Abstract
Introduction
Chemotherapy- and radiotherapy-induced mucositis is a significant burden for cancer patients (Table 1). Symptoms of oral mucositis become apparent 5 to 10 days after chemotherapy and may progress from erythema, cracking, and inflammation to pain, bleeding, ulceration,1,2 and pain.1,3 Pelvic radiotherapy is reported to induce changes in the bowel habits of 90% of patients, with half of all patients reporting that quality of life is significantly adversely affected, 4 and that serious complications can persist decades post treatment cessation.4-6 Epidemiological studies on cancers of the head and neck report a prevalence of oral mucositis of 80% for patients undergoing radiotherapy and 40% of patients receiving chemotherapy. 3 Moreover with high-dose chemotherapy, mucositis can develop in 100% of bone marrow transplant patients. Mucositis is the most frequent and serious reported side effect in the first 3 months following a transplant and is the most common condition requiring systemic analgesics during cancer therapy. 3 The frequency of mucositis is also higher in patients receiving continuous infusion therapy for breast and colon cancer. 7
Chemoradiotherapy-Induced Gastrointestinal Toxicities.

Abbreviations: IBS, irritable bowel syndrome; ISBO, intermittent small bowel obstruction; TJ, tight junctions; 5-FU, 5-fluorouracil; SIBO, small intestinal overgrowth syndrome.
Unlike oral mucositis, clinical data reflecting the long-term effects of treatment-induced gastrointestinal mucositis are lacking. An important and debilitating symptom of intestinal mucositis is diarrhea. Gastrointestinal mucositis has been reported in 80% of patients treated with 5-fluorouracil (5-FU). 8 The frequency of chemotherapy-induced diarrhea depends on the drug administered and the schedule implemented. The highest rate of diarrhea has been reported to occur with a weekly regimen of irinotecan and 5-FU bolus with 10% of patients going on to develop grade 3 to 4 mucositis. Late-onset diarrhea may occur within a week following higher dosages of irinotecan and after approximately 2 weeks following a weekly administration of lower doses. 9 In stage III colorectal cancer (CRC), chemotherapy with FOLFOX induced diarrhea in 56% of patients, yet with FOLFIRI, the prevalence of diarrhea increased to 89%. The risk of a first episode was highest during the first cycle (35 %) and dropped to less than 10% during subsequent cycles. 10
The frequency of treatment-induced gastrointestinal toxicity in CRC has been posited to likely increase with the introduction of novel drugs and the use of more intense combination regimens of polychemotherapy combined with monoclonal antibodies.6,10 Targeted therapies, including erlotinib, gefitinib, lapatinib, sorafenib, and sunitinib, have been associated with a 2- to 8-fold increased risk of all or high-grade diarrhea compared with conventional chemotherapy regimens. 11
Despite several interventions being available, including cryotherapy and loperamide, for the control of oral mucositis and diarrhea, respectively, there is a need to further explore additional safe and effective preventative and treatment options for treatment-induced mucositis and related symptoms. The goal of this narrative review was to present an overview of the safety and efficacy reported for various interventions posited to reduce the adverse effects of antineoplastic agents. The majority of clinical research has focused on prevention or treatment of oral mucositis, while intestinal toxicity has been less well reported.
Methods
The inclusion criteria were any type of clinical trial examining the use of any oral or topical probiotic or nutritional intervention with both the abstract and the journal article written in English. All clinical trial designs and methodology were included, namely, prevention as well as treatment studies. The following databases were used to retrieve journal articles: PubMed, the Cochrane Library, Science Direct, Scopus, Embase, and Google Scholar, and searches were current up to November 2016.
Electronic databases were searched using the following search string:
Probiotics OR Diet OR Food OR Nutrition OR Micronutrients OR Vitamins OR Minerals OR Dietary supplements OR Functional foods OR Honey AND Chemotherapy AND Mucositis AND Chemotherapy AND Diarrhea AND Radiotherapy AND Mucositis AND Radiotherapy AND Diarrhea.
Examination of references in retrieved articles was also conducted.
