Abstract
Background:
Breast cancer is the most common cancer type in women and quality of life an essential part of patients’ well-being. Although the treatment with mistletoe extracts is covered by multiple cancer guidelines and reviews, it is uncertain whether mistletoe extracts can improve the quality of life in breast cancer patients. We therefore performed a systematic review and meta-analysis on this topic.
Methods:
This systematic review included randomized clinical trials (RCTs) and non-randomized studies of intervention (NRSIs) comparing the quality of life in breast cancer patients treated with mistletoe extracts as add-on therapy to control groups treated conventionally. We searched previous systematic reviews and multiple databases until January 2023. We conducted a meta-analysis and assessed the risk of bias according to the Cochrane Handbook via RoB 2 and ROBINS-I and the certainty of evidence via GRADE, respectively.
Results:
Nine RCTs and 7 NRSIs with 833 and 2831 participants, respectively, were included. The pre-post changes for the quality of life resulted in a pooled standardized mean difference for RCTs of SMD = 0.61 (95% CI 0.47-0.75; P < .0001) and for retrospective NRSIs of SMD = 0.46 (95% CI 0.10-0.82; P = .01). The risk of bias was low to high for the RCTs and serious for all NRSIs. The certainty of evidence was moderate for RCTs and very low for NRSIs.
Discussion:
Our results indicate a clinically relevant, medium-sized effect of mistletoe extracts on the quality of life in breast cancer patients which may be based on the immunomodulating effects of mistletoe extracts during chemotherapy. The limitations of evidence include the risk of bias which is mainly caused by the difficulty of blinding. Further RCTs and real-world evidence need to confirm this result, especially in the setting of neoadjuvant chemotherapy and in breast cancer survivors.
Introduction
Breast cancer is the most frequent type of invasive cancer in women, with 2.3 million new cases worldwide in 2020. 1 Breast cancer and its standard medical treatment lead to an important decrease in quality of life (QoL), amounting to 19.6 million disability-adjusted life years globally for 2016. 1 QoL has been defined as “an overall general well-being that comprises objective descriptors and subjective evaluations of physical, material, social and emotional well-being together with the extent of personal development and purposeful activity, all weighted by a personal set of values.”2,3 Apart from antineoplastic therapy, QoL in breast cancer patients is also influenced by multiple factors such as age, disease stage, and social and educational status.4,5 In order to support QoL in breast cancer patients pharmacological and non-pharmacological interventions have been investigated. 6
First developed by Wegman and Steiner, extracts from the European mistletoe (Viscum album L.) are authorized medicinal products with indications in supportive cancer care.7,8 Mistletoe extracts (ME) are injected subcutaneously 2 to 3 times a week, during and beyond standard oncologic treatments. 9 Recent meta-analyses of clinical studies concluded that application of ME produces a significant, medium-sized effect on QoL in cancer and may prolong survival.10 -12 The most studied bioactive components are viscotoxins and lectins, but ME also contain other compounds such as flavonoids, phenolic acids and polysaccharides. 13 One possible clinically observed mode of ME is immunomodulation, which may reduce inflammatory markers and the rate of neutropenia. This, together with a peripheral endorphin-release, may form the basis for an improvement of quality of life and reduction of side effects related to standard oncological therapies.14,15 Further research is necessary, as modes of action are not fully elucidated due to the complexity of the bioactive compounds and since the phytochemical composition depends on the host tree and the way of pharmaceutical preparation 13 Different preparations of ME (eg, Abnobaviscum, Helixor, Iscador, Iscucin, Lektinol) are used in clinical practice for various cancer entities, including breast cancer. Mistletoe therapy is individualized according to each patient’s clinical case and treatment response. 16
ME are included in multiple guidelines,17 -19 but certain aspects of their efficacy continue to be discussed. ME are noted in the recommendations of the German Working Group on Gynecological Oncology, the guidelines of the American Society of Integrative Oncology (endorsed by the American Society for Clinical Oncology) and of the German Cancer Society.17 -19 In all 3 of these, ME are indicated as optional treatment to improve QoL of solid tumor patients. This indication comes with a level of evidence 1a, attributed to therapies assessed via systematic reviews of randomized clinical trials (RCTs). 19 The main points of discussion are the lack of double-blinded RCTs and the heterogeneity of results.20,21 The heterogeneity could be explained by the broadness of previous meta-analyses since none of the previous meta-analyses focused on 1 cancer entity only.12,22 However, a subgroup analysis yielded smaller heterogeneities for breast or lung cancer studies when compared to all cancer types. 12 In addition, our earlier work included prospective studies only 12 and in turn neglected retrospective non-randomized studies of intervention (NRSIs) that may contribute to the evidence. This is particularly relevant in the light of a recent GRADE guideline regarding the integration of NRSIs into the assessment of evidence certainty 23 when no high level of evidential certainty from RCTs alone exists and NRSIs provide complementary data. 23 The latter is the case here since individualization is common among ME treatment and real-world data, as provided by NRSIs, are an important complement to RCTs in personalized medicine. 24
Therefore, the aim of this study is to systematically review the literature on ME treatment in breast cancer patients in order to synthesize both RCTs and NRSIs through meta-analysis and evidence assessment.
