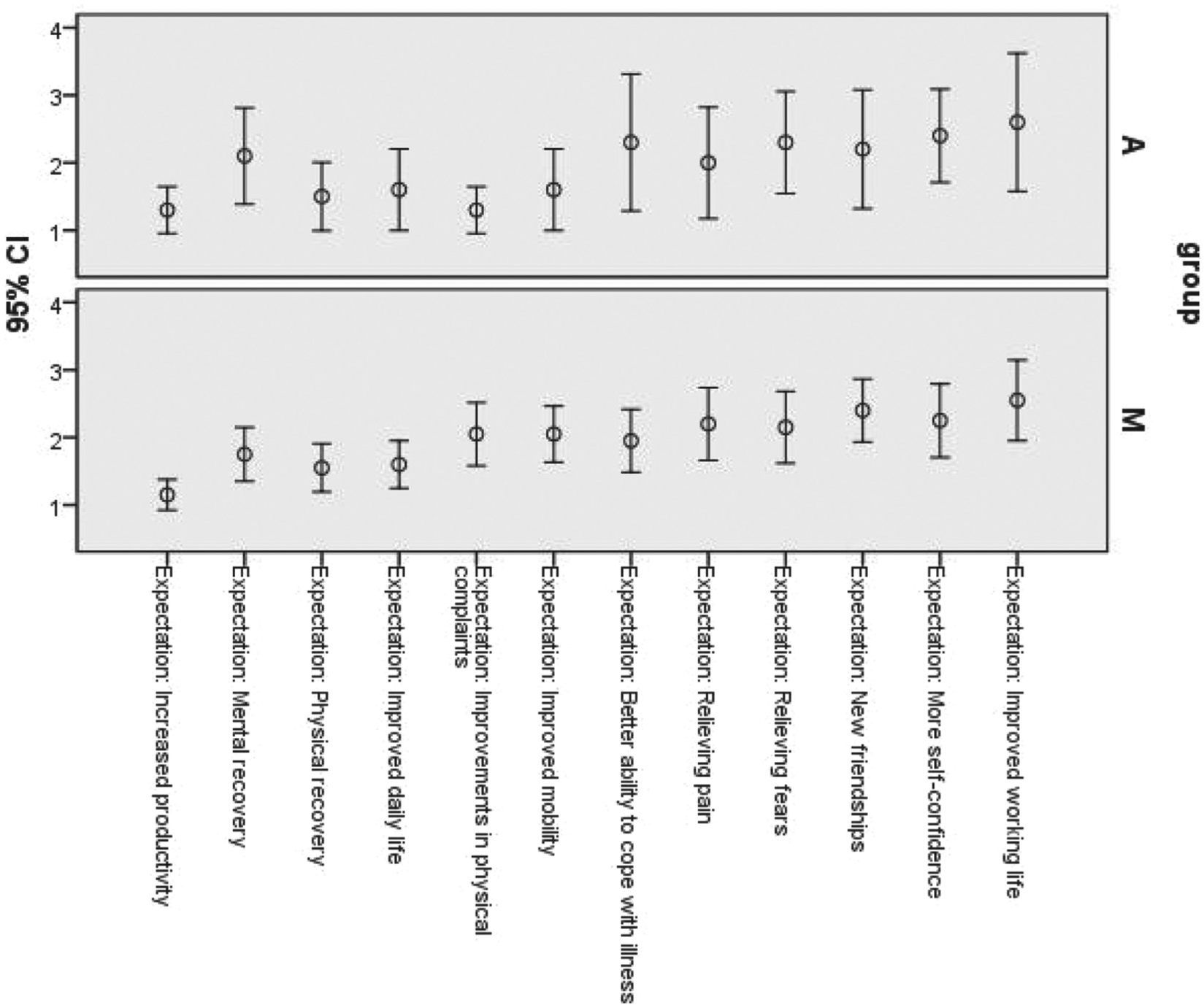
Patients’ expectations.
In the MM group, a significant reduction in the CFS-D sum-scale (baseline [T01], mean ± SD = 33.5 ± 8.1, after intervention [T1], 30.1 ± 9.2) and the CFS-D physical fatigue (T01, 15.8 ± 3.6; T1, 13.7 ± 4.9) was detected and an improved global sleep quality (T01, 10.0 ± 4.0; T1, 8.0 ± 4.1) with all Ps ≤ .05. We found further improvements in the multimodal group in the autonomic regulation sum-scale (T01, 35.9 ± 4.4; T1, 39.2 ± 5.1) and in the rest/activity scale (T01, 13.7 ± 3.2; T1, 16.7 ± 3.2; all Ps ≤ .05). Sleep efficiency (T01, 1.5 ± 1.4; T1, 0.9 ± 1.1) and daily sleepiness (T01, 2.0 ± 0.9; T1, 1.5 ± 0.7) showed a trend toward improvement. CFS-D cognitive and affective, PSQI sleep latency and sleep quality, and aR orthostatic-circulatory and digestive regulation did not change significantly (Table 3).
