Abstract
Background:
Cancer and its treatment significantly impact the quality of life (QOL) of adolescent and young adult (AYA) patients. This meta-analysis examined the effects of exercise interventions on the generic and cancer-specific QOL of AYA cancer patients and survivors.
Methods:
Four databases were searched from January 2010 to September 2019. RevMan 5.3 was used to synthesize the effects of exercise intervention on the QOL of AYA cancer patients and survivors.
Results:
A total of 11 eligible studies have been included in this paper. Direct outcome comparisons found that pooled overall effects on generic QOL were in favor of exercise interventions, but only with marginal significance (Z= 1.96, P = 0.05). When performing the subscale analysis of the generic QOL, three trials assessed the effects of exercise interventions on the emotional domain of QOL, and the weighted mean difference (WMD) for the overall intervention effect was 3.47 (95% confidence interval [CI] = 0.42 to 6.51). Additionally, exercise interventions increased the minutes of physical activity per week undertaken by AYA cancer patients and survivors (Z= 2.88, P = 0.004).
Conclusion:
Exercise interventions had positive effects on generic QOL and increased the minutes of physical activity per week undertaken by AYA cancer patients and survivors. In addition, exercise intervention programs appear to be safe, as there were no studies that reported adverse events. Future research, with rigorous methodological standards and larger sample sizes, should be designed to confirm the positive effects of exercise interventions on the QOL of AYA cancer patients and survivors.
Introduction
Cancer is the most common cause of death among adolescent and young adult (AYA; aged 13 to 39 years) cancer patients, and there is an increasing number of AYAs being diagnosed with cancer. In the United States, about 70 000 AYAs are diagnosed with cancer annually. 1 During the past 2 decades, research has shown that disease-free survival rates for AYA cancer survivors have improved from 80% to 88% in Australia. 2 The overall 5-year survival rates of AYA cancer survivors have increased to 80% in several high-income countries, and this has been attributed to progressive improvements in active treatment and supportive care. 3 However, improved survival rates are often accompanied by cancer-related sequelae, resulting in AYA cancer survivors living with potentially lifelong adverse health consequences. 4 Common cancer-related sequelae include physical deconditioning, cognitive changes, functional deficits, and limitations resulting from extensive cancer therapies, and a negative impact on psychosocial functioning. 4 All of these sequelae result in a deterioration in the quality of life (QOL) of AYA cancer survivors.
While there is growing recognition that exercise is effective as a nonpharmacological intervention in diminishing cancer-related sequelae for AYA cancer survivors, 5 more than half of AYA cancer survivors nevertheless remain sedentary. 6 According to the physical activity guidelines of Centers for Disease Control and Prevention, AYAs are advised to engage in at least 150 minutes of moderate physical activity per week. 7 However, most AYA cancer survivors do not meet these physical exercise guidelines. 8 Recent evidence suggests that regular physical exercise can improve both physical and psychosocial health in cancer survivors, and there has been widespread attention on the health benefits of exercise interventions on cancer survivors. 9 Assessing the QOL outcomes and its domains can provide prognostic and predictive information for clinicians, helping them understand the experiences of AYA cancer survivors and in the developing of effective lifestyle interventions for this target study population.10,11
There have been previous systematic reviews investigating the effects of exercise interventions on the QOL of cancer survivors. One review 10 examined the effects of exercise interventions on the QOL of adolescent cancer survivors, aged 13 to 19 years. This systematic review reported that exercise intervention appears to be safe and feasible, and improve the QOL of adolescent cancer survivors. However, this meta-analysis did not include all AYA cancer survivors. Because AYAs are biologically, physically, and psychosocially vulnerable to cancer in a unique way, they may respond differently to exercise interventions. 3 Furthermore, the recent systematic review by Munsie and colleagues, 4 investigating the health benefits of exercise interventions on AYA cancer patients, did not perform quantitative analysis for the pooled effects of exercise intervention on this target study population. The aim of the present meta-analysis, therefore, is to quantitatively evaluate the most recent studies on the effects of exercise interventions on the QOL of AYA cancer patients and survivors, and to identify the implications for future research.
