Abstract
Hepatocellular carcinoma (HCC) is one of the common cancers and lethal diseases worldwide. Both oxidative stress and chronic inflammation contribute to the pathogenesis of HCC. Because of limited treatment options and a grave prognosis of HCC, preventive management has been emphasized. The marine macroalgae Ulva lactuca (Ulvaceae) is consumed by humans and livestock because of its nutritional value. Recent studies showed that various extracts of U. lactuca possess antiviral, antiplasmodial, antinephrotoxic, antioxidant, and anti-inflammatory properties. However, very limited information is available on anticancer potential of U. lactuca with no reports on liver cancer chemopreventive efficacy of this marine algae. Accordingly, the present study was initiated to evaluate the possible antihepatocarcinogenic effects and antioxidant mechanisms of action of various U. lactuca extracts against a clinically relevant rodent model of HCC. Initiation of hepatocarcinogenesis was performed in Sprague-Dawley rats by a single injection of dietary carcinogen diethylnitrosamine (DENA, 200 mg/kg, intraperitoneally), followed by promotion with phenobarbital (0.05%) in drinking water. The rats were fed with daily oral dose (50 mg/kg) of polysaccharide sulfate or aqueous extract of U. lactuca for 2, 12, and 24 weeks. At these timepoints, blood samples were taken to measure hepatic injury markers, including alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, γ-glutamyl transferase, and bilirubin. The liver tissue was harvested for measurement of hepatic oxidative indices, including lipid peroxidation, reduced glutathione, nitric oxide, catalase, superoxide dismutase, glutathione reductase, and glutathione S-transferase. Hepatic histopathology, immunohistochemical analysis of cell proliferation and apoptosis by DNA fragmentation assay were performed. Our results clearly indicate that sulfated polysaccharides of U. lactuca exert a marked chemoprevention of DENA-initiated hepatocarcinogenesis through inhibition of abnormal cell proliferation and induction of apoptosis. A modest inhibition rat liver carcinogenesis was observed with the aqueous extract. The sulfated polysaccharides altered serum parameters of hepatic damage and modulated various components of the hepatic enzymatic and nonenzymatic antioxidant defense systems. The sulfated polysaccharides from U. lactuca may have unique properties of providing protection against DENA-induced oxidative stress which could contribute to chemoprevention of experimental hepatocarcinogenesis. U. lactuca sulfated polysaccharides could be developed as chemopreventive and therapeutic drug against human HCC.
Keywords
Introduction
Carcinogenesis is an active process induced by one or several genotoxic or carcinogenic agents and is generally preceded in sequential steps from the time of administration to the progression of visible preneoplastic lesions. The multistep process of neoplasia has been demonstrated in several organ systems, most notably in the rat liver. 1 Nitrosamines are an important class of environmental carcinogen owing to their carcinogenic and mutagenic properties. The sources of human exposure to nitrosamines include agricultural chemicals, tobacco products, pharmaceutical preparations, cosmetics, and food preservatives. 2 Diethylnitrosamine (DENA) is frequently used to induce hepatocarcinogenesis in experimental animal models.3,4
Hepatocellular carcinoma (HCC) accounts for 80% to 90% of all liver cancers and is the third most common cause of cancer morbidity, 5 resulting in almost one million deaths every year. 6 Lack of effective diagnostic tools for the detection and limited surgical resection of advanced HCC contribute to a grave prognosis connected with high mortality from this disease. Recently, much attention has been directed to the role of oxidative stress and apoptosis evasion in the pathogenesis of HCC and these events are being widely recognized as the central features of the disease. 7 Therefore, efforts for developing less toxic therapies targeting antioxidant system and behavior of malignant cells are necessary to manage hepatocellular cancer. A critical need also exists for the discovery and development of novel preventive therapies without adverse side effects to reverse the current mortality associated with HCC.8-11
Recently, identification of bioactive constituents from medicinal and dietary plants to suppress hepatic tumorigenesis is gaining considerable attention.12-16 Marine algae have long been used as a fresh food with potential nutritional benefits and treatment for various disorders. 17 Various marine bioactive compounds such as phenolics, carotenoids, terpenoids, and polysaccharide sulfates, exhibit potent antioxidant activities. The antioxidant properties of these compounds are attributed to the ability of scavenging superoxide and hydroxyl radicals, quenching singlet and triplet oxygen and exhibiting reducing power.18-20
Ulva lactuca (family: Ulvaceae) is a prevalent macroalgae present in the intertidal zone, in calm and protected seaports as deep as 10 meters and in northern climates. U. lactuca grows along rocky or sandy shores of oceans. In some parts of the world, the seaweed is greatly consumed by humans and livestock because of its nutritional value. Recent studies have showed that various extracts of U. lactuca possess biological and pharmacological activities, such as anticoagulant, antiviral, antiplasmodial, antinephrotoxic, protein kinase A–inhibitory, antioxidant, and anti-inflammatory properties.20-25 However, there are only few reports on the antitumor effect of U. lactuca in the literature. In one study, sulfated polysaccharides from U. lactuca inhibited the proliferation and differentiation of Caco-2 colon cancer cells without any effect on normal colonocytes. 26 At least in one in vivo study, an extract from U. lactuca showed antineopastic activity against Ehrlich ascites carcinoma in mice. 27 In spite of significant promise of U. lactuca as an important marine source of novel anticancer agent, chemopreventive efficacy of this alga has not been investigated against HCC to the best of our knowledge and belief. Hence, the present study has been undertaken and designed to evaluate the possible antihepatocarcinogenic effect of various extracts of U. lactuca against a sophisticated and clinically relevant rodent model of liver cancer. The possible involvement of antioxidant activities of U. lactuca constituents has also been explored by analyzing serum and hepatic biochemical parameters in experimental animals.
Materials and Methods
Chemicals
All the chemicals and reagents were obtained from Sigma-Aldrich (St Louis, MO, USA), Randox chemical company (Antrim, Northern Ireland, UK), Bio-Merieux Company (Marcy l’Etoile, France), and Spinreact (Girona, Spain).
Preparation of U. lactuca Aqueous Extract and Sulfated Polysaccharides
Fresh seaweed samples were collected in polythene bags from the intertidal regions of the Red Sea western coastal regions of Egypt, particularly from Marsa Alam (Lat. 25° 04′ 0.48″ N; Long. 34° 54′ 7.2″ E) and El-Qusair district (Lat. 26° 06′ 54″ N; Long. 34° 16′ 58.08″ E). The air shade-dried algae were roughly cut and minced using a mechanical blender. The dried seaweed (100 g) was rehydrated with 1:1 of distilled water and heated at 100°C for 1 hour, respectively. After centrifugation, the supernatant was precipitated with ethyl alcohol (3 volumes) and then freeze-dried to yield the polysaccharide sulfate extract. The precipitate was then washed with distilled water and ethyl alcohol several times to remove salts and minerals and subsequently dried to remove any traces of alcohol. The polysaccharide sulfate fraction was dissolved in boiling quality water (qH2O) and used for animal experimentation. The aqueous extract was prepared by adding 50 mg of the powdered algae to 100 mL of distilled water followed by boiling for 15 minutes. The solution was then brought to room temperature, filtered, and orally administered to experimental animals.
Analysis of Sugar and Sulfate Contents in Polysaccharide Extract
The sugar content of polysaccharide sulfate fraction was analyzed by the phenol–sulfuric acid method. 28 The sulfate content was determined by the turbidimetric method. 29
Measurement of antioxidant activities
The free radical scavenging activity for the sulfated polysaccharide extract was determined by using a stable free radical, α,α-diphenyl-β-picryhydrazyl (DPPH). 30 Briefly, 1 mg extract was dissolved in 1 mL qH2O followed by addition of DPPH (1×10−4 M in methanol). After 30 minutes at room temperature, the amount of DPPH was determined spectrophotometrically at 517 nm. The capability of scavenging free radicals was calculated using the following equation:
The superoxide radical scavenging ability of the sulfated polysaccharide fraction was assessed by the method of Liu and Ng. 31 The reaction mixture, containing the ulvan sulfated polysaccharide extract (1 mg/mL), 0.1 M Tris-HCl (pH 8.0), 936 μM NADH, 300 μM nitroblue tetrazolium, and 120 μM phenazine methosulfate, was incubated at room temperature for 5 minutes and the absorbance was read at 560 nm. The capability of scavenging superoxide radical was calculated using the following the equation:
Animals and Maintenance
Pathogen-free male Sprague-Dawley rats, initially weighing 90 to 100 g, were used in the experiment and obtained from the National Research Center (Dokki, Giza, Egypt). Experimental animals were housed in a conventional animal facility. The animals were acclimatized to standard conditions (temperature 24°C ± 1°C, relative humidity 55% ± 5% and a 12-hour photoperiod) and were housed in stainless steel cages (4-6 rats per cage) for 2 weeks before commencement of the experiment. The animals were kept under observation for this period before initiating the experiment to exclude any recurrent infection. During the entire period of study, the rats were provided with normal basal diet (El-Nasr, Giza, Egypt) and water ad libitum. The animal procedures were conducted according to the Canadian Committee for Animals Use and Care 32 and were approved by Faculty of Science, Beni-Suef University.
