Abstract
Introduction
Cancer is still the number one killer in the world, and most treatments for the disease depend on the use of cytotoxic substances and other drugs as chemotherapy that have a toxic effect on normal cells. Therefore, an alternative approach to cancer treatment should involve the drugs or other substances that block the growth and spread of cancer by interfering with specific molecules targeted drugs that are involved in the development of cancer growth. Furthermore, the new generation of drugs should be nontoxic to the normal cells and reasonable for the patients. 1
Actually, more than 47% of anticancer therapies on the market are natural products, their derivatives or natural substances synthetic simulate,2,3 and over 25 000 identified phytochemicals have been shown to possess effective anticancer activities.2,4 Graviola is a small deciduous tree of the Annonaceae family broadly distributed in tropical countries, that is well known by several names including Annona muricata, soursop, Brazilian paw, and guanabana.4,5 Annona muricata is a natural plant source of anticancer phytochemicals, with more than 212 phytochemicals identified in the various extracts of graviola, found in fruits, seeds, bark, roots, and pericarp. It is used to treat a number of illnesses and diseases like cough, skin disorders, diabetes, and cancer.5,6
The diverse constituents’ metabolites of graviola such as acetogenins are understood to play a central role in its anticancer properties on different human cell lines, as are many other constituents such as flavonoids, sterols, alkaloids, and others.5,7 Different studies in non–skin cancer cell lines show that graviola suppresses cancerous cells without harmful effects on normal cells. This therapeutic action of graviola might be due to its ability to selectively inhibit cancer.5,8
Several studies have shown that low-dose radiation (LDR) has the potential to stimulate response to anticancer drugs and reduce side effects by improving therapeutic efficiency through enhancement of different phenomena such as adaptive response and cell-cell communication.9,10 Additionally, response of the immune system11,12 and inhibition of metastasis subsequently have been observed with LDR. 13 These findings suggest that LDR stimulates natural molecular barriers against oxidative damage that were observed in earlier mouse studies showing that the levels of natural barriers increased in liver cells after whole-body exposure to LDR, 14 helping protect or treat sporadic cancer. This study was aimed to explore the basis of graviola’s antitumor and apoptogenic effect, as well as the role of LDR as an adjuvant in increasing the action of graviola and vice versa. Among these, we determined CD44 growth rate, mitotic index by cell cycle, apoptotic index by annexin V, and proliferation by transforming growth factor β (TGF-β) and Bcl-2 in tumor tissue.
Materials and Methods
The study was performed in the National Center for Radiation Research and Technology (NCRRT), Atomic Energy Authority, Cairo, Egypt.
Animals
Female Swiss albino mice weighing 27 to 30 g were obtained from the breeding unit at the NCRRT, Cairo, Egypt. Mice (10 animals/cage) were housed and maintained under proper environmental conditions, that is, controlled air, temperature, and relative humidity. Mice were provided with pellet diet and free access to water. Animal experiments were consistent with the ethics guidelines of the Public Health Guide for the Care and Use of Laboratory Animals (National Research Council), 15 in accordance with the recommendations for the proper care and use of laboratory animals approved by the Animal Care Committee of NCRRT, Cairo, Egypt.
Chemicals
All chemicals used in the present investigation were of analytical grade and purchased from Sigma Chemical Company (St Louis, MO). Graviola supplement capsules (50 mg) were purchased from Raintree (Carson City, NV).
Graviola Preparation
Graviola, a product of Raintree, consists of capsules weighing 50 mg, consisting of 100% pure graviola leaf/stem powder without binders or fillers. The capsule contents were suspended in dimethyl sulfoxide (50 mg/mL). After incubating for 5 minutes, the suspension was centrifuged and the supernatant was filtered to remove any remaining particles. Subsequent dilutions were prepared in Dulbecco’s modification of Eagle’s medium supplemented with 10% of fetal bovine serum. Stock solutions and respective dilutions were freshly prepared prior to treatment.
Irradiation Procedures
Whole-body γ irradiation was performed with a Canadian Cs116 Gammacell 40 biological irradiator at the NCRRT, Cairo, Egypt, at a dose rate of 0.006 Gy/s. After 10 days of tumor inoculation, mice were irradiated with a fractionated dose of 2 Gy delivered per week with a total cumulative dose of 6 Gy after 3 weeks, according to the experimental design.
