Abstract
Background:
Chemoradiotherapy-induced gastrointestinal toxicity may lead to a significant impairment of the oncological patient’s quality of life, as well as to reduced adherence to the treatment, which may have a negative impact on survival and mortality rates.
Objective:
The aim of this review was to investigate whether oral probiotic administration prevents chemotherapy (± radiotherapy)-induced gastrointestinal toxicity, particularly diarrhea.
Methods:
We searched the MEDLINE, Web of Science, and SCOPUS databases for randomized controlled trials in English published between 1990 and 2020. We conducted statistical data analyses expressing the treatment effect size as a risk ratio (RR) together with a 95% confidence interval (CI). Implications are based on trials rated as having a low risk of bias (RoB).
Results:
We included 8 trials (n = 697 participants), from which 3 studies rated as low RoB contained primary endpoint data; the risk of developing grade 3/4 diarrhea in patients receiving probiotics was reduced by 78% compared to the control group (RR = 0.22 [95% CI 0.05-1.08]; P = .06; n = 114 participants). Probiotics showed preventive effects in patients treated with chemotherapy alone (RR = 0.34 [0.12-0.94]; P = .04, n = 121 participants) and in patients with colorectal cancer (RR = 0.56 [0.34-0.92]; P = .02; n = 208 participants). The reduction in the incidence of overall diarrhea was not significant.
Conclusions:
Probiotics failed to prove a preventive effect of statistical significance against the development of severe and overall diarrhea in cancer patients treated with chemotherapy (± radiotherapy). However, we cannot rule out that the effects of probiotics are clinically relevant, especially in certain subgroups of patients. This needs to be clarified in further well-performed studies.
Introduction
Worldwide, the incidence of newly diagnosed cancer cases for 2020 was estimated at 19.3 million, which means an increase of 12% from 2016 to 2020.1,2 The use of oral chemotherapy is also increasing, as it represents an irreplaceable method for the treatment of oncologic diseases. 3 Apart from the primary targets, it also interferes with the metabolism of rapidly dividing healthy tissues, which often results in heterogeneous adverse effects. Along with hematological, neurological, and skin toxicity, the most frequently observed effect is gastrointestinal toxicity. Chemotherapy is often combined with radiotherapy, which can increase the incidence or intensity of adverse effects.
Gastrointestinal toxicity manifests itself as nausea, vomiting, diarrhea, constipation, mucositis, enterocolitis, abdominal pain, gastrointestinal leukocytoclastic vasculitis, autoimmune colitis, and ischemic colitis.4,5 From the viewpoint of the cancer patient and most likely due to their frequency of occurrence, nausea and vomiting are the most unpleasant events, 5 while diarrhea represents one of the most common and troublesome adverse effects related to chemotherapy or radiotherapy. 6
A number of factors or their combinations contribute to the development of diarrhea: mucositis and consequent malabsorption, dysbiosis, immunosuppression, tight junction dysfunction, and impaired barrier function. Some cytostatics and their metabolites, such as irinotecan, induce diarrhea directly.7,8 The incidence of grade 3/4 diarrhea related to chemotherapy regimens ranges from 5% to 47%. 4 When a bolus of 5-FU is used in combination with irinotecan, the overall incidence of diarrhea can reach 50% to 80%. 9 Regarding synthetic molecules, this state is most often induced by 5-fluorouracil (5-FU), capecitabine, irinotecan, and platinum derivatives. 4 Gastrointestinal toxicity could lead to a significant impairment of the oncological patient’s quality of life. 10
Several strategies have been proposed to prevent and reduce chemotherapy-related gastrointestinal toxicity.4,11-14 Experimental research has indicated that the host response to cytotoxic agents is modulated by the gut microbiota. 14 In cancer patients, gut microbiota manipulation is considered to be generally safe; however, it is accompanied by risks and controversies that can potentially introduce clinical complications. 15 Oral administration of beneficial live microorganisms (probiotics) is among the most accepted strategies for gut microbiota modulation.
A number of reviews have shown that probiotics are effective and safe in the prevention of diarrhea in both adults and children.16-20 Certain probiotic strains are recommended in the treatment of acute diarrhea,21-23 although the recent findings of Collinson et al 24 do not support the use of probiotics in such a manner.
