Abstract
Purpose. Despite advanced medical treatment options, many prostate cancer patients are still confronted with unfavorable physical and psychological burdens. Physical exercise has proven to be beneficial for prostate cancer patients, yet specific exercise offers are rare. The ProRehab Study aimed to evaluate the exercise program offered in rehabilitative prostate cancer sports groups in Germany and determine whether it is beneficial for patients following prostatectomy. Methods. Eighty-five prostate cancer patients were recruited for a multicenter, 2-armed, nonrandomized controlled trial 6 to 12 weeks after prostatectomy. The intervention group (n = 56) took part in a 15-month supervised multimodal exercise program. Exercise sessions took place once a week for 60 minutes at a moderate intensity (3.84-4.84 MET-hour). The control group (n = 29) received no intervention. Outcomes included aerobic fitness, activity levels, quality of life, disease- and treatment-related adverse effects, such as urinary incontinence and erectile dysfunction, and relapse-relevant blood values. Intention-to-treat analysis was performed. Results. A significant between-group difference was observed in the urinary symptom score (P = .027). Physical fitness, urinary incontinence, physical, role, emotional, and social functioning, as well as further disease- and treatment-related side effects (dyspnea, urinary, and bowel symptoms) significantly improved within the intervention group. Erectile dysfunction and physical activity levels improved similarly in both groups. Conclusions. The presented data hint at the potential of rehabilitative sports groups for prostate cancer patients. However, according to the current state of the art, exercise intensity and volume may need to be increased to enhance the effects. A number of shorter studies (8-24 weeks) have proven significant between-group differences in quality of life, incontinence, and fitness outcomes when patients exercised 2 to 3 times per week. This is the first exercise intervention study with prostate cancer patients that was conducted over 15 months. Further studies are necessary to investigate whether prostate cancer patients recover sooner when receiving a supervised exercise program.
Keywords
Introduction
Despite advanced medical treatment options, most prostate cancer (PCa) patients are confronted with unfavorable disease- and treatment-related side effects. Curative treatment for local PCa generally involves radical prostatectomy and/or irradiation and may cause adverse effects such as erectile dysfunction (ED), urinary incontinence (UI), and bowel problems. 1 Together with further burdens such as fatigue, pain, and a reduced physical functioning, activity levels and quality of life (QoL) may be negatively affected long after primary treatment.1-3
Exercise interventions have proven to be a beneficial and safe supportive measure for PCa patients, by improving physical fitness and QoL and reducing fatigue and other treatment-related side effects.4-6 Pelvic floor exercises, for example, can counteract postoperative UI.5,7 Yet, despite the increasing evidence on physical activity (PA) in PCa patients, specific exercise offers for the men concerned are rare.4,5 Among others, Parsons recently stated, in an editorial article in the Journal of Clinical Oncology, that the routine implementation of PA for PCa patients needs to be considered and will require a novel infrastructure that ensures equitable access for all patients. 8 In Germany, such a supportive care structure was initiated over 30 years ago by founding rehabilitative sports groups for cancer patients. The exercise offer (50 exercise sessions in 18 months) is incorporated in the German social security statutes and financially subsidized by the statutory health insurances. 9
Therefore, the aim of the ProRehab study was to evaluate the exercise program offered in rehabilitative sports groups particularly for PCa patients in Germany. In this context, our objective was also to improve the knowledge on the influence of a multimodal exercise program on physical fitness, QoL, and treatment-related adverse effects in PCa patients following prostatectomy. To date, only one other study has examined the effect of a combined exercise intervention in PCa patients following surgery. 10 Almost all other postoperative studies focus on the effect of pelvic floor exercises on UI. Furthermore, the influence of PA on ED in PCa patients remains uncertain, although previous studies suggest a positive association.11,12
Methods
Participants
Participants were recruited from 4 German hospitals: Heilig-Geist-Hospital Cologne, Marien-Hospital Bergisch Gladbach, Clinical Centre Leverkusen, and University Hospital Essen. Eligibility criteria included men with a malignant PCa disease who underwent radical prostatectomy or combination therapy (surgery + radiation) and received surgery 6 to 12 weeks before the scheduled enrollment. Patients were excluded if they had a metastatic disease, were scheduled to receive hormone treatment or chemotherapy, had a severe cardiac disease (New York Heart Association class III-IV), mental illness or chronic disease that ruled out regular PA, abused alcohol, drugs, or medication, had insufficient German language skills or exercised regularly (>1 hour/week).