Solid Tumor Induction
Ehrlich ascites carcinoma (EAC) cell line was obtained from the Pharmacology and Experimental Oncology Unit of the National Cancer Institute, Cairo University, Giza, Egypt. The tumor cell line was maintained in the experimental female Swiss albino mice by weekly intraperitoneal injection of 2.5 × 106 cells/mouse. EAC cells are of mammary origin. 16 The viability of the cells was 99% as judged by trypan blue exclusion assay. The xenograft model of Ehrlich solid tumor (EST) was induced in female Swiss albino mice by viable EAC cells (about 2.5 × 106/mouse) in 0.2 mL isotonic saline implanted subcutaneously (sc) into the right thigh of the hind limb of each mouse.17,18 The tumor developed in 100% of the mice with a palpable solid tumor mass (>1 cm3) was achieved within 7 days postimplantation. The day of tumor implantation was assigned as day 0. On the seventh day, animals were randomized into 4 experimental groups.
Survival Rate Analysis
The day of implantation was considered as the 0 point of the experiment. Mice were monitored for recording and analysis of the survival rate daily for 37 days by registering mortalities occurring in EST-bearing untreated and treated groups according to Abdin et al. 19 After 1 week of tumor growth, oral gavage treatment of phosphate-buffered saline (PBS)–suspended graviola extract was given daily for 30 days. The doses of graviola extract at 50 mg/kg for these studies were based on previous in vivo studies, and on the recommended dose for human consumption.20-23
Experimental Design and Treatment Protocols
Sixty mice were divided into 4 groups as follows: (1) Normal control group (C; 10 mice): animals received 2 mL/kg PBS orally at corresponding times along the experimental time course; (2) EST group (20 mice): animals were inoculated intramuscularly with 0.2 mL (containing 2.5 × 106 EAC cells) in the right thigh once and received 2 mL/kg PBS orally at corresponding times along the experimental time course; (3) graviola-treated EST-bearing mice group (EG; 15 mice): EST-bearing mice were gavaged with graviola at a dose of 50 mg/kg body weight/day for 30 days; (4) graviola-treated and irradiated EST-bearing mice (EGR; 15 mice): EST-bearing mice were administered graviola at a dose of 50mg/kg body weight/day orally for 30 days and exposed to fractionated whole-body low-dose γ radiation at a dose level 2 Gy once a week for 3 weeks.
Mice were sacrificed after 37 days of tumor growth and 30 days of treatment with graviola and LDR. Changes in tumor growth were screened in each treatment group. Body weights and tumor volume of mice were measured weekly and before the treatment. Tumor weight was measured after removal at the end of the experiment. For tumor volume monitoring and tumor growth inhibition determination, the volume of solid tumor was measured by using micro calipers (Vernier, Shanghai, China) after 7, 14, 21, and 28 days from inoculation of EAC. The tumor volume was calculated after 28 days by the following equation:
Tumor growth inhibition rate (%) was determined according to the following equation 24 :
Tumor Tissue Sample Collection and Preparation
After 37 days and 16 hours of fasting, animals of each group were sacrificed under gentle ether anesthesia and tumor tissues were dissected immediately and weighed, then washed with PBS for biochemical analyses.
Flow Cytometry Assays
Cell Cycle
One hundred microliters of cell suspension (1 × 106 cell/mL) was prepared by isolation of mononuclear cells from the processing of tissue with Tris-EDTA buffer. One hundred microliters of cell suspension was added to PI buffer (propidium iodide with RNase) and incubated for at least 1 hour in the dark at +4°C. After incubating, the cells were acquired by using BD Flow cytometry Accurie C6Plus version (Becton Dickinson, San Diego, CA). The DNA content of 10 000 cells was analyzed by FACS Caliber flow cytometry to calculate the percentages of cells occupying the different phases of the cell cycle by using the BD Accuri C6 Plus software files, which can import data into flow cytometry software for seamless software and FlowJo software. 25 Samples were run in triplicate, and each experiment was repeated 3 times.