Results
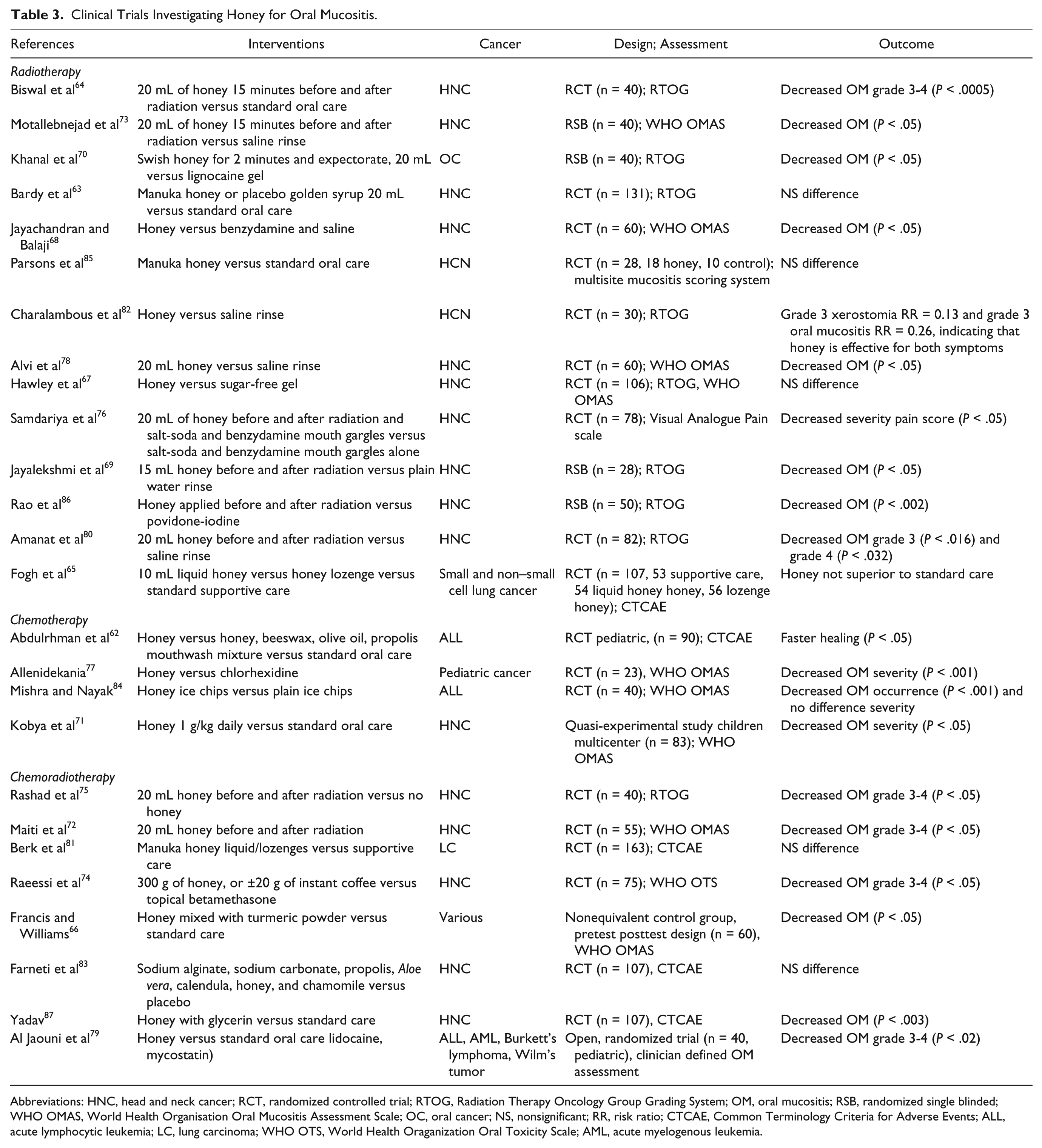
Fifty articles examining various nutritional compounds were identified, 49 from PubMed and 1 from Embase. 12 Reports included 1 study on activated charcoal, 13 1 study on β-glucan, 14 2 studies on multinutrient formulations,15,16 2 studies on an amino acid–rich oral formulation,17,18 1 study with folic acid with B12, 19 5 studies on vitamin E,12,20-23 17 intervention studies containing minerals,24-40 and 21 studies with glutamine (Table 2).41-61 Moreover, 25 clinical trials were identified examining honey alone or in combination as a prophylaxis/intervention for oral mucositis (Table 3).62-87
Clinical Trials Investigating Nutrients for Oral and Intestinal Mucositis.

Abbreviations: CRC, colorectal cancer; CTCAE, Common Terminology Criteria for Adverse Events; OM, oral mucositis; HNC, head and neck cancer; OC, oral cancer; Tx, treatment; 5-FU, 5-fluorouracil; IM, intramuscular; NS, nonsignificant; ALL, acute lymphocytic leukemia; AML, acute myelogenous leukemia; CML, chronic myelogenous leukemia; RCT, randomized controlled trial; RTOG, Radiation Therapy Oncology Group; OeC, esophageal cancer; HCC, hepatocellular carcinoma; WHO OMAS, World Health Organization Oral Mucositis Assessment Scale; im, intramuscularly; OC, oral cancer; AL, acute leukemia; NHL, non-Hodgkin’s lymphoma; HDC, high-dose chemotherapy; HSCT, hematopoietic stem cell transplantation; equiv, equivalent; NPC, nasopharyngeal carcinoma; tid, three times a day; bid, two times a day; HM, hematologic malignancies; HL, Hodgkin’s lymphoma; MM, multiple myeloma; MS, myelodysplastic syndrome; BC, breast cancer; OvC, ovarian cancer; NIDCR, National Institute of Dental and Craniofacial Research; PBSCT, peripheral blood stem cell transplantation; Mel, melanoma; LC, lung carcinoma; Lym, lymphoma; RR, risk ratio; CI, confidence interval; mCRC, metastatic colorectal cancer; KS, Kaposi sarcoma; CALGB, Cancer and Leukemia Group B scale; Sar, sarcoma; NB, neuroblastoma; mBC, metastatic breast cancer; BMT, Stanford University Bone Marrow Transplant toxicity scale; IV, intravenous; bw, body weight; AST, aspartate aminotransferase; NSCLC, non–small cell lung cancer, AC, abdominal cancer; PC, pelvic cancer.