Design
The study was designed to be a prospective, multicenter, 2-armed, partially randomized and controlled patient preference trial. A protocol paper has already been published elsewhere. 13 Patient preference was chosen for ethical reasons and because rehabilitative sports groups are accessible and eligible for all cancer patients in Germany. Furthermore, we assumed that recruitment and adherence would be challenging due to the long intervention period of 15 months. The time period was chosen in order to evaluate the standard exercise offer for PCa patients, which is currently subsidized by the statutory health insurance companies in Germany. Based on the procedure of a patient preference trial, patients that refused randomization due to a strong group preference were to receive the intervention of their choice while patients who gave consent to randomization were to be allocated randomly. 14 Since all patients preferred a group, the study has to be classified as a nonrandomized controlled trial. Ethical approval was obtained by the ethics committee of the German Sport University Cologne. All patients provided written informed consent prior to participation. Eligible patients were identified by their urologists and invited for baseline testing at the German Sport University Cologne if interested in the study.
Exercise Intervention
After baseline testing, patients in the intervention group (IG) exercised for 15 months in 1 of 4 community-based rehabilitative sports groups that were implemented particularly for PCa patients. The multimodal exercise program is based on the given regulatory framework of the German social security statutes and was financially subsidized by the patient’s statutory health insurance. Exercise sessions took place once a week for 60 minutes and were supervised by qualified trainers. They mainly included aerobic, resistance and pelvic floor exercises, as well as games and exercises that promote flexibility, coordination, relaxation skills, cognitive abilities, interaction, cooperation, and communication; all adding up to moderate exercise sessions of approximately 3.84 to 4.84 MET-hours (Metabolic Equivalent of Task per hour) per week. Trainers chose the training content from a pool of exercises, and the training intensities were adjusted individually based on the patient’s condition and experience. A detailed description of the program was published previously. 13 Additionally, subjects exercised another 60 minutes per week at home. Control group (CG) patients did not participate in the intervention.
Assessment of Primary and Secondary Endpoints
The primary endpoint, aerobic fitness, was evaluated using a spiroergometry (ZAN 600 USB CPX, Oberthulba, Germany) and submaximal test protocol on a treadmill (WOODWAY, Lörrach, Germany). The initial speed of 0.8 m/s was increased moderately by 0.2 m/s every 3 minutes until one of the following conditions occured: the minute ventilation or ventilation equivalent increased excessively, the ventilation equivalent reached a value between 28 and 30, the metabolic respiratory quotient exceeded 1.0, muscular fatigue set in, the patient felt exhausted (Borg scale: ≥17), or any complications interfered with the testing procedure. The full protocol has been published previously. 13 Relative peak oxygen uptake (VO2peak; in mL/kg/min) and maximum pace (in m/s) were assessed. VO2peak was defined as the highest value of VO2 attained in the test. Not all patients reached the point of complete exhaustion due to comorbidities or individual complaints. UI was determined with the 20-minute pad test.15,16 Prostate-specific antigen (PSA) and testosterone values were evaluated by taking blood samples.
QoL outcomes as well as disease- and treatment-related side effects were assessed by the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) and its PCa-specific module (EORTC QLQ-PR25). 17 ED was assessed using the International Index of Erectile Function (IIEF). 18 Overall PA levels were recorded by means of the Freiburger Questionnaire of Physical Activity. 19 Assessing PA levels was particularly important to verify whether home-based exercises were performed in the IG and CG and contamination occurred in the CG.
All objectively measured and patient-rated outcomes were assessed at baseline (before the intervention) and at posttesting, after the 15-month intervention in both groups. In the IG outcomes were additionally assessed during the intervention; however, these data are not presented.