Cell Surface Markers
One hundred microliters of cell suspension (1 × 106 cell/mL) was prepared by isolation of mononuclear cells from the processing of tissue with Tris-EDTA buffer. The cells were washed with PBS/BSA (bovine serum albumin) with 2 mL and then centrifuged at 2000 rpm for 5 minutes. The supernatant was discarded, and the pellet resuspended in 100 µL of PBS. Seven microliters of a marker (CD44 [BD Pharmingen, Cat. No. 564392]; TGF-β [BD Pharmingen, Cat. No. 555053]; and BcL-2 [BD Pharmingen, Cat. No. 610539]) was mixed well, and the tube was incubated for 30 minutes at room temperature in the dark. Cells were washed twice with 2 mL PBS/BSA, and centrifuged at 2000 rpm for 5 minutes and the supernatant discarded. Finally, cells were resuspended in 200 µL of 4% paraformaldehyde in PBS for fixation until acquired by using BD Flow cytometry Accurie C6 Plus version (Becton Dickinson). The DNA content of 10 000 cells was analyzed by FACS Caliber flow cytometry to calculate the percentages of cell surface markers by using the Intuitive Software. BD Accuri C6 Plus software files can be exported in FCS 3.1 format for seamless data import into flow cytometry analysis programs such as FCS Express software and FlowJo software. 25 Samples were run in triplicate, and each experiment was repeated 3 times.
Annexin V
One hundred microliters of cell suspension (1 × 106 cell/mL) was prepared by isolation of mononuclear cells from the processing of tissue with Tris-EDTA buffer. One microliter of cell suspension was added to a 5 mL tube and then resuspended in 2 mL 1× binding buffer (1 mL of 10 × buffer +99 mL distilled H2O) for a good mix. One hundred microliters of cell suspension were taken in another 5 mL test tube then 5 µL of annexin V (FITC label; Cat. No. 556547 BD pharmingen FITC apoptosis Kit) was added, and directly 5 µL PI (PE label). The incubation time was for at least 15 minutes in the dark at room temperature. After the incubation time, the cells were resuspended in 200 µL 1× binding buffer. The cells were ready to acquire on flow cytometry by using BD Flow cytometry Accuri C Plus version (Becton Dickinson).The DNA content of 10 000 cells was analyzed by FACS Caliber flow cytometry to calculate the percentages of cells occupying the different phases of the cell cycle by using the Intuitive Software. BDFCS 3.1 format for seamless data be exported in programs such as FCS Express software and FlowJo software. 25 Samples were run in triplicate, and each experiment was repeated 3 times.
Statistical Analysis