Experimental Design for Chemopreventive Study
Pathogen-free rats were randomly divided into 4 experimental groups (18 rats in each). Group A (normal control) rats were maintained on normal basal diet and drinking water and injected with 1 mL of saline (0.9% NaCl). Group B rats were designated as the carcinogen control and received a single necrogenic, intraperitoneal injection of DENA (200 mg/kg body weight) dissolved in 0.9% saline at the age of 9 weeks. After 1 week, all DENA-initiated rats were given phenobarbital (PB) as the promoter in drinking water (0.05% w/v) and it was continued thereafter till the end of the study. Groups C and D rats were treated exactly as group B and additionally fed with oral dose (50 mg/kg body weight) of polysaccharide sulfate or aqueous extract of U. lactuca (50 mg/kg body weight), once a day, respectively, throughout the entire period of study. Food and water intake as well as behavioural changes were monitored every day, and the body weights of animals were recorded every week. All the animals were sacrificed at 2, 12, and 24 weeks following the initiation of the study by decapitation under light ether anaesthesia in order to measure various endpoint biomarkers (Figure 1). For the last 4 days, PB was removed from the drinking water and the animals did not receive the polysaccharide fraction or aqueous extract. The animals were fasted overnight prior to being sacrificed.

Schematic view of experimental design.
Blood Sampling for Biochemical Assays
At the end of each experimental period (ie, 2, 12, or 24 weeks), 6 rats from each group were sacrificed under light ether anaesthesia following overnight fasting state. Blood samples were collected through the punctured jugular vein in chilled nonheparinized tubes, allowed to coagulate at room temperature, and then centrifuged at 3000 rpm for 20 minutes. The clear, nonhemolyzed supernatant was quickly removed and stored at −20°C for subsequent biochemical analysis.
Estimation of Liver Injury Markers in Blood
Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were measured based on the principles described by Reitman and Frankel. 33 The activity of alkaline phosphatase (ALP) was estimated according to the method described by Belfield and Goldberg. 34 γ-Glutamyl transferase (γ-GT) was determined by the method of Shaw et al. 35 The total bilirubin level (TBL) was determined on the basis of its coupling with diazotized sulfanilic acid in the presence of caffeine to yield an azo-dye according to the method of Jendrassik and Grof. 36 Total protein and albumin concentrations were determined colorimetrically by the methods adopted by Henry 37 and Doumas et al, 38 respectively.
Tissue Processing
After sacrifice, liver tissue was immediately excised and divided into 2 parts. One part was kept at 10% neutral-buffered formalin for histopathological investigation at Histology Unit, National Cancer Institute, Cairo, Egypt. The other part was rinsed with saline (0.9% NaCl), then homogenized in phosphate buffer (10 mM, pH 7.4) which contained 1.15% potassium chloride and 1.15% ethylenediamine tetraacetic acid (EDTA) and centrifuged at 3000 rpm for 20 minutes to obtain tissue homogenate supernatant. The supernatant was collected and used for the estimation of several endpoint parameters as described below.
Biological Assays
Lipid Peroxidation
A modified method of Preuss et al 39 was used to measure the level of lipid peroxides by measuring the malondialdehyde (MDA) formed in the liver tissue. Briefly, 1 mL aliquots of liver homogenate supernatant were precipitated with 2 ml of 7.5% trichloroacetic acid (TCA), then centrifuged at 1000 rpm for 10 minutes, and the supernatant was collected. One milliliter of 0.7% thiobarbituric acid was added to the supernatant and incubated at 80°C for 30 minutes. 1,1,3,3-Tetramethoxy propane was used as the standard. The amount of MDA formed was determined by measuring the optical density of the supernatant at 532 nm. The results were expressed as nanomoles of MDA formed per gram tissue.
Reduced Glutathione
Reduced glutathione (GSH) level was measured by the modified method of Beutler et al. 40 Briefly, 200 µL of the liver homogenate was mixed with 1.5 mL of precipitating solution (1.67 g glacial metaphosphoric acid, 20 mg EDTA, and 30 g NaCl per 100 mL of distilled water) and allowed to stand for 5 minutes. After centrifugation at 3000 rpm for 20 minutes, the supernatant was collected, mixed with Ellman’s reagent (5,5-dithio-bis[2-nitrobenzoic acid]), and the absorbance was measured at 412 nm.
Nitric Oxide
Nitric oxide (NO) is relatively unstable in the presence of molecular oxygen. With a short half-life of 3 to 5 seconds, it is rapidly oxidized to nitrite and nitrate, which are collectively designated as NOx. A high correlation between endogenous NO production and nitrite/nitrate (NOx) levels has been established. The measurement of these levels was performed by the method of Montgomery and Dymock. 41
Superoxide Dismutase
The activity of superoxide dismutase (SOD) in the liver was assayed by the technique of Marklund and Marklund 42 based on the rapid auto-oxidation of pyrogallol in aqueous solution. To 1 mL of the homogenate supernatant, 100 µL Tris-buffer was added followed by 5 μL pyrogallol. The change in absorbance at 430 nm was determined by subtracting the initial absorbance immediately measured after the addition of pyrogallol from the final absorbance measured after incubation for 10 minutes.
Catalase
Catalase (CAT) activity was assayed by the method described by Cohen et al. 43 To the aliquot of liver supernatant, ethanol was added to a final concentration of 0.17 M and the samples were incubated for 30 minutes in an ice-water bath. After 30 minutes, 10% Triton X-100 was added to a final concentration of 1%. Cold isotonic buffer was then added to produce a 100-fold dilution. Five hundred microliters of sample was added to 5 mL of H2O2 and then vortexed for 3 minutes. One milliliter of H2SO4 was added followed by 7 mL of KMnO4 and the resultant solution was read at 480 nm.
Myeloperoxidase
Myeloperoxidase (MPO) activity was assayed using Manoranjan and Mishra’s 44 method, which is based on measurement of pyrogallol oxidation to purpurogallin by peroxidase when catalyzed by myeloperoxidase at 420 nm at 25°C. To 1 mL of liver homogenate supernatant, 3 mL phosphate-buffered saline (PBS), 315 μL pyrogallol, and 154 μL H2O2 were added. The mixture was incubated for 15 minutes at 25°C, the reaction was stopped by addition of 500 µL of 5% (v/v) H2SO4 and the absorbance was read at 420 nm.
Glutathione Reductase
Glutathione reductase (GR) catalyzes the reduction of oxidized glutathione (GSSG) in the presence of NADPH, which was oxidized using a reagent kit. The oxidation of NADPH to NADP was accompanied by a decrease in the absorbance at 340 nm and was directly proportional to the GR activity in the sample. 45
Glutathione S-Transferase
Glutathione S-transferase (GST) activity was measured by method described by Habig et al. 46 based on conjugation of 1-chloro-2,4-dinitro benzene (CDNB) with GSH that produced dinitrophenyl thioether, which was detected spectrophotometricaly at 340 nm.
Histopathological Assessment
Tissue specimens were dehydrated in a graded ethanol series, cleared in xylene, immersed in Paraplast wax, and sectioned at 5 µm thickness using a microtome. Liver sections were mounted on coated slides (Thermo Scientific, Menzel-Gläser, Braunschweig, Germany). The tissue sections were consequently deparaffinized and then stained by haematoxylin and eosin for histopathological assessment.
