Abstract
Background. Patients receiving chemotherapy for hematological malignancies are at high risk for febrile neutropenia (FN). Garlic extracts (GEs) are natural food substances showing antimicrobial effects in vivo. Objectives. We explored whether adding GE may be efficacious in reducing the risk or severity of infections. Design. This was a placebo-controlled double-blind randomized study. Results. Of 95 patients randomized to receive GE or placebo following chemotherapy, a febrile episode was documented in 50% of patients receiving GE and 63.3% receiving placebo (P = .89). There was a higher risk of developing a third and fourth febrile episode in the GE group (P = .01). However, among those at a lower risk for FN, those receiving GE developed fewer FN episodes (P = .075), especially those with severe neutropenia (P = .05). Major adverse events were distributed equally, but nonadherence was more common in the GE than in the placebo group: 19.5% versus 4%, respectively (P = .05). Conclusions. GE was safe and did not reduce FN risk in the entire cohort, but yet appeared to exert a protective effect in the lower-risk subgroup. We do not recommend the use of GE for FN prevention in higher-risk patients. A larger-scale clinical trial for the lower-risk subgroup of patients is advocated. (This trial was registered at www.clinicaltrials.gov as NCT00247039.)
Introduction
Neutropenia is an invariable result of high-dose chemotherapy given to cancer patients and is particularly common in patients treated for hematological malignancies. As the neutrophil count drops, patients are susceptible to both conventional and opportunistic infections. Thus, neutropenia is a major risk associated with cancer treatment, and is the most dangerous and severe immediate side effect of chemotherapy.1,2 When fever occurs (ie, febrile neutropenia [FN]), patients with hematological malignancies require hospitalization and administration of empirical broad-spectrum antimicrobials. The range of pathogens causing infections in these patients is wide and includes bacteria, fungi, and rarely viruses. 3 A microbiologically documented infection is identified in approximately 30% of febrile neutropenic episodes. 4 Prophylactic measures such as granulocyte colony stimulating factor (GCSF) are routinely required.5,6 However, the use of antibiotic prophylaxis for patients at risk for FN is controversial. Although mortality reduction has been reported with administration of necessarily broad-spectrum prophylactic antibiotics, this benefit is countered by deleterious effects of drug toxicity, 7 emergence of antibiotic-resistant bacteria, and fungal overgrowth.8-10
Garlic compounds are natural food additives with antimicrobial effects.11-15 In many experiments, mostly performed in vitro, they have been shown to have wide antibacterial range, covering both Gram-negative and -positive species.11,14 Garlic also has antifungal effects, including against Aspergillus species.13,15 Allicin, a major ingredient of fresh garlic extract (GE) that is produced during the crushing of garlic cloves, exerts various beneficial biological effects, including a broad spectrum of antimicrobial activity and antihyperlipidemic and antihypertensive effects.12,16,17 Furthermore, allicin has an antioxidant and cytoprotective effect, may delay onset and/or progression of breast cancer, 12 and has been found to suppress carcinogenesis in combination with selenium in the Chinese population. 18 A review of the literature shows some evidence from animal studies reporting protective effects of garlic against colon cancer. 19 The research cited above as well as other studies and observations performed with oral ingestion of garlic and its derivatives over the years have shown that overall, as a therapeutic agent, garlic appears to be remarkably safe. 16
Although many clinical trials with herbal/traditional compounds and substances are currently being performed, most are not randomized or placebo controlled. Robust studies of both safety and efficacy are lacking.20-22 The rationale for use of garlic natural compounds in FN patients include, on the one hand, the frequency of FN in the setting of treatment of hematological malignancies and, on the other, garlic’s purported antimicrobial and anti-inflammatory properties. Adding a nontoxic and possibly effective therapy may delay or reduce the risk and severity of infections.
Methods
Trial Design
We conducted a parallel, double-blind, randomized, placebo-controlled trial to study the safety and efficacy of GE in chemotherapy-induced FN in hematological patients. The trial was performed with the approval of the local institutional review board committee of Hadassah Medical Center, and all patients signed an informed consent before randomization and treatment.