Statistical analysis of data was performed by 1-way analysis of variance (ANOVA) using the Graph Pad Prism software package 5.0 for Windows from Graph pad Software Inc. Results were expressed as the mean ± standard error of mean for the 10 mice in each group. Values of P <.05 were considered statistically significant.
Results
Graviola and combination of graviola with LDR improves survival rate and induces tumor volume regression in EST.
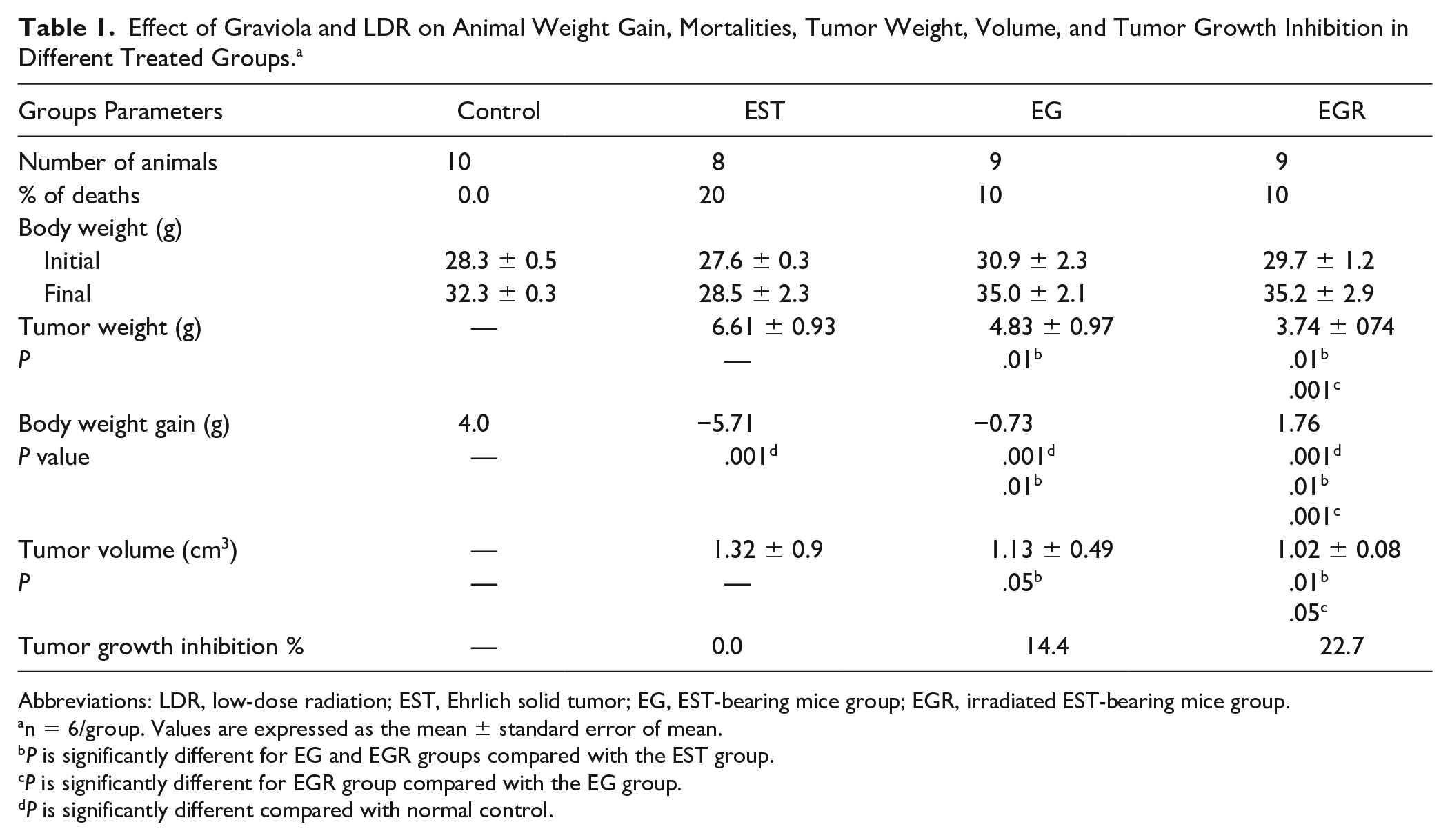
The change in body weight gain, mortalities, tumor volumes, tumor weight, and % of tumor volume inhibition of each group were recorded during the experimental period. As shown in Table 1 and illustrated by Figure 1, the results displayed final body weight gain resulting in nonsignificant changes in the groups under investigation when compared with the initial body weight in all groups, but when subtracting tumor weight from final body weight, the results recorded amelioration in the body weight in groups treated with graviola alone or accompanied with radiation exposure compared with the group bearing tumor without treatment. Concerning animal mortalities, none were observed in the normal control group, and ≈20% died throughout the experimental period in the EST group, and ≈10% in the EG and EGR groups.
Effect of Graviola and LDR on Animal Weight Gain, Mortalities, Tumor Weight, Volume, and Tumor Growth Inhibition in Different Treated Groups. a
Abbreviations: LDR, low-dose radiation; EST, Ehrlich solid tumor; EG, EST-bearing mice group; EGR, irradiated EST-bearing mice group.
n = 6/group. Values are expressed as the mean ± standard error of mean.
P is significantly different for EG and EGR groups compared with the EST group.
P is significantly different for EGR group compared with the EG group.
P is significantly different compared with normal control.

Animal mortalities, tumor weight, tumor volume, and tumor growth inhibition in differently treated groups.
Table 1 and Figure 1 clarified the great variation in the tumor volumes and % tumor growth inhibition between the examined groups. At the end of 37 days of the experiment, the average tumor volume reached up to 1.32 ± 0.9 cm2 in the EST group. Graviola treatment significantly reduced the tumor volume up to 1.32 ± 0.9 cm2 and inhibited tumor growth by 14.4% with P value <.05 compared with the EST group. Combination of both therapies (graviola and LDR) significantly reduced the tumor volume to 1.02 ± 0.08 cm2 and inhibited tumor growth by 22.7% with P value <.01 and <.05 compared with the EST and EG groups, respectively.