Methods
The review has been reported according to PRISMA, 25 and the protocol was registered with the PROSPERO database prior to analysis (CRD42023388900).
Literature Search
Studies were searched for in 2 previous systematic reviews.12,22 In addition, Medline, Embase, the Cochrane Central Register of Controlled Trials, Clinicaltrials.gov, Google scholar, and the database of the Society for Cancer Research 26 were screened. Our search strategy combined the 3 terms “breast cancer,” “quality of life” and “mistletoe” including their synonyms and was adopted for each database. As an example, the search for Medline can be seen in the Supplemental (see Supplemental 1).
We included RCTs as well as NRSIs which compared breast cancer patients treated with ME to a control group. In case of insufficient data we contacted the authors of Oei et al for additional information. 27
Extraction
Data was extracted from each study independently by 2 reviewers (HW, ML) and covered the following items: lead author, year of publication, country where the study was conducted, patient characteristics (age, sex, number of patients in each arm), duration of study, type of study (interventional vs non-interventional, randomized vs non-randomized, blinded vs not blinded, single vs multi-center); additional therapy (eg, chemotherapy); number of drop-outs in each study arm; ME preparation (eg, Eurixor); control treatment (eg, placebo); effect size of primary outcomes plus standard deviation; applied instrument to measure primary outcomes; statistics according to intent-to-treat analysis (yes/no); sponsoring of study. Two reviewers (HW, ML) compared the independent extractions and resolved differences by discussion.
Bias Assessment
Risk of bias assessment was performed by 2 authors (HW, ML) according to the Cochrane handbook, that is, Rob-2 for RCTs and ROBINS-I for NRSIs.28 -30
Statistical Analyses
Data from RCTs and from NRSIs were analyzed separately according to chapter 24 of the Cochrane Handbook. 28 The effect sizes are presented as standardized mean differences (SMD) of the changes from baseline to post-intervention between the verum and the control group for RCTs and NRSIs. Odds ratios (OR) were converted to SMD following the Hasselblad and Hedges and Hedges’ method if necessary.31,32 The Mann Whitney statistics were transformed to SMD as described by Rahlfs et al. 33
The data were analyzed using R version 4.2.2 including the packages meta as well as Comprehensive Meta-Analysis V.2 to perform a meta-analysis in order to obtain a pooled estimate of the effect of ME on QoL. We assessed the heterogeneity between studies using the Cochrane Q test and quantified it by the index of heterogeneity (I2), the heterogeneity variance τ2 and a prediction interval.34,35 If I2 was 50% or higher and the confidence interval of τ2 did not contain 0, we applied a random-effect meta-analysis in accordance with the study protocol, otherwise we used a fixed-effect model. We conducted a meta-regression with RCTs and regressed the age of the patients, the study year, and the study duration on effect-size.
Sensitivity Analysis
Sensitivity analyses were conducted to examine the robustness of the results. To reduce heterogeneity, 2 prospective NRSIs36,37 were not included in the main analyses, following Higgins et al 28 but added in the sensitivity analyses. In addition, we excluded 2 studies that measured self-regulation37,38 and one study which applied ME i.v., 39 respectively. If studies reported no validated scale for measuring QoL but multiple surrogate parameters,40 -42 we used the measure with the highest comparability to the other effect sizes in the analyses (eg, symptom scores over performance status) and inserted other measures for sensitivity analyses.
Subgroup Analyses
Subgroup analyses were conducted for the following moderators: blinding status, tumor stage, ME preparation, and risk of bias.
Publication Bias
We checked for signs of a publication bias by examining funnel plots, Egger’s test 43 and Duval and Tweedie’s trim-and-fill procedure. 44
Certainty Assessment
The certainty of evidence was assessed with GRADE. 45 In short, the rating starts at “high” for RCTs and at “low” for NRSIs and can be rated down 1 or 2 levels for the 5 domains risk of bias, inconsistency, indirectness, imprecision, and publication bias. The quality of evidence can be rated up for NRSIs in case of large effect sizes, a dose-response gradient or if residual confounding supports inferences regarding the treatment effect.
Results
Study Selection and Characteristics
The literature search combined the search of database with the results of 2 earlier systematic reviews.12,22 The search flow is shown in Figure 1.