The objective of our systematic review with meta-analysis was to investigate the efficacy of oral probiotic administration in the prevention of chemotherapy (± radiotherapy)-induced gastrointestinal toxicity, particularly diarrhea.
Methods
Eligibility Criteria
In this systematic review and meta-analysis, we followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 25 We established the protocol and clinical questions prior to a comprehensive literature search and registered the protocol at the PROSPERO International Register of Systematic Reviews (National Institute for Health Research, University of York, UK; https://www.crd.york.ac.uk/prospero/) as CRD42021248390. We have defined the criteria for considering studies for this review as follows:
Population: Cancer patients undergoing chemotherapy ± radiotherapy
Intervention: Orally administered live microorganisms
Control: Placebo or active treatment
Outcome: Reduction in the incidence of gastrointestinal adverse effects, particularly diarrhea
Study design: Randomized controlled trials (RCTs) published between 1.1.1990 and 31.12.2020
Language: Trials published in English
Inclusion Criteria
We set the inclusion criteria as follows: 1. original articles of randomized controlled trials, 2. patients with diagnosed primary tumors undergoing chemotherapy ± radiotherapy, 3. patients receiving defined chemotherapy, 4. patients using orally administered live microorganisms (probiotics) for the prevention of gut-associated adverse effects, and 5. trials evaluating chemotherapy ± radiotherapy-induced gastrointestinal adverse effects, particularly diarrhea.
Exclusion Criteria
We set the exclusion criteria as follows: 1. experimental trials and animal model trials, 2. trials assessing oral mucositis and/or constipation or the effects of heat-killed and/or tyndallized microorganisms or the effects of microorganism derivatives, 3. trials reporting insufficient or incomparable data, and 4. trials in which the microorganisms used were not reported at the strain level.
We excluded constipation since it has not yet been demonstrated that probiotics could prevent (or cure) constipation in noncancer (or cancer) patients. Next, constipation represents a condition that is very difficult to attribute to chemotherapy (or radiotherapy) and distinguish it from secondary constipation caused by agents given to control, for example, tumor-induced symptoms (such as antiemetics against nausea/vomiting or opioids against pain) or a special diet.
Trials reporting insufficient or incomparable data include trials not reporting relevant outcomes, trials reporting changes in stool consistency/stool frequency or trials reporting proportions of patients with improved diarrhea according to a questionnaire.
Primary and Secondary Endpoints
As a primary endpoint, we set the incidence of chemotherapy (± radiotherapy)-induced grade 3/4 diarrhea.
As secondary endpoints, we set 1. the incidence of diarrhea of any grade (the overall diarrhea) and 2. the rescue medication intake (number of patients on loperamide-based rescue medication).
Search Strategy
We performed 2 independent systematic searches of the literature (RD and MW) in the MEDLINE, Web of Science, and SCOPUS databases. We used the following search terms: (chemotherapy OR radiotherapy OR cytostat* OR cytotox*) AND (carcinoma OR cancer OR tumor OR malign*) AND (probiotics OR lactobacill* OR bifidobacter*) AND (toxicit* OR prevent* OR adverse OR side-effects OR diarrhea OR diarrhoea). In SCOPUS, we set the limitations as follows: (TITLE-ABS [multicent* OR clinical trial OR trial* OR rct* OR random* OR blind* OR control* OR placebo*]). We limited the searches with language restriction to English and article type to clinical trial. We identified 240 references in total (MEDLINE 106, Web of Science 91, and SCOPUS 43). We extracted the data and moved it to the reference manager Mendeley by Elsevier. After removing duplicates (n = 66), we screened the abstracts of 174 references. A total of 148 of the 174 references did not meet the inclusion criteria or met the exclusion criteria.
Data Extraction
We comprehensively and systematically read 27 articles and added 2 references by a manual search using Google Scholar. Of 29 articles, we excluded 21 based on the following reasons: incomparable data (n = 11), full text in Chinese (n = 1), failed to fulfil randomized controlled trial (n = 3), letter to editor (n = 1), inappropriate drug form of active substance (n = 2), strains not reported (n = 2), chemotherapy not reported (n = 1). If a discrepancy occurred, we resolved it through a discussion between RD, MM, MA, and MW. We summarized the data extraction process in a flowchart (Figure 1).

Flowchart of the search strategy and data extraction based on PRISMA 2009. RCT, randomized controlled trial.