Participants
A total of 101 patients were enrolled and randomized to receive GE or placebo capsules after the course of chemotherapy and during their expected neutropenia nadir, for 1 cycle. Eligible patients had to be older than 18 years, with a confirmed diagnosis of hematologic malignancies requiring chemotherapy (either newly diagnosed or previously treated). Exclusion criteria were the following: inadequate liver (aspartate aminotransferase/alanine aminotransferase > 3 times the upper limit of institutional laboratory normal) and kidney functions (blood urea nitrogen and creatinine > 3 times the upper limit of normal). Risk for FN was assessed,6,23 and eligible patients had to receive a chemotherapy protocol expected to exert at least a moderate risk for neutropenia. Risk groups were classified according to the National Comprehensive Cancer Network (NCCN) growth factor guidelines 6 and further defined as intermediate (I), high (H), and very high (VH) if any patient-related factors upgrading the risk were present (ie, previous neutropenia with the same protocol and doses, extensive prior chemotherapy, marrow radiation prior to treatment, pathological evidence for bone marrow involvement by disease, age >65 years). Growth factors (GCSF) were administered according to the NCCN guidelines6,23 as well as standard prophylactic antibiotic treatment as assigned by the treating physician (in certain protocols, acyclovir for herpes viruses and trimethoprim/sulfamethoxazole for Pneumocystis jirovecii pneumonia prevention). Prophylaxis with broad-spectrum antibiotics is not routinely used at our institution (however, patients were not excluded if they were already treated for an active infection with antibiotics). Patients were excluded if they were on full-dose anticoagulation therapy for safety because of a possible bleeding tendency related to excessive garlic ingestion.24-26 In addition, patients taking 5 or fewer days of treatment, who thus may not benefit from the medical effect of allicin, were also excluded.
Outcomes and Response Assessment
Responses were continuously evaluated by daily blood counts (complete blood counts) for hospitalized patients and at least twice weekly for ambulatory patients, until the resolution of neutropenia or white blood cell count reconstituting >3000 white blood cells /mm3. Primary end points were the onset of fever >38°C and time to first fever during the study time period (thus, requiring the administration of empirical antibiotic treatment). Secondary end points were the length of each febrile episode and number of recurrent febrile episodes. We planned to assess these outcomes for subgroups with different risks for FN.
Intervention: Dosage and Administration
Garlic compounds are not considered drugs by the Israeli health administration but rather food supplements. In accordance with the above information, it is reported to be safe and effective17-19 to administer garlic compounds to patients at doses of 900 mg a day in 2 divided doses of 450 mg active compound (allin 9.9 mg, allicin 4.5 mg) or placebo. Garlic and placebo tablets were purchased from a commercial herbal manufacturer in compliance with good manufacturing practice and ISO 9001:2000 regulations (Superherb, Nazareth-Illit, Israel) in sealed identical bottles that contained a 3-week supply. The patients were randomized to begin therapy after the last day of the chemotherapy course and until the resolution of neutropenia.
Randomization
As noted, the patients were stratified using standard NCCN criteria of FN6,23 then randomized. Randomization in blocks was used to allocate patients 1:1 to 1 of the 2 treatment arms. This randomization procedure was conducted for each of the 3 risk groups separately. The randomization and codes were kept by an independent personnel blinded to the patient selection process.
Sample Size Considerations
The sample size calculations were based on a comparison of the probabilities of documented infections during the neutropenic episode. The basic hypothesis was that treatment with GE would result in 20% fewer FN episodes than placebo. For 80% power and a 2-sided Z-test with α set at .05, we estimated that we would need 50 participants in each arm.
Statistical Analysis
We used 2-sample independent t tests or the Wilcoxon test for continuous variables and χ2 or Fisher’s exact test, as appropriate. Kaplan-Meier and log-rank tests were implemented for the analysis of time to first fever. All tests were 2 sided, with a .05 significance level. Statistical analyses were done using SAS software version 9.2. An interim analysis was performed, after enrolling 59 patients, which demonstrated balanced randomization and no safety issues.
Blinding
As noted, GE and placebo capsules were identical. Both investigators and participants were blinded as to treatment allocation.