Measurements at Baseline (T00, T01) and After Intervention (T1) for the Multimodal and Aerobic Treatment Groups a .

Abbreviations: MM, multimodal treatment group; AT, aerobic treatment group; CFS-D, German version of the Cancer Fatigue Scale; PSQI, Pittsburgh Sleep Quality Index; T aR, Trait Autonomic Regulation Scale; SD, standard deviation; CI, confidence interval.
There were no significant differences between treatment groups at time T01. Significant (P ≤ .05) changes within the treatment group from T01 to T1 are shown in boldface.
Significant (P ≤ 0.05) differences between MM and AT at T00.
In the AT group, only the aR sum-scale (T01, 36.0 ± 5.4; T1, 38.0 ± 5.5), specifically the orthostatic-circulatory scale (T01, 15.5 ± 2.3; T1, 16.6 ± 2.9) improved significantly. We could not detect any significant improvement in case of the CFS-D and the PSQI and their subscales or aR digestive regulation for the AT group.
Patients’ satisfaction with the MM therapy concept was good to very good (3.47 ± 0.53), particularly for eurythmy therapy (3.81 ± 0.40) and for painting therapy (3.71 ± 0.64). In contrast to that, satisfaction in the AT group was rather moderate with a mean of only 2.67 ± 0.71 (Table 4).
Patients’ Satisfaction With the Intervention a .

Patients’ satisfaction with the intervention was assessed at the end of the intervention with a 5-point Likert-type scale (0 = not at all satisfied, 1= not particularly satisfied, 2 = moderately satisfied, 3 = quite satisfied, 4 = very satisfied). Mean, standard deviation, and range of each therapy component are presented.
Discussion
In this pilot study, we were able to show the feasibility to implement a new multimodal 10-week intervention program for women with breast cancer with a chronic, CRF syndrome by applying psychoeducation and sleep education in combination with eurythmy and painting therapy. We detected significant improvements in global CRF, physical fatigue, global sleep quality, autonomic regulation, and particularly in the rest/activity regulation, with high satisfaction on the side of the patients. In the AT group significant improvements were confined to autonomic regulation particularly in orthostatic-circulatory regulation, but cancer fatigue remained unaffected, although the pre- to posttreatment difference effects were comparable to the MM group.
In this study, we treated patients with long-term CRF with a disease duration of 3 years on the average in the AT group and 3.9 years in the MM group. In patients with such chronic CRF, spontaneous improvement is extremely unusual and a moderate effect-size in the MM group of 40% in CRF and 50% to 75% in global sleep quality and autonomic regulation points to a moderate to strong clinically relevant improvement. 44 As we did not have a waiting list design in our study, we measured baseline CFS-D and PSQI twice within 1 to 3 weeks to ensure stability for the baseline assessment and to allow differentiation from possible intervention effects.
Despite convincing evidence based on 11 randomized controlled studies dealing with the importance of physical exercise training for breast cancer patients suffering from chronic CRF syndrome and an estimated effect of −0.32, 45 it has become apparent during the past few years that this intervention shows limitations. Although lasting improvements with respect to physical fatigue could be seen, there were no or only minor improvements in case of cognitive fatigue, distress 46 or insomnia. 18 These findings are compatible with the results of our pilot study, where the physical exercise group showed no improvement with regard to sleep quality. The improvement in overall fatigue was not significant, although this may have been possibly because of the small sample size of our current study. Furthermore, we have to point to the design of our AT intervention. We used a mixture of trainer-led exercise interventions alongside a home-based exercise therapy. The adherence to home-based training for at least 3 times per week—documented with a training book—was 70%. It is conceivable that more frequent trainer-led exercise sessions might improve the current outcome as can be derived from the results of a new meta-analysis only showing minor effects for purely home-based exercise programs and better effects for trainer-based exercise programs in breast cancer patients with CRF. 47
When comparing the patients’ expectations of aerobic training to multimodal intervention it has to be underlined that in the former, more emphasis is put on attending to physical complaints whereas in the latter, the focus is on mental recovery and support of self-confidence. This reflects 2 aspects, first classical complementary and alternative medicine user expectation to participate in decision making for allocation and second the individualization of their salutogenetic approach.48,49 Therefore, we decided at this early stage of development of our new therapeutic trials not to randomize but to accept the patients’ preferences.