Sample Size Calculation and Statistical Analyses
Based on previous studies, a moderate improvement in physical performance after a physical exercise intervention can be assumed when compared to a CG (effect size δ/σ = 0.5).20,21 To detect an effect using a 2-tailed t test, with a power of (1 − β) = 80% and α = .05, 64 patients per group were required. Assuming an attrition rate of 30%, the aim was to recruit 182 patients.
Analyses included descriptive statistics, independent and paired t tests, χ2 test, and analysis of covariance (ANCOVA). Student’s t test and the χ2 test were used to compare both groups at baseline. ANCOVA was performed to analyze the differences in the outcome measures between baseline and posttesting. The model was adjusted for medical treatment, recruitment center, baseline values, and a propensity score, in order to account for patient preference. The propensity score is a balancing score that is defined as the probability of treatment assignment conditional on observed baseline characteristics. It therefore allows one to analyze a nonrandomized study because conditional on the propensity score, the distribution of observed baseline covariates will be similar between treated and untreated subjects. Some of the particular characteristics of a randomized controlled trial can therefore be simulated. 22 The propensity score was estimated using a logistic regression model that included the parameters recruitment center, Gleason score, age, and body mass index. Missing values of the primary and secondary outcome parameters were assumed to be missing at random and replaced by multiple imputation. In the imputation process, we used the full conditional specification (FCS) method and computed 5 MI data sets, after a burn-in of 200. We transformed log transformations to deal with nonnormality of continuous variables. The multiple imputation model included all baseline variables that were likely to have affected the probability to respond. Variables with more than 40% missing values or low variance (resulting in convergence problems) were not imputed. These parameters were analyzed using Student’s t test. As a sensitivity analysis, a complete cases analysis was done. The intention-to-treat principle was used for all analysis. No adjustment for multiplicity was made. P values <.05 are considered significant. To help data interpretation Cohen’s d effect sizes were calculated. Based on Cohen’s standard guidelines, effect sizes were defined as follows: d = 0 to 0.2 (trivial effect size), d = 0.2 to 4.49 (small effect size), d = 0.5 to 0.79 (moderate effect size), and d = 0.8 and above (strong effect size). Data were analyzed using IBM SPSS Statistics for Windows, Version 21.0.
Results
From October 2007 to October 2011, 257 eligible patients were approached by the directing institution. Eighty-five (33%) patients agreed to participate in the study and attended baseline testing. Due to financial reasons the trial had to be stopped after 5 years. Fifty-six patients preferred the IG and 29 the CG. Seventy patients attended follow-up testing, 50 in the IG and 20 in the CG. Dropouts in the IG (n = 6) were not due to adverse effects of the training intervention. The flow of the participants through the study is depicted in Figure 1.

Flow of participants through the study.
At baseline groups were balanced regarding anthropometric and medical data (Table 1). Although patients were recruited 6 to 12 weeks following prostatectomy, the mean time between surgery and intervention start was 15.96 weeks in the CG and 17.99 weeks in the IG, due to logistical difficulties concerning the scheduling of baseline testing. On average patients in the IG attended 69.18% of their supervised exercise sessions. All recruitment centers (hospital) were located in a larger city and not far from an intervention site (sports club). Therefore, the number of patients per intervention site was closely related to the recruitment rate of the hospital.
Baseline Characteristics of the Intervention Group and the Control Group.

Abbreviations: BMI, body mass index; SD, standard deviation; TNM, tumor, node, metastases.
Changes in Objectively Measured Outcomes
Table 2 shows the objectively measured outcomes. Group differences over time did not reach statistical significance in any of the outcomes. Relative VO2peak (primary endpoint), maximum pace, and UI improved significantly from baseline to posttesting in the IG (P = .011, P = .035, and P = .005, respectively). Changes in the CG were not significant. PSA and total testosterone stayed nearly unchanged in both groups.
Effects of a 15-Month Exercise Intervention on Objective Outcomes (Aerobic Fitness, Blood Values, and Urinary Incontinence) in Prostate Cancer Patients Following Prostatectomy a .