Cell Proliferation Assay by Immunohistochemistry
Immunohistochemical detection of proliferating cell nuclear antigen (PCNA) as a cell proliferation marker in liver sections was performed by the modified method of Hsu et al. (2002) 47 as previously described by Bishayee and Dhir (2009). 48 Briefly, the sections were hydrated in PBS 1×, incubated for 10 min at 80°C in 10 mM sodium citrate buffer (pH 6.0) for antigen retrieval, and slowly cooled to room temperature for 20 min. The tissue sections were washed with PBS and the endogenous peroxidases were blocked by 1% H2O2 followed by incubation with primary anti-PCNA antibody (mouse monoclonal from Santa Cruz Biotechnology, Santa Cruz, CA) at a 1:1000 dilution for 30 minutes. The sections were incubated in a secondary antibody (rabbit anti-mouse IgG-B, 1:200 dilution) for 30 minutes using a mouse ABC staining system (sc-2017 from Santa Cruz) and treated with avidin and biotinylated horseradish peroxidase (ABC reagent). The chromogenic reaction was developed by 3,3-diaminobenzidine tetrahydrochloride solution for 3 min, and all sections were counterstained with 0.1% fast Mayer reaction solution (Sigma-Aldrich). The sections were dehydrated and mounted. Brownish yellow particles covering the nuclei, visualized under a light microscope (Lyca Vision, LWR, Wetzlar, Germany) at a magnification of 400×, represented the immunopositive PCNA signal. In order to determine the PCNA labeling index (LI), a total of 1,000 hepatocytes per animal were analyzed using a light microscope. The LI was expressed as the number of PCNA-positive hepatocytes × 100/total number of hepatocytes analysed.
Apoptosis by DNA Fragmentation Test
It was measured following the method of Mahvash and Azra. 49 Approximately, 1 mL homogenate containing 100 mg tissue was mixed with 500 µL lysis buffer and centrifuged at 10, 000 rpm for 20 minutes. Two hundred microliters of supernatant was suspended in 200 μL of TCA followed by centrifugation at 5000 rpm for 10 minutes. Both supernatant and pellets were individually added to 1 mL diphenylamine. The supernatant was boiled in a water bath for 10 minutes, cooled in ice, and the resultant blue color was read at 575-600-nm wavelength to estimate fragmented DNA.
Expression of Results and Statistical Significance
All data are presented as mean ± standard error of mean (SEM). Data were analyzed with GraphPad Prism V6.01 software packages (San Diego, CA, USA). One-way analysis of variance (ANOVA) was used to test the significance of differences along the time between treatments and the control as well. Tukey-Kramer multicomparison test was also performed to estimate the changes among different time intervals within the variable by using the error calculated from ANOVA. Percentage change is calculated by comparing DENA-exposed group with normal control and DENA plus U. lactuca-treated groups with DENA control. Pearson’s correlation was used to analyze the relationship between variables. Statistical significance was set at P<0.05.
Results
Chemical Composition and Antioxidant Activities of U. lactuca
The polysaccharide sulfate fraction of U. lactuca was subjected to hot water extraction at 2 different temperatures to obtain A1 (60°C) and A2 (100°C) fractions. The total sugar present in polysaccharide sulfate extracts were determined to be 201.34 and 267.19 μg/mL, while sulfate contents were estimated to be 15.17% and 12.25% for fractions A1 and A2, respectively. The A2 fraction showed greater and statistically significant (P<0.0001) free radical and superoxide anion scavenging activities than A1 fraction (Table 1).
Chemical Analysis of the Sulfated Polysaccharide Extracts Obtained From U. lactuca. a
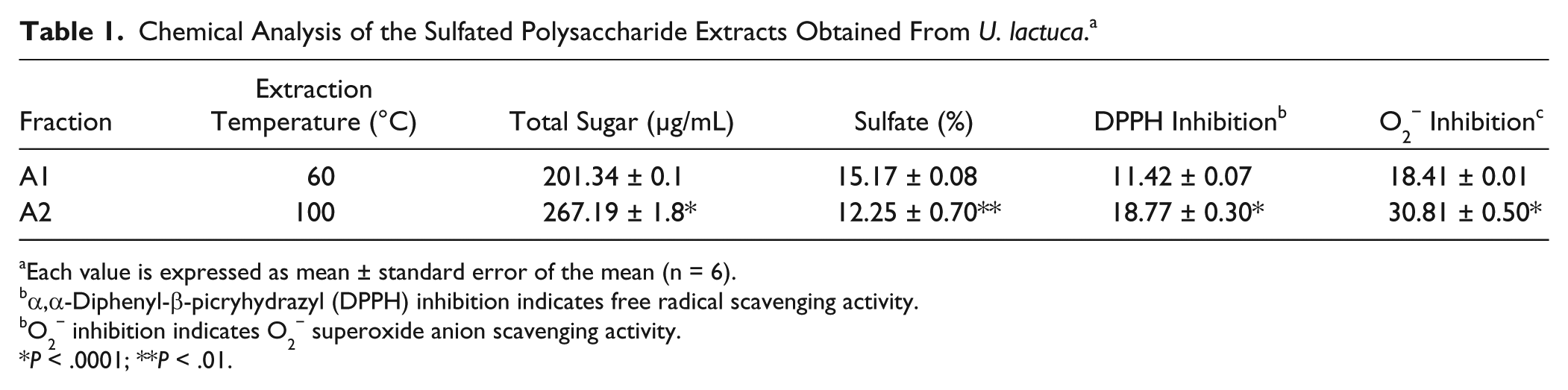
Each value is expressed as mean ± standard error of the mean (n = 6).
α,α-Diphenyl-β-picryhydrazyl (DPPH) inhibition indicates free radical scavenging activity.
O2− inhibition indicates O2− superoxide anion scavenging activity.
P < .0001; **P < .01.
Effects of U. lactuca on Serum and Hepatic Markers of Liver Damage and Hepatocarcinogenesis
Data presented in Table 2 indicate that serum AST, ALT, ALP, and γ-GT activities were mostly significantly (P<0.0001) increased in DENA control rats when compared with the corresponding normal control group at 12 or 24 weeks. The administration of DENA-treated rats with both polysaccharide sulfate and aqueous extract of U. lactuca at daily doses of (50 mg/kg body weight, orally) showed significant (P<0.0001) decrease of these markers as compared with their corresponding DENA control rats.
Effects of Sulfated Polysaccharides and Aqueous Extract of U. lactuca on the Activities of Serum Biomarker Enzymes of Liver Damage During Experimental Rat Hepatocarcinogenesis.*

Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; γ-GT, γ-glutamyl transferase; DENA, diethylnitrosamine.
Data are expressed as mean ± standard error of the mean (n = 6). In the columns, mean values with the same superscript letters are nonsignificant, whereas with different superscript letters are significantly different. The values in parentheses indicate percentage change compared with the corresponding control. Various groups are as follows: group A, normal control; group B, DENA control; group C, DENA + polysaccharide sulfate; group D, DENA + aqueous extract.
As depicted in Figure 2, significant increases were observed in the total bilirubin level at 2, 12, and 24 weeks after DENA administration particularly at 12 and 24 weeks postdose, where the elevation was double and 3-fold in comparison with normal control rats, respectively. On the other hand, abrogation of this elevation by the polysaccharide sulfate treatment to near normal level at all 3 time points was noticed, which was better than the aqueous extract. Significant depletion of total protein (Figure 3) and albumin levels (Figure 4) were detected in DENA-exposed groups compared with normal control at 2, 12, and 24 weeks postdose. Interestingly, treatment with polysaccharide sulfate increased the total protein (Figure 3) and albumin levels (Figure 4) at various time points throughout the study over the DENA-treated groups, and these effects were greater than the treatment with the aqueous extract.