Treatment with graviola or as an adjuvant with LDR suppresses proliferation by inhibiting CD44, TGF-β, and Bcl-2 signaling in EST The flow cytometry analysis was used to demonstrate the expression of CD44, TGF-β, and Bcl-2 in Table 2 and Figure 2. The results clarified that the mean values of CD44, TGF-β, and Bcl-2 of the normal control group were 5.2 ± 0.7, 5.4 ± 0.9, and 15.2 ± 1.4, respectively. Meanwhile, the mean values of CD44, TGF-β, and Bcl-2 expression were significantly increased up to 75.7 ± 3.9, 66.8 ± 3.8, and 76.4 ± 4.5, respectively, in the EST group compared with the normal control group with P value <.0001. Otherwise, the graviola-alone treatment significantly brought down the CD44, TGF-β, and Bcl-2 expression to 28.2 ± 2.1, 31.6 ± 3.4, and 31.1 ± 4.1, respectively, with P value <.0001 compared with the EST group. At the same time, the combined treatments (graviola and LDR) applied more pronounced effects indicated by the significant decrease in CD44, TGF-β, and Bcl-2 expression to 22.7 ± 2.2, 28.7 ± 2.5, and 25.2 ± 2.8, respectively, compared with EST group (P < .001).
Effect of Graviola and LDR on Expression of CD44, TGF-β, and Bcl-2. a

Abbreviations: LDR, low-dose radiation; EST, Ehrlich solid tumor; EG, EST-bearing mice group; EGR, irradiated EST-bearing mice group.
Values are expressed as the mean ± standard error of mean.
P is significantly different compared with normal control.
P is significantly different for EG and EGR groups compared with the EST group.
P is significantly different for EGR group compared with the EG group.

Flowchart showing the percentage CD44, TGF-β, and Bcl-2 expression.
Graviola and LDR induce cell cycle arrest at G1 and G2/M phase and inhibit DNA synthesis in EST. We have previously shown that treatment with graviola alone or as an adjuvant to LDR reduces the numbers of mortalities, tumor volume, and tumor growth % in a dose-dependent manner (50 mg/kg body weight/day and 2 Gy/week for 3 weeks). To determine whether graviola and LDR regulate cell cycle progression in EST, the polymorphic nuclear cells were extracted and the DNA was stained with PI, followed by FACS analysis. The treatment of EST with graviola alone or as an adjuvant with LDR resulted in dose-dependent significant increases in the percentage of cells in G1and G2/M phases causing the majority of cell cycle arrest with a concomitant reduction in cell numbers in the S phase compared with the EST group with P value <.0001 (Table 3 and Figure 3). However, the combination treatment with graviola and LDR increases significantly the G1 and G2/M phase with a reduction in S phase with P values .03, .01, and .04, respectively, compared with graviola only.
The Percentage of Cells Appearing in Each Phase of the Cell Cycle as Analyzed by Flow Cytometry. a