Methods
Data Sources and Search Strategy
This meta-analysis involved a literature search in 4 electronic databases: PubMed, EMBASE, Scopus, and CNKI, from January 1, 2010, to September 30, 2019. Articles published in both English and Chinese were included. Search terms used a combination of key words and medical subject headings. These included adolescent, young adult, cancer, cancer treatment, cancer survivorship, exercise, physical exercise, physical activity, motor activity, and quality of life.
Eligibility Criteria
Patients and survivors with childhood-onset cancer were included. In line with the conventional age-related definition, AYAs were defined as persons aged between 13 and 39 years. 3 Patients with adulthood-onset cancer were excluded. Interventions included all exercise interventions. Controls included other active or supportive care interventions, wait-list controls, or usual/standard care. The primary outcome was generic or cancer-specific QOL outcome measures. Secondary outcomes included the minutes of moderate physical activity per week, and any adverse event directly due to exercise interventions. Study designs included case-controlled and randomized controlled trials (RCTs).
Data Extraction and Risk of Bias Assessment
For each included study, data were extracted by 2 independent investigators (XZ, MX), who extracted the characteristics of each included trial, including key information on the QOL outcomes. In discussing any disagreements that arose, a third investigator (YZ) was always involved. For the methodological quality assessment, risk of assessment for each included trial used the Cochrane Risk of Bias Assessment tool, including random sequence generation, allocation concealment, blinding participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. 12 Similarly, any disagreement in terms of methodological quality assessment was resolved by discussion. In these cases, the third investigator (YZ) spoke with both parties in order to achieve a consensus.
Data Synthesis and Analysis
The Cochrane Collaboration’s Review Manager (RevMan 5.3) was used to generate the pooled effect of exercise interventions. 13 Statistical heterogeneity was calculated with χ2 test and I2 statistics. A χ2 test with a P value >.1 and an I2 value <50% were considered indications of statistical homogeneity. 12 If the heterogeneity was high, the random effect model was used to estimate the intervention effect size. Otherwise, the fixed effect model was used to estimate the intervention effect size. If data for standard deviations were missing, they were estimated using the standard deviation from other related information such as standard errors or confidence intervals (CIs). 13 Values of P < .05 were considered statistically significant.
Results
Characteristics of Included Studies
Figure 1 shows the full study selection procedure. A total of 707 articles were identified. After removing the duplicates, 397 titles and abstracts were screened, 42 full-text articles were assessed for eligibility, and 11 studies6,14-23 were included in this meta-analysis.
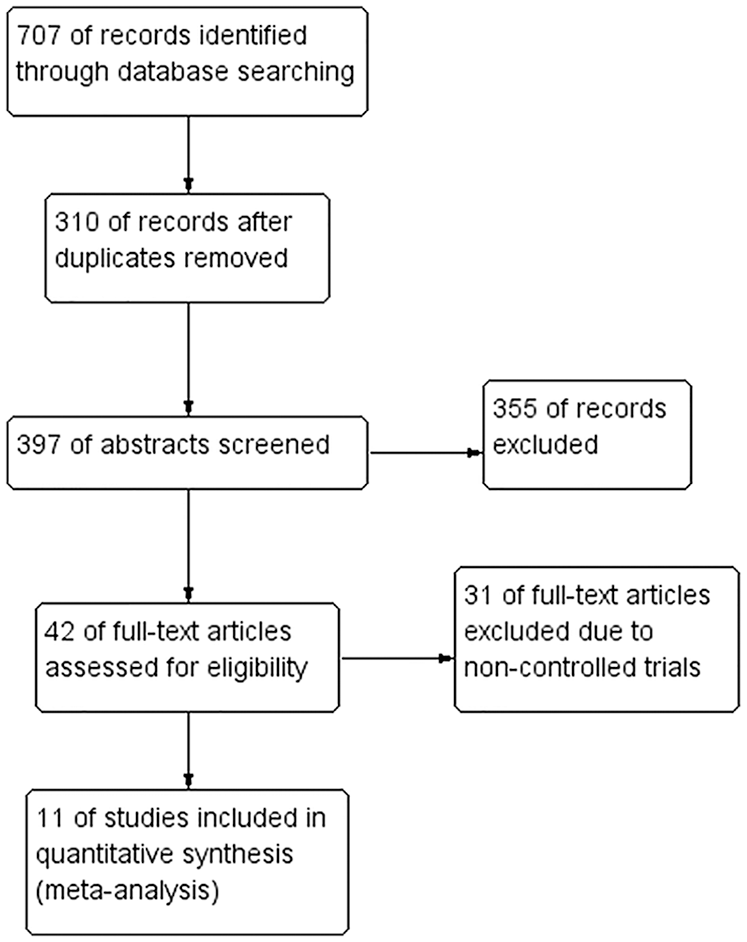
PRISMA flow diagram of study selection.