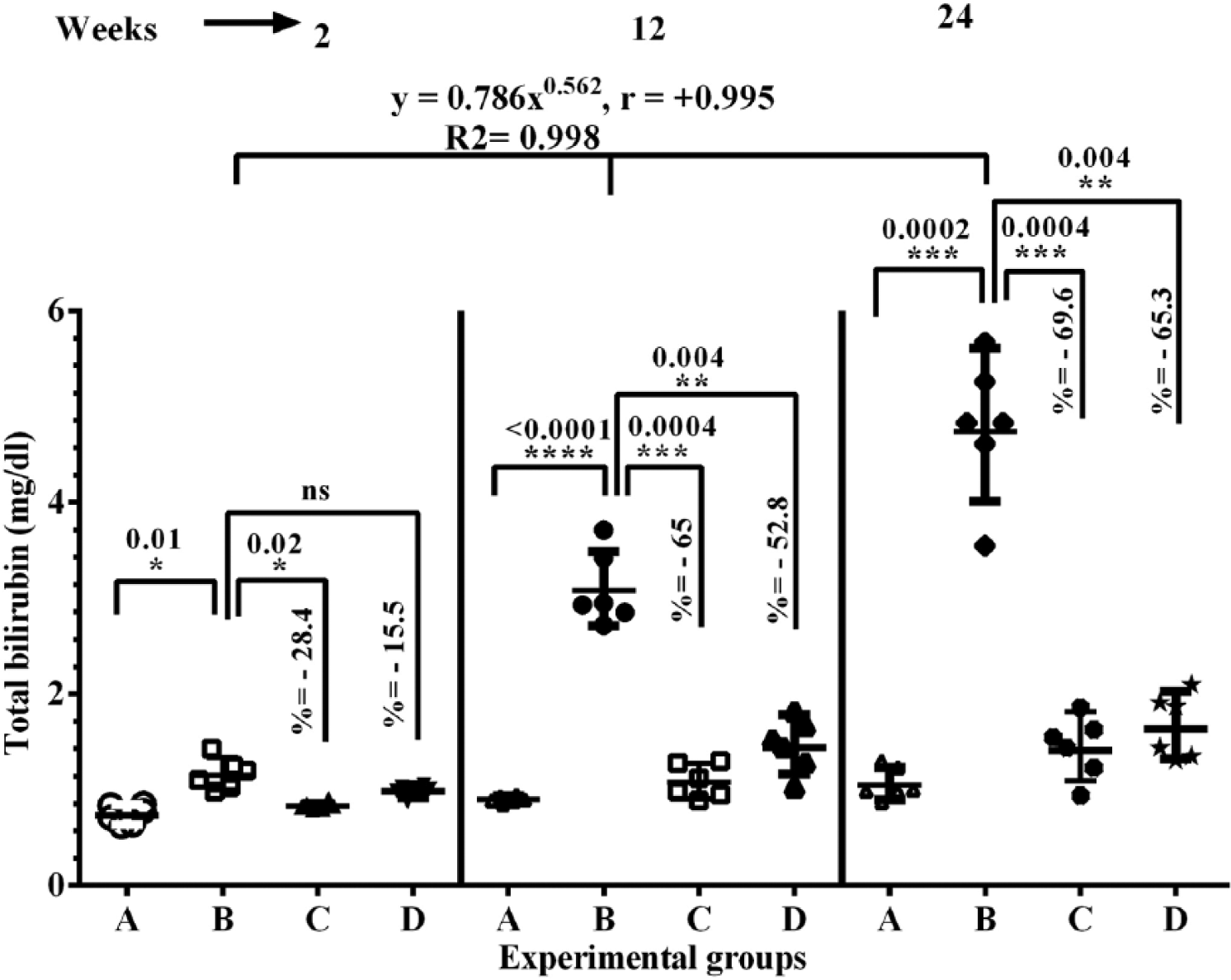
Effects of sulfated polysaccharides and aqueous extract of U. lactuca on the activity of serum total billirubin of the experimental groups. Data are expressed as mean ± standard error of the mean (n = 6). Various experimental groups are: A, normal control; B, DENA control; C, DENA plus sulfated polysaccharides; and D, DENA plus aqueous extract. Comparisons were made between group A and group B (DENA control), and also between group B and group C or D.

Effects of sulfated polysaccharides and aqueous extract of U. lactuca on serum total protein of the experimental groups. Data are expressed as mean ± standard error of the mean (n = 6). Various experimental groups are: A, normal control; B, DENA control; C, DENA plus sulfated polysaccharides; and D, DENA plus aqueous extract. Comparisons were made between group A and group B (DENA control), and also between group B and group C or D.
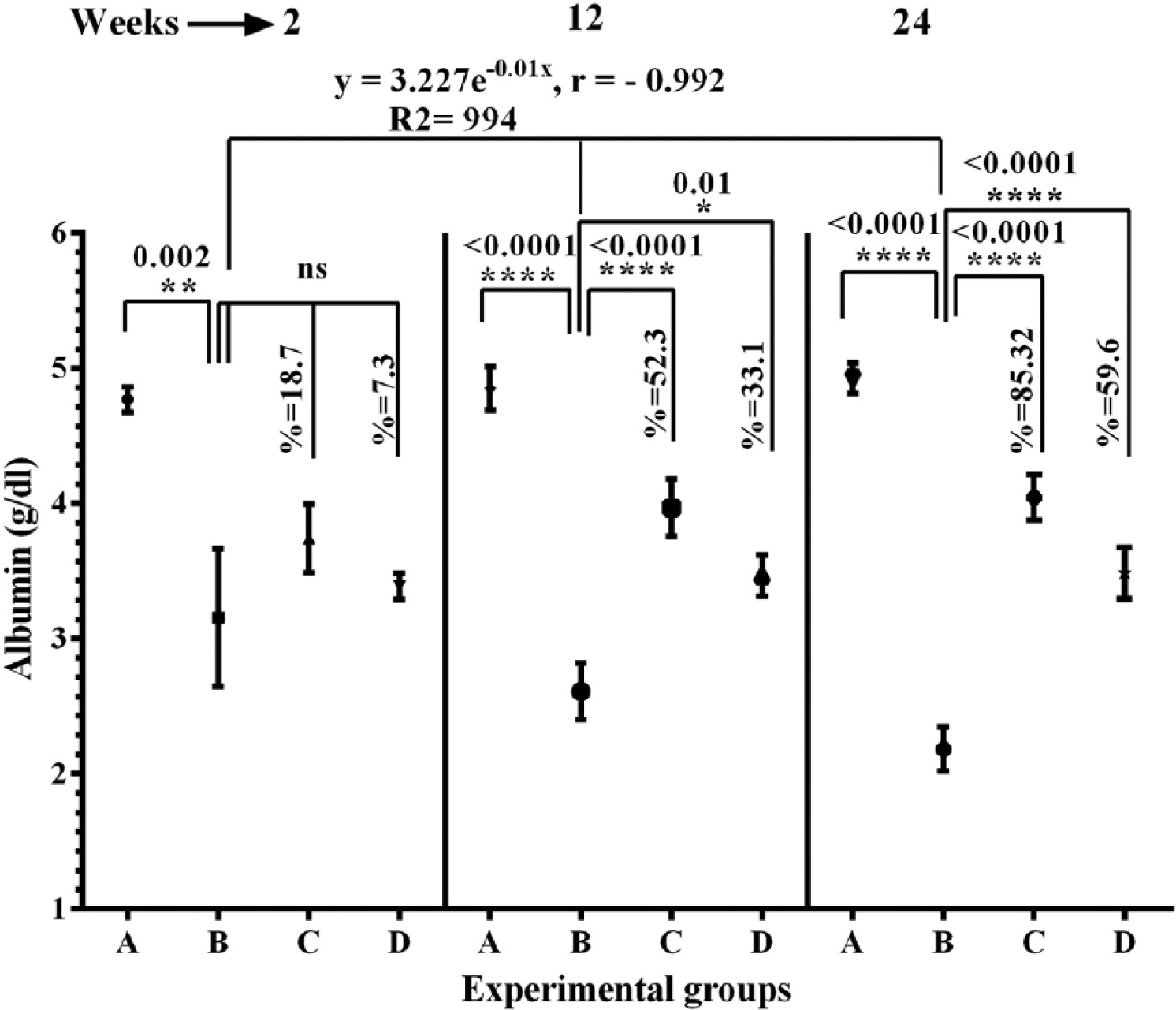
Effects of sulfated polysaccharides and aqueous extract of U. lactuca on serum albumin level of the experimental groups. Data are expressed as mean ± standard error of the mean (n = 6). Various experimental groups are: A, normal control; B, DENA control; C, DENA plus sulfated polysaccharides; and D, DENA plus aqueous extract. Comparisons were made between group A and group B (DENA control), and also between group B and group C or D.