Results
Patient Characteristics and Stratification
A total of 101 adult patients with hematological malignancies (leukemias and lymphomas) were enrolled. Of these, 95 were eligible for analysis, as shown in the flow diagram (supplementary Figure 1; available at http://ict.sagepub.com/content/by/supplemental-data). 27 Three declined treatment just before randomization, and 3 stopped the treatment after <3 days and, thus, were excluded from the analysis. Patient and disease characteristics by study arm are shown in Table 1. The distribution across the various protocols, according to risk subgroup, is presented in supplementary Table 1 (available at http://ict.sagepub.com/content/by/supplemental-data). All patients at very high risk for FN had acute leukemia, requiring induction or consolidation high-dose therapy, whereas patients in other risk groups had mostly aggressive lymphomas requiring standard protocols for newly diagnosed and relapsed patients.
Baseline Characteristics of Evaluable Participants in Both Study Arms.

Abbreviations: AML, acute myeloid leukemia; ALL, acute lymphoid leukemia; NHL, non-Hodgkin’s lymphoma; HL, Hodgkin’s lymphoma; Other, multiple myeloma, chronic lymphocytic leukemia, multicentric Castleman’s disease; FN, febrile neutropenia; BM, bone marrow; CR, complete remission; PR, partial remission; CVC, central venous catheter; BMI, body mass index; SD, standard deviation.
The patients in the 2 groups were well matched for baseline characteristics (except for BMI, which was slightly higher in the control group) as well as disease-related and risk factors for infection (such as bone marrow involvement in >40% of patients, low number of patients in complete remission at the time of enrolment (<25%), and occurrence of neutropenia in previous chemotherapy cycles in 60% of patients). In general, patients enrolled had good performance and nutritional status. Most patients (>60%) were receiving P jirovecii prophylaxis with trimethoprim/sulfamethoxazole; only a minority (5% of both arms) were receiving other antibacterial medications at the time of enrollment.
Treatment Outcomes