We conducted independent data extractions (RD and MW) and recorded the details from each trial included. The obtained data consisted of the name of the first author and year of publication, population characteristics, type of trial, cancer type, cancer treatment, probiotic characteristics, dose per day in colony-forming units (CFU), duration of intervention, control characteristics, diarrhea assessment tool, number of patients in the probiotic/synbiotic group, number of patients in the control group, and reported outcomes. We present these data in a summary Table 1.
Basic Characteristics of the Studies Included in the Systematic Review.

Abbreviations: 5-FU, fluorouracil; CFU, colony forming unit; NCI-CTCAE, National Cancer Institute—Common Terminology Criteria for Adverse Events; DBPC, double-blind placebo controlled; NSS, not statistically significant; RT, radiotherapy; WHO, World Health Organization.
Risk of Bias Assessment
Two authors independently assessed the quality of the included trials (RD and RH). For risk of bias (RoB) assessment, we used the Cochrane Handbook for Systematic Reviews of Interventions. 26 The discrepancies were resolved through a discussion between the authors RD, RH, and MA.
Statistical Data Analysis
Using RevMan 5.4.1, 27 we expressed the treatment effect size as a risk ratio (RR) together with a 95% confidence interval (CI). For every selected study, we calculated a natural logarithm of the RR estimate and a corresponding standard error (SE) and entered it into a statistical model; we applied the inverse variance statistical method. If I 2 = 0 (meaning homogenous data), we used and reported a fixed-effects model; if I 2 > 0 (meaning heterogeneity was present), we used and reported a random-effects model. I 2 ≥ 75% indicated a considerable level of heterogeneity. We calculated the pooled effect estimate as RR with 95% CI. We created forest plots representing effect estimates and 95% CIs for each included study, as well as pooled effect estimates.
We calculated the weight given to each study as the inverse of the variance of the effect estimate. Thus, we assigned larger studies with smaller SEs more weight than smaller studies, which had larger SEs. This minimizes the imprecision (uncertainty) of the pooled effect estimate.
We assessed heterogeneity, including the between-study variance τ 2 (applicable for the random-effects model only), χ 2 test and visual inspection of forest plots. We used the I 2 statistic to assess the impact of heterogeneity on the meta-analysis. When we detected heterogeneity, we attempted to determine potential reasons. RevMan 5.4.1 also automatically performed a test for the overall effect, which provides the p value from the Z-test to examine whether the pooled estimate of the effect is statistically significant. For the subgroup analysis, we provided a p value for subgroup differences.
We assessed reporting bias via funnel plots only if the number of included studies was ≥5. We did not perform Egger’s test since the number of selected studies was not sufficient for this purpose.
First, we analyzed the primary endpoint in studies having low overall RoB evaluations, and then we performed a sensitivity analysis on all the studies. Next, if applicable, we performed subgroup analyses on all the studies based on RoB overall assessment, study blinding type, cancer type, control treatment type, and cancer therapy type.
Results
Summary of Obtained Evidence
In this systematic review, we included 8 RCTs published in English from 2007 to 2019, in which the subjects (n = 697) underwent cytostatic treatment (Figure 1). Along with chemotherapy, cancer patients concomitantly used probiotics (n = 400) or control treatment (n = 297). Trials were controlled by placebo in 5 cases. We judged the control treatments in the rest of the studies to be possibly active, comprising heat-killed bacteria, guar gum, and Enterobacterium faecalis culture in daily doses of 3 × 109 CFU. Gynecologic (with or without rectal and prostate) cancers were studied in 4 out of the 8 trials, colorectal cancer in 3 trials, and 1 trial was conducted in patients with thoracic esophageal cancer. Six trials were carried out in a double-blind, placebo-controlled fashion.8,28-32 Two trials were open label.33,34 Platinum derivatives (cisplatin n = 5, oxaliplatin n = 1) were the most prevalently used cytotoxic agents, followed by 5-FU (n = 4) and capecitabine (n = 3; Table 1).
We rated the overall RoB as “low” in 4 trials,8,28,31,32 as “some concerns” in 1 trial, 33 and as “high” in 3 trials29,30,34 (Figure 2).

Risk of bias assessment for relevant domains of all included studies.