Administration of DENA significantly depleted hepatic MPO levels in group B rats at 12 and 24 weeks compared with nomal control rats in group A (Table 3). Oral polysaccharide sulfate showed significant increase in MPO by 110.81% and 122.95% at 12 and 24 weeks, respectively, compared to DENA control. These findings seem to be more efficient than the aqueous extract of U. lactuca. DENA treatment (group B) decreased the activities of hepatic GR and SOD at various time points (Table 3). However, polysaccharide sulfate significantly improved their levels with greater efficiency than the aqueous extract of U. lactuca at all 3 time points. Regarding the catalase, its activity was declined at 12 and 24 weeks of postdosing with DENA (Table 3). However, its activity was increased at 2 weeks after DENA treatment. The aqueous extract seems to be more effective in reversing the catalase activity at 2 and 24 weeks than polysaccharide sulfate. The activity of GST decreased at 2 weeks and then moderately increased at 12 and 24 weeks after DENA treatment (Table 3). The aqueous extract of U. lactuca showed better results than polysaccharide sulfate in normalizing GST activity at 2 weeks. However, both algal products exerted similar results at subsequent time points.
Effects of Sulfated Polysaccharides and Aqueous Extract of Ulva lactuca on the Activity of Antioxidant Enzymes During Rat Liver Carcinogenesis.

Abbreviations: MPO, myeloperoxidase; SOD, superoxide dismutase; CAT, catalase; GR, glutathione reductase; GST, glutathione S-transferase; DENA, diethylnitrosamine.
Data are expressed as mean ± standard error of the mean (n = 6). In the columns, mean values with the same superscript letters are non-significant, whereas with different superscript letters are significantly different. The values in parentheses indicate percentage change compared with corresponding control. Various groups are as follows: group A, normal control; group B, DENA control; group C, DENA + polysaccharide sulfate; group D, DENA + aqueous extract.
Administration of DENA significantly increased hepatic MDA levels in group B rats after 12 and 24 weeks of our study compared with group A (Figure 5). The polysaccharide sulfate treatment showed significant depletion in MDA content by 57.34%, 46.45%, and 68.37% at 2, 12, and 24 weeks postdose, respectively. These findings seem to be more efficient than the aqueous extract of U. lactuca at 2 and 24 weeks, whereas the aqueous extract showed slightly better result at 12 weeks. DENA treatment (group B) decreased of hepatic GSH levels at 2, 12, and 24 weeks postdose, with a dramatic reduction at 24 weeks (Figure 6). Administration of polysaccharide sulfate restored GSH levels noticeably at 2 and 24 weeks postdose. On the other hand, the aqueous extract elevated the GSH level at 12 and 24 weeks better than the polysaccharide sulfate. The present study showed that declining intramitochondrial GSH level at 24 weeks of DENA administration was closely correlated (r = −0.6) with the increases in lipid peroxidation (Figure 7). The level of NO was significantly increased in DENA-induced rats at 2 and 12 weeks postdose and insignificantly decreased at 24 weeks post-dose in comparison with normal control (Figure 8). The polysaccharide sulfate significantly decreased NO level at 2 and 12 weeks and increased the same after 24 weeks compared with DENA control. Almost comparable results were obtained with the aqueous extract. Interestingly, a strong inverse relationship (r = −0.9) was found between NO level and SOD in DENA control animals at 24 weeks of DENA administration (Figure 9).

Effects of sulfated polysaccharides and aqueous extract of U. lactuca on hepatic MDA level of the experimental groups. Data are expressed as mean ± standard error of the mean (n = 6). Various experimental groups are: A, normal control; B, DENA control; C, DENA plus sulfated polysaccharides; and D, DENA plus aqueous extract. Comparisons were made between group A and group B (DENA control), and also between group B and group C or D. LPO, lipid peroxide; MDA, malondialdehyde.

Effects of sulfated polysaccharides and aqueous extract of U. lactuca on hepatic GSH level of the experimental groups. Data are expressed as mean ± standard error of the mean (n = 6). Various experimental groups are: A, normal control; B, DENA control; C, DENA plus sulfated polysaccharides; and D, DENA plus aqueous extract. Comparisons were made between group A and group B (DENA control), and also between group B and group C or D. GSH, reduced glutathione.

Relationship between hepatic GSH and MDA content 24 weeks following the administration of DENA in rats. The correlation coefficient is −0.6, that is, inverse relationship between the GSH and MDA at 24 weeks of DENA administration. The dotted curves are the border of correlation area; the line is the indicator of correlation strength, that is, the closer the data and line, the stronger the correlation. GSH, reduced glutathione; MDA, malondialdehyde.

Effects of sulfated polysaccharides and aqueous extract of U. lactuca on hepatic NO level of the experimental groups. Data are expressed as mean ± standard error of the mean (n = 6). Various experimental groups are: A, normal control; B, DENA control; C, DENA plus sulfated polysaccharides; and D, DENA plus aqueous extract. Comparisons were made between group A and group B (DENA control), and also between group B and group C or D. NO, nitric oxide.

Relationship between hepatic NO content and SOD activity 24 weeks following the administration of DENA in rats. The correlation coefficient is −0.9, that is, strong inverse relationship between NO level and SOD in DENA control animals at 24 weeks. The dotted curves are the border of correlation area; the line is the indicator of correlation strength, that is, the closer the data and line, the stronger the correlation. NO, nitric oxide; SOD, superoxide dismutase.
Effects of U. Lactuca on Hepatic Histopathological Indices During Hepatocarcinogenesis
The liver section of the normal control rats showed normal hepatic architecture with distinct hepatic cells, sinusoidal spaces, space of Disse, Kupffer cells, and unremarkable central vein (Figure 10A). Examination of liver sections from DENA control rats at 24 weeks postdose revealed the presence of clear cell foci associated with multifocal necrosis, pyknotic nuclei, and the abnormal cell architectural, severe necrosis, karyocytomegaly, prominent Kupffer cell hyperplasia, hepatocytes hypertrophy represented by nuclear cytoplasmic ratios, and hydropic degeneration with pyknotic nuclei. Furthermore, lymphocytes and eosinophilic infiltration associated the granuloma within the liver parenchyma, bile duct hyperplasia in the portal area with inflammatory cell infiltrate and macrovesicular fatty changes were also noticed. Moreover, the macrovesicular fatty changes associated with the angiectasis (cavernous widening of liver sinusoids) were also found that indicated hepatocellular neoplasm (Figure 10B1-B8). Administration of polysaccharide sulfate for 24 weeks resulted in a significant decrease in liver inflammation and necrosis with the induction of apoptosis as well. Moreover, the polysaccharide sulfate improved the liver lobule through normal hepatic strands with mild dilatation of sinusoids. The nuclei were normal, indicating the recovery of the liver tissues with no congestion as compared with the DENA group (Figure 10C1 and C2). On the other hand, the aqueous extract of U. lactuca treated rats for 24 weeks showed nonsignificant improvement in hepatocellular architecture as compared to DENA control (Figure 10D).