The therapy approach of our concept is multimodal, therefore we cannot deduce the effect of individual therapeutic components. However, our data support the idea that sleep education could have a relevant impact on the improvement of global sleep quality, which is mainly because of elevating sleep efficiency and lowering daily sleepiness, and in turn favorably affects rest/activity regulation. This view is supported by a series of studies investigating sleep hygiene, sleep restriction and stimulus control showing strong and sustained effects on sleep quality as well as minor to moderate affects on CRF in breast cancer,28,50,51 The psychoeducation in our multimodal concept focuses on a better understanding and disease management of breast cancer and stress management. Even if its impact remains unclear there is some probability that it could contribute to better relaxation and stress management with a possible co-influence on CRF, sleep, and autonomic regulation. This is in line with a meta-analysis that reports a low effect size of −0.16 of cognitive behavioral therapies in breast cancer patients with CRF. 45 We invited participants to repeat their exercises in order to further improve mindfulness, relaxation, and their handling of emotions at home. Unfortunately, we did not use a homework book and evaluated rather a moderate adherence qualitatively in a statement round at the beginning of every intervention day. Recently, another group published a comparable self-management program consisting of 6 modules (forms of fatigue, etiology, and treatment of CRF, management, sleep, handling of emotions, and individual daily adaption) for patients suffering from CRF resulting in good patient satisfaction. 52
In our multimodal therapy concept with eurythmy and painting therapy, we integrated these 2 therapies that are standard treatments in anthroposophical medicine for breast cancer patients with CRF, but for which there is as yet insufficient evidence. 22 The traditional clinical and outpatient use of eurythmy therapy is an add-on therapy in the context of the integrative medicine approach of anthroposophical medicine for a large number of indications, and particularly for oncology, although there is still insufficient evidence for its individual, specific application. 53 However, eurythmy therapy integrates meditative exercises with specific therapeutic movement sequences with a salutogenic objective, which can result in a significant improvement of heart rate variability 24 and distress, 23 underlining its potential health-promoting influence. An outcome study captured a positive long-term effect for eurythmy therapy in case of different chronic conditions. 54 First studies on cancer patients are currently ongoing. In addition to salutogenic effects, art therapy may help in the psychological management of, and readjustment to, cancer, with general effects on health-related quality of life and coping. 55 There are first hints from a pilot study on cancer patients undergoing chemotherapy that the anthroposophical approach of painting therapy in addition to traditional treatments can improve depressive symptoms and influence CRF. 25 An outcome study showed that health-related quality of life and symptoms improved in chronic conditions. 56 We integrated both these therapies—eurythmy and painting therapy—in our concept to lower CRF in general and cognitive fatigue and distress in particular. As we were unable to show a significant reduction of cognitive fatigue, we intend to strengthen further research elements, such as fine motor skills in eurythmy therapy and form drawing in our art therapy setting. Despite the fact that their significance in the treatment of CRF in general and in our multimodal therapy concept in particular is still unresolved, these therapies did in fact achieve the highest patient satisfaction rates, which encourages us to specify them as an integral component in our multimodal approach and to conduct further research on this topic. Another aspect which should be taken into account is that the multimodal approach obviously requires more financial resources compared to aerobic training, but as pointed out earlier, aerobic training is a useful method only for reducing physical but not cognitive fatigue and limited for reducing distress. 45 CRF is indeed a complex syndrome, including cognitive fatigue, distress, and insomnia with an important socioeconomic impact (sickness related, nonproductive time, invalidity pension, etc.), 57 and therefore a multimodal approach with therapies focusing on all these symptoms is imperative. It will have to be seen in which way a compromise can be achieved by securing maximal health benefits for the patients’ undergoing multimodal therapy and the need to spare financial resources to attain cost-effectiveness (essential for the measurement of efficiency in further research).
Finally, it needs to be stressed that our study still possesses certain limitations, which are because of the pilot character of the current investigation, for example, small number of participants (particularly in the aerobic treatment arm), a possible undertreatment of the partially home-based physical exercise group, and of course yet unknown effects of the different treatment modules, which have neither been used in the present form nor in their specific combination. Furthermore, the lack of randomisation may have resulted in consecutive selection bias, even if we did not find any indication for systematic differences between therapy groups regarding sociodemographic and efficacy parameters at baseline. In addition, we did not measure adherence to the therapies in a standardized way, although the aerobic training group filled out a training protocol and the multimodal group filled out a sleep agenda for 2 weeks before and after sleep intervention. (Homework adherence with regards to psychoeducation in particular was rather moderate.) In a next intervention study, adherence documentation and adherence quality will be improved to rule out possible distortions.
Conclusion
In this pilot study, we were able to implement a new 10-week multimodal intervention program for breast cancer patients with CRF. This research supports first evidence that CRF, sleep quality, rest/activity, and autonomic regulation can be improved. The next step should be to evaluate this in a randomized controlled trial.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mahle Foundation, Stuttgart. Matthias Kröz and Roland Zerm received financial support by the Humanus Institute, Berlin.