Table 1 summarizes the characteristics of these trials. The data are organized under the headings of study design, characteristics of participants, intervention details, outcome measures, and main findings. Two of these15,19 were case-controlled trials, while other included studies were RCTs.6,13,14,16-18,20-23 There were both generic and cancer-specific QOL measures used for measuring the QOL of AYA cancer patients and survivors. Of these 11 studies, 1 study used generic QOL measures of Short Form Health Survey-36, 14 or the PedsQL4.0 (Pediatric Quality of Life Inventory4.0) Generic Core Scale, 16 1 study used cancer-specific QOL measures such as FACT-G (Functional Assessment of Cancer Therapy–General), 21 and only 1 study used both generic and cancer-specific QOL measures such as the PedsQL4.0 Generic Core and Cancer Module Scales. 18 The total duration of interventions ranged from 10 to 24 weeks, frequency of interventions varied from 3 times/week to daily session, time per session ranged from 20 to 60 minutes. The main mode of intervention delivery was by the Internet (Table 1).
Characteristics of 11 Included Studies.
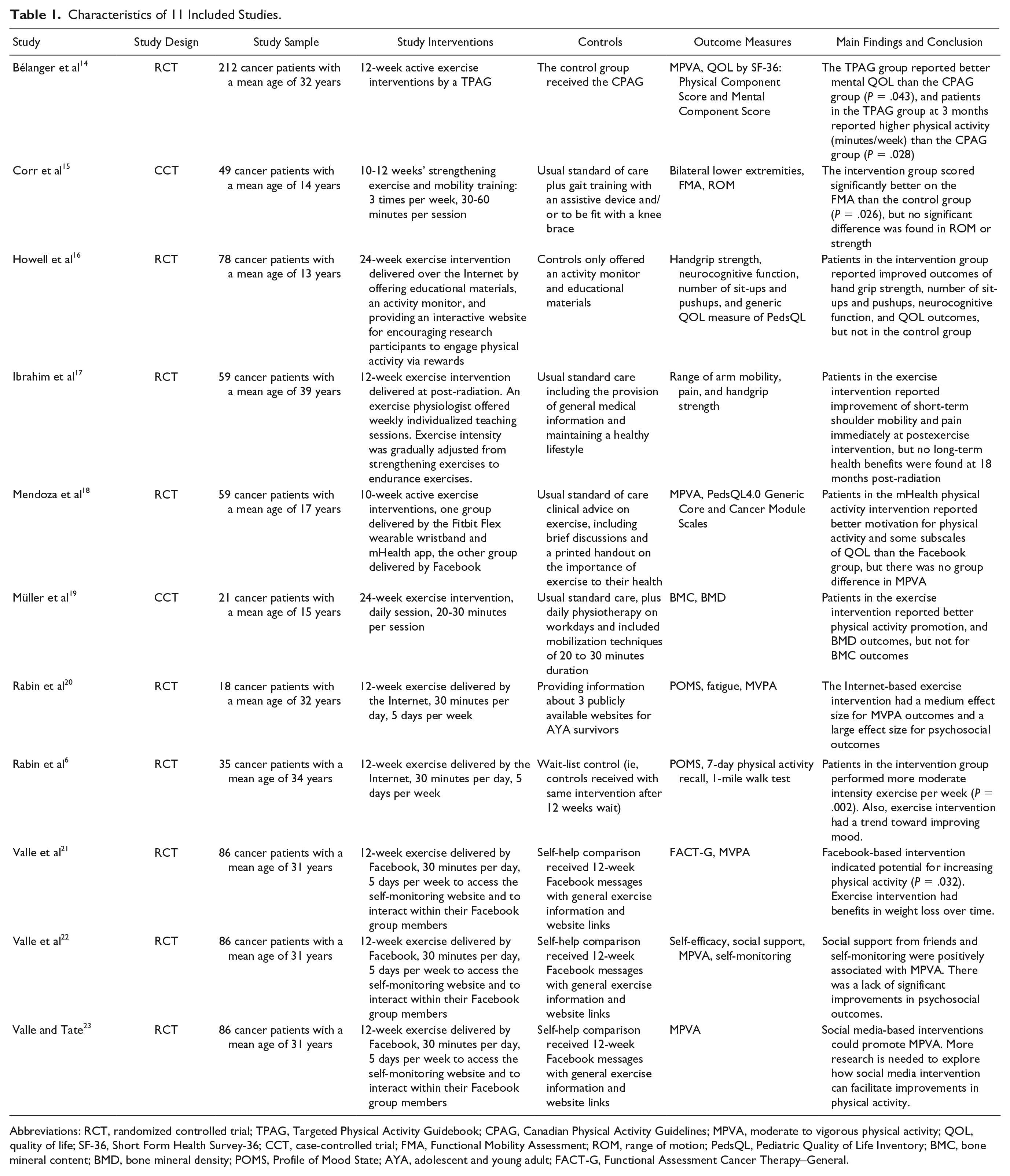
Abbreviations: RCT, randomized controlled trial; TPAG, Targeted Physical Activity Guidebook; CPAG, Canadian Physical Activity Guidelines; MPVA, moderate to vigorous physical activity; QOL, quality of life; SF-36, Short Form Health Survey-36; CCT, case-controlled trial; FMA, Functional Mobility Assessment; ROM, range of motion; PedsQL, Pediatric Quality of Life Inventory; BMC, bone mineral content; BMD, bone mineral density; POMS, Profile of Mood State; AYA, adolescent and young adult; FACT-G, Functional Assessment Cancer Therapy–General.
