Abstract
Objectives
Acute leukemia often leads to severe complications such as febrile neutropenia. Mortality rates remain high, underscoring the need for novel prognostic markers. Regulatory T cells (Tregs) have not been extensively studied in this context.
Methods
This prospective observational, single-center study included 114 patients diagnosed with acute leukemia. Tregs percentages were measured using flow cytometry. Statistical analyses involved logistic regression to identify factors significantly associated with survival.
Results
Of the 114 patients, 78 recovered, while 36 died. The median Treg percentages were 5.9% in patients under 65 years and 5.38% in those 65 and older. A lower percentage of Tregs was associated with higher mortality in the older group (p = 0.04). Multivariate analysis highlighted the presence of comorbidities, documented infections, and day seven C-reactive protein levels as significant predictors of survival.
Conclusion
This study supports the importance of monitoring chronic diseases and infection foci alongside traditional markers like C-reactive protein. Future research should explore the mechanistic roles of Tregs in immunosuppression and survival in this vulnerable population.
Introduction
Acute leukemias are a group of diseases with an extremely poor prognosis, characterized by the clonal growth of immature progenitor cells in the bone marrow. The most common type of acute leukemia in adults is acute myelocytic leukemia (AML), and the age-adjusted incidence of acute leukemia is 3.6/100,000 in the population. The median age at diagnosis in patients with AML is 68 years, and the incidence increases in the elderly group. A total of 70% of newly diagnosed AML cases are over the age of 55 years. 1 Febrile neutropenia (FN), which increases morbidity and mortality rates in acute leukemia patients, occurs as a direct result of using treatments intended to be myeloablative. Mortality rates between 10% and 30% have been reported in the literature for acute leukemia patients who developed FN.2–4 Among the factors cited to impact the survival status of acute leukemia patients adversely are advanced age, accompanying chronic disease, delayed antibiotic treatment, and abnormalities in laboratory test results, e.g. low serum albumin level, anemia, and increased lactate dehydrogenase level. 4
FN constitutes a medical emergency in acute leukemia patients. In this context, its rapid diagnosis and early initiation of empirical antibiotic therapy are essential to improve survival outcomes. However, signs and symptoms of infection may be subtle in patients with acute leukemia due to both leukemia and FN, and it may be difficult to diagnose patients who are prone to the development of sepsis. 5 Given the variability of the clinical course of patients with FN, various biomarkers have been sought to predict the risk of developing severe infections and survival outcomes. Acute phase reactants such as C-reactive protein (CRP) and procalcitonin (PCT) are among the most well-known biomarkers routinely used to monitor patients. However, the increases in CRP and PCT levels are not specific to infection. Therefore, several other biomarkers, including presepsin, interleukin (IL)-8, and expression of cluster of differentiation (CD) 64 in neutrophils, have also been investigated and reported to be helpful.6–8 However, tests involving such biomarkers are not likely to be performed in health institutions in countries with limited resources.
Regulatory T cells (Tregs) are a subset of CD4+ T lymphocytes with adverse immunomodulatory functions. The anti-inflammatory cytokines secreted by Tregs, including IL-10, IL-35, and transforming growth factor-β, help create a balanced immune response by regulating inflammation through mechanisms such as cell-cell interaction. A total of 5% to 10% of CD4+ cells in peripheral blood are Tregs. Over the past two decades, Tregs have become the focus of studies on sepsis. Tregs are thought to contribute to immunosuppressive conditions during the course of sepsis.9,10 Although the Treg percentage in peripheral blood is routinely measured within the scope of flow cytometry, it is not a parameter used in the follow-up of patients with FN and in immunosuppressed patients. As a matter of fact, we could not find any study on the relationship between Tregs and FN in the literature. In this context, we conducted this study to investigate the relationship between the percentage of Tregs in peripheral blood and the survival outcomes in acute leukemia patients who developed FN.