Abbreviations: CG, control group; IG, intervention group; Max., maximum; Mdn, median; CI, confidence interval; PSA, prostate-specific antigen; SD, standard deviation; Rel. VO2peak, relative peak oxygen uptake.
Mean change is based on posttest score minus baseline score but may not precisely reflect this difference, given that mean change is estimated after multiple imputation using analysis of covariance. The model was adjusted for recruitment center, baseline value of the outcome, and propensity score for group preference. P value <.05 is significant.
Data for testosterone and PSA were skewed and logarithmized before analysis.
Due to low variance, multiple imputation was not performed. Results for complete case analysis are presented.
Due to over 40% missing data, multiple imputation was not performed. Results for complete case analysis are presented.
According to the calculated effect sizes, a small effect on relative VO2peak and maximum pace can be suggested in favor of the IG. The effect sizes of the other objective outcomes are trivial.
Changes in Subjectively Measured Outcomes
Table 3 and 4 present the patient-rated outcomes. Significant between-group differences were not observed in any of the EORTC QLQ-C30 scales (Table 3). The functional scales of the EORTC QLQ-C30 reveal significant improvements in physical, role, emotional, and social functioning (P < .000, P < .000, P = .020, and P = .003, respectively) within the IG. Only role and social functioning also significantly improved in the CG (P = .002 and P = .015, respectively). Regarding the symptom scales, dyspnea significantly decreased in the IG (P = .009). Results of the EORTCQLQ-PR25 showed a significant group difference over time in favor of the IG in the urinary symptom score (P = .027; Table 4). Significant within-group improvements were observed in urinary symptoms, bowel symptoms, and sexual function (P = .001, P = .007, and P = .008, respectively) in the IG from baseline to posttesting. The CG also improved significantly in sexual function (P = .008). In order to obtain a valid assessment, the IIEF evaluation of erectile function only included patients that were sexually active at baseline and posttesting. There were no significant within- or between-group differences in the erectile function scale (Table 4). When looking at all patients that filled in the IIEF questionnaire both at baseline and posttesting whether or not sexually active, we found that the number of patients becoming sexually active increased in the IG (14 at baseline, 16 at posttesting), while it decreased in the CG (10 at baseline, 8 at posttesting). The results of this subgroup analyses are not presented. Overall PA levels improved in both groups, without reaching statistical significance.
Effects of a 15-Month Exercise Intervention on Patient-Rated Quality of Life Outcomes (EORTC QLQ-C30) in Prostate Cancer Patients Following Prostatectomy a .

Abbreviations: EORTC QLQ-C30, European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire; CG, control group; IG, intervention group; N, sample size; CI, confidence interval; QoL, quality of life; SD, standard deviation.
Mean change is based on posttest score minus baseline score but may not precisely reflect this difference, given that mean change is estimated after multiple imputation using analysis of covariance. The model was adjusted for recruitment center, baseline value of the outcome, and propensity score for group preference. P value <.05 is significant.
Due to low variance, multiple imputation was not performed. Results for complete case analysis are presented.
Effects of a 15-Month Exercise Intervention on Patient-Rated Disease- and Treatment-Related Side Effects (EORTC QLQ-PR25), Erectile Function (IIEF; in Sexually Active Patients at Baseline and Posttest), and Physical Activity Levels in Prostate Cancer Patients Following Prostatectomy a .
