Abstract
Recent reports suggest the existence of a subpopulation of stem-like cancer cells, termed as cancer stem cells (CSCs), which bear functional and phenotypic resemblance with the adult, tissue-resident stem cells. Side population (SP) assay based on differential efflux of Hoechst 33342 has been effectively used for the isolation of CSCs. The drug resistance properties of SP cells are typically due to the increased expression of ABC transporters leading to drug efflux. Conventionally used chemotherapeutic drugs may often leads to an enrichment of SP, revealing their inability to target the drug-resistant SP and CSCs. Thus, identification of agents that can reduce the SP phenotype is currently in vogue in cancer therapeutics. Withania somnifera (WS) and Tinospora cordifolia (TC) have been used in Ayurveda for treating various diseases, including cancer. In the current study, we have investigated the effects of ethanolic (ET) extracts of WS and TC on the cancer SP phenotype. Interestingly, we found significant decrease in SP on treatment with TC-ET, but not with WS-ET. The SP-inhibitory TC-ET was further fractionated into petroleum ether (TC-PET), dichloromethane (TC-DCM), and n-butyl alcohol (TC-nBT) fractions using bioactivity-guided fractionation. Our data revealed that TC-PET and TC-DCM, but not TC-nBT, significantly inhibited SP in a dose-dependent manner. Furthermore, flow cytometry–based functional assays revealed that TC-PET and TC-DCM significantly inhibited ABC-B1 and ABC-G2 transporters and sensitized cancer cells toward chemotherapeutic drug-mediated cytotoxicity. Thus, the TC-PET and TC-DCM may harbor phytochemicals with the potential to reverse the drug-resistant phenotype, thus improving the efficacy of cancer chemotherapy.
Introduction
Chemotherapy remains the major mode of anticancer treatment for a variety of cancers. However, the development of drug resistance continues to act as a major deterrent in cancer chemotherapy, leading to treatment failure and/or cancer recurrence. Recent identification of a subpopulation of innately drug-resistant cancer stem cells (CSCs) within several cancers provides one possible explanation for cancer drug resistance and recurrence. The CSCs show properties of extensive self-renewal and differentiation and are chiefly responsible for tumor initiation, maintenance, and progression, analogous to the functions of normal adult stem cells in tissue homeostasis. 1 It is believed that although conventional chemotherapy kills the bulk of tumor cells, leading to tumor shrinkage, it leaves behind the CSCs unscathed, thus leading to cancer relapse. 2 Indeed, CSCs have been identified in cancer patients after chemotherapy. 3 CSCs were first documented in hematological malignancies 4 ; such tumorigenic and drug-resistant CSCs have now been documented in a wide variety of solid tumors, including breast cancer, brain tumor, pancreatic cancer, colon cancer, head and neck squamous cell carcinoma, melanoma, prostate cancer, and others. 5
The ATP-binding cassette (ABC) families of transporters are molecular pumps that contribute to drug resistance by ATP-dependent drug efflux. 6 Interestingly, normal stem cells express high levels of ABC family of transporters that is believed to help them efflux cellular toxins. 7 Indeed, one of the early strategies to isolate stem cells exploited their efflux properties. 8 Stem cells from multiple tissues have been identified based on their ability to efflux the lipophilic dye Hoechst 33342, which is a fluorescent dye that binds to the AT regions of the DNA. 9 Typically, this molecule is excited at 355 nm and emits at 440 to 460 nm with a weak emission in the red range. When cells are stained with Hoechst 33342 and analyzed by a flow cytometer equipped with a ultraviolet (UV) laser, most of the cells show high emission in a flow cytometry plot (with red emission on the x-axis and blue emission on the y-axis), but stem cells appear as a hypodiploid, low fluorescing “dull” population on the far left side. This Hoechst-low population has been termed the side population (SP). 10 This method was first described in mouse bone marrow cells, where it was demonstrated that the SP is highly enriched for stem cells because it had the ability to reconstitute both lymphoid and myeloid lineages and was also enriched at least 1000-fold for in vivo reconstitution in lethally irradiated mice. 9 Similarly, SP has been identified in many normal tissues, including mammary gland, lung, skeletal muscle, heart, liver, brain, and skin in both human cells and animal models. 10