Quality of Study Methods
Figure 2 presents the results of the risk of bias assessment. The major sources of potential bias identified were a failure to blind study subjects or research personnel; incomplete outcome data; and selective outcome reporting. Due to the nature of exercise intervention, it may be difficult to blind participants in terms of intervention delivery. Of 11 trials, 4 trials14,15,19,20 failed to provide clear information about blinding the outcome assessors to minimize potential methodological bias. For bias of incomplete outcome data and selective outcome reporting, 3 trials21-23 came from the same RCT study but reported different study outcomes.

Risk of bias summary.
Effects of Exercise Interventions on Study Outcomes
Direct outcome comparisons found that the pooled overall effects of generic QOL were in favor of exercise interventions, but only with marginal statistical significance (Z = 1.96, P = .05; Figure 3). When performing the subscale analysis of the generic QOL, 3 trials assessed the effects of exercise interventions on the emotional domain of QOL, and the weighted mean difference for the overall intervention effect was 3.47 (95% CI = 0.42-6.51; Figure 3). For cancer-specific QOL, there were no statistically significant differences between exercise intervention and controls (Figure 4). Additionally, exercise interventions increased the minutes of physical activity per week undertaken by AYA cancer patients (Z = 2.88, P = .004; Figure 5).

Generic quality of life (QOL) comparison between exercise intervention and controls at postintervention.

Cancer-specific quality of life comparison between exercise intervention and controls at postintervention.