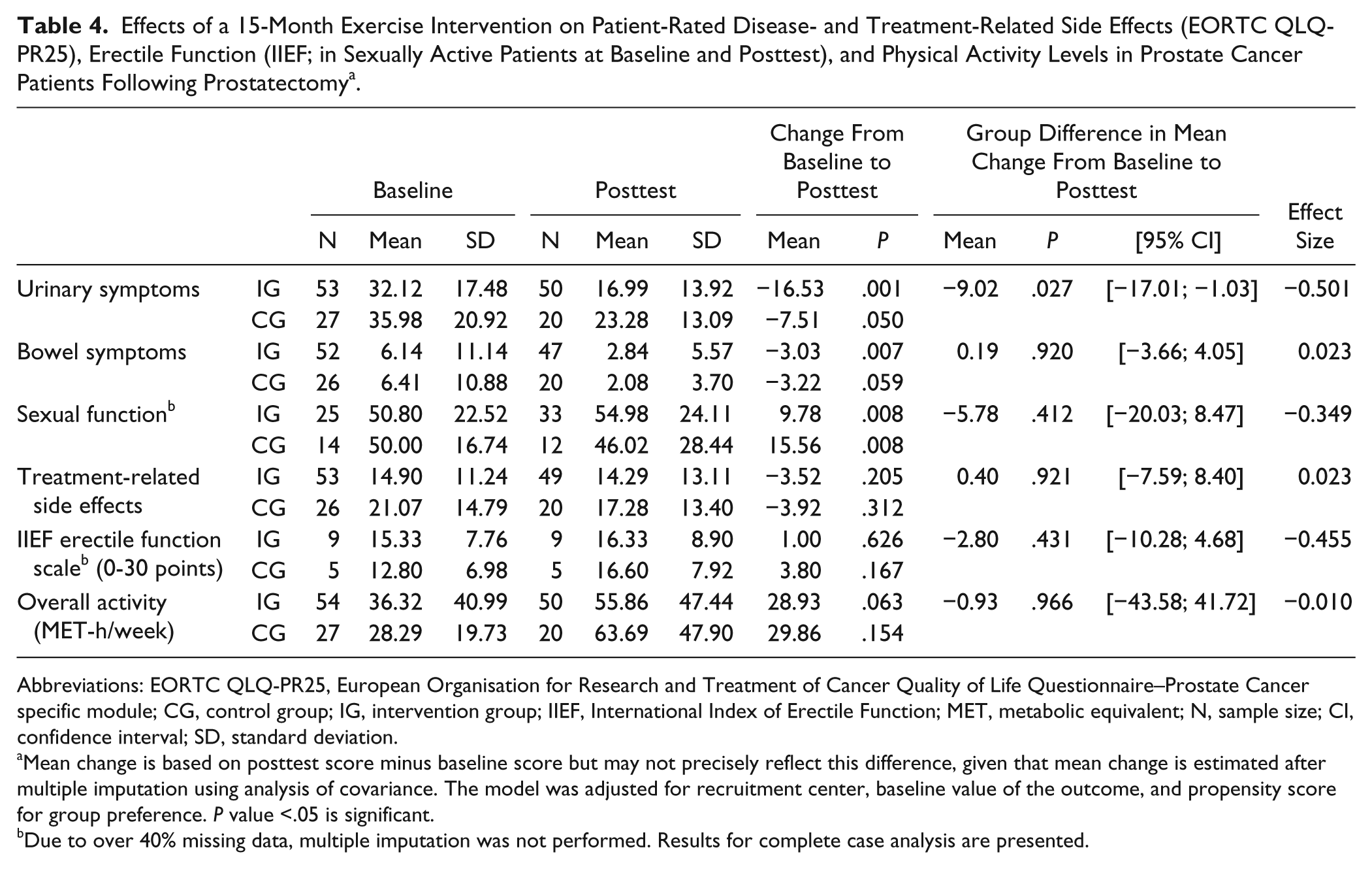
Abbreviations: EORTC QLQ-PR25, European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire–Prostate Cancer specific module; CG, control group; IG, intervention group; IIEF, International Index of Erectile Function; MET, metabolic equivalent; N, sample size; CI, confidence interval; SD, standard deviation.
Mean change is based on posttest score minus baseline score but may not precisely reflect this difference, given that mean change is estimated after multiple imputation using analysis of covariance. The model was adjusted for recruitment center, baseline value of the outcome, and propensity score for group preference. P value <.05 is significant.
Due to over 40% missing data, multiple imputation was not performed. Results for complete case analysis are presented.
According to the calculated effect sizes of the patient-rated outcomes (Tables 3 and 4), a moderate effect in favor of the IG can be observed in the urinary symptom scale. Furthermore, small effects in favor of the IG were determined on the following: global health status, physical function, and fatigue. Small effects were also observed on cognitive function, insomnia, sexual function, and erectile function, yet in favor of the CG. Based on Cohen’s definition, all other effect sizes have to be classified as trivial.