Primary endpoint: Incidence of chemotherapy ± radiotherapy induced grade 3/4 diarrhea—low RoB studies
We identified 7 articles (n = 634 participants) containing primary endpoint data, from which we classified 3 studies (n = 114 participants) as overall low RoB. The weights assigned to each study for further statistical analysis were roughly equal. The 95% CIs calculated for RR in each study were quite wide because of small sample sizes and/or low or zero number of grade 3/4 diarrhea events in the compared treatment groups. The assessment of heterogeneity indicated that the included studies were homogenous, and the P-value of the χ 2 test did not reject the null hypothesis (that there was no heterogeneity between the studies). Based on that, we used a fixed-effects model. The risk of developing severe diarrhea grade 3/4 in patients receiving probiotics was reduced by 78% in comparison with the control group (Figure 3). Although the statistical significance of the pooled RR cannot be confirmed at a significance level of α = .05, we consider the RR value to be clinically significant.

Studies rated as having a low overall risk of bias.
Primary endpoint: Incidence of chemotherapy ± radiotherapy induced grade 3/4 diarrhea—all studies
The sensitivity analysis of all studies also produced nonsignificant (NSS) results (Table 2). It should be noted that we assigned the lowest weights to those studies evaluated as having a low RoB because these studies have smaller sample sizes, larger SEs, and fewer grade 3/4 diarrhea events.
Complementary Subgroup Analyses for Primary and Secondary Endpoints.
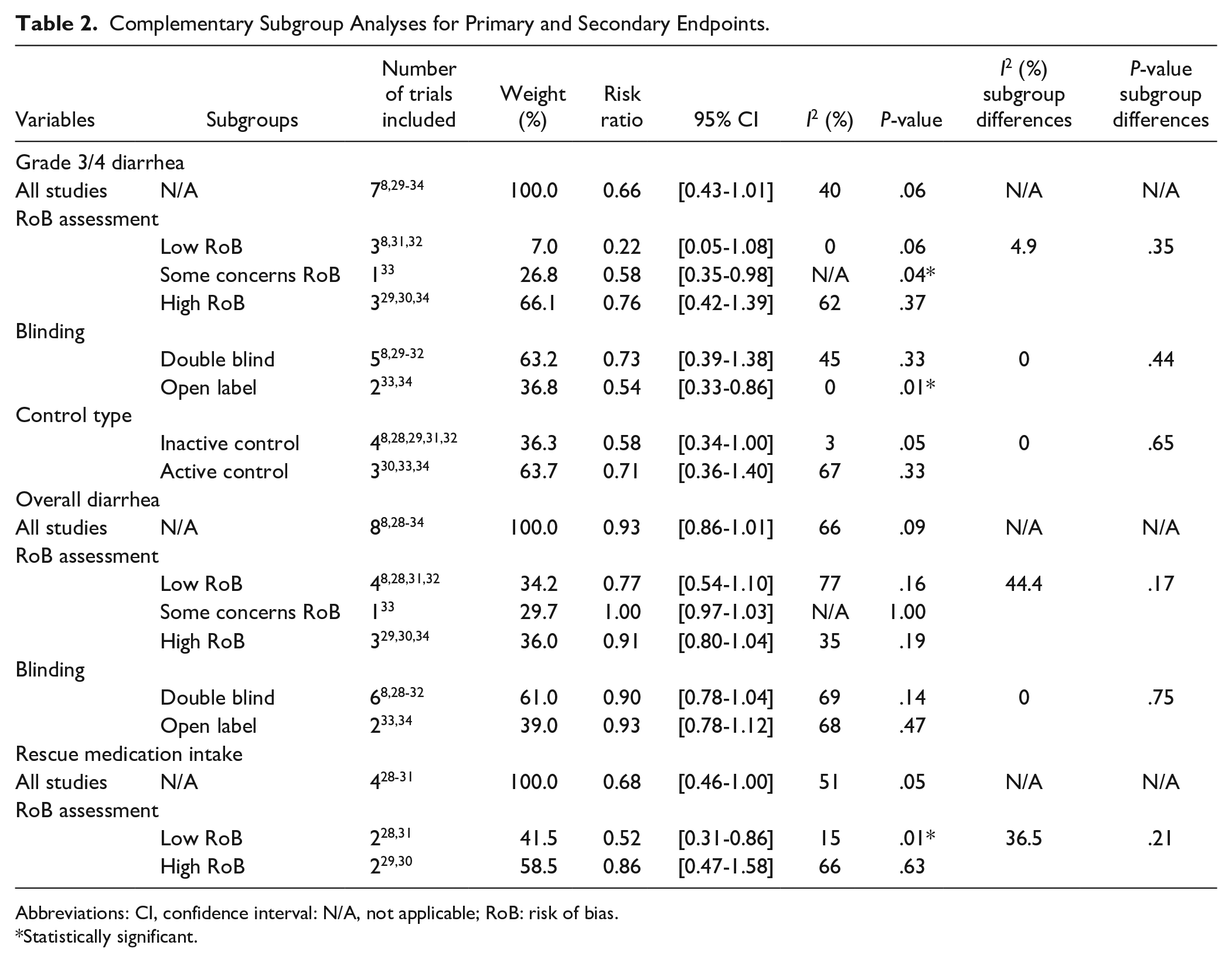
Abbreviations: CI, confidence interval: N/A, not applicable; RoB: risk of bias.