Methods
This study was designed as a prospective observational, single-center study. The reporting of this study conforms to the STROBE guidelines. 11 The study protocol was approved by the Izmir Bozyaka Training and Research Hospital Ethics Committee (approval no. 2022/125, approval date: 8 October 22). Izmir Bozyaka Training and Research Hospital is located in the western region of the country and is the only state hospital with a Hematology and Bone Marrow Transplantation Unit in this region. The study population consisted of patients who received chemotherapy with a diagnosis of acute leukemia in a training and research hospital and had an FN attack between 2022 and 2024. Since acute leukemia is an uncommon disease, all patients who were followed up with a diagnosis of acute leukemia during the determined study period and wanted to participate in the study were included. Patients signed the informed consent form before participating in the study. Patients under the age of 18 years and those with one or more missing blood samples taken during follow-up were excluded from the study.
Study protocol
Patients’ quick Sequential Organ Failure Assessment (qSOFA) scores were calculated by the attending physician at the onset of FN attacks. 12 In line with the international FN guidelines, patients were divided into three diagnostic groups: patients with microbiologically documented infection, patients with clinically documented infection, and patients with fever of unknown cause. 13 Blood samples were taken from patients who developed FN at three different time points: at the onset of fever (D0) and three and seven days after D0. At D0, CRP, PCT, and Treg tests were conducted. CRP tests were repeated three (D3-CRP) and seven (D7-CRP) days after D0.
Patients were started on empirical treatment after their blood and urine cultures were taken. According to the definitions of the Republic of Turkey Ministry of Health, age 65 years and above is considered old age. 14 Therefore, patients were divided into two groups: <65 years old and ≥65 years old. Patients’ data were obtained from patient files and an electronic data recording system. Patients’ survival outcomes were monitored for 30 days from the day they were diagnosed with FN.
Data collection
Patients’ data, including demographic and clinical characteristics such as age, gender, comorbidities, vital signs, focus of infection, qSOFA score, laboratory, radiological and microbiological data, antimicrobial treatment, and treatment response data were recorded in the case follow-up forms from the first day of hospitalization. An improvement in patients’ clinical and laboratory parameters within 72 hours after the initiation of antimicrobial treatment was deemed a positive response to antimicrobial treatment. In patients with prolonged neutropenia and more than one fever attack, data pertaining to the first attack were recorded.
Laboratory analysis
CRP test was performed with the immunoturbidimetric method using the CRP4 kit on the c 502 module of the Cobas 8000 Modular Analyzer System (Roche®, Mannheim, Germany). Results are expressed as mg/L. Cutoff: 5 mg/L.
The PCT test was performed by electrochemiluminescence immunoassay using the Elecsys BRAHMS PCT Kit on the Cobas 8000 Modular Analyzer System e602 module (Roche®, Mannheim, Germany). Results are expressed as ng/mL. Cutoff: 0.5 ng/mL.
Blood cultures used in laboratory measurements conducted using conventional microbiological methods and automated systems were incubated in an automatic hemoculture system (BacT ALERT 3D®, Becton-Dickinson Diagnostic Systems, France) for seven days.
Flow cytometry
An ethylenediamine tetraacetic acid anticoagulated peripheral blood sample was taken from each acute leukemia patient who developed FN on D0 to determine the percentage of Tregs. To this end, a three-laser, eight-color, and 10-parameter flow cytometry device (Beckman Coulter®, Dxflex, USA) and CD4-fluorescein isothiocyanate, CD25-allophycocyanin, and forkhead box P3 (FOXP3)-phycoerythrin fluorescent dyes were used. CD4, CD25, and FOXP3 readings were expressed in each patient’s mean of fluorescence intensities. The Treg value was calculated as a percentage in accordance with the manufacturer’s recommendations.
Statistical analysis
Statistical analyses were performed using SPSS 26.0 (Statistical Product and Service Solutions for Windows, version 26.0, IBM Corp., Armonk, NY, USA, 2019) software package. The conformity of the variables to the normal distribution was assessed using analytical methods, i.e. Kolmogorov-Smirnov and Shapiro-Wilk tests. Descriptive statistics were expressed as mean ± SD, median with interquartile range, minimum (min)-maximum (max) values for continuous variables, and frequency and percentage values for categorical variables. The t-test was used to compare independent groups in terms of continuous variables determined to conform to the normal distribution, and the Mann-Whitney U test was used to compare binary groups, e.g. exitus versus alive patient groups in terms of non-parametric continuous variables. Pearson’s chi-square or Fisher’s exact tests were used to compare groups in terms of categorical variables. Probability (p) statistics of ≤0.05 were deemed to indicate statistical significance. Parameters found to be statistically significant in the univariate analysis were further evaluated using a multivariate logistic regression model to predict the mortality risk.