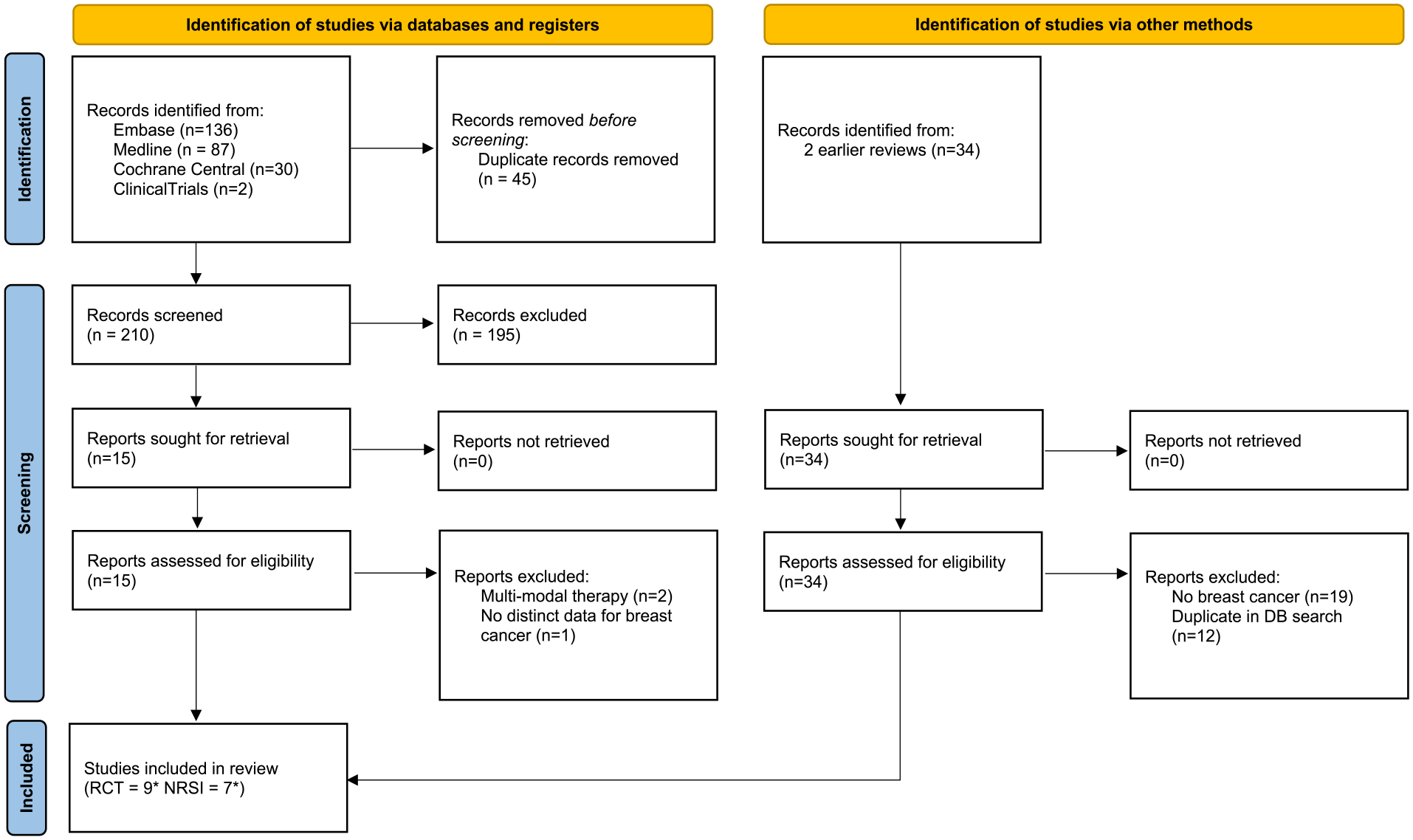
Search flow (* 37 with 2 cohorts (1 RCT and 1 NRSI) in 1 publication).
Sixteen studies presented in 15 publications have been included of which 9 are RCTs37 -39,46 -51 7 are NRSIs.27,36,37,40 -42,52 One study included 2 cohorts with one being a RCT whereas the other is a NRSI. 37 Two RCTs were double-blinded48,49 7 are unblinded. Two NRSIs were prospective36,37 while the remaining 5 were retrospective. The characteristics of all studies are presented in Table 1.
Characteristics of Included Studies.