Statistically significant.
The funnel plot on Figure 4 did not indicate severe asymmetry; however, the number of included studies was too low to conclude either the presence or absence of a publication bias.

Funnel plot of sensitivity analysis—grade 3/4 diarrhea—all studies.
The subgroup analysis based on RoB assessment did not show a statistically significant difference between the subgroups; however, we observed a decrease in the treatment effect with a higher RoB assessment (Table 2). We decided to exclude the high-RoB studies to explore the source of heterogeneity.
After the exclusion of high RoB studies, probiotics significantly reduced severe diarrhea by 47% compared to the control group (RR = 0.53 [0.32-0.87]; P = .01), while heterogeneity was no longer present (I 2 = 0%; Figure 5).

Sensitivity analysis—studies with overall “low” and “some concerns” RoB evaluation.
Subgroup analysis by cancer type
Subgroup analyses revealed that probiotics significantly reduced the risk of developing grade 3/4 diarrhea in colorectal cancer patients (RR = 0.56 [0.34-0.92]; P = .02; Figure 6).

Sensitivity analysis—colorectal cancer versus gynecological cancer.
Subgroup analysis by cancer treatment type
In patients treated with chemotherapy alone in comparison with the control group, probiotics significantly reduced the risk of developing grade 3/4 diarrhea (RR = 0.34 [0.12-0.94]; P = .04; Figure 7).

Sensitivity analysis—radiotherapy (+ chemotherapy) versus chemotherapy (alone).
Subgroup analysis: By control treatment type
We also carried out a subgroup analysis based on active and inactive control treatment. No statistically significant subgroup differences were detected (Table 2).
Subgroup analysis: Double-blind versus open-label studies
A comparison of open-label studies with double-blind studies did not produce statistically significant subgroup differences (Table 2).
Secondary endpoint: Incidence of chemotherapy ± radiotherapy induced overall diarrhea—low RoB studies
The heterogeneity assessment indicated considerable heterogeneity (I 2 = 77%; P = .005; Figure 8). Since we excluded the Chitapanarux et al study from the calculation of the primary endpoint, we decided to run the analysis of this endpoint without this particular study to discover if it changed the heterogeneity. The heterogeneity declined substantially (I 2 = 14%; Figure 9), and the risk of developing overall diarrhea in patients receiving probiotics was significantly reduced by 36% (RR = 0.64 [0.48-0.86]; P = .003).
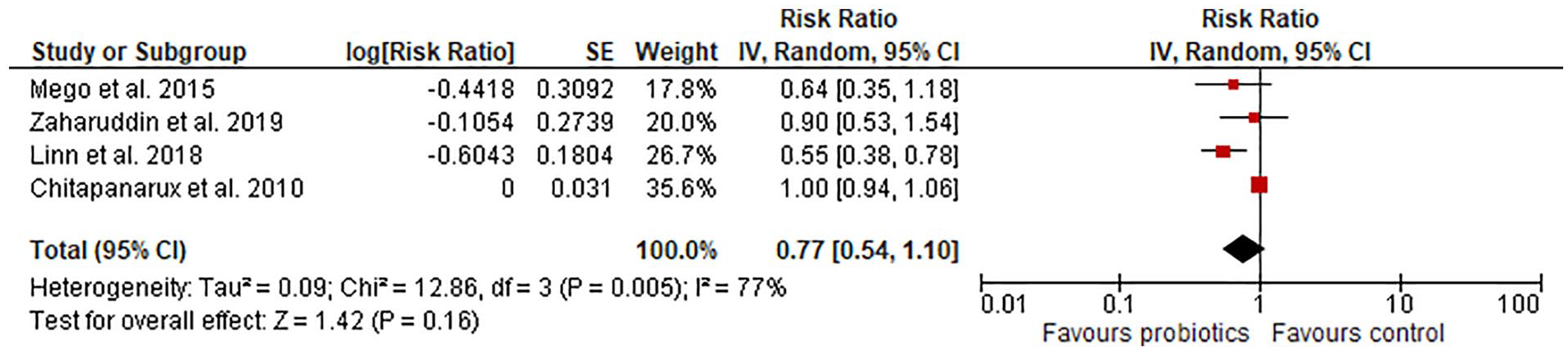
Studies rated as having a low overall risk of bias.