Results
A total of 114 patients, 62 (54.4%) male and 52 (45.6%) female, were included in the study sample. The mean age of the sample was 57.77 ± 14.89 (min: 18; max: 86) years. Fifteen patients were ≥65 years of age. Of the 114 patients, 104 (91.2%) patients were followed up with a diagnosis of AML, and 10 (8.8%) were followed up with a diagnosis of acute lymphocytic leukemia. In terms of disease status, 48 (42.1%) patients each were newly diagnosed and in remission/consolidation, while 18 (15.8%) had relapse/refractory disease. Thirty-two (28.1%) patients had at least one accompanying chronic disease. Of the 36 comorbidities in these 32 patients, 29 (80.6%) had diabetes mellitus (DM), 4 (11.1%) had chronic obstructive pulmonary disease, 2 (5.6%) had congestive heart failure, and 1 (2.7%) had cerebrovascular disease. The rates of patients with qSOFA scores of 0, 1, 2, and 3 were 33.3% (n = 38), 35.1% (n = 40), 19.3% (n = 22), and 12.3% (n = 14), respectively. While 68.4% (n = 78) of the patients recovered, 31.6% (n = 36) died. Univariate analysis revealed a significant negative relationship between survival and being newly diagnosed (p = 0.01), having a documented focus of infection (p = 0.002), and a qSOFA score ≥ 2 (p < 0.001). Demographic and clinical characteristics of patients who developed FN are shown in Table 1.
Demographic and clinical characteristics of patients who developed febrile neutropenia.
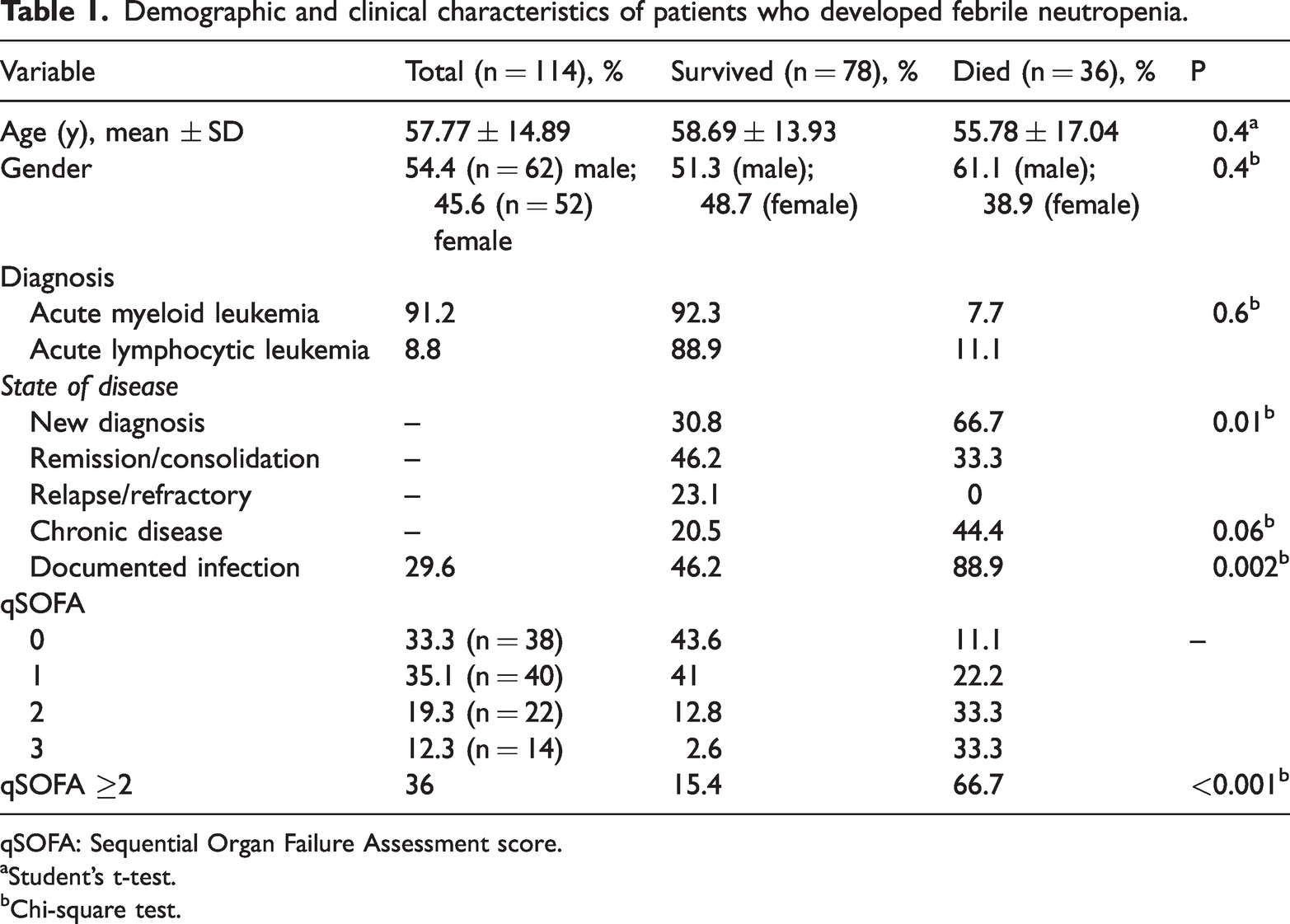
qSOFA: Sequential Organ Failure Assessment score.
Student’s t-test.
Chi-square test.
In terms of the documented focus of infection and growth in culture, it was determined that 46 (40.4%) of the patients had a fever of unknown origin, 42 (36.8%) had clinically documented infection, and 26 (22.8%) had both clinically and microbiologically documented infection. Sixty-eight (59.6%) patients had a clinically or both clinically and microbiologically documented focus of infection. Of these patients, 36 (52.9%) had pneumonia, 12 (17.6%) had bloodstream infections, 10 (14.7%) had soft tissue infections, 4 (5.9%) had gastroenteritis and tonsillopharyngitis, and 2 (3%) had urinary tract infections. Analysis of the relationships between survival and laboratory parameters revealed a significant relationship between survival and only one of the laboratory parameters, D7-CRP (p = 0.01) (Table 2).