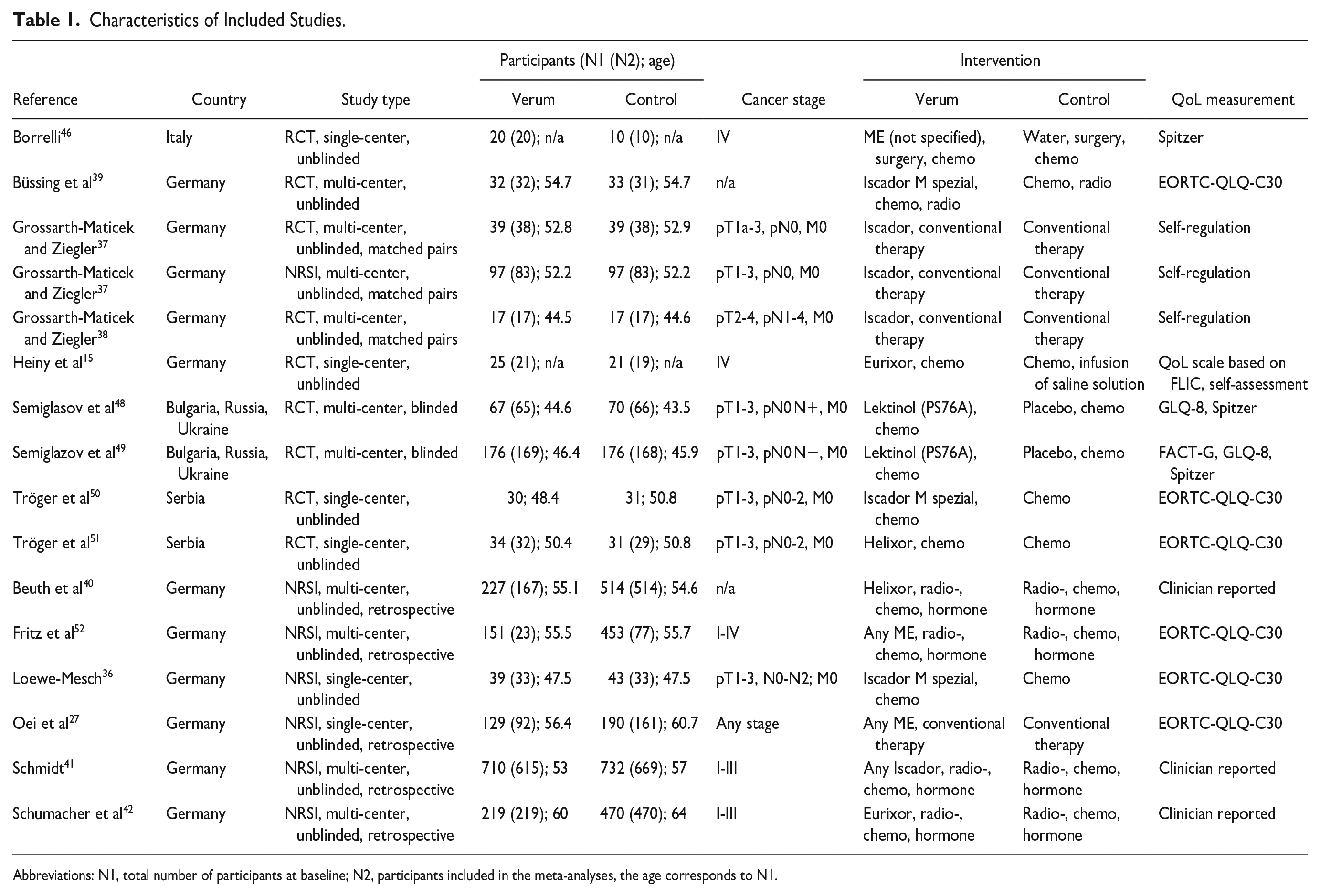
Abbreviations: N1, total number of participants at baseline; N2, participants included in the meta-analyses, the age corresponds to N1.
Results of Synthesis
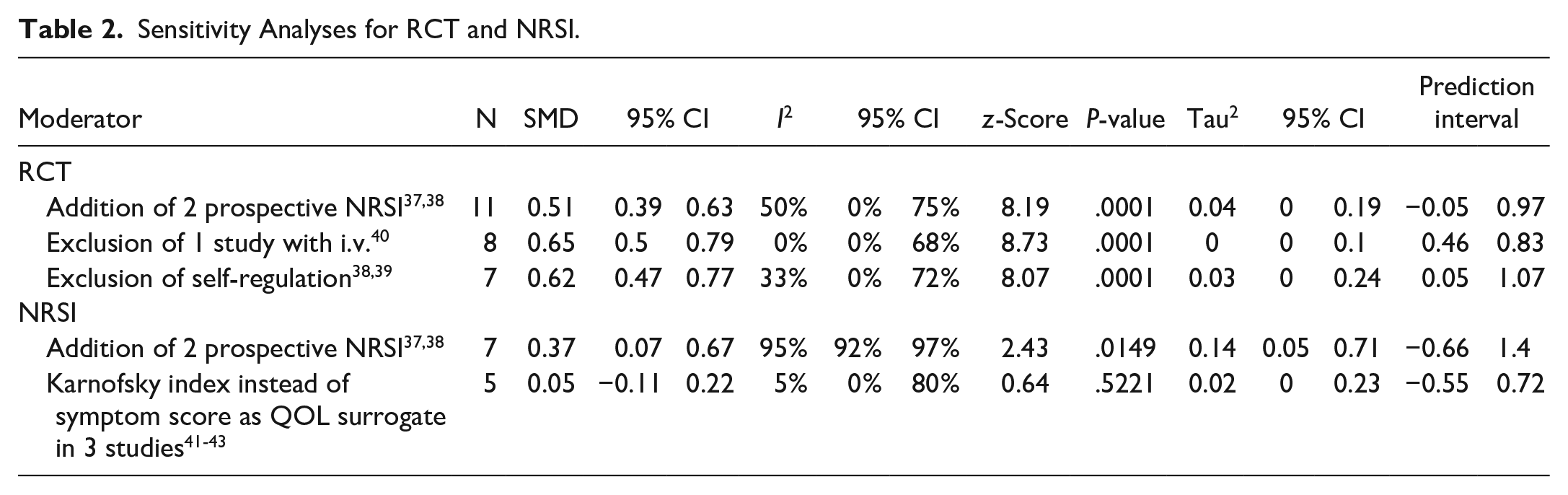
For RCTs, a fixed-effect meta-analysis was performed, shown in Figure 2. The estimated pooled effect size is SMD = 0.61 (95% CI 0.47-0.75). The between-study heterogeneity was estimated with I2 = 12.3 % (95% CI 0.0%-54.1%). The heterogeneity variance was τ2 = 0.01 (95% CI 0.000-0.13) and the prediction interval ranged from 0.31 to 0.86, indicating neither heterogeneity nor negative intervention effects for future studies. The sensitivity analyses (see Table 2) and the subgroup analyses (see Table 3) shows that the analysis is robust against alternative sets of conditions and in subdomains of the total population. There is a tendency for methodologically more rigorous studies to yield higher or equally high effect sizes compared to less rigorous ones. Blinded studies showed a tendency for a higher effect size (SMD = 0.69, 95% CI 0.38-0.97) than unblinded ones (SMD = 0.47, CI 0.27-0.68).