Clinical Trials Investigating Honey for Oral Mucositis.
Abbreviations: HNC, head and neck cancer; RCT, randomized controlled trial; RTOG, Radiation Therapy Oncology Group Grading System; OM, oral mucositis; RSB, randomized single blinded; WHO OMAS, World Health Organisation Oral Mucositis Assessment Scale; OC, oral cancer; NS, nonsignificant; RR, risk ratio; CTCAE, Common Terminology Criteria for Adverse Events; ALL, acute lymphocytic leukemia; LC, lung carcinoma; WHO OTS, World Health Oraganization Oral Toxicity Scale; AML, acute myelogenous leukemia.
We also identified 19 probiotic studies (Table 4).88-105 Nine placebo-controlled clinical trials88-92,95-97,106 examined the prophylactic use of probiotic in intestinal mucositis induced by chemotherapy, radiotherapy, or a combination of both. Two trials did not include a comparator group.98,104 Four studies examined the use of a probiotic in oral mucositis,99,100,103,105 3 studies were post-chemoradiotherapy treatment trials,93,94,101 and 2 studies examined the febrile incidence during chemotherapy. 102
Clinical Trials Investigating Probiotics for Oral and Intestinal Mucositis/Diarrhea.

Abbreviations: CFU, colony-forming unit; HNC, head and neck cancer; RCT, randomized controlled trial; CTCAE, Common Terminology Criteria for Adverse Events; HSCT, hematopoietic stem cell transplantation; MM, multiple myeloma; HL, Hodgkin’s lymphoma; NHL, non-Hodgkin’s lymphoma; AML, acute myelogenous leukemia; RMS, rhabdomyosarcoma; CML, chronic myelogenous leukemia; OM, oral mucositis; NS, nonsignificant; qd, one a day; RR, risk ratio; CI, confidence interval; NK, natural killer; VSL#3, L casei, L plantarum, L acidophilus, L delbruekii subsp bulgaricus, B longum, B breve, B infantis, Streptococcus salivarius subsp thermophiles; Colon Dophilus, B breve HA-129 (25%), B bifidum HA-132 HA (20%), B longum HA-135 (14.5%), L rhamnosus HA-111 (8%), L acidophilus HA-122 (8%), L casei HA-108 (8%), L plantarum HA-119 (8%), S thermopilus HA-110 (6%), L brevis HA-112 (2%), B infantis HA-116; Infloran, 1 billion viv Lyophilisat and 1 billion B bifidum viv Lyophilisat; Dixentil, L acidophilus and L casei (strains not provided), zinc, galacto-oligosaccharides, and vitamins B1/B2/B6 and nicotinamide; tid, three times a day; QOL, quality of life; bid, two times a day; 5-FU, 5-fluorouracil; RNB, randomized nonblinded; IBS, irritable bowel syndrome; FACT, Functional Assessment of Cancer Therapy; WBC, white blood cells.
Oral mucositis was most commonly assessed according to the Radiation Therapy Oncology Group/European Organization for Research and Treatment of Cancer objective grading system (RTOG/EORTC), the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), or the World Health Organization Oral Mucositis Toxicity Scale. Intestinal mucositis was assessed by changes in stool consistency, incidence and severity of diarrhea, and the use of antidiarrheal medication. The most frequently used assessment scales are the CTCAE diarrhea grade and the Bristol stool charting system.
Bias and quality analysis were not performed as part of this review, as several reviews have already reported on bias and quality of studies for minerals, glutamine, honey, and probiotics, the 4 types of interventions with sufficient number of trials to attempt conducting a systematic review or meta-analysis. Although some studies had low bias, the overall assessment is that many studies have high bias and most studies suffered from some methodological weaknesses.107-112 The systematic review and meta-analysis of 9 probiotic trials for the prevention of chemoradiotherapy-induced diarrhea found that while the trials were of generally good methodological quality, there were significant blinding issues, and 1 study was published as a poster abstract only. Ambiguous handling of incomplete outcome data and lack of intention-to-treat analysis were noted as further bias risks. 112
Reviewed Interventions
Prophylactic activated charcoal has been shown to reduce dose-limiting chemotherapy-induced diarrhea, thereby optimizing irinotecan therapy, while reducing antidiarrheal medication in an open-label, single-arm study. The study involved 28 patients with advanced CRC receiving irinotecan 125 mg/m2 intravenously once a week for 4 weeks every 6 weeks. In cycle 1, patients received irinotecan plus activated charcoal (5 mL aqueous solution containing 1000 mg activated charcoal in 25 mL of water), given the evening before the irinotecan dose and then 3 times daily for 48 hours after the dose. The use of activated charcoal in the first cycle was associated with a significant reduction in the incidence and severity of diarrhea, reduced the use of loperamide as a rescue medication, and was well tolerated with excellent compliance 13 (Table 2).