Similar to normal stem cells, the existence of SP has been demonstrated in a variety of tumors and cell lines, including neuroblastoma, 8 C6 glioma cell line, 11 human retinoblastoma, 12 human gastrointestinal cancer cell lines, 13 ovarian cancers, 14 melanoma, 15 leukemia, 16 MCF7 breast cancer cell line, 17 and lung cancer cell line. 18 Isolated tumor SP has been found to be more tumorigenic than the corresponding non-SP 19 and has also been shown to generate non-SP that forms the bulk of the tumor,8,11 thus revealing their cancer stem-like properties. The CSCs have high expression of ABC transporters (ABC-B1 or multidrug resistance [MDR]-1, ABC-G2 or BCRP, ABC-C1 or MRP-1) on their surfaces, 20 which likely contributes to their inherent drug resistance as well as the SP phenotype. Indeed, in several cancers, SP marks a subpopulation of CSCs. 10 Treatment with standard chemotherapeutic agents has been shown to lead to an increase in the SP. For example, treatment of neuroblastoma cells and neurospheres with mitoxantrone (MXR) led to an increase in the SP cells and afforded them a survival advantage.8,21 Similarly, significantly higher drug efflux from leukemic SP cells, compared with non-SP cells, was demonstrated in acute myeloid leukemia. 3 HL60 leukemia cells and MCF7 breast cancer cells, chronically exposed to a low concentration of doxorubicin (DOX), showed enrichment for SP.19,22 Temozolomide treatment of human gliomas led to a significant increase in the SP cells. 21 In most of these cases, the drug killed a large number of bulk cells but left behind the resistant ones. Thus, there is an urgent need to target the SP cells, such that, together with conventional drugs that target the bulk of cancer cells, one can aim to effectively control cancer relapse.
Phytochemicals are natural products found in plants; they show promise as a complement to current chemotherapy and may be used along with conventional therapy to target cancer stem-like cells. 23 Various anticancer drugs are derived from plant sources, leading to scientific interest in the discovery of anticancer drugs from plants. 24 Phytochemicals have been in vogue for anticancer treatment for a long time owing to fewer associated side effects, and in this study, we have explored the anti-SP activity of 2 well-known medicinal plants used in Indian traditional medicine: Withania somnifera (WS) of the family Solanaceae and Tinospora cordifolia (TC) of the family Menispermaceae. Both WS and TC have been used in the Indian traditional system of medicine for the treatment of cancer. 25 Indeed, the anticancer potential of both WS and TC has been demonstrated by various in vitro studies and animal models. For example, WS root extracts have been shown to prevent squamous cell carcinoma and skin carcinogenesis in mice and possess antitumor, antiangiogenic, and radiosensitizing effects in experimental tumors in vivo without any systemic toxicity. 26 Similarly, TC extracts have been topically used to treat various tumors, have been shown to be clinically effective against throat cancer, and inhibit skin carcinogenesis and experimental metastasis. 27 More recently, we have reported the anticancer effects of the ethanolic extracts (ETs) of WS and TC against human breast cancer cells. 28 However, a detailed investigation of the effects of WS and TC extracts against cancer stem-like cells or SP remained unexplored. Because conventionally used anticancer drugs enrich the SP, we designed this study such that the goal was to evaluate the ability of the ETs of WS and TC to deplete or prevent the enrichment of SP in human epithelial cancer cells treated with anticancer drugs. Furthermore, aiming to isolate the bioactive phytochemicals, we used a bioactivity (SP-based assay)-guided fractionation strategy, and fractions of ETs of WS and TC were tested for anti-SP activity. Because the SP phenotype is strongly mediated by ABC transporters (ABC-B1 and ABC-G2), a direct functional assay was performed to evaluate the ABC-B1 (MDR1) and ABC-G2 (BCRP) inhibitory effects of various fractions in human epithelial cancer cells. We show that some fractions of the ETs of TC possess SP inhibitory properties, mediated, at least in part, by the inhibition of ABC transporters.
Materials and Methods
Cell Culture
HeLa (human cervical carcinoma), A549 (human lung adenocarcinoma), BT474, MDA-MB-231, MDA-MB-453, T47D (human breast carcinoma), MDA-MB-435S, G361, and WM 266.4 (melanoma) cells were cultured in Dulbecco’s modified eagle medium (DMEM) supplemented with penicillin (100 IU/mL), streptomycin (100 µg/mL), and 10% fetal bovine serum (FBS) in a humidified CO2 incubator at 37°C.