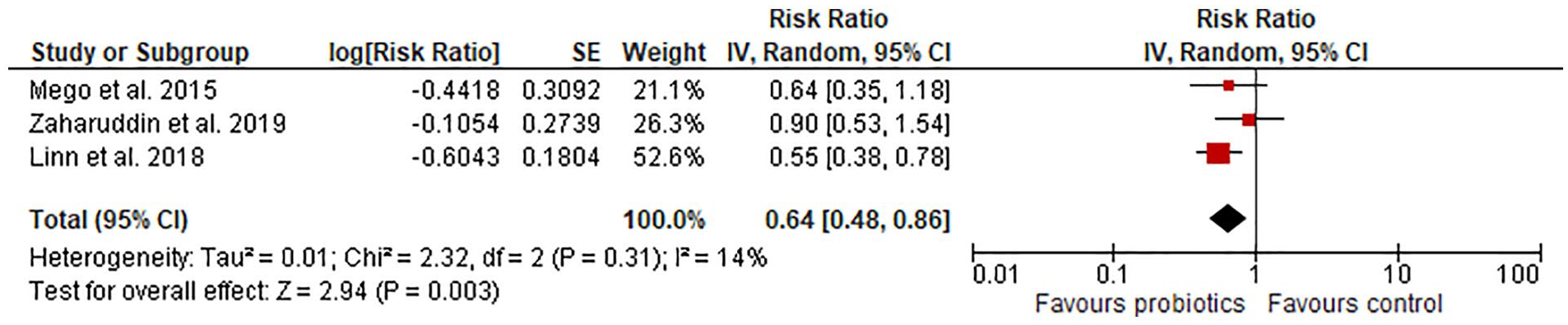
Studies rated as having a low overall risk of bias—Chitapanarux et al excluded.
Secondary endpoint: Incidence of chemotherapy ± radiotherapy induced overall diarrhea—all studies
In the sensitivity analysis of all studies, probiotics failed to prove their beneficial effects (Table 2). No statistically significant subgroup differences were detected, either in the subgroup analysis based on the RoB assessment or in the comparison of open-label studies with double-blind studies (Table 2).
Secondary endpoint: Rescue medication intake
We identified only 2 studies that had low RoB and contained rescue medication intake data. We used a random-effects model since the assessment of heterogeneity indicated a moderate amount. In low-RoB studies, probiotics reduced rescue medication intake by 48% (RR = 0.52 [95% CI 0.31-0.86]; P = .01). In the sensitivity analysis of all studies, probiotics reduced the risk of rescue medication intake by 32% compared to the control group (RR = 0.68 [0.46-1.00]; P = .05); a result is on the edge of the P value threshold for statistical significance (Table 2).
Discussion
Based on RoB evaluation, we performed a synthesis of data gathered from original articles that studied cancer patients treated with chemotherapy (± radiotherapy) and concomitantly with well-identified live orally administered microbial strains.
In comparison with other meta-analyses,6,35-38 the present work includes the Motoori et al 34 study and 2 others published recently.31,32 Next, we drew a conclusion based on the low-bias studies. Finally, we included studies with the well-defined strains only; since the probiotic effects are strain specific, this criterion filtered out studies that may have been performed with low-quality products that might have biased the outcome. We also defined the route of administration that excludes microbes acting in the oral cavity, which are unlikely to have any substantial impact on the gut environment.