Beta-glucan is an immune-modulating polysaccharide that was shown in a retrospective, controlled trial of 62 patients with CRC to prevent significant reductions in leucocyte and neutrophil counts compared with chemotherapy alone with a FOLFOX-4 regimen. The addition of β-glucan was also associated with a lower incidence of oral mucositis and diarrhea (Table 2). 14
Concurrent administration of folate and cobalamin failed to reduce mucositis in a pilot study of 39 patients with non–small cell lung cancer treated with pralatrexate. Mucositis remained the dose-limiting toxicity of pralatrexate treatment 19 (Table 2).
A meta-analysis that assessed the effectiveness of oral glutamine in radiotherapy-induced mucositis in head and neck cancers reported that in 5 clinical studies (234 patients total),41,43,44,52,59 glutamine was shown to reduce the risk and severity of radiotherapy-induced oral mucositis compared with either placebo or no treatment (risk ratio [RR] = 0.17, 95% confidence interval [CI] = 0.06-0.47). 107 Oral glutamine was also shown to be beneficial in 1141,44,46,49,54-60 of 15 studies investigated in a systematic review investigating the effects of glutamine for chemotherapy- or radiotherapy-induced oral mucositis. Glutamine significantly reduced the incidence of grade 2, 3, or 4 mucositis and/or reduced weight loss as well as the duration, time of onset, and/or maximum grade of mucositis. 108 Four studies showed no effect45,48,50,53 (Table 2).
A recent study found that 9 g glutamine in combination with an elemental diet was associated with a significant reduction in chemotherapy-induced oral mucositis in esophageal cancer compared with no treatment or glutamine alone. The incidence of grade 2 or higher of oral mucositis was 60% in the control group, 70% in the glutamine group, and 10% in the glutamine plus elemental diet group. 42 A further review of 9 randomized, controlled studies concluded that glutamine may reduce gastrointestinal mucositis and diarrhea and improve nitrogen balance, immune imbalance, and wound healing in chemotherapy-induced toxicity 113 (Table 2).
Glutamine has been shown to be a principal nutrient with glucose supporting survival of mammalian cells and, unfavorably, cancer cells. However, oral glutamine has been reported to be unlikely to contribute significantly to tumor growth, local invasion, and metastatic dissemination. 114 High baseline consumption of dietary glutamate has been shown in humans to be associated with a lower risk for CRC, 115 and oral glutamine 2 days before tumor implantation has been shown in rodents to increase natural killer cell activity, upregulate intestinal glutathione metabolism, and decrease tumor growth by 40% to 50%.116,117 Oral glutamine administered during chemoradiotherapy did not negatively affect tumor control and survival in patients with stage IIIB non–small cell lung cancer. 56 As cancer cells can manipulate host metabolism favoring tumor growth, deprivation of dietary glutamine or the use of oral supplementation for mucositis was reported unlikely to adversely affect tumor growth during chemotherapy treatments. Glutamine, however, has been shown to be ineffective in controlling acute radiation-induced intestinal mucositis.61,118
A Cochrane review of 3 studies64,73,75 investigating honey concluded that it was associated with a weak to moderate benefit in the prevention of radiotherapy-induced oral mucositis. 119 Three subsequent meta-analyses concluded that oral administration of honey could prevent the incidence of radiotherapy-induced oral mucositis in head and neck cancers.109,110,120 Cho et al concluded that oral administration of honey after radiotherapy could prevent the development of moderate to severe mucositis and associated weight loss. 109 Xu et al concluded that, compared with no treatment, honey could reduce the incidence of oral mucositis after chemoradiotherapy (RR = 0.35, 95% CI = 0.18-0.70, P = .003). 110 Honey was also found to decrease treatment interruptions, weight loss, and delay the onset of oral mucositis. Honey, however, was inefficacious in decreasing the peak mucositis score. Co et al reported statistical pooling showing that the risk ratio of having a treatment interruption was significantly lower with the use of honey versus control 0.11 (95% CI = 0.02-0.58) with a risk ratio of developing severe mucositis when honey was administered as 0.45 with a CI of 0.09 to 2.21. 120
Friend et al specifically examined pediatric trials and identified 4 trials62,71,77,79 with grade C evidence that honey was effective as a preventative and adjunctive therapy for chemotherapy-induced oral mucositis in children. 121 Honey was found to reduce the frequency, duration, and stage of chemotherapy-induced mucositis.