Fixed-effect meta-analysis pooling standardized mean differences from RCT regarding the effect of mistletoe extracts versus control on quality of life in breast cancer patients.
Sensitivity Analyses for RCT and NRSI.
Subgroup Analyses for RCT and NRSI.
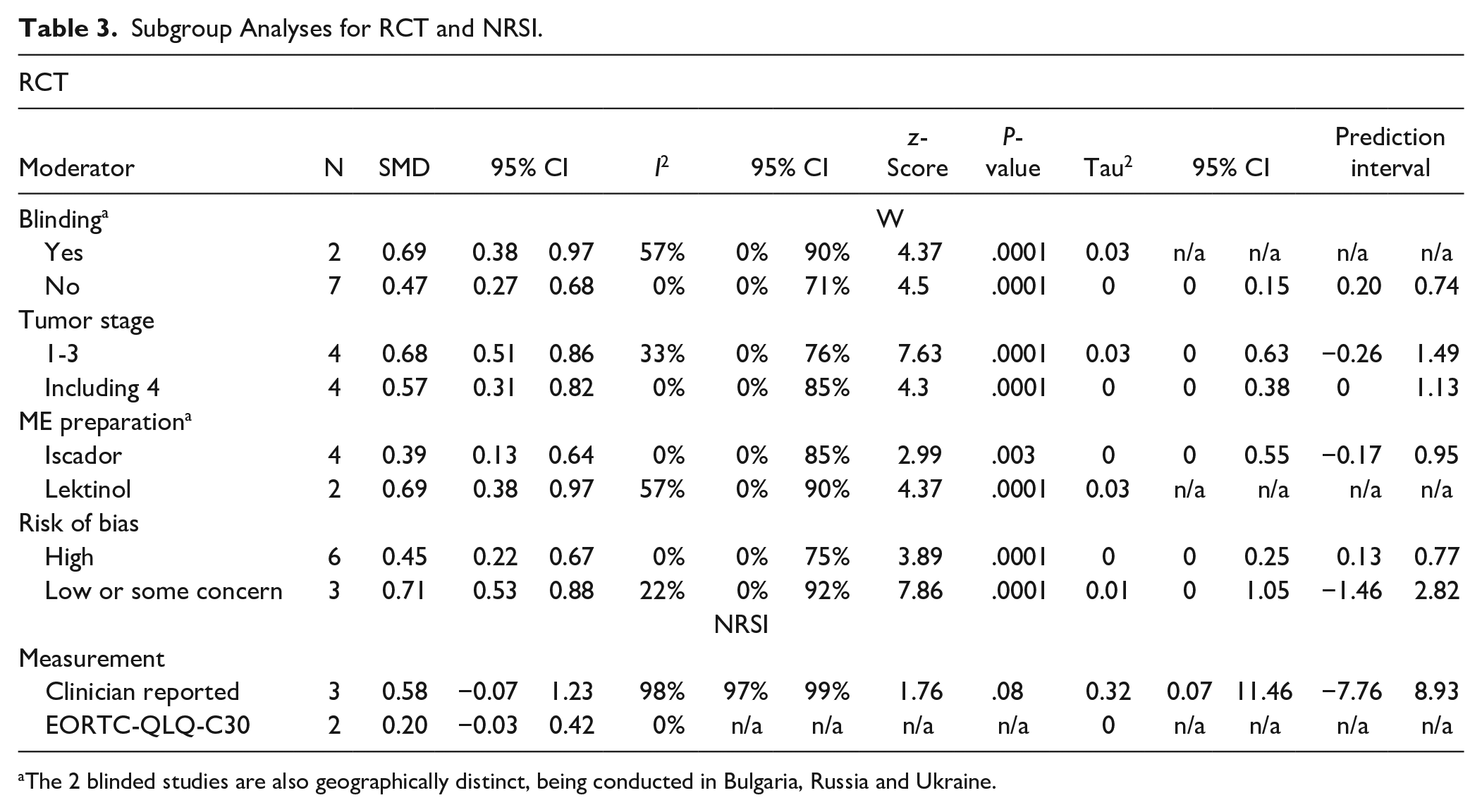
The 2 blinded studies are also geographically distinct, being conducted in Bulgaria, Russia and Ukraine.