Our findings are almost consistent with those of previous systematic reviews, specifically that certain microbial strains or their combinations may provide mild benefits to certain groups of cancer patients in the form of reducing the severity of the adverse effects of anticancer therapy on the gastrointestinal system.6,35-38
Our analysis of low RoB studies showed that the risk of developing grade 3/4 diarrhea in patients receiving probiotics was reduced by 78% compared to the control group (NSS). The sensitivity analysis of all studies showed a reduction of 34% (NSS). It seems that probiotics have beneficial effects, particularly in patients with colorectal cancer. Lu et al 37 reported a significant decrease in the incidence of grade 3/4 diarrhea by 84% in a sample of 186 patients. The majority of the publications included in the meta-analysis of Lu et al were in Chinese, which explains the larger sample size and statistically significant outcome. Furthermore, Lu et al excluded RCTs that combined chemotherapy with radiotherapy. Our subgroup analysis of patients treated with chemotherapy alone produced similar results. Lu et al 37 outlined the poor methodological quality of the studies included; statistical data analyses were performed regardless of RoB assessment outcomes. In patients treated with chemoradiotherapy, the authors Lin and Shen 38 observed a significant decrease in grade 3/4 diarrhea in the probiotic group compared to the control group. In addition to the fact that the analysis included patients treated with radiotherapy alone, the sample size was much larger (n = 1259). Wang et al 35 published results where the effect of probiotics was expressed as an odds ratio (OR). A significant reduction in the incidence of NCI-CTCAE grade 3/4 diarrhea in patients treated with radiotherapy (± chemotherapy) was found (OR = 0.35 [95% CI 0.13-0.96]; P = .04), although for those patients treated with chemotherapy alone, the results were NSS.
Regarding the secondary endpoint related to the overall diarrhea incidence, the effects of probiotics were NSS, and the heterogeneity assessment indicated considerable heterogeneity. To decrease heterogeneity, we ran the analysis without the Chitapanarux et al study. The analysis showed that the risk of developing overall diarrhea in patients receiving probiotics was reduced by 36% (P = .003; I 2 = 14%). This outcome is consistent with the finding of Lu et al, 37 who reported a significant reduction in the total diarrhea rate of 53% in a sample of 441 patients. Wei et al 6 conducted a meta-analysis in regard to control treatment type, distinguishing between active and placebo control. Considering the placebo-controlled trials, 3 studies with 771 participants contained data relevant for the prevention of radiotherapy (± chemotherapy)-induced overall diarrhea; however, no totals were calculated. For chemotherapy-induced overall diarrhea, a beneficial effect of probiotics was found. The incidence of overall diarrhea was also significantly reduced in 2 further meta-analyses.36,38
The analysis of low RoB trials (n = 117 participants) suggests that probiotics may reduce the risk of rescue medication intake; on the other hand, the analysis of all studies produced results on the edge of statistical significance. In other reviews, it was not possible to either demonstrate or refute a difference in effect for this endpoint.6,36
No significant increase in the incidence of AEs was found in the probiotic groups,37,38 suggesting that the employment of probiotics should be further researched.
The strength of our systematic review lies in the inclusion of studies having overall low RoB assessments that provide more accurate statistical evidence. We also performed sensitivity and subgroup analyses, when applicable. Next, we rigorously specified parameters, formulated clinical questions, and submitted methodological and analytical criteria to PROSPERO prior to a comprehensive search of the literature in predefined databases. Furthermore, we provided a precise implementation of the methodology in line with the Cochrane Collaboration recommendations in the Cochrane Handbook for Systematic Reviews of Interventions. If concerns were raised, we contacted some of the authors of the original articles (n = 5) for further information to assess the quality of the studies correctly. Probiotic properties, dosages, and effects seem to be strain specific.39-41 Hence, we excluded trials testing microorganisms that were not identified at the strain level and trials investigating tyndallized (heat-killed) microorganisms. We also decided not to perform any subgroup analysis based on probiotic dosage (CFU). In addition, we would like to emphasize the importance of taking into account the doses of the specific strains that have been shown to be effective in individual studies (Table 1).