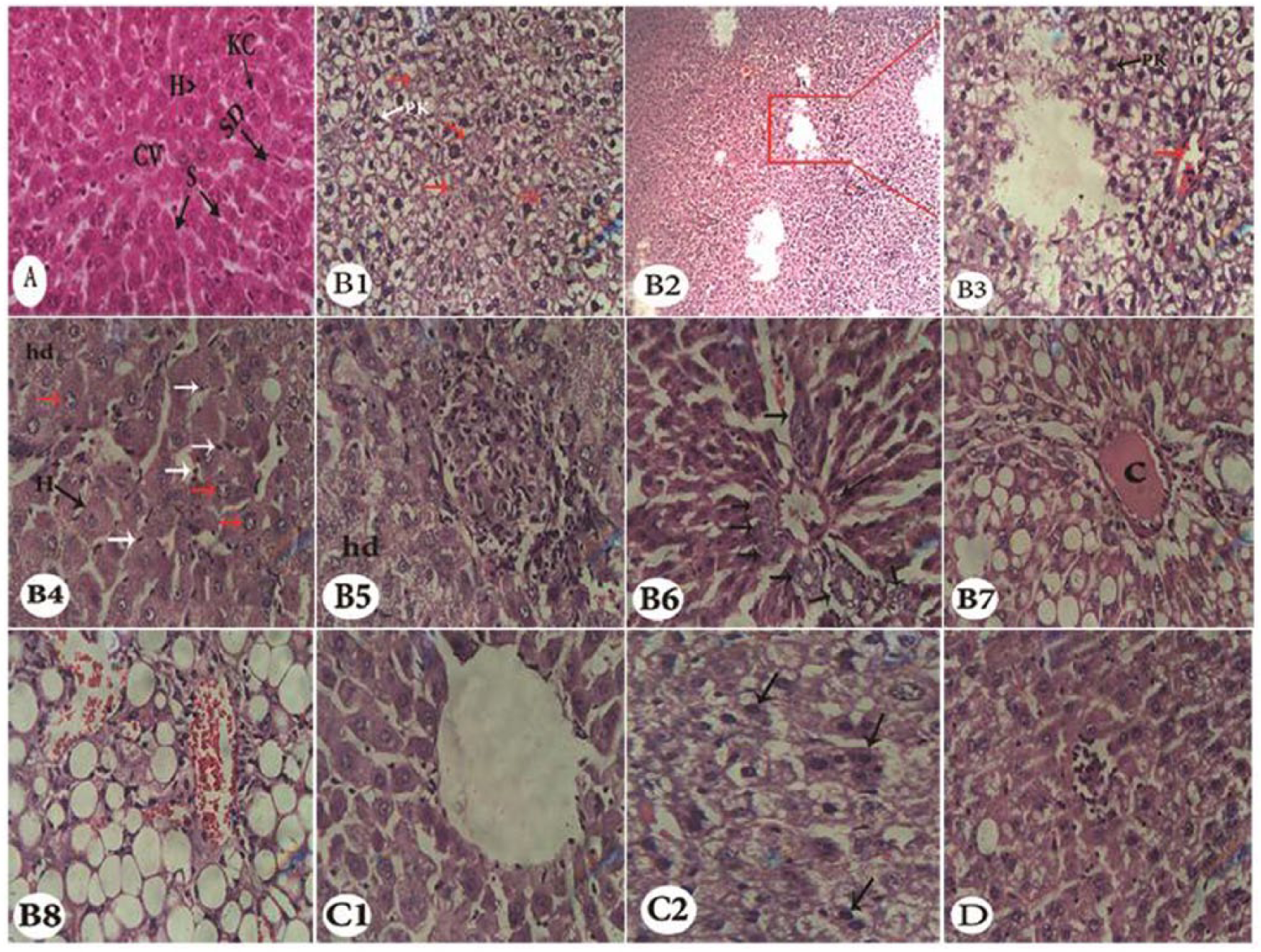
Photomicrographs of liver specimens stained with hematoxylin and eosin at 24 weeks postdose. (A) Liver from normal control rat (saline treated) showing normal liver histology with unremarkable central vein (CV), hepatic cells (H), hepatic sinusoids (S), space of Disse (SD), and Kupffer cells (KC) (400×). (B) Liver from rat treated with diethylnitrosamine (DENA) showing multifocal necrosis (red arrows), clear cell foci, pyknotic nuclei (PK), and disturbances of cell architectural (B1, 400×); extensive severe necrosis (B2, 200×), and clear cell foci associated with pyknotic cells (B3, 400×); considerable karyocytomegaly and Kupffer cells hyperplasia represented by red and white arrows, respectively, focal dilatation of sinusoids, hepatocytes hypertrophy (H), hydropic degeneration (hd) (B4, 400×); hydropic degeneration, granuloma within parynchymatous liver tissue with lymphocytic and eosinophilic infiltration (B5, 400×); bile ducts hyperplasia represented by arrows in the portal area (B6, 400×); clear congestion of portal vessel associated with macrovesicular fatty changes and preportal inflammatory cell infiltrate (B7, 400×) and angiectasis (cavernous widening of liver sinusoids) associated with hepatocellular neoplasm; macrovesicular fatty changes (B8, 400×). (C) Liver from treated rats with DENA plus polysaccharide sulfate, showing normal arranged hepatocytes strands with normal architectures with remarkable central vein, but still with minor sinusoidal dilatation (C1, 400×) and considerable apoptotic bodies represented by arrows (C2, 400×). (D) Liver from treated rats with DENA plus aqueous extract showing marginal improvements of liver tissues with a focus of neutrophil infiltrate associated with hepatocyte necrosis (400×).
Effects of U. lactuca on Cell Proliferation During Hepatocarcinogenesis
Figure 11 depicts the immunohistochemical staining of differential expression levels of the proliferative marker, PCNA, in liver sections obtained from various experimental rat groups at the 3 time points. Near to complete absence of PCNA-positive cells were observed in normal control at 2, 12, and 24 weeks postdose (Figure 11A-C). A substantial increase in PCNA expression was contrastingly observed in animals treated with DENA alone at the 3 time points (Figure 11D-F). The elevated PCNA levels were shown to be reversed in the sections obtained from rats treated with polysaccharide sulfate in addition to DENA treatment (Figure 11G-J) at 2, 12, and 24 weeks, respectively, whereas the aqueous extract treatment in conjunction with DENA exposure resulted in insignificant decreases in PCNA-positive hepatocytes (Figure 11K-M) at 2, 12 and 24 weeks, respectively. Figure 12 represents the qualitative analysis performed on PCNA immunohistochemical data, which shows significant elevation in the frequency of PCNA-positive hepatocytes (PCNA LI) in the DENA-treated animals compared with the normal control group at various time points. In comparison with the DENA-control rats, polysaccharide sulfate treatment had shown significantly decrease of the number of PCNA-positive cells at all 3 time points.
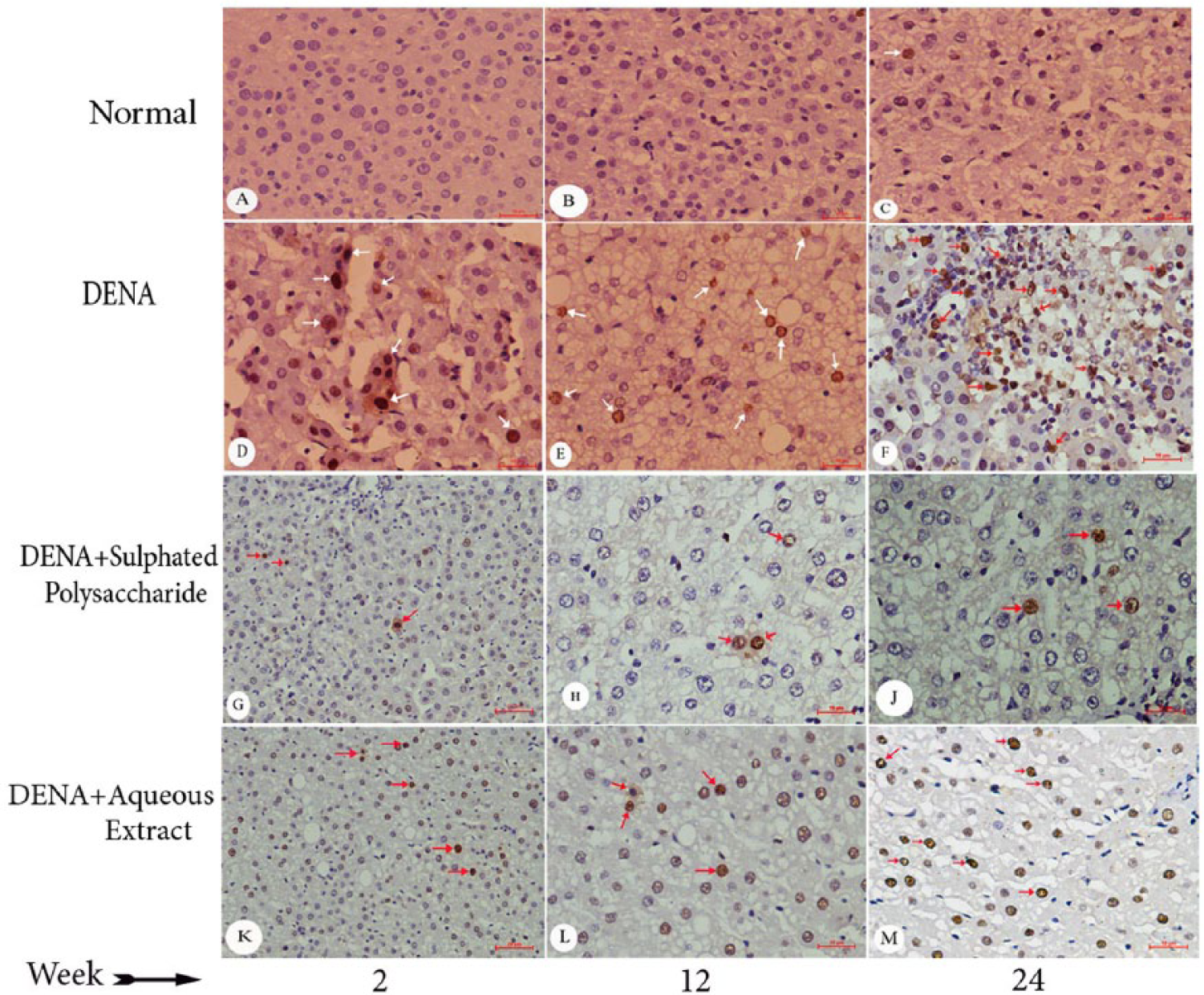
Immunohistochemical staining of PCNA in liver tissue of various groups of experimental animals along the selected time points. Immunohistochemistry was performed as described in the Materials and Methods section. White and red arrows point to PCNA-positive cells. (A, B, C) Normal liver sections at the three time points (2, 12, and 24 weeks, respectively) showing absence of PCNA staining (400×). (D, E, F) PCNA staining in DENA control liver at 2, 12, and 24 weeks, respectively, showing numerous PCNA-positive nuclei (400×); (G, H, J) Liver sections from polysaccharide sulfate plus DENA at 2, 12, and 24 weeks, respectively, showing very less frequent immunostaining of PCNA. (K, L, M) Numerous frequent immunostaining for PCNA of aqueous extract plus DENA compared with DENA control at 2, 12, and 24 weeks, respectively. PCNA, proliferating cell nuclear antigen.