Abbreviations: EST, Ehrlich solid tumor; EG, EST-bearing mice group; EGR, irradiated EST-bearing mice group.
Values are expressed as the mean ± SEM.
P is significantly different in relation to the normal control.
P is significantly different for EG and EGR in relation to the EST group.
P is significantly different for EGR group compared with the EG group.

Flowchart showing the cell cycle analysis in experimental groups.
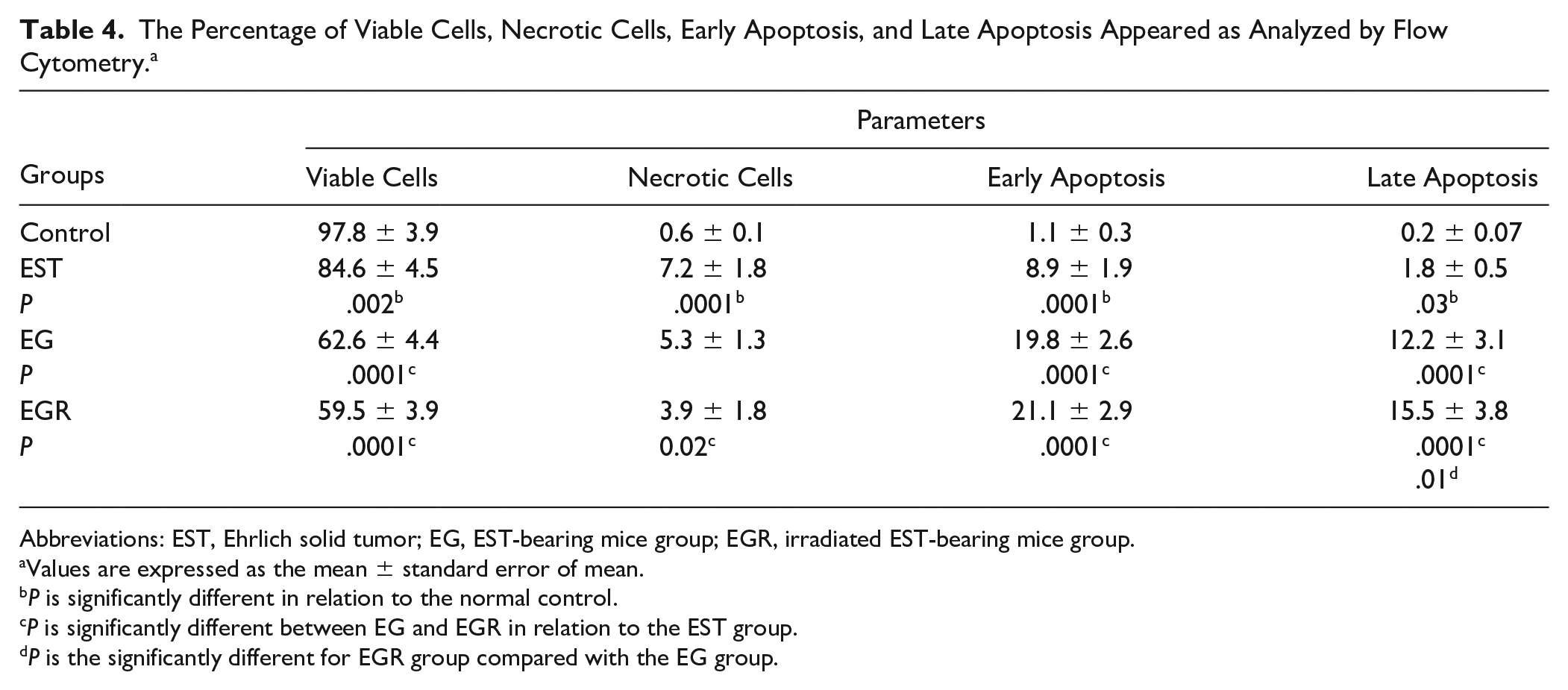
Graviola and LDR induces apoptosis in EST categorization of the early apoptotic cells, late apoptotic cells, dead cells, and also the intact cells could be carried out with PI-annexin V double staining; thus, we performed this assay to further explore cell apoptosis. To explore the potential role that graviola and LDR could play in the apoptosis of EST cells, the ApopNexin FITC apoptosis detection kit has been used to identify the formation of apoptotic cells in the 4 groups. A typical set of results for the ApopNexin FITC apoptosis detection kit is illustrated in Table 4 and Figure 4. The lower left quadrant of both histograms shows the population of the viable cells, while the lower right quadrant represents the population of late apoptotic cells. The upper left quadrant represents the necrotic cells, while the upper right quadrant represents the early apoptotic cells. The early and late apoptotic cell population downregulates significantly in the tumor group to 8.9 ± 1.9 and 1.8 ± 0.5 with P value <.0001 and <.03, respectively, and a significant increase in the necrotic cell population to 7.2 ± 1.8 with P value <.0001 compared with normal control. Meanwhile, graviola alone or as an adjuvant with LDR therapy substantially reduces in necrotic cells to 5.3 ± 1.3 but with no significant difference, and 3.9 ± 1.8 with P value <.02, respectively, compared with the tumor group. Otherwise, the graviola-alone treatment significantly increased the early and late apoptosis to 19.8 ± 2.6 and 12.2 ± 3.1, respectively, with P value <.0001 compared with the tumor group. Also, combination therapy resulted in a significant elevation in early and late apoptosis to 21.1 ± 2.9 and 15.5 ± 3.8, respectively, with P value <.0001 compared with the tumor group, and with P value <.01 compared with the graviola-alone therapy in late apoptosis.
The Percentage of Viable Cells, Necrotic Cells, Early Apoptosis, and Late Apoptosis Appeared as Analyzed by Flow Cytometry. a
Abbreviations: EST, Ehrlich solid tumor; EG, EST-bearing mice group; EGR, irradiated EST-bearing mice group.
Values are expressed as the mean ± standard error of mean.
P is significantly different in relation to the normal control.
P is significantly different between EG and EGR in relation to the EST group.
P is the significantly different for EGR group compared with the EG group.

Flowchart showing Annexin V in experimental groups.