Drugs and Chemicals
DOX, MXR, Verapamil, Fumitremorgin C (FTC), Rhodamine 123 (Rho 123), DMEM, Trypsin-EDTA, Hank’s balanced salt solution (HBSS), HEPES buffer, Hoechst 33342 (bisbenzimide trihydrochloride), Thiazolyl Blue Tetrazolium Bromide (MTT), and Propidium Iodide (PI) were purchased from Sigma Chemical Co (St Louis, MO). FBS was purchased from Gibco (Invitrogen, USA). Dimethyl sulfoxide (DMSO) was purchased from Calbiochem.
Preparation of Plant Extracts, Fractions, and HPTLC Finger Printing
The roots of WS Dunal of the family Solanaceae and the stem part of TC Hook F and Thomas of the family Menispermaceae were used for the study. The taxonomic identification and the preparation of ETs were carried out as reported earlier. 28 The yields of ethanolic (ET) extract of WS (WS-ET) and TC (TC-ET) were 7.12% and 4.94%, respectively. The TC-ET (80 g) was further fractionated by dispersing in methanol-distilled water and extracted successively with solvents of increasing polarity index such as petroleum ether (PET), dichloromethane (DCM), and n-butyl alcohol (nBT) to get TC-PET (TC petroleum ether fraction), TC-DCM (TC dichloromethane fraction), and TC-nBT (TC n-butyl alcohol fraction), respectively. Each fraction was concentrated using a rotary evaporator under reduced pressure and completely dried by lyophilization. The yields of TC-PET, TC-DCM, and TC-nBT were 15.19%, 11.40%, and 18.50%, respectively. For biological studies, the lyophilized ETs and their fractions were dissolved in DMSO. HPTLC finger printing of TC-ET, TC-PET, and TC-DCM was carried out as described earlier. 29 The HPTLC finger printing of the bioactive TC-ET, TC-PET, and TC-DCM revealed sharp, symmetrical, and well-resolved peaks in the solvent system of toluene:ethyl acetate:formic acid (6:3:1) with maximum UV absorbance at 280 nm (data not shown).
Side Population Analysis
C57BL/6 mice (6-8 weeks of age) were killed humanely, and bone marrow cells were isolated from the femur and tibia. SP assay was performed as described before with modifications. 9 Briefly, 1 × 106 cells/mL nucleated cells were incubated in a 1.5-mL Eppendorf tube in prewarmed DMEM containing 2% FBS, 10 mM HEPES, and freshly added Hoechst 33342 (5 µg/mL) for 90 minutes at 37°C with intermittent mixing in a circulating water bath. A parallel control set was prepared in which ABC transporter inhibitor verapamil (50 µM) was added along with Hoechst 33342. At the end of 90 minutes of incubation, cells were washed twice in ice-cold HBSS (by centrifugation at 1200 rpm for 4 minutes at 4°C), and cell pellets were resuspended in ice cold HBSS containing 2% FBS and 10 mM HEPES and maintained in cold until analysis to prevent further efflux of Hoechst 33342. PI (2 µg/mL), which allows for the discrimination of live and dead cells, was added just before FACS analysis. Cells were analyzed in a flow cytometer (MoFlo-Beckmen Coulter, USA) equipped with an UV laser. Hoechst 33342 was excited with the UV laser at 355 nm, and emissions were detected at 450/50 nm (Hoechst Blue) and 670/30 nm (Hoechst red). A total of 1 × 105 events were recorded and data analyzed in Summit v4.3 software (Beckmen Coulter, USA). Dead cells (PI positive cells) were excluded from the analysis based on PI gating. SP appeared as a distinct and small population of cells on the left corner of the dot plot as compared with the bulk cells. SP was gated based on its disappearance in the presence of verapamil (ABC transporter inhibitor) in the control set.
For the cancer cell lines, cells at 70% to 80% confluency were trypsinized, and single-cell suspensions (1 × 106 cells/mL) were incubated in DMEM containing Hoechst 33342, as described above. A parallel control set was prepared in which ABC transporter inhibitor verapamil (50 µM) or FTC (10 µM) was added along with Hoechst 33342. At the end of incubation, cells were washed twice with ice-cold HBSS containing 2% FBS, and analyzed in a flow cytometer as described above.