Quantitative analysis cell proliferation during DENA-induced rat liver carcinogenesis in the presence or absence of various treatments at different time points. PCNA LI was determined by immunohistochemistry as described in the Materials and Methods section. The LI was expressed as the number of PCNA-immunopositive cells × 100/total number of tumor cells analyzed. Data are expressed as mean ± standard error of the mean (n = 6). Bars with different letters indicate statistically significant differences. DENA, diethylnitrosamine; PCNA proliferating cell nuclear antigen; LI, lableing index.
Effects of U. lactuca on Apoptosis During Hepatocarcinogenesis
Based on data presented in Table 4, we can conclusively state that DENA caused moderate to substantial increase in apoptotic hepatocytes in rats as compared to normal rats at different periods of the experiment (ie, 2, 12, and 24 weeks). A further increase in apoptotic cells was achieved by both polysaccharide and aqueous extract treatment. Nevertheless, the most prominent result was obtained with the polysaccharide fraction at 12 weeks of the study.
Qualitative Assay of DNA Fragmentation–Based Apoptosis in Various Experimental Groups.*

Data are expressed as mean ± standard error of the mean (n = 6). In the columns, mean values with the same superscript letters are nonsignificant, whereas with different superscripts letters are significantly different. The values in parentheses indicate percentage change compared with corresponding control. Various groups are as follows: group A, normal control; group B, DENA control; C, DENA + polysaccharide sulfate; group D, DENA + aqueous extract.
Discussion
The use of animal models of chemical carcinogenesis is considered to be one of the desirable goals for identification of multistep process of carcinogenesis and also to understand the risk caused by environmental agents, and in turn, develop effective means of diagnosis and management of cancers. The higher risks of malignancy are due to sedentary life styles and environmental pollution, and effective preventive agents could be developed to halt or reverse tumor progression. 50 Retarding tumor incidence and reducing onset of tumor by natural agents alone or in combination may be a valuable strategy in cancer chemoprevention.51-54
The major findings of the current study indicate chemopreventive efficacy of sulfated polysaccharides and aqueous extract of U. lactuca. Interestingly, both U. lactuca fractions showed no adverse effect on the liver or other organs of rats when compared to the corresponding normal control animals (data not shown). The results showed that sulfated polysaccharides provided a better chemoprevention of hepatocarcinogenesis initiated by DENA and promoted with PB. Following its metabolic biotransformation, DENA produces promutagenic products, including O 6 -ethyldeoxyguanosine as well as O 4 - and O 6 -ethyldeoxythymidine in liver, which are responsible for their carcinogenicity. 55 PB induces hepatotoxicity by metabolic activation and elevates a number of hepatic enzymes, including certain cytochrome P450s, glucuronosyl transferases, and glutathione S-transferases. 56
The instability of liver cell metabolism caused by DENA and PB generally reflects the critical changes in serum enzymes activities. 57 Serum transaminases (AST and ALT), ALP, TBL and γ-GT are representative of liver functions and their increased levels at various selected time points are indicators of gradual liver damage. The 2-fold increase in ALT activity at 24 weeks following DENA exposure was indicative of hepatocellular damage, which could be associated with a rise in AST. 58 Similarly, gradual increase of ALP activity reflects pathological alteration in biliary outflow as well as nonspecific alteration in the plasma membrane integrity. These findings correlated well with the liver histopathology. Quantitation of serum bilirubin represents an index of hepatic function and, thus any abnormal increase in its level refers to severe hepatocellular dysfunction. 59 Furthermore, DENA induces necrosis of bile duct epithelial cells, cessation of bile flow, and consequently hyperbilirubinemia. 60 This is in line with our findings, which showed 3- and 4-fold increase in the level of serum bilirubin at 12 and 24 weeks postdose, respectively. γ-GT is an enzyme embedded in the plasma membrane canalicular domain of hepatocytes and considered to be one of the best indicators of liver damage. 61 In our study, its gradual liberation into serum indicates hepatocyte damage and consequently injury to liver. 62 The significant reduction of serum total protein and albumin levels (hypoalbuminemia) at various time points as observed here indicate severity of cellular dysfunction in chronic liver disease. 63 Moreover, the reduction is attributed to endoplasmic reticulum damage, which in turn, decreases the protein synthesis and aggregation of triglycerides, which eventually leads to fatty liver. 64
The recorded increase in most serum indices of liver functions could be a secondary event following DENA-induced lipid peroxidation of plasma membranes of hepatocytes. Since the antioxidant activity of various seaweeds has been documented,65,66 the restorative effect of U. lactuca extracts could be owing to their ability to prevent or reduce oxidative DENA metabolism into more toxic metabolites. This could be supported by our in vitro results, particularly with sulfated polysaccharides, which showed scavenging activities of DPPH and superoxide anion radicals. Our results also suggest that sulfated polysaccharides aids in parenchymal cell regeneration in liver more efficiently than the aqueous extract, thereby protecting membrane integrity by decreasing enzyme leakage. This may be related to its unique chemical compositions, such as sulfated polyaldobiuronan, rhamnosylated saccharides, and iduronic acid which are required for the synthesis of heparin analogs with antithrombotic activities. 67 Furthermore, the ulvan and its oligosaccharides have antitumor and immune system modulatory properties. 68
One of the useful markers of oxidative stress is the increased levels of lipid peroxides. Interestingly, we found an increased level of MDA (indicative of lipid peroxidation) in DENA control rats when compared with the normal control group corroborating the results of earlier studies.20,69 The products of lipid peroxidation are mutagenic and carcinogenic as they cause serious damage to cellular macromolecules like proteins, lipids, and DNA by generating reactive oxygen species (ROS).59,70 The current results with sulfated polysaccharides are in confirmation with previous studies on Lycium chinensis polysaccharides, 71 which showed protection against lipid peroxidation during hepatocarcinogenesis.