Median serum procalcitonin, C-reactive protein levels, and regulatory T cell percentages in three patient groups with febrile neutropenia.

CRP: C-reactive protein; D0: onset of fever; D3: 3 days after the onset of fever; D7: 7 days after the onset of fever; max: maximum; min: minimum; PRC: procalcitonin; Treg: regulatory T cell.
Bold value was found to be statistically significant.
Kruskall-Wallis H test.
One-way analysis of variance test.
Univariate analysis revealed a significant relationship between survival and D3-CRP (mg/L) (p = 0.009), D7-CRP (mg/L) (p < 0.001), and PCT > 0.5 ng/mL (p = 0.028) levels in acute leukemia patients who developed FN (Table 3).
Laboratory parameters of patients who developed febrile neutropenia.

CRP: C-reactive protein; D0: onset of fever; D3: 3 days after the onset of fever; D7: 7 days after the onset of fever; INR: international normalized ratio; max: maximum; min: minimum; PCT: procalcitonin; PRC: procalcitonin; Treg: regulatory T cell.
Bold values were found to be statistically significant.
Mann-Whitney U test.
Student’s t-test.
The relationship between the percentage of Tregs and survival was not significant in patients who developed FN (p = 0.5). Patients were divided into two groups: <65 years old (n = 84) and ≥65 years old (n = 30). The median percentage of Tregs in patients aged <65 and ≥65 was 5.9 (min: 0; max: 18) and 5.38 (min: 1; max: 9), respectively. There was no significant difference between the percentage of Tregs in patients who died and survived in the <65-year-old patient group (p = 0.6). In patients aged 65 years and above, the percentage of Treg cells was 2.5 (min: 1; max: 4) in the group that died (n = 8), while it was 5.96 (min: 2; max: 9) in the surviving group (n = 22) (p = 0.04).