Sensitivity Analysis
Complete case analysis revealed a significant between-group difference in VO2peak in favor of the IG (mean change: IG = 3.19 vs CG = −1.00; P = .018; 95% confidence interval [0.73; 0.77]). Results for all other outcomes were similar to the primary analysis.
Discussion
A 15-month multimodal exercise intervention within a rehabilitative PCa sports group proved to significantly reduce urinary symptoms in PCa patients following prostatectomy. The sensitivity analysis also revealed a significant group difference in VO2peak in favor of the IG. Significant within-group improvements in the IG from baseline to posttesting were observed in physical fitness, UI, physical, role, emotional, and social functioning, as well as further disease- and treatment-related side effects (dyspnea, urinary and bowel symptoms). Effect size calculations suggest a moderate effect on urinary symptoms and a small effect on VO2peak, maximum speed, global health status, physical function, and fatigue in favor of the IG. The positive trends within this study hint at the potential of rehabilitative PCa sports groups. No meaningful changes in PSA or testosterone occurred, indicating that the program is safe regarding possible relapse. Although 95% confidence intervals are wide, PA levels suggest that the CG started exercising independently. This may account for the improvements in some of the outcomes (eg, role and social functioning) in the CG. Both groups improved in sexual functioning, including erectile function, yet effect sizes hint at a greater effect in favor of the CG.
Whether the between-group differences in VO2peak and the urinary symptom scale are clinically relevant can be discussed. The mean group difference of 2.33 in VO2peak implies a difference of approximately 8.5% when assuming that the VO2max of healthy men aged between 50 and 70 years may lie between 16 and 43 mL/kg/min. When considering a change of ≥10 points (on a 100-point scale) clinically relevant in a score of the EORTC questionnaires, then the group difference in the urinary symptom scale of −9.02 (~9%) is close to being so. With group differences of almost 10% these 2 variables tend to be clinically relevant. This is supported by the calculated effect sizes, although comparable studies including effect size calculations are missing and therefore the interpretation was based on Cohen’s rough guidelines. Nevertheless, specific exercise programs addressing specific outcomes, for example, a comprehensive aerobic training to improve aerobic fitness, would probably bring about greater effects over a period of 15 months. However, the exercise program in rehabilitative cancer sports groups is based on a holistic approach.
To our knowledge, this is the first intervention study with PCa patients involving a supervised exercise program that was conducted for more than 12 months. The only comparable, although shorter, study by Park et al demonstrated that a 12-week combined resistance, flexibility, and pelvic floor muscle training can positively influence functional fitness, flexibility, balance ability, UI, and QoL following prostatectomy. 10 Studies examining the effects of a supervised aerobic or resistance training in PCa patients during radiation have also shown significant between-group differences in QoL and fitness outcomes. However, in contrast to our study, these interventions lasted for only 8 to 24 weeks and patients exercised 2 to 3 times per week.5,10,23,24 Aside from higher exercise volumes, exercise adherence is presumably better in shorter interventions, which may enhance the effects. Another important note is that baseline fitness levels and QoL outcomes (EORTC QLQ-C30) of the patients in our study were comparable to the general age-related population and therefore on an average level. Possibly patients with more profound impairments or patients receiving hormone treatment would have shown greater benefits. Regarding UI, previous studies reveal that a supervised pelvic floor exercise program enhances early continence, compared to a nonsupervised program.25,26 In further long-term pelvic floor exercise studies, group differences in UI rates were pronounced in the first months but then declined at 6 to 12 months.27,28 This may explain why we were not able to observe a significant between-group difference in UI over the 15 months. Possibly PCa patients recover “automatically” over time; however, a supervised exercise program hastens the process. Also, the influence of PA on sexual function and ED in patients following prostatectomy remains uncertain. Our findings correspond with the observations by Jones et al, who found no significant group differences in PCa patients following an intensive postoperative nonlinear aerobic exercise program. 29 Our IIEF analysis only involved patients that were sexually active at baseline and posttesting, which reduced the sample size and therefore the validity of the results considerably. However, it prevented misinterpretation of the data and revealed that a great proportion of PCa patients following prostatectomy are not sexually active.