Discussion
Murine-type tumors are common malignant tumors. The primary therapy for those tumors includes surgery, radiotherapy, chemotherapy, immunotherapy, and targeted therapy. Though these therapies have been very successful in the treatment of early carcinoma, the prognosis for advanced and recurrent diseases remains very guarded. The basic target in the use of anticancer agents is to inhibit the propagation of tumor cells or destroy them without damaging the normal cells.26,27 One such strategy to control cancer growth and metastasis without affecting normal cells is the use of phytochemicals along with low-dose ionizing radiation. It could be mentioned that graviola might be such a novel dietary agent that was initially identified with anticancer potential through preliminary screening of hundreds of food extracts and compounds for growth inhibition of cancer cells.2,26 Researchers have demonstrated that LDR-induced accelerated epigenetic changes include mi RNA expression that involved stimulating DNA repair, suppressing cell lethality, intercellular induction of apoptosis, and suppressing cancer progression.28,29
In this study, we found that graviola treatment only (EG group) at a dose of 50 mg/kg body weight/day for 30 days or as an adjuvant with LDR (EGR group) at a dose level 2 Gy once a week for 3 weeks decreased tumor volume and inhibited tumor growth. According to a study on breast cancer, treatment with Annona muricata fruit extract for 5 weeks induced tumor regression. 30 In another study, the therapeutic effects graviola treatment in mice bearing 4T1-induced tumors were assessed, and it was found that the mean tumor volume of the group treated was smaller than the untreated group. 31
Our study using low-dose γ rays with graviola in animals bearing EST demonstrated a decrease in the growth rate of tumors as well as inhibition of metastasis; these findings could be attributed to the enhancement of anticancer immunity. 13 These results are associated with expression of key cancer progression markers such as downregulation and maintenance of CD44, TGF-β, and Bcl-2.
CD44 is the most common cancer stem cell marker in various types of cancer, including breast cancer. 32 CD44 plays an indispensable role in activating survival pathways that protect cancer cells from apoptosis. 33 We have found that graviola treatment alone or as adjuvant with LDR inhibits tumor growth and represses CD44 expression in vivo without causing toxicity symptoms. Furthermore, combining treatment (graviola and LDR) appeared more efficient compared with treatment with graviola alone. This could be interpreted as suggesting graviola and LDR might prevent hyaluronic acid (HA) and CD44 binding, the event that constitutively activates phosphoinositide 3-kinase (PI3K)/AKT signaling. Several studies reveal that PI3K/AKT signaling progresses the cancer cascade and also enhances P-glycoprotein activity in doxorubicin-resistant human breast carcinoma cells. 34 The expression of P-glycoprotein coded by the multidrug resistance (MDR) gene reduces drug uptake or causes efflux of the drug out of cancer cells and also increases HA binding with CD44. 35 Furthermore, HA-CD44 binding activates protein kinase C, which in turn phosphorylates the stem cell maintenance transcription factor resulting in the upregulation of ATP-binding cassette B1 (ABCB1), a drug efflux pump inducing MDR. 36 Interestingly, cells expressing CD44 could produce a higher amount of the cytokine TGF-β, which has been shown to aid epithelial-mesenchymal transition (EMT), and promotes invasion of various tumors, including breast cancer. Previous studies have demonstrated that inhibition of the CD44 gene by RNAi method decreased TGF-β expression on HA stimulation, and subsequently inhibited breast cancer cell invasion in vitro.37,38 The data of the present study pointed to significant downregulation of TGF-β in the EG and EGR groups. However, the downregulation is more noticeable in EGR compared with EG mice. TGF-β downregulation might be due to knockdown of DNA-binding protein-1 (Id1)-CD44. It was reported that anti–TGF-β compound could decrease the glioma-initiating cell population by inducing the trans differentiation of the CD44-high/Id1-high population into a CD44-low/Id1-low population. 39 The potential synergism between graviola and LDR could be contributed to the extra manifested downregulation of TGF expression over its expression in EG mice. The combined therapy resulted in activation of intercellular signaling leading to enhancement of natural anticancer mechanisms (cancer barrier). 40 Our results are confirmed by the finding of Kim et al, 41 where the high level of TGF-β expression was associated with relapse-free and overall survival of breast cancer, and cancer proliferation is reduced by blocking of TGF-β. Also, a decrease in tumor growth–promoting Tregs-altered cytokine responses (eg, a decrease in TGF-β levels).13,42
The previous study indicates that epidermal growth factor receptor (EGFR) has been identified as a promising target for breast cancer therapies. Binding of a ligand such as EGF or TGF results in a signaling cascade that produces diverse effects, including cell migration, maturation, differentiation, metastasis, angiogenesis, and inhibition ofapoptosis. 43 The progress of solid tumor in the EST group is associated with increased expression levels of several typical oncogenes, such as CD44 and Bcl-2 (Figure 2). In many studies the increase in the expression of CD44 and Bcl-2 have been observed to be highly correlated with proliferative ability and blocking the apoptosis pathway of many humancancers.44-46 Bcl-2 neutralizes the proapoptotic effect of Bax, whereas CD44 has a role in cell survival and proliferation involving alteration of multiple molecules that regulate the antiapoptotic processes such as Bcl-2.33,47 Treatment with graviola alone or in combination with LDR significantly inhibits the expression of Bcl-2 and CD44 (Figure 2).