Forward stepwise logistic regression analysis revealed a significant relationship between survival and the presence of comorbidity (p = 0.015; odds ratio [OR], 14.8; 95% confidence interval [CI], 1.67–131.062), having a documented focus of infection (p = 0.002; OR, 9.7; 95% CI, 2.3–41.07), and D7-CRP level (p = 0.021; OR, 1.01; 95% CI, 1.002–1.023).
Discussion
FN constitutes a medical emergency in acute leukemia patients. In this context, its rapid diagnosis and early initiation of empirical antibiotic therapy are essential to improve survival outcomes.1–5 In this study, we prospectively evaluated 114 acute leukemia patients who developed FN. A total of 31.6% died during the follow-up period. A negative correlation was found between the presence of accompanying chronic disease, which was detected in 28.1% of the patients, and survival outcomes. The most common accompanying chronic disease was DM, which in the literature was associated with higher mortality rates in patients diagnosed with various cancers. Hyperglycemia and DM may increase early mortality rates by increasing the risk of infectious complications through their immunosuppressive effects. 15
In our study, forward stepwise logistic regression analysis revealed a documented focus of infection and D7-CRP level as the parameters that predicted the survival outcomes in this patient group. Similarly, international FN guidelines also defined having a clinically documented focus of infection as a high risk for the development of sepsis. 16 For this reason, monitoring CRP levels is deemed helpful in the follow-up of patients with FN. CRP, as an indicator of chronic inflammation in the body, has been found to be a biomarker significantly predicting survival outcomes in patients with various cancers.17,18 PCT is the most evaluated acute phase reactant in terms of its predictive value for sepsis and survival in patients with FN. Some of these studies found that an early increase in PCT levels predicted the development of FN, while others reported contradictory results regarding the predictive power of PCT for microbiologically documented infection.19,20 In our study, we found the PCT level at the onset of FN to be higher than the 0.5 ng/mL PCT threshold value in 12.8% of the survivors and 27.8% of the deceased (p = 0.028). However, further analysis did not reveal a significant relationship between PCT level and survival, probably due to the fact that the PCT level is a sensitive indicator measured in milligrams, and thus, serum PCT levels were detected over a wide range.
Studies conducted to date have shown that the percentage of Tregs is higher in patients who develop sepsis than in those who do not. However, the relationship of Tregs with sepsis during the hyperinflammatory phase and the subsequent course of the disease is still unclear. There are only a handful of studies in the literature investigating the effect of the percentage of Tregs on the development of sepsis and survival in patients with FN.21–23 Our study contributed to the literature with its finding of a negative relationship between the low percentage of Tregs and survival in elderly acute leukemia patients who developed FN. Given that chronic inflammation is common in older individuals and Tregs are more associated with the stimulation of anti-inflammatory events, it is not surprising that patients with a low Treg percentage have a higher mortality rate than others. Considering that AML constitutes the majority of adult acute leukemias and is often a disease of advanced age, determining the percentage of Tregs at the onset of the FN attack may be helpful.
The primary limitations of this study include its design as a single-center study and the relatively small sample size, which may restrict the generalizability of the findings. Further studies involving multicentric data and larger patient cohorts are required to validate these findings and to explore the underlying mechanisms of Treg influence on survival in this patient population more comprehensively.
Conclusion
Our findings demonstrate that lower Treg percentages are significantly associated with increased mortality in elderly patients aged 65 and older. This suggests that Treg quantification could serve as a valuable prognostic tool in this subset of patients. Additionally, the identification of comorbidities, the presence of a documented infection focus, and elevated CRP levels are key predictors of mortality.
Footnotes
Acknowledgements
We extend our sincere gratitude to all the patients who participated in this study.
Author contributions
S.C. had full access to all of the data in the study and was responsible for study conception. S.C., İ.D., S.T., and Y.O. drafted the manuscript. S.C., I.D., and O.B. had a role in collecting the data. Y.O. did the statistical analyses. All authors read and approved the final manuscript.
Data availability statement
All data generated or analyzed during this study are included in this published article. Further inquiries may be directed to the corresponding author.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This manuscript was supported by Eriskin Asi Dernegi (no. 1).