Seven trials have been published since the meta-analyses report.109,110,120 Four trials examined honey in radiotherapy-induced mucositis in head/neck and lung cancers,65,69,80,86 1 trial in chemotherapy-induced mucositis in acute lymphocytic leukemia, 84 and 2 trials in chemoradiotherapy-induced mucositis in children with various cancers.79,87 Only one trial 65 reported no effect. Medical manuka honey administered as a liquid or as a lozenge was not superior to best supportive care in preventing radiation esophagitis. 65
A single-blinded randomized controlled trial (n = 28) found that 15 mL of natural honey was associated with a statistically significant difference in degree of oral mucositis between the experimental and control groups in weeks 4, 5, and 6 (P < .01). 69 Compared with the active comparator, povidone-iodine, honey significantly reduced radiation-induced oral mucositis, decreased the incidence of intolerable mucositis, treatment breaks, loss of treatment days (P < .0001 and < .0003), and did not affect the radiation-induced tumor response. 86 Honey significantly reduced the severity of mucositis (grades 3 and 4) compared with control group at the end of 6 weeks of radiation treatment. 80 Honey ice cubes were shown to significantly reduce the occurrence of chemotherapy-induced oral mucositis in pediatric cancer patients compared with plain ice cubes on the 5th (P = .001) and 15th (P = .001) days of assessment. 84 A significantly higher number of patients developed grade 2 or above chemoradiotherapy-induced mucositis in the control arm compared with the experimental arm (P = .003). 87 Absolute risk reduction between honey and control for developing grades III and IV oral mucositis was found to be significant in a study of pediatric cancer patients receiving chemoradiotherapy (P < .05 79 ; Table 3).
A meta-analysis of randomized controlled trials (RCTs) examining oral mucositis induced by chemoradiotherapy, or hematopoietic stem cell transplantation (HSCT), found that patients pretreated with mineral supplementation delayed the onset of mucositis and that fewer patients experienced less peak oral mucositis compared with controls. 111 The analysis examined 7 studies with zinc,26-31,33 3 with calcium,36-38 2 with selenium,24,25 and 2 with iodine.39,40 Significant study bias was observed though and study heterogeneity, making it difficult to make specific clinical recommendations. Mineral formulations did not overall significantly reduce mean duration of mucositis, pain duration, or use of analgesics. 111 Of the 14 studies included in the meta-analysis, the 3 excluded26,32,122 and 2 recent studies34,98 are presented in Table 2. Zinc is essential for proper immune function and for the integrity of connective tissue and cell membranes, and 50 to 150 mg elemental zinc daily was reported to reduce oral mucositis27-29,31 (Table 2).
Four studies examining the effects of multinutrient formulations that consisted of a mixture of open-label, retrospective, and prospective studies were identified. In a small, open-label study, it was reported that oral or parenteral administration of a multinutrient formulation was well tolerated in patients with head and neck cancers treated with chemoradiotherapy without developing severe mucositis. 15 In another, multinutrient formulation composed of amino acids, omega-3 fatty acids, ribonucleic acids, vitamins, and antioxidants was shown to be associated with less severe mucositis in patients with head and neck cancers treated with chemoradiotherapy. 16 In 2 other studies that examined the same amino acid–rich oral formulation, the first study found that the formulation was associated with reduced severity of mucositis in squamous cell carcinoma treated with radiotherapy with or without chemotherapy when compared with no formulation administration. The nutrient formulation was also associated with improved completion rates of chemoradiotherapy. 17 The second study was a prospective pilot study in CRC treated with 5-FU-based chemotherapy. This study reported that the multinutrient formulation was associated with a decrease in the severity of oral mucositis in approximately 90% of the patients during the first course of treatment (P = .0002) and maintained in the second course of treatment (P <.0001 18 ; Table 2).
Vitamin E may reduce mucositis by regulating nrf2 activation. Gamma-tocotrienol has been shown to prevent 5-FU-induced redox signaling by regulating nrf2 activation and cell survival in human oral keratinocytes. 123 Applying vitamin E directly to the mucous membranes may be more effective than orally administered. Oral vitamin E (400 mg twice daily) was shown to have no effect on the incidence or severity of mucositis in an RCT of 60 patients with leukemia receiving allogenic bone marrow transplantation. 12 Despite this, topical vitamin E was shown to be beneficial in the treatment of oral lesions associated with mucositis in an RCT of patients with solid tumors (n = 17) or leukemia (n = 1). Six of 9 patients receiving vitamin E had complete resolution of oral lesions compared with 1 out of 9 in the control group (P = .025). 21 In another study with 80 patients who developed oral mucositis, 100 mg of a topical, but not oral, application of vitamin E was shown to improve mucositis. 22 In a further study with 54 patients with head and neck cancers it was found that vitamin E before, and for the duration of radiotherapy, decreased the incidence of mucositis. 20 Topical vitamin E reduced the risk of mucositis by 36%. 20 Both topically applied vitamin E and the oligomeric procyanidin known as pycnogenol were shown in an RCT to reduce mucositis in 72 children although pycnogenol was not effective for severe, grade 4 mucositis 23 (Table 2).