Despite the novel approach of this study, there are some limitations. For the involved hospitals recruiting PCa patients for an exercise intervention trial was new. Recruitment rates varied considerably between the recruitment sites. Since the process of incorporating patient recruitment in their standard routine was often troublesome, the calculated sample size of 182 patients could not be reached in the allowed time. Therefore, the lack of significant group differences may result from limited power. Nevertheless, the sample size is comparable to other studies. Since rehabilitative sports groups are accessible for all cancer patients in Germany, the knowledge on the beneficial effects of PA is spreading, and recruitment was challenging due to the long intervention period, a patient preference design was chosen over a conventional randomized controlled design. As a consequence, the study group sizes were not balanced. The chosen design is certainly a limitation, although baseline characteristics and values (except social functioning) were comparable between the groups. Also, alternative designs are increasingly discussed because poor adherence and exercise contamination in randomized controlled trials is a common challenge and can skew the results in trials with exercise interventions.30,31 We considered the possible bias that emerges within a patient preference trial in the statistical analyses by including a propensity score. There is a lack of consensus as to which baseline variables to include in a propensity score model. 22 We chose the baseline variables that were given prior to baseline testing and when patients were allocated to the CG or IG. Including additional baseline characteristics may be meaningful in future studies, since further baseline covariates may affect treatment assignment and outcome. For further research the comparison of 2 exercise interventions may also be advantageous for long-term studies. In this context, additional assessment time points, especially in the CG, should also be considered to allow a more detailed comparison of the group developments and to further examine whether (supervised) exercise programs hasten PCa recovery. To examine the effectiveness of a specific exercise program on specific outcomes, a single-center trial would be more suitable. In this study, the different intervention sites put together an exercise program based on a pool of exercises. Although the exercise choices and intensities were similar, the exercise modalities differed slightly from site to site. A final shortcoming of the trial is the low pad test compliance and the missing pretreatment data.
Conclusion
The presented data provide preliminary evidence on the effectiveness of rehabilitative PCa sports groups in Germany. For the first time a comprehensive evaluation of the exercise program was conducted; an important step, when aiming at administering efficacious and standardized programs to increase PA in PCa patients. In principle, the German model of rehabilitative sports groups provides an ideal infrastructure for PA programs in cancer survivors due to the supervised setting and financial support given by the statutory health insurance companies. Recruitment in this trial was challenging because the sports groups that we implemented were the first ones nationwide particularly for PCa patients, and it took some time for the information to spread. In the meantime, however, over 85 PCa sports groups exist in Germany and other countries, such as New Zealand, have started to consider the exercise offer. Nevertheless, exercise intensities and possibly supervised sessions need to be increased to enhance the effects. Although the most beneficial exercise modes and dosages for PCa patients remain mostly unclear, the current state of the art and exercise recommendations suggest that supervised programs and higher exercise intensities and volumes are more effective.5,6,32 Kenfield et al, 33 for example, showed that patients who engaged in moderate PA may improve overall survival; however, to improve PCa-specific survival at least 3 hours of vigorous activity per week (≥6 MET-h) are necessary. To further examine whether PCa patients increase their PA levels sooner and therefore recovered faster, when exercising in a supervised setting, further studies with long intervention periods, additional assessment time points, and patients with more profound adverse effects are necessary. Since UI and ED, beyond other adverse events, can persist for many months or even years in PCa patients, it is, in general, essential to consider long-term interventions and support for the men concerned.
Footnotes
Acknowledgements
We would like to thank Prof Klaus Schüle, Dr Bernhard Brehmer, the involved sport clubs, the Cancer Society North-Rhine Westphalia (Krebsgesellschaft NRW e.V.), the State Sport Association of North-Rhine Westphalia (Landessportbund NRW e.V.), and the Barmer GEK Health Insurance for supporting this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Cancer Society North-Rhine Westphalia (Krebsgesellschaft NRW e.V.) and the Barmer GEK Health Insurance.