Changes in the minutes of physical activity per week between exercise intervention and controls.
In order to determine the strength of confidence in the intervention effect size, publication bias for the included trials was visually inspected by the criterion of symmetry. From Figure 6, the funnel plot of the pooled intervention effect size of the generic QOL was with symmetry distribution, indicating that these trials had limited publication bias. Of the 11 trials that were included, there were no studies reporting any adverse event directly related to exercise interventions.

Funnel plot of comparison based on generic quality of life (QOL) outcome.
Discussion
This meta-analysis examined the pooled effects of exercise intervention on the QOL in AYA cancer patients. Of the 11 trials, only 4 of them were included in statistical pooling because other trials did not present enough data about the QOL of AYA cancer patients. Overall, the pooled effects of generic QOL were in favor of exercise intervention, especially in the subdomain of emotional functioning. The pooled results of 3 studies involving 349 subjects showed that the weighted mean difference for the emotional QOL was 3.47 (0.42, 6.51). Cancer-specific QOL was not in favor of exercise interventions, which may be due to using insensitive QOL measures in AYA cancer patients. Nevertheless, this meta-analysis found that participants in the exercise intervention group reported significantly increased engagement in physical activity, consistent with the findings of previous research. 4
Across the trials included in this meta-analysis, the number of research participants varied from 18 to 212. The mean age of participants ranged from 13 to 39 years. There were different QOL outcome measures, including pediatric QOL scales and cancer-specific QOL for general age population. In addition, a wide range of cancer diagnoses, coupled with different study stages (ie, implementing exercise intervention either during cancer treatment or else after treatment was completed) were noted. Moreover, the intensity of exercise intervention programs varied from light physical activity like walking to vigorous physical activity such as aerobic exercise. Consequently, these variations may contribute to the overall cancer-specific QOL without statistical significance.
Several limitations of this meta-analysis should be noted. First, most trials included in this meta-analysis had a high risk of bias. Future trials with higher standards of methodological quality are needed in order to minimize such methodological bias. Second, although this meta-analysis included AYA-specific cancer patients based on the international definition of AYAs from 13 to 39 years old, future studies should perform subgroup analysis in terms of different age groups, such as 13 to 25, and 26 to 39 years, as in a cancer context the transition period of AYA patients poses unique challenges, 4 so they may respond to exercise interventions differently during different transition stages. And, third, this meta-analysis included trials with small sample sizes, and used insensitive QOL measures (eg, FACT-G, not specific to AYA cancer patients). This limits the generalizability of the study’s findings.
This meta-analysis also had several important implications for future research and clinical practice. First, exercise intervention programs were varied in terms of delivery mode (group-based vs individual-based; delivered through online social media vs off-line intervention programs; printed exercise intervention materials vs self-help intervention); the intervention duration varied from 10 to 24 weeks; the intensity of exercise varied from weekly sessions to daily sessions. Therefore, future research is needed to determine the essential attributes of exercise (mode, duration, and intensity) for maximizing the positive effects on the QOL of AYA cancer patients. Second, this meta-analysis included trials using nonspecific QOL measures to evaluate cancer-specific QOL outcomes for AYA cancer patients. It is recommended that future research uses more sensitive QOL measures (eg, PedsQL–Cancer Module Scales) to capture exercise interventions for the cancer-specific QOL in AYA cancer patients. And, third, considering the positive effects of exercise interventions on the generic QOL and increased physical activity levels of AYA cancer patients, oncologists should adopt exercise interventions as an integral component of the management of QOL in this target study population.
Conclusion
Exercise interventions led to improvements in generic QOL and increased the minutes of physical activity per week undertaken by AYA cancer patients and survivors. In addition, exercise intervention programs appear to be safe, as there were no studies that reported adverse events. Future research of rigorous methodological quality and employing larger sample sizes should be conducted to confirm the positive effects of exercise interventions on the QOL of AYA cancer patients and survivors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 71804073). All authors have no conflict of interest.