For retrospective NRSIs, the pooled effect size has been calculated using a random-effect model, yielding a SMD = 0.46 (95% CI 0.1-0.82), as shown in Figure 3. The between-study heterogeneity was estimated with I2 = 96.5 % (95% CI 94.1%-97.9%), the heterogeneity variance was τ2 = 0.15 (95% CI 0.05-1.25) and the prediction interval ranged from −0.92 to 1.84. Thus, all 3 parameters reveal statistical heterogeneity regarding the NRSI analysis. A subgroup analysis, shown in Table 3, reveals that different types of measurement (self-reported vs clinician-reported) result in tendentially different effect sizes.

Random-effect meta-analysis pooling standardized mean differences from NRSI regarding the effect of mistletoe extracts versus control on quality of life in breast cancer patients.
Meta-regression identified age of the patients as a potential predictor. The meta-regression model for the group of RCTs was not significant (P = .06), the negative slope indicates a linear decrease of QoL by 0.04 standard deviations for each additional year of age (Supplemental Figure 3). The meta-regression of study year on effect size was not significant and the relationship was a flat line (data not shown). A meta-regression of study duration on effect size yielded no significant effect as well.
Risk of Bias
For RCTs, the risk of bias was assessed with Cochrane’s ROB2 tool. Six studies had a “high” risk of bias, 2 resulted in an overall rating of “some concern,” and 1 study was assessed with a “low” risk of bias (see Figure 4 for a summary and Figure 5 for a domain- and study-specific view of the ratings). The subgroup analysis (Table 3) showed a tendency for larger effects for studies with low risk of bias or some concern (SMD = 0.71, 95% CI 0.53-0.88) compared to studies with high risk of bias (SMD = 0.45, 95% CI 0.22-0.67). The risk of bias of the NRSI was estimated using Cochrane’s ROBINS-1. The overall risk of bias was “serious” for all 6 studies (Figure 6).
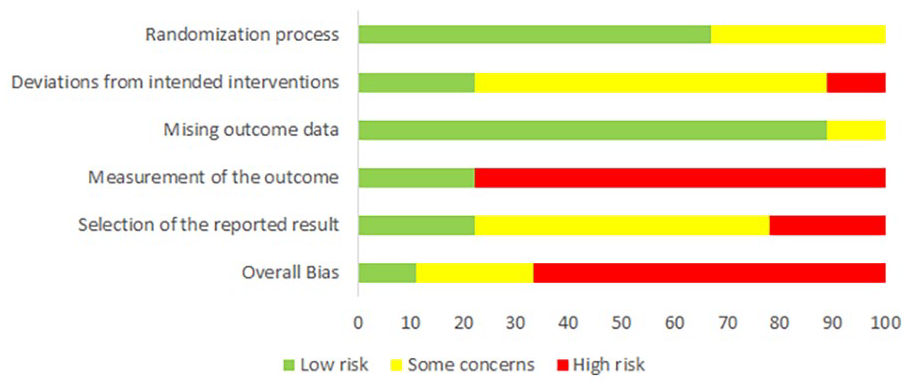
Summary of risk of bias assessment of RCTs with ROB2 as percentage (intention-to-treat).
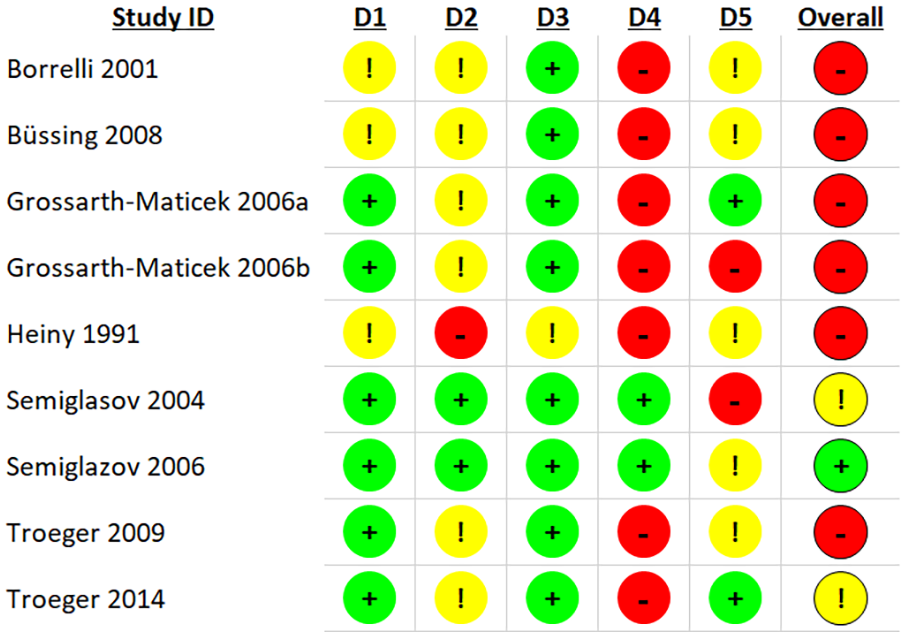
Details of risk of bias assessment of RCT according to Cochrane RoB 2 tool (intention-to-treat).