Diethylnitrosamine significantly increased NOx and decreased GSH, GR, SOD, MPO, GST, and CAT in hepatic tissues, indicating that reactive oxygen and nitrogen species induced by DENA play an important role in hepatic carcinogenesis. Continuous ROS generation and decreased antioxidant enzymes in liver tissues has long been reported in many chemically induced models of HCC.61,72-74 NOx is known to inhibit DNA repair proteins; thereby inhibiting the ability of the cell to repair damaged DNA.75,76 Data outlined here demonstrated that both sulfated polysaccharide and aqueous extract of U. lactuca attenuated the NOx level and increased the GSH, SOD, CAT, GR, MPO, and GST in liver tissues. The scavenging activity of sulfated polysaccharides for DPPH and free radical ions are consistent with previous studies showing that algal polysaccharides play a major role as a free radical-scavengers in vitro and antioxidants for retardation and prevention of oxidative damage in living tissues. 77 Moreover, their activities depend on various structural parameters such as the degree of sulfation, molecular weight, the sulfation position, sugar type, and glucosidic branching. 78 Also, the polysaccharides might behave as biological response modifiers, fostering the immune system by stimulating the splenocytes and macrophages and inducing anti-inflammatory cytokines which could suppress the growth of cancer cells. 79
Reduced glutathione is known to be involved in many cellular processes including endogenous and exogenous compound detoxification. DENA, an electrophilic carcinogen, may interact with the large GSH nucleophilic pool, thereby reducing its level. This finding is in line with the current results, which showed a reduced level of GSH following DENA treatment and a negative correlation between GSH and MDA contents. On sulfated polysaccharides and aqueous extract treatment, a significant elevation of hepatic GSH was observed as compared with DENA-induced rats, which is consistent with the idea of reduced DNA-carcinogen interaction averting a procarcinogenic environment.
Superoxide dismutase acts as the first line of defense against the oxidative damage of superoxide anions, which dismutates 2 superoxide radicals to H2O2 and O2. In addition, CAT acts as supporting antioxidant enzyme by converting H2O2 to H2O, thereby reducing the cellular destruction and providing protection against ROS. The reduction in SOD and CAT activities may be caused by the increase in ROS generation during DENA metabolism 69 and we also observed similar results in DENA-treated rats. In the present investigation, an increase in MDA formation was perhaps associated with increased ROS, in line with the observation that free radicals decrease the activity of hepatic SOD. 80 Furthermore, the negative correlation between SOD and NO may suggest that SOD is involved in the antioxidant defense system against ROS supporting other previous results. 81
Glutathione S-transferase is a soluble protein located in the cytosol and plays a major role in detoxification and excretion of xenobiotics. 82 Furthermore, it stimulates the conjugation of the thiol groups of GSH to the electrophilic pole of xenobiotics and consequently increases solubility. The xenobiotic-GSH conjugate is then either eliminated or converted to mercapturic acid. 70 As per our present results, the activity of GST was increased in sulfated polysaccharide-treated rats, which reveals a novel role of the experimental agent by offering a greater coupling of GSH with electrophilic intermediates. There is other evidence suggesting the antioxidant effect of oral administration of sulfated polysaccharide extract of U. lactuca for 21 days in rats. 83
Our 2-week study may be considered the assessment of the initiation phase, 12 weeks as premalignant phase and 24 weeks as late premalignant transformation phase of hepatocarcinogenesis with subsequent hepatocellular carcinoma development according to previous reports.84-87 Treatment with nitrosamines or other carcinogens caused an induction of foci, including basophilic and clear cell foci. 88 Foci of cellular alteration represent a localized proliferation of hepatocytes developing under conditions of initiator-promoter administration; an increase in focal cell proliferation comparative to the normal control, nonfocal hepatocytes can be noticed following carcinogen administration. Accordingly, administration of promoting agents (eg, PB) after DENA causes selective enhancement of initiated cell proliferation over noninitiated cells in the target tissue resulting in the appearance of neoplastic hepatic foci.89-91 It has also been reported that the promoter of hepatocellular neoplasm causes bile duct hyperplasia in the portal regions and angiectasis as well. In this study, long-term treatment with sulfated polysaccharides reduced the incidence of altered hepatocytes as well as basophilicity with a reversal of heterogeneity toward normal cellular architecture. The aqueous extract showed less improvement of hepatocellular degeneration with neutrophil infiltration associated with hepatocyte necrosis.
Cellular proliferation plays a principal role in the multistep carcinogenesis process, including initiation, promotion, and progression, and has emerged as an important mechanism for HCC. 92 PCNA, a cofactor of DNA polymerase, is directly implicated in DNA replication and represents an important marker for estimating the proliferation during hepatocarcinogenesis. 93 In addition, it has been usually used as a reliable marker for assessment of tumor progress and clinical prognosis of patients with different malignancies. The high PCNA expression and LI in liver sections of DENA-treated group indicate accelerated cell proliferation during the early phase of rat liver tumorigenesis in agreement with previous observations.48,94,95 Interestingly, a significant decrease in the number of PCNA-positive hepatocytes and consequently PCNA LI due to sulfated polysaccharide co-treatment clearly indicates a decrease in the proliferation of the hepatocyte, possibly through inhibition of DNA synthesis during experimental hepatocarcinogenesis. Interestingly, sulfated polysaccharides from other natural products previously showed in vitro and in vivo antiproliferative effects in several tumor models.18,96-98
Apoptosis is marked by distinct morphological alterations characterized by nuclear, cytoplasmic condensation, blebbing of plasma membrane and DNA fragmentation. 99 Deregulation of apoptosis disrupts the balance between cell death and cell proliferation and eventually contributes to hepatic malignancy development.100,101 In the current study, DNA fragmentation was detected to identify cells undergoing apoptosis. The results showed either small or moderate increase of DNA fragmentation in DENA-exposed rat livers compared to normal livers indicating the attempt of carcinogen-challenged hosts to remove transformed cells. Sulfated polysaccharides co-treatment showed a substantial increase in the formation of DNA fragments thereby providing remarkable evidence of cell death induction by apoptosis and subsequently reduction of a large proportion of transformed hepatocytes in DENA-treated animals. Similar effects, albeit of lower magnitude, were observed with the aqueous extract. The apoptosis induction during experimental hepatocarcinogenesis in rats by sulfated polysaccharides may be related to various phytoconstituents, namely iduronic acid and aldobiouronic acid as previously identified. 102 Sulfated polysaccharides isolated from U. lactuca can significantly suppress the proliferation of several human cancer cells in vitro 26 and this present report suggests a potential therapeutic role of polysaccharides isolated from seaweed in the treatment of hepatocellular carcinoma. These observations are in agreement with a previous study 103 that investigated the apoptosis-inducing efficacy of polysaccharides isolated from Angelica sinensis in cervical cancer cell line. Interestingly, our results concur with previous communications which reported the antiproliferative activity of fucan sulfated polysaccharide from Sargassum filipendula against HeLa cells. 104
In summary, our results clearly indicate that sulfated polysaccharides extracted from U. lactuca exert a marked chemoprevention of hepatocellular carcinogenesis in rats initiated with DENA and promoted by PB. Under the same experimental conditions, the aqueous extract elicited a moderate inhibition of rat liver carcinogenesis. The chemoprotective effect of sulfated polysaccharides may be mediated through inhibition of abnormal cell proliferation and induction of apoptosis. The sulfated polysaccharides also modulated various components of the enzymatic and non-enzymatic antioxidant defense system. We propose that sulfated polysaccharides abolish a severe oxidative injury induced by the initiating agent DENA and substantiated by PB by interacting directly with ROS (eg, OH·) as well as indirectly by activating the antioxidant defense system. The potential chemoprevention effect of sulfated polysaccharidess fraction might be due to synergistic effect of the phytomolecules of various acids present in this fraction. Ancillary experiments to unveil precise molecular mechanism of sulfated polysaccharides against DENA-induced liver cancer are currently under way.
Footnotes
Acknowledgements
The authors wish to acknowledge staff members of Zoology Department, Faculty of Science, Beni-Suef University for their help and expertise in animal experimentation and handling of rats. The first author is grateful to Dr Kumaraiah Palle for his encouragement and mentorship during the period he spent in his laboratory and to Dr Kaushlendra Tripathi for advice and useful discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Faculty of Science, Beni-Suef University, Egypt.