SP Analysis in Cell Lines Following Treatment With DOX, WS-ET, and TC-ET
HeLa, A549, and MDA-MB-435S (5 × 105 cells) were seeded in 60-mm tissue culture plates, and cells at 50% to 60% confluency were treated with sublethal concentrations of DOX (0.50 µM, where 70% of cells are viable at 48 hours) for 96 hours, as shown previously.3,28,30 Untreated cells served as controls. An SP analyses was undertaken after 96 hours of drug treatment, as described above. A parallel control set was prepared in which ABC transporter inhibitor verapamil (50 µM) or FTC (10 µM) was added along with Hoechst 33342.
Similarly, cells were treated with sublethal concentrations of WS-ET (20 µg/mL) and TC-ET (50 µg/mL) 28 for 96 hours, and SP assays were undertaken at the end of the treatment period. DMSO-treated cells served as vehicle control.
Assays for ABC Transporter Inhibitory Activity of TC Fractions
To gauge if the TC extract and its fractions functioned to inhibit ABC transporter activity, HeLa, A549, and MDA-MB-435S (1 × 106 cells/mL) cells were incubated with Hoechst 33342 for 90 minutes (as for a standard SP assay) in the presence of TC-ET (50 µg/mL) and its fractions TC-PET, TC-DCM, and TC-n-BT at 50 µg /mL along with verapamil and FTC (known ABC transporter inhibitors), which served as positive controls, and DMSO, which served as vehicle control. To gauge dose-dependent inhibitory effects on SP, cells (HeLa and A549) were also incubated with varying concentrations (10, 25, and 50 µg/mL) of TC-ET, TC-PET, and TC-DCM. At the end of incubation, cells were scored for SP phenotype by flow cytometry.
ABC-B1 (MDR1) inhibitory function of TC extract and its fractions was determined by Rho efflux assay as described earlier, with modifications. 31 Briefly, HeLa cells (1 × 106 cells/mL) in DMEM containing 2% FBS were incubated with Rho 123 (0.50 µM) at 37°C, CO2 incubator for 30 minutes (accumulation phase). After centrifugation and removal of supernatant, cells were reincubated in DMEM containing 2% FBS at 37°C, CO2 incubator for an additional 60 minutes (efflux phase) with or without verapamil (50 µM), DMSO (vehicle control), TC-ET, TC-PET, and TC-DCM (50 µg/mL each). At the end of incubation, cells were washed twice with ice-cold HBSS and kept on ice until analyses by flow cytometry (FACSCanto II, Becton Dickinson, USA). Rho 123 was excited with the laser at 488 nm and emission was detected at 530/30 nm (FL-1). Mean fluorescence intensity (MFI) was determined for vehicle control (DMSO), verapamil, TC-ET, TC-PET, and TC-DCM treated samples.
Similarly, ABC-G2 inhibitory potential of TC extracts and fractions was evaluated in A549 cells using MXR efflux assay as described earlier with modifications. 32 Briefly, A549 cells (1 × 106 cells/mL) were incubated in DMEM containing 2% FBS with MXR (10 µM) at 37°C, CO2 incubator for 30 minutes (accumulation phase). After centrifugation and removal of supernatant, cells were reincubated for an additional 60 minutes (efflux phase) with or without FTC (10 µM), DMSO (vehicle control), TC-ET, TC-PET, and TC-DCM (50 µg/mL each). At the end of incubation, cells were washed twice with ice-cold HBSS and kept on ice until analyses by flow cytometry (FACSCanto II, Becton Dickinson, USA). MXR was excited with the laser at 633 nm, and emission was detected at 660/20 nm (FL-3). MFI was determined for vehicle control (DMSO), FTC, TC-ET, TC-PET, and TC-DCM treated samples.
Cytotoxicity Assay
Dose-dependent cytotoxic effects of TC-ET, TC-PET, and TC-DCM were measured by MTT assay as described earlier. 28 Briefly, HeLa (6000/100 µL) and A549 (8000/100 µL) cells were seeded in 96-well microtiter tissue culture plates for 24 hours. Cells were then treated with varying concentrations of TC-ET, TC-PET, and TC-DCM (50, 100, and 200 µg/mL) in triplicates for 48 hours. DMSO served as vehicle control. At the end of treatment, 20 µL of MTT solution (5.00 mg/mL in PBS) was added to each well and further incubated for 4 hours. Thereafter, medium containing MTT was gently replaced by 200 µL DMSO, and the absorbance values were measured by a microtiter plate reader at 570 nm. Dose-response curves were generated by plotting percentage cell viability (calculated as [Optical density of test/Optical density of vehicle control] × 100) on the y-axis and concentration in (µg/mL) on the x-axis. Cell viability in vehicle control was normalized to 100. IC50 values of TC-ET, TC-PET, and TC-DCM were obtained from the graph as the concentration that decreases percentage cell viability to 50% (using the curve-fit model).