Details of risk of bias assessment of NRSI according to Cochrane ROBINS-1 tool.
Reporting Biases
A possible publication bias was assessed for both RCTs and NRSIs by visual examination, by Egger’s test and by the Duval & Tweedie’s trim-and-fill procedure. For RCTs, the Egger’s test resulted in an intercept of I = −1.6 (95% CI −3.36-0.16) and is not significant (P = .068). Duval and Tweedie’s trim and fill procedure did not identify any study to the left, that is, to the more negative side of the mean effect size to be trimmed, but 4 to the right side, which would adjust the effect size more positively to SMD = 0.74. As it is unlikely that researchers would have missed publishing more positive studies, we see this as a support of a lack of publication bias. There was no indication for a visual funnel plot asymmetry (see Supplemental Figure S1 in the Supplemental). We used all 7 NRSIs for a publication bias analysis. Duval and Tweedie’s trim and fill method yielded no studies to be trimmed and filled to either direction of the mean, and Egger’s intercept test was not significant (I = 0.97 (95% CI −8.6-10.5; P = .8)), whereas visual inspection of the funnel plot (see Supplemental Figure S2 in the Supplemental) neither favors nor disfavors a publication bias.
Certainty of Evidence
The certainty of evidence was assessed with the GRADE approach. We downgraded the quality of evidence for the RCT analysis from “high” to “moderate” by one point due to the risk of bias. The risks of inconsistency, indirectness, imprecision, and publication bias were not downgraded. For NRSI, the certainty of evidence starts at “low” according to GRADE and was further downgraded to “very low” by one point regarding the risk of bias and for inconsistency.
Discussion
General Interpretation in Context of Other Evidence
Our analysis is, to our knowledge, the first meta-analysis to focus on the effect of ME on 1 cancer entity. We found a medium effect size of ME on the QoL of breast cancer patients with little heterogeneity between studies, compared to larger heterogeneity in an earlier study where different types of cancer were included in an overall meta-analysis. 12
The magnitude of the effect size is comparable to, or larger than those of other interventions for breast cancer: Physical activity improves quality-of-life scores compared to control with a mean difference of 6.78 (95% CI 2.61-10.95) which corresponds to SMD = 0.08 (0.03-0.13) with a “probable” likelihood of causality. 53 Cognitive behavioral therapy (CBT) was reported to improve QoL with an SMD of 0.65 (95% CI 0.07-1.23) and a very low quality evidence in 1 study, 54 whereas Getu et al estimated an effect of SMD = 0.39 (95% Ci 0.12-0.66) and concluded that CBT is effective in improving the QoL of breast cancer patients. Compared to no therapy, yoga enhanced quality of life in breast cancer patients (SMD = 0.22 (95% CI 0.04-0.40)) with moderate-quality evidence. 55 Mindfulness interventions improved quality of life in breast cancer patients by SMD = 0.21. 56
Our findings suggest that the effect is robust: heterogeneity was low for the randomized studies, so a fixed effect model could be applied. There was no effect of the years the studies were conducted in on effect size, implying that methodological improvements that are assumed to be put in place over the years did not impact effect sizes. Effect sizes for blinded randomized studies, that is, methodologically more reliable studies were highest. However, this might be confounded with the fact that these studies were run in Russia, Bulgaria or Ukraine, where the effect size was SMD = 0.69 as opposed to studies run in Germany, Switzerland or Austria where effect size was SMD = 0.47. The advantage of studies performed in Eastern Europe may be that ME are much less known and could not be acquired in pharmacies, lowering the risk of unidentified application in the control groups. The ensuing lack of knowledge of side effects (eg, skin reactions) increases the probability that blinding was successful. Possibly lower health standards in Eastern Europe, with less supportive care, might suggest that the effect size shown in this setting is the “ME only” effect, while this effect is diminished in Western Europe, where patients have access to other supportive therapies. The effect size of all randomized studies is numerically higher (SMD = 0.61) than that of the non-randomized studies (SMD = 0.46), albeit not significant. Apparently, real world settings do not seem to increase the effectiveness of ME with regards to overall QoL. This might have to do with the fact that such real-world documentations are often more susceptible to implementation difficulties regarding QoL data-collection procedures and unidentified influences compared to randomized studies. Nonetheless, the NRSIs provide higher external validity by the real world setting and support the results deriving from RCTs that have inherently higher internal validity.