Our review has several limitations. The first is clinical heterogeneity. In the systematic summary, we included a variety of diagnoses, predominantly pelvic cancers and colorectal cancer, and 1 study evaluating patients with esophageal cancer. The heterogeneity of the cancers in the individual studies is followed by heterogenic cytostatic regimes, whereas the effectiveness of probiotics may vary in distinct regimes. Moreover, we could not determine which study patients received concomitant biological therapy that could possibly enhance the gastrointestinal toxicity of the chemotherapy. 42 It is not clear which patients underwent pelvic radiotherapy along with chemotherapy. This kind of radiotherapy can either directly cause gastrointestinal toxicity or contribute to its development. 4
As a second limitation, we identified the variety of probiotic cultures, the timing of their administration, their dosages, and concomitant treatments (Table 1). The beneficial properties of microorganisms are strain specific, so it is imperative that the results of the meta-analysis be interpreted carefully. In an ideal case, the data should be synthesized on the basis of the strain/combination of strains used, as in recently published meta-analyses.43,44
A third limitation is that the tools used for the evaluation of severity of the adverse effects vary across the individual studies. In 3 of the studies, the severity of diarrhea was evaluated using NCI-CTCAE 2.0; in 1 study, it was evaluated using NCI-CTCAE 3.0; in another 3 studies, it was evaluated using NCI-CTCAE 4; and in 1 study, it was evaluated using the WHO tool. The differences in the evaluation grades are not only between NCI-CTCAE and WHO but also between the individual versions of NCI-CTCAE. We think that a potential for bias can exist mainly in reporting lower grades of diarrhea (eg, no diarrhea vs grade 1), leading to misreporting of the “overall incidence of diarrhea” rather than severe diarrhea. For example, NCI-CTCAE 4 defines diarrhea as “a disorder characterized by frequent and watery bowel movements,” while the newest version of NCI-CTCAE 5 defines diarrhea as “a disorder characterized by an increase in frequency and/or loose or watery bowel movements.” In the latter, the “and/or” is especially confusing for the grade 1 diarrhea definition “Increase of <4 stools per day over baseline, mild increase in ostomy output compared to baseline.” Due to the difference, there is a potential for confusion. The investigators evaluating diarrhea according to the NCI-CTCAE 5 may report grade 1 diarrhea even when 2 solid stools occur in a particular day in a patient who has been regularly having a 1 bowel movement a day. Herein, we want to point out the urgent need for using unified criteria throughout future studies.
The inclusion of studies in which a sterilized active product 30 or guar gum 33 were used as controls may not be an ideal approach—these substances can also possibly exhibit health effects. This was not confirmed by the subgroup analysis (Table 2), which did not show a statistically significant difference between groups.
Ultimately, the number and sample size of the included studies were low, which could be attributed to language limitations, since there exist a number of trials published in Chinese. 37 In addition, I 2 -values should be interpreted cautiously when a meta-analysis has few studies. 45
The interpretation of NSS results obtained from a small sample size data analysis is always challenging. 46 Present findings in favor of using probiotics to reduce the adverse effects of chemotherapy are currently inapplicable to practice, mostly due to heterogeneity in the reported data, such as regimens (chemotherapy ± radiotherapy) and their duration, population, cancer type, microbial strains, duration of probiotic intervention, and diarrhea assessment tools. Nevertheless, the current data support the assumption that in the future, the administration of live microorganisms could become a potent tool for improving the safety of specific chemotherapy-based regimens.
Conclusions
We have shown that in trials rated as low RoB, orally administered probiotics failed to prove a preventive effect of statistical significance against the development of severe and overall diarrhea in cancer patients treated by chemotherapy (± radiotherapy). The sensitivity analyses of all studies for severe and overall diarrhea produced similar results. Despite its statistical insignificance, we cannot rule out that the effects of probiotics are clinically relevant; when we excluded the high-RoB studies, the risk of developing grade 3/4 diarrhea in patients receiving probiotics was significantly reduced, and it seems that probiotics exhibit larger effects in patients with colorectal cancer and those treated with chemotherapy alone. Furthermore, the analysis of low-RoB trials suggests that probiotics may reduce the risk of rescue medication intake. Additional well-designed and well-executed trials are required to enable the analysis of a larger sample size and consequently the achievement of conclusive answers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported from the state budget through the Ministry of Education, Youth and Sports by project VI CZECRIN (LM2018128) and from the European Regional Development Fund – project CZECRIN_4 PATIENTS (CZ.02.1.01/0.0/0.0/16_013/0001826). The article processing charge is covered by the authors.