To assess the effects of fractions of TC-ET on cytotoxicity mediated by chemotherapeutic drugs (DOX and MXR), HeLa and A549 cells were treated with TC-ET, TC-PET, and TC-DCM (50 µg/mL) in the presence of DOX (1.00 µM) and/or MXR (0.50 µM) in 96-well microtiter tissue culture plates for 48 hours. DMSO served as vehicle control. At the end of the treatment, percentage cell viability was determined by MTT assay (as described above).
Statistical Analysis
All values are expressed as the mean ± standard error of the mean. Data were analyzed using One-way ANOVA, followed by a Dunnett’s post hoc multiple comparisons test. A value of P < .05 was considered to be statistically significant compared with the vehicle control.
Results
Establishing the Hoechst 33342 Efflux-Based SP Assay
To screen for plant extracts that deplete the SP phenotype, we first established the SP assay protocol in mouse (C57BL/6) bone marrow–derived cells as an experimental control, where the SP assay was first established. 9 We detected 0.10% SP in the bone marrow–derived cells (Figure S1), which disappeared in the presence of verapamil (an ABC-B1 inhibitor), thus, validating our SP assay protocol. 9 We next proceeded to establish the SP assay in cultured HeLa cells. As reported in the literature,11,19 we detected 1.43% SP in HeLa cells (Figure 1A), which disappeared in the presence of verapamil (Figure 1A), an ABC-B1 inhibitor. Following suit, we evaluated the presence of SP in a battery of cancer cell lines, including A549, BT474, MDA-MB-231, MDA-MB-435S, MDA-MB-453, T47D, G361, and WM 266.4. We detected the presence of a distinct SP in A549 (9.08%) and MDA-MB-435S (0.31%), which disappeared in the presence of FTC, which is an ABC-G2 inhibitor (Figures 1B and 1C). However, we failed to detect SP in the other cell types tested (Figure 1D). Accordingly, in all our experiments, we used verapamil as an ABC transporter inhibitor for HeLa and FTC as an ABC transporter inhibitor for A549 and MDA-MB-435S. The SP gates in each experiment were set based on its disappearance in the presence of verapamil or FTC in the DMSO-treated control samples (Figures 1A-1C). Based on these pilot experiments, we chose HeLa, A549, and MDA-MB-435S cells for our further studies.