Limitations of the Evidence Included in the Review
Several limitations of the underlying evidence should be kept in mind. First, the reliability of measurement of QoL in breast cancer patients has improved strongly, 6 but some of the included studies reported on parameters which can only be regarded as surrogates of QoL. While the exclusion of studies that measured “self-regulation” showed no substantial impact compared to the main RCT-analysis, the sensitivity analyses for NRSIs revealed that the alternation of surrogate parameters shifted the results into non-significance. One has to bear in mind, however, that the alternate effect measure was the Karnofsky index which is not a uniformly reliable QoL scale, and is applicable particularly for patients with terminal cancers, 57 while the majority of the patients in the NRSIs were in stages I-III.
Second, the included studies were heterogeneous regarding for example, tumor stages, QoL scales, blinding, and risk of bias. In contrast, the RCT-analysis showed no sign for between-study heterogeneity for all 3 statistical parameters. Third, the risk of bias was low in 1 RCT only, yet medium to high in 8 RCTs and serious for all NRSIs, respectively, which resulted in the downgrading of the quality of evidence according to GRADE. This risk of bias assessment is persistently rated as “medium” even in well-conducted ME studies, because of its therapy-inherent difficulty to blind the treatment. Subcutaneous injections of ME regularly lead to local skin reactions which makes them discernible, and active placebos that would mimic such irritations are normally not accepted by ethical committees or clinicians for ethical reasons. This should be borne in mind when assessing the RoB-status of the evidence.
Limitations of the Review Process
Our approach was to include both RCTs and NRSIs into the analysis in order to cover the clinical evidence from trials and real-world practice. This leads to unresolved issues regarding the GRADE approach, 58 since it remains unclear whether RCTs and NRSIs should be considered en bloc if the certainty of evidence is assessed and how the certainties of different study types are balanced out to achieve an overall assessment.23,58 In practice, only very few reviews pool the evidence syntheses 59 which necessitates proper guiding. 60
Furthermore, the GRADE approach dictates to start the rating at a “high” level for RCTs and, as in the present case, at a “low” level for NRSIs. 61 For RCTs, the certainty of evidence was assessed as “moderate” and for NRSI “very low.” Although the statistical results of our analysis needs to be seen in this light, it is also important to remember that the GRADE framework has been criticized to lack a theoretical and empirical basis showing that it helps therapists to make better treatment decisions for their patients.62,63
Another limitation of the review process is that the adverse events and interactions with co-medications are not covered by this review. A recent analysis regarding the effects of ME on fatigue found few adverse events reported by studies that are also included here. 22
Since a recent comment questioned the usefulness of the I2 statistics, 64 we calculated a prediction interval which provides a range in which future studies can be expected to fall based on the current evidence. For RCTs, it doesn’t include negative intervention effects [0.12; 0.93] which is further supported by our trim-and-fill analysis that indicated the true effect size to be even higher than that reported in the main analysis. The reliability of the trim-and-fill approach may be, however, limited due to the low number of included studies.
Implications for Practice, Policy, and Future Research
In general, the QoL of patients with breast cancer improved over the last decade 65 and our analysis indicates that compared with other potential interventions, ME may be an effective additional treatment which, due to their immunomodulating effects, may reduce side effects of other anti-cancer treatments. We feel that our data supports the inclusion of ME into current guidelines as optional additive treatments with a likely positive effect on QoL. These first results, with a low heterogeneity despite including different ME preparations, seem to confirm the current clinical practice that different ME preparations can be used for the same cancer entity.
Our results will need verification in light of future trials that overcome the above-mentioned limitations. 66 Future research should focus, in our view, on solid prospective real-world evidence in large cohorts of patients whose baseline status is well described such that predictive assessments and analyses can be calculated to figure out what types of patients, for example, which cancer stage and breast cancer subtype, will benefit from ME treatment. Also, such studies might be helpful in putting the evidence from randomized studies into context. 67 Particular attention should be paid to ME effects for breast cancer patients undergoing neoadjuvant chemotherapy and breast cancer survivors, as their numbers are increasing and no study in this setting could be found in this meta-analysis.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231198074 – Supplemental material for Quality of Life in Breast Cancer Patients Treated With Mistletoe Extracts: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ict-10.1177_15347354231198074 for Quality of Life in Breast Cancer Patients Treated With Mistletoe Extracts: A Systematic Review and Meta-Analysis by Martin Loef, Daniela Paepke and Harald Walach in Integrative Cancer Therapies
Footnotes
Availability of Data
Additional data of this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The review has been supported by the “Stiftung Integrative Medizin & Pharmazie” (Stuttgart).
Data Support
We are grateful for additional data provided on request by Oei et al. 27
Protocol
The corresponding protocol was registered at PROSPERO with the number CRD42023388900.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