This might imply the induction of apoptosis (Figure 4) and decrease of tumor growth (Figure 1) observed in the present study. In agreement of our finding, previous reports showed that extracts of different graviola parts induced apoptosis in cancer cell lines (breast). 31 The capacity of the graviola to induce apoptosis could be due to the presence of naturally produced secondary metabolites.48,49
It is worth mentioning that knocking down of CD44 from colon cancer cells leads to reduced expression of antiapoptotic molecules like Bcl-2, Bcl-XL, and increased level of apoptotic molecules like Bax and caspases-3/8/9. 47 Moghadamtousi et al reported that graviola induced apoptosis through activation of caspases 3/7 and 9, upregulation of BAX, and downregulation of Bcl-2 at the mRNA and protein levels. 50 Graviola leaf might induce apoptosis in MCF-7 cells by an intrinsic pathway related to cytochrome c migration to the cytosol as the result of Bax protein binding and Bcl-2 suppression. The ability to block cell cycle progression in cancer cells can effectively elevate the anticancer potential of natural products. 51 In mice that received graviola alone or graviola combined with LDR, the increase of G0/G1 phase cells was accompanied by a significant decrease of S-phase and G2/M phase cells (Figure 3), which means that graviola or graviola/LDR might block the G1/S transition and induce dose-dependent G0/G1 cell cycle arrest. Significant increases of cell population at the sub G0/G1 phase could suggest the incidence of cell cycle arrest in the EG and EGR mice groups. Moghadamtousi et al stated that graviola extracts were shown to have the potential to induce G1 cell cycle arrest. 52 The significant increases in the G1/G2 population in EGR mice more than EG mice could be interpreted in the view that DNA mismatch repair (MMR) proficiency appears to be associated with radiosensitivity. LDR enhances sensitivity in G2/M checkpoint and p53-caspase-3-dependent apoptosis as well as persistent RAD 48 leading to cell cycle arrest and regulation. 53 Evidently, graviola extracts have the capacity to regulate the cell cycle machinery, leading to cell cycle arrest and inhibition of cell proliferation. 54 Also, graviola leaves could inhibit the proliferation of human breast cancer cells by arresting the cell cycle and inducing apoptosis. 30 A dysfunction or inability of cells to execute to remove cancer cells was observed in multiple cancer types as defects within the intrinsic pathway in breast cancer progression. 55 Flow cytometric analysis of annexin V/FITC distinguished a separate population of early apoptotic, late apoptotic/necrotic cells, and living cells as a result of the employment of the high affinity binding of annexin V to phosphatidylserine (PS), 52 which is exposed at the surface of apoptotic cells and has broadly been used as a probe to measure apoptosis in vivo and in vitro. 56 As depicted in Table 4 and Figure 4, the total apoptotic percentages (early apoptotic and late apoptotic cells) in EG mice and EGR mice are higher than EST mice, rather than the necrotic cells percentage in EG and EGR groups, which were lesser than in EST mice. Corsten et al reported induction of cancer cells apoptosis in a time-dependent manner, whereas the combined treatment of graviola and LDR augment apoptosis. 56 In other studies, graviola extract exaggerates the inhibition of growth capabilities as well as surging apoptosis in lung A549 and breast MDAMB-468 cells.30,57
It could be suggested that graviola inhibits tumor growth via its capabilities to downregulate the proliferation signals (CD44 and TGF-β), suppress cell cycle G0/G1 phase, and arrest G2/M phase, as well as to stimulate cancer cells apoptosis through stimulation of PS expression and suppression of antiapoptotic Bcl-2. It seems that combining LDR with graviola is more efficient in controlling tumor growth compared with graviola alone. Therefore, this combination between graviola and LDR might be considered as a promising anticancer strategy and could represent a beneficial base for clinical applications. However, further studies applied to different types of tumors and cancer cells in response to the graviola/LDR treatment are strongly recommended to complete the picture and confirm the results emerging from the present study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