A probiotic containing lozenge, specifically with Lactobacillus brevis, has been shown in an RCT to reduce radiation- and chemotherapy-induced oral mucositis in 200 patients with head and neck cancers. Use of the probiotic was associated with a reduced incidence of mucositis grades III and IV (P = .001). Supportive treatment with the probiotic was administered during treatment and for 1 week post treatment completion (radiotherapy and weekly cisplatin). 99 The same probiotic lozenge formulation was also examined in 3 studies of patients undergoing HSCT for a variety of cancers including multiple myeloma. In the first pilot study, of 21 patients, only 19% developed grade III or IV mucositis compared with the expected 60%. No adverse events except occasional grade I/II nausea due to study drug were noted. 105 The second pilot study by the same research group found that 20% developed grade III or IV mucositis. 100 In a repeat pilot study, patients treated with HSCT were given the probiotic lozenge 4 to 7 days before initiation of chemotherapy and continuing until resolution of mucositis or till day 24. The single-arm, single-center, phase II study found that of the 31 patients enrolled, 7 (22.6%) patients did not develop any mucositis, 6 (19.4%) patients developed grade 1 mucositis, 12 (38.7%) patients developed grade 2 mucositis, and 4 (12.9%) and 2 (6.5%) patients developed grade 3 and grade 4 mucositis, respectively. Median time to onset and for resolution of mucositis was 6 days and 8 days, respectively. No adverse events were reported with usage of study drug. 100 However, in the fourth pilot study by another group using the same formulation in patients undergoing HSCT, all 16 patients developed various grades of mucositis with no statistical difference between the probiotic lozenge and standard treatment 103 (Table 4).
The majority of studies investigated the prophylactic use of probiotics for diarrhea, while a few investigated the efficacy of probiotics in the treatment of irritable bowel syndrome or diarrhea, weeks to years post treatment (Table 4). The studies used a variety of probiotic interventions and protocol designs and outcomes, making it difficult to identify which type of intervention and protocol was most beneficial. A systematic review and meta-analysis of probiotics for prevention of chemoradiotherapy-induced diarrhea in people with abdominal and pelvic cancers found that probiotics were generally beneficial in treatment-induced diarrhea, especially for grades 2 and 3. 112 A recent meta-analysis that grouped 7 studies for the prophylactic use of probiotics for cancer therapy–induced diarrhea 124 concluded that current evidence does not support widespread implementation of probiotics for the management of diarrhea secondary to cytotoxic therapy and the tyrosine kinase inhibitor, dacomitinib. The administration of probiotic was begun on the first day of cancer therapy initiation and as such referring to prophylactic interventions may be inaccurate. The administration of prophylactic probiotics must begin ideally 1 month prior to chemotherapy/radiotherapy initiation.
In order to investigate the probiotic effect to reduce diarrhea induced by chemotherapy and/or radiotherapy, relative risks were calculated for the 9 RCTs, and the results showed that 7 studies favored probiotics for preventing chemotherapy- and/or radiotherapy-induced grade ⩾2 diarrhea (Figure 1). The co-administration of probiotics with radiotherapy shows enhanced efficacy in preventing intestinal adverse effects induced by radiotherapy compared with chemotherapy.
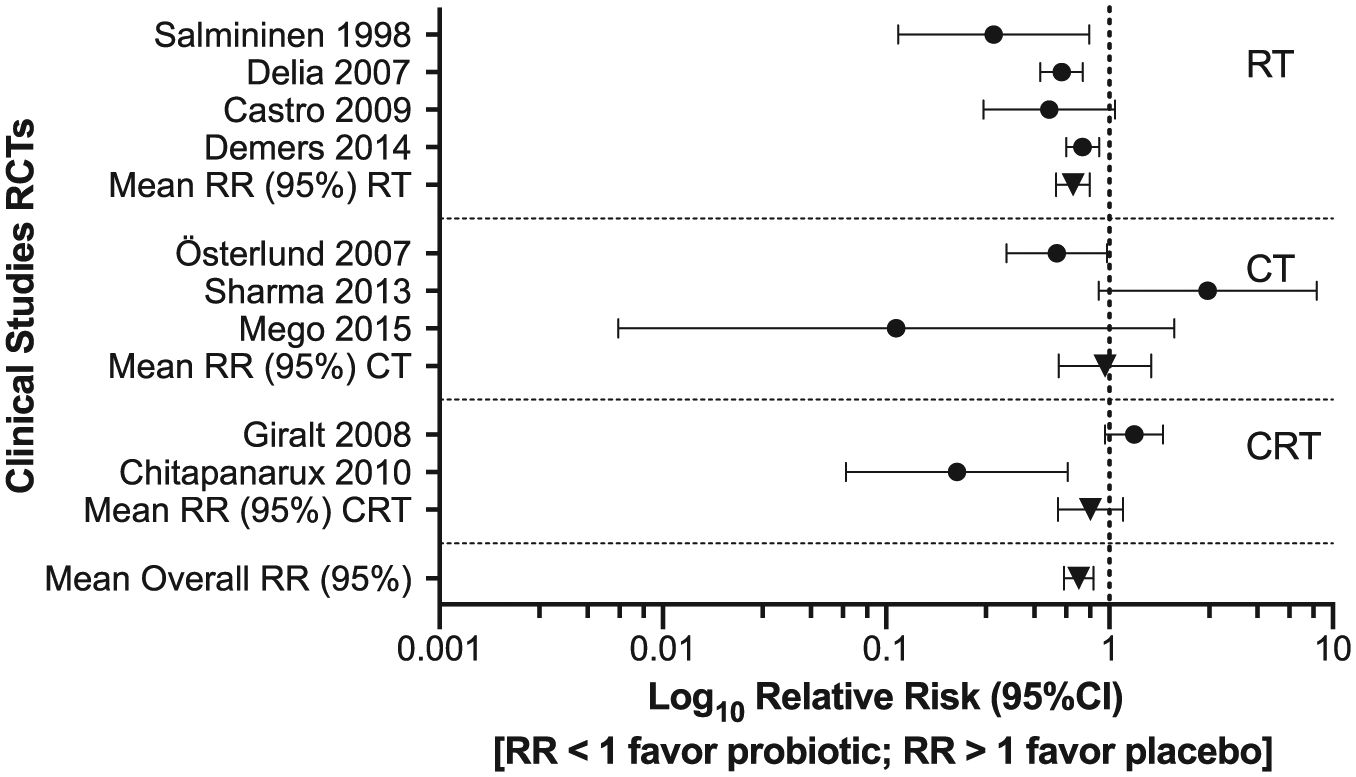
Forest plot of RCTs of chemotherapy-/radiotherapy-induced diarrhea.