The majority of patients in both groups ultimately developed neutropenia at similar rates (Table 2); 75.5% in the placebo and 76.1% in GE patient groups developed significant neutropenia (<500 neutrophils/mm3) during the course of treatment. The median lowest neutrophil count reported was 100/mm3 in both groups. Other blood counts (Hb/platelets) were not affected differentially when comparing the 2 treatment groups. The overall febrile episode rate varied as expected among the 3 risk groups (Figure 1A): intermediate risk, lowest; very high risk, highest; and high risk, between the 2 extremes. Most patients (>50%) eventually developed a febrile episode during their neutropenia (Figure 1B, P = .89), and this did not differ between patients receiving GE (50%) or placebo (63.3%).
Clinical Outcomes in Each Treatment Arm.
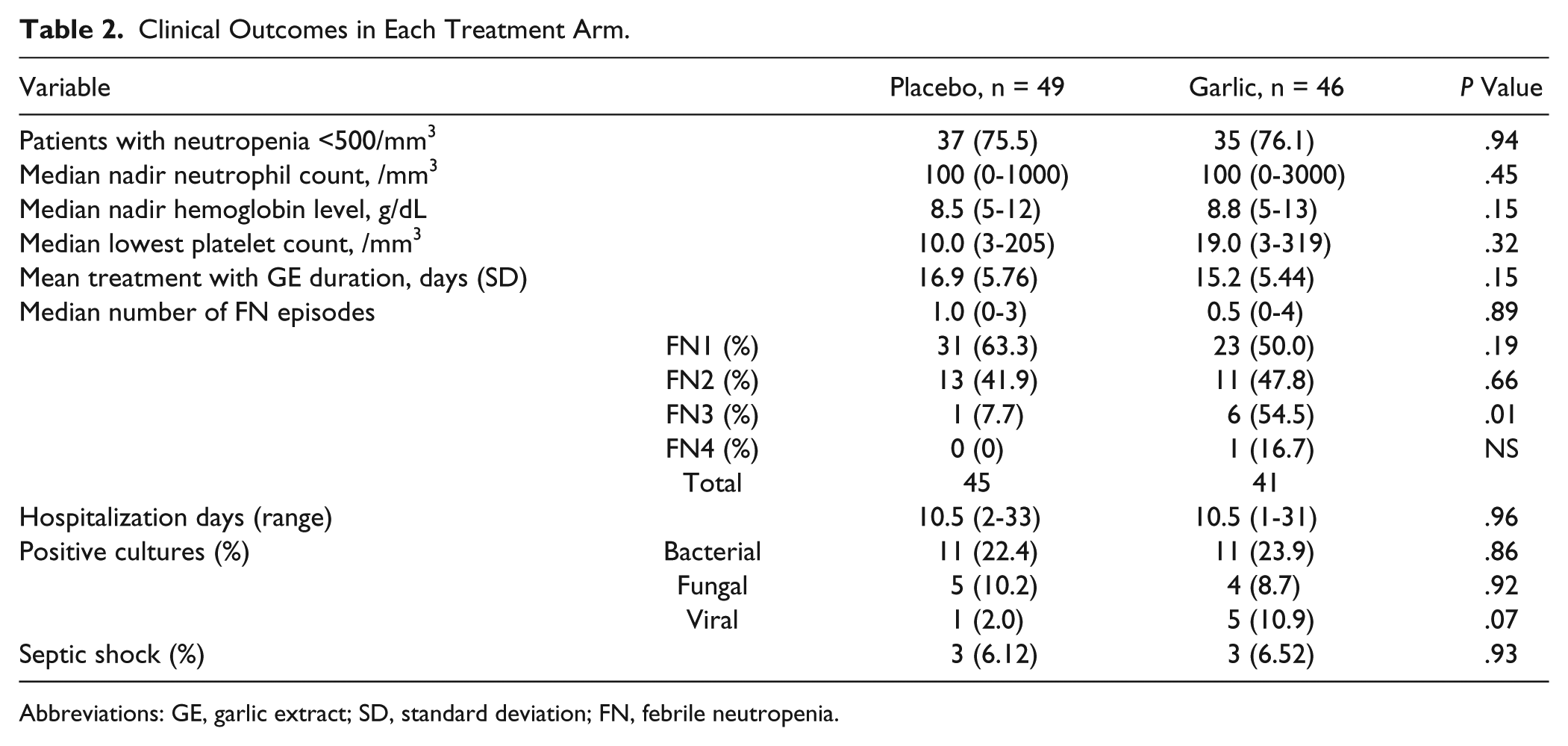
Abbreviations: GE, garlic extract; SD, standard deviation; FN, febrile neutropenia.

A. Time to fever of all patients. B. Time to fever, according to risk group.
Overall, the number of positive cultures (bacteria and fungi) was similar. However, there was a trend (P = .077) for more infections of viral origin in the GE group (Table 2). The use of antimicrobial, antibacterial, and antiviral agents was similar (not shown). Severe infections manifested by septic shock were observed at a similar low rate of 6% in both groups (P = .93).
Results by Risk Groups
Patients in different risk groups, I, H, and VH, had varying periods of neutropenia with fever (mean of 2.9 days as compared with 3.8 days and 16 days in the 3 risk groups, respectively), as shown in Table 3. The risk of developing first and second FN episodes was similar. However, the GE group actually had a higher risk of developing a third and fourth febrile episode (P = .01). The latter patients exclusively belonged to the very-high-risk group (Table 2). In contrast, intermediate-risk patients receiving GE showed a trend (P = .075) toward fewer FN episodes (Figure 2). Moreover, this became nominally statistically significant (P = .053) when analyzing only patients in the lower risk subgroups (I and H) who developed neutropenia (Table 3).
Results by Risk Group.

Abbreviations: I, intermediate; H, high; VH, very high; FN, febrile neutropenia; P, placebo; GE, garlic extract.

Time to fever, intermediate risk group.