Adjuvant Interventions Safety
One study implementing manuka honey mouthwash found that while it demonstrated benefit in ameliorating radiation-induced weight loss and increase quality of life in the absence of cisplatin chemotherapy, it was also reported that undiluted manuka honey caused severe nausea, vomiting, and severe stinging. 85 In an additional study, Bardy et al found no difference between golden syrup and manuka honey, 63 suggesting that perhaps the high sucrose content was responsible for the antibacterial effect observed. Neither study showed improvement in mucositis; however, compliance associated with the taste and texture of the interventions was an issue, which may have influenced the study outcome.
The common dose administered in clinical trials was 20 mL of honey, 3 times daily. At this dose, honey did not affect blood sugar levels when initial fasting blood sugar level was below 150 mg/dL. 72 Patients undergoing radiotherapy for head and neck cancers have been reported to be prone to a range of dental complications, 125 and a concern was the added risk of developing dental caries, in spite of research suggesting otherwise. 126 Radiation-related caries are related to hyposalivation, shifts in the oral microbiota, and altered saliva composition. The rapid onset and progression often leads to extensive loss of dentition within short periods of time. Honey contains known cariogenic substances including glucose, fructose, sucrose, and numerous acids, including gluconic, acetic, lactic, butyric, and formic acids, that may contribute to cariogenic increased risk. 127 However, honey has also been shown to prevent radiation-induced decrease enamel microhardness in xerostomic patients compared with patients with normal salvia. 128 None of the trials reported that the use of honey predisposed to the development of caries.
The use of probiotics as an adjunctive medicine in oncology to enhance treatment or reduce adverse events associated with chemotherapy or radiotherapy is not part of standard practice. The principal concern being that patients treated with chemotherapy are frequently immune-compromised and, therefore, are at increased risk of sepsis from administering probiotic formulations. A systematic review of 17 studies (N = 1530) found no reports of significant side effects such as serious localized or systemic infections when administered to patients receiving cancer treatments. Five case reports showed probiotic-related bacteremia, fungemia, or positive blood cultures. 129 Wang et al included 11 studies in its safety analysis. Seven studies did not report any adverse events. Four studies reported various adverse events. The reporting of adverse effects was, however, very inconsistent and poorly documented. Although some infections were reported, no probiotic bacteria growth could be found in blood cultures. Other adverse effects included mild gastrointestinal upsets, fever, and anorexia, which were also observed in the control groups. 112 Okawa et al reported 1 death but no evidence of an association with the probiotic intervention was reported. 130 A few probiotic trials have been performed in children.102,131 A single-blinded study found that Bifidobacterium breve reduced the frequency of fever, which was associated with a lower use of intravenous antibiotics compared with placebo in children receiving chemotherapy (1-13 years of age, n = 42). No adverse events were reported. 102 A safety and feasibility study did not report Lactobacillus plantarum–associated bacteremia in children undergoing allogenic hematopoietic cell transplant; 131 however, a case of septic shock caused by yogurt-derived Lactobacillus species was recently reported in a 54-year-old male patient with acute promyelocytic leukemia. The bacteremia developed a week after the patient underwent high-dose chemotherapy and autologous peripheral blood stem cell transplantation. The pathogen was identified by strain-specific polymerase chain reaction analysis to be identical to the Lactobacillus rhamnosus GG found in the yogurt. 132
Discussion
Recent interventions have continued to explore and to further build the scientific and clinical understanding of oral and intestinal mucositis preventative and treatment options that may be available to clinicians and patients.13,112,121,133 The prevention and treatment of oral and intestinal mucositis that seeks to decrease the risk of formation and/or progression of these deleterious sequela of chemoradiotherapy regimens are important factors that impinge on patient quality of life and clinical decisions relevant to treatment. While it is acknowledged that oral and intestinal mucositis represent significant burdens of antineoplasitic therapies, the implementation of adjunctive treatments still remain a challenge. 134
Chemotherapy and radiotherapy have significant adverse effects on the microbiota of the oral and gastrointestinal mucosa. Oral mucositis is strongly associated with bacteremia and sepsis due to Escherichia coli, Pseudomonas aeruginosa, and Candida albicans. 103 How probiotics were postulated to overcome the side effects of chemotherapy and/or radiotherapy was advanced from observations that Lactobacillus brevis–containing lozenges produced anti-inflammatory metabolites. 103 It was reported that L brevis produced arginine deiminase and sphingomyelinase, which hydrolyses platelet-activating factor known to be associated with oral mucositis in radiation therapy. 103 Arginine deiminase then converts arginine to ammonia and citrulline, reducing the amount of available arginine to be converted to nitric oxide—a major mediator of inflammation. Furthermore, the appeal of probiotics administered for oral mucositis was enhanced when they were demonstrated to have no serious adverse effects.99,103,105 This local oral benefit did not, however, extend to the intestines, with 14 out of 16 patients developing diarrhea. 103
In the intestines, cancer therapy has been found to decrease commensals such as Bifidobacteria, Clostridium cluster XIVa, and Faecalibacterium prausnitzii, combined with increases in Enterobacteriaceae and Bacteroides. These changes induce intestinal dysbiosis and contribute to the development of mucositis, particularly diarrhea and bacteremia. 135 Adverse shifts in the intestinal microbiota has led to the notion that the administration of probiotics could reduce the side effects of chemotherapy and radiotherapy.