Adverse Events
Compliance, as evaluated by nonadherence (nonspecific complaints and noncompliance), differed between the GE and placebo groups significantly (P = .054). Although patients treated with GE did not complain of a significant garlic odor and the median duration was not different either—16 days (range = 5-32) versus 14 days (range = 5-30)—more patients stopped the treatment before the end of the neutropenic period—19.5% in the GE versus 4% in the placebo arm. This was for nonspecific reasons (nonadherence). Severe adverse events were distributed equally between the 2 groups (Table 4). None seemed to be related to the study GE or placebo. Because of previous reports relating garlic intake to bleeding,24 -26 we specifically assessed the rate and risk of bleeding in both groups. The mean number of bleeding episodes was similar in both groups. In addition, the mean blood and platelet transfusion rates (either prophylactic or on an as-needed basis) did not differ. Only 1 patient died in each group while receiving the study preparation, and neither death was related to the GE or placebo. It should be noted that GE did not affect treatment outcomes for the underlying diseases as assessed by the clinical response.
Adverse Events.

Abbreviations: SD, standard deviation; CR, complete remission; PR, partial remission.
Only 1 patient in each group had major bleeding.
Discussion
FN is a major side effect in patients treated by chemotherapy.1,2,4 Garlic was shown in vitro to exert antimicrobial effects,11 -15 and oral ingestion of garlic is safe.12,16,17 Moreover, it may change the antibiotic resistance pattern for multidrug-resistant pathogens, thus complementing a possible role in infection treatment with current antibiotics.28,29 We hypothesized that the use of garlic natural compounds in FN patients would be safe, would reduce the risk of infection, and might lessen the occurrence of more severe infections. Clinical trials with herbal/traditional compounds that test both safety and efficacy are lacking.20-22 Use of food supplements and complementary medicine is growing among oncology patients. This was the rationale for conducting a double-blind, randomized trial to assess the safety and efficacy of GE in FN-prone patients.
Our group of patients indeed belonged to a population that is at relatively high risk to develop FN among patients treated for hematological cancer (75% of patients having a neutrophil count below 500 /mm3), as evidenced by the frequency of febrile episodes and recurrent episodes in >50% of patients.
In accordance with other clinical reports,12,17-19 we did not find any safety issues with the use of GE in our cohort. However, we were not able to reach our primary objective of showing evidence for the efficacy of GE in all FN-prone patients. We found that GE did not reduce FN risk overall. Moreover, in the very-high-risk group, patients in the GE arm experienced more episodes of recurrent FN during their neutropenic nadir. This can be related to minor mismatching of the 2 groups in this risk arm. Alternatively, it may reflect GE interference with the use of broad spectrum antibiotics. However, we did not observe more aggressive infections in this group or differences in the rates of isolations between the treatment groups. In contrast to in vitro studies showing antifungal properties for GE,13,15 we did not observe any such benefit in our treated patients. This may be explained by either lack of in vivo function or insufficient antifungal activity. Thus, for the very-high-risk patient subgroup, we do not propose the use of GE as a preventive method for FN, in particular considering the higher nonadherence encountered in a significant number of GE-treated patients as compared with controls.
However, when analyzed according to subgroup, GE did appear to exert a protective effect (P = .075) in the lowest-risk patient group (mostly lymphoma patients). This lack of statistical significance is most probably a result of the small sample size in each risk group. Indeed, when we assessed FN episodes occurring only in patients who essentially developed significant neutropenia (<500 neutrophils/mm3), these differences became marginally statistically significant (P = .05). This may attest to the hypothesized antimicrobial effects of GE, as suggested by in vitro studies.12,14 In addition, the lower-risk patients had a shorter duration of FN, allowing for less possibility for GE to interact with broad spectrum antibiotics (if such an interaction exists), and this shorter time period also diminishes any compliance difficulties.
Conclusion
This study has shown that oral GE is safe in chemotherapy-treated hematological cancer patients at risk for FN. However, its efficacy in preventing FN was borderline and only demonstrated in a lower-risk patient subgroup. Thus, we advocate confirming this observation in a larger-scale clinical trial restricted to the intermediate-risk subgroup of patients and prudently recommend the use of GE for FN prevention only in this risk category.
Footnotes
Acknowledgements
MEG, NS, OBP designed research; MEG, JS, DL, NG, YK, AG conducted research; MEG, NS, OBP analyzed data; MEG, JS, NG, OBP wrote paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial was supported by the Dunsky Foundation for Cancer Research, Israel.