There is moderate evidence that zinc, selenium, vitamin E, glutamine, and honey may be beneficial for the prevention or treatment of oral mucositis. However, the low numbers and heterogeneity of the studies reviewed generally makes it difficult to offer specific clinical recommendations. In one review, mineral supplementation, including zinc, did not overall significantly reduce mean duration of the mucositis, pain incidences, or use of analgesics. 111 The recommendation for zinc supplementation is currently restricted for patients with oral cancer having treatment with chemotherapy or radiation according to the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) guidelines. One drawback of using zinc supplementation is that it may induce nausea and even vomiting. Zinc supplementation should not be taken on an empty stomach, as it increases the adverse effects. The studies mostly used 220 mg zinc sulfate, equivalent to 50 mg elemental zinc, 2 to 3 times daily as a mouthwash or capsule/tablet.
The administration of selenium in clinical studies employed doses that ranged from 200 µg elemental selenium twice daily 25 to sodium selenite oral fluid 200 to 500 µg an hour prior to radiotherapy sessions. 24 Applying vitamin E directly to the oral mucosa may be more effective than orally administered.
Glutamine was found to be the most studied nutritional intervention and despite evidence suggesting that glutamine may reduce gastrointestinal mucositis and chemotherapy-induced diarrhea, the European Society for Clinical Nutrition and Metabolism (ESPEN) 133 stated in its recent guidelines on nutrition in cancer patients that “there are insufficient consistent clinical data to recommend glutamine to prevent radiation-induced enteritis/diarrhea, stomatitis, esophagitis, or skin toxicity.” The MASCC/ISOO guideline has been updated from a recommendation against glutamine to “no guideline possible” for glutamine for oral or gastrointestinal mucositis. 136 The safety of glutamine has also been reviewed in view of emerging evidence that malignant cells can utilize glutamine as a mitochondrial substrate. 137 The glutamine doses investigated have ranged from 9 to 30 g daily in divided doses. As the lower dose has been shown to be beneficial in oral mucositis, it may be prudent to use the lower end of the dosage spectrum. Good oral hygiene is essential if honey-based interventions for mucositis are recommended. Activated charcoal may reduce symptoms associated with chemotherapy-induced mucositis including diarrhea. Clinical trials investigating the administration of probiotics to prevent treatment-induced intestinal toxicity has produced mixed results. 124 Studies have used various end-point parameters including stool frequency and stool consistency (described separately or as diarrhea grade 2-4), use of rescue anti-diarrheal medications, and microbiome shifts induced by chemotherapy or radiotherapy. The use of probiotics in the prevention or treatment of chemotherapy and/or radiotherapy-induced gastrointestinal toxicity appears to be beneficial and without significant side effects. The MASCC/ISOO guideline suggests that probiotics containing Lactobacillus species be used to prevent diarrhea in patients receiving chemotherapy and/or radiation therapy for a pelvic malignancy, 136 while the ESPEN guideline states that there are insufficient consistent clinical data to recommend probiotics to reduce radiation-induced diarrhea.
Conclusion
There is plausible clinical evidence for the administration of honey, zinc, selenium, topical vitamin E, and glutamine as an adjuvant treatment to reduce the risk of developing oral mucositis during chemotherapy or radiotherapy. Activated charcoal, glutamine, and probiotics may also be beneficial in chemotherapy-induced diarrhea. Considering the excellent safety profile and resulting high therapeutic index, further research examining the mechanism of action and clinical efficacy of probiotics in chemotherapy- and radiotherapy-induced intestinal mucositis is warranted. Given that adverse disturbances in the oral and intestinal microbiomes can promote immune dysregulation and increase the risk of patient mortality, there is need for further research in this area.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Luis Vitetta has received National Institute of Complementary Medicine and National Health and Medical Research Council of Australia competitive funding and industry support for research into probiotics and other compounds and is currently Director of Medical Research at Medlab Clinical and with Michael Thomsen investigating the formulation and administration of probiotics. Michael Thomsen is funded by the Francis M Hooper scholarship, Sydney Medical School Foundation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