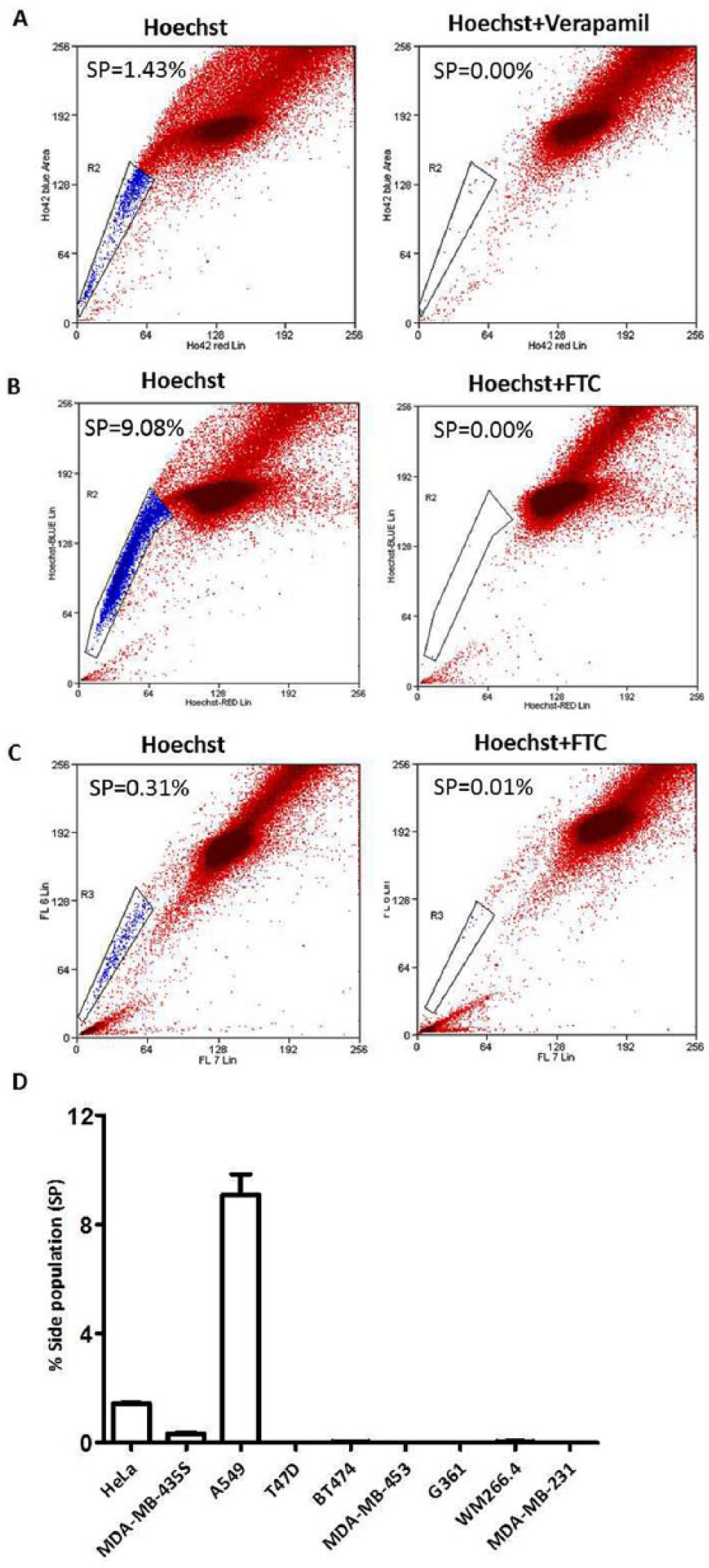
Establishment of side population (SP) assay in epithelial cancer cell lines. HeLa (human cervical carcinoma), A549 (human lung adenocarcinoma), BT474, MDA-MB-231, MDA-MB-453,T47D (human breast carcinoma), MDA-MB-435S, G361, and WM 266.4 (melanoma) cells were incubated with Hoechst 33342 alone, or Hoechst 33342 plus verapamil (50 µM) or fumitremorgin C (FTC, 10 µM) for 90 minutes at 37°C and analyzed by flow cytometry for the detection of SP. (A) to (C) are representative dot plots of SP assay in HeLa, A549, and MDA-MB-435S, respectively. The graph in D shows percentage SP in each of the cell lines tested. Error bars represent ±standard error of the mean; n = 3.
Establishing a Bioactivity-Guided Assay to Screen for SP-Inhibitory Property of WS-ET and TC-ET
Recent literature has revealed that treatment with commonly used anticancer drugs such as DOX enriches for SP.3,19,30 To see if this observation holds true in the 3 cell lines that we have chosen for the study—HeLa, A549, and MDA-MB-435S—we first investigated the behavior of these cell lines after DOX treatment. To do so, these cells were treated with DOX (0.5 µM) for 96 hours (see Methods section). At the end of the treatment period, cells were analyzed for their SP content. In untreated control cells, we detected 1.37%, 10.35%, and 0.23% of SP in HeLa, A549, and MDA-MB-435S cells, respectively, which disappeared in the presence of ABC transporter inhibitors (verapamil or FTC; Figures 2A-2C). However, in the DOX-treated cells, we detected a significant increase in SP: 6.27%, 20.57%, and 0.65%, respectively (Figures 2A-2C). Thus, our data revealed a 4.57-, 1.98-, and 2.82-fold enrichment of SP in the 3 cell lines HeLa, A549, and MDA-MB-435S, respectively, after treatment with DOX. These results confirmed that treatment of cancer cells with a conventionally used anticancer drug, such as DOX, results in an enrichment of SP, consistent with previous reports.3,19,30 Thus, we designed the goal of our study to screen for anticancer plant extracts that prevent the enrichment of SP, or deplete the SP, in contrast to DOX, which enriches it.
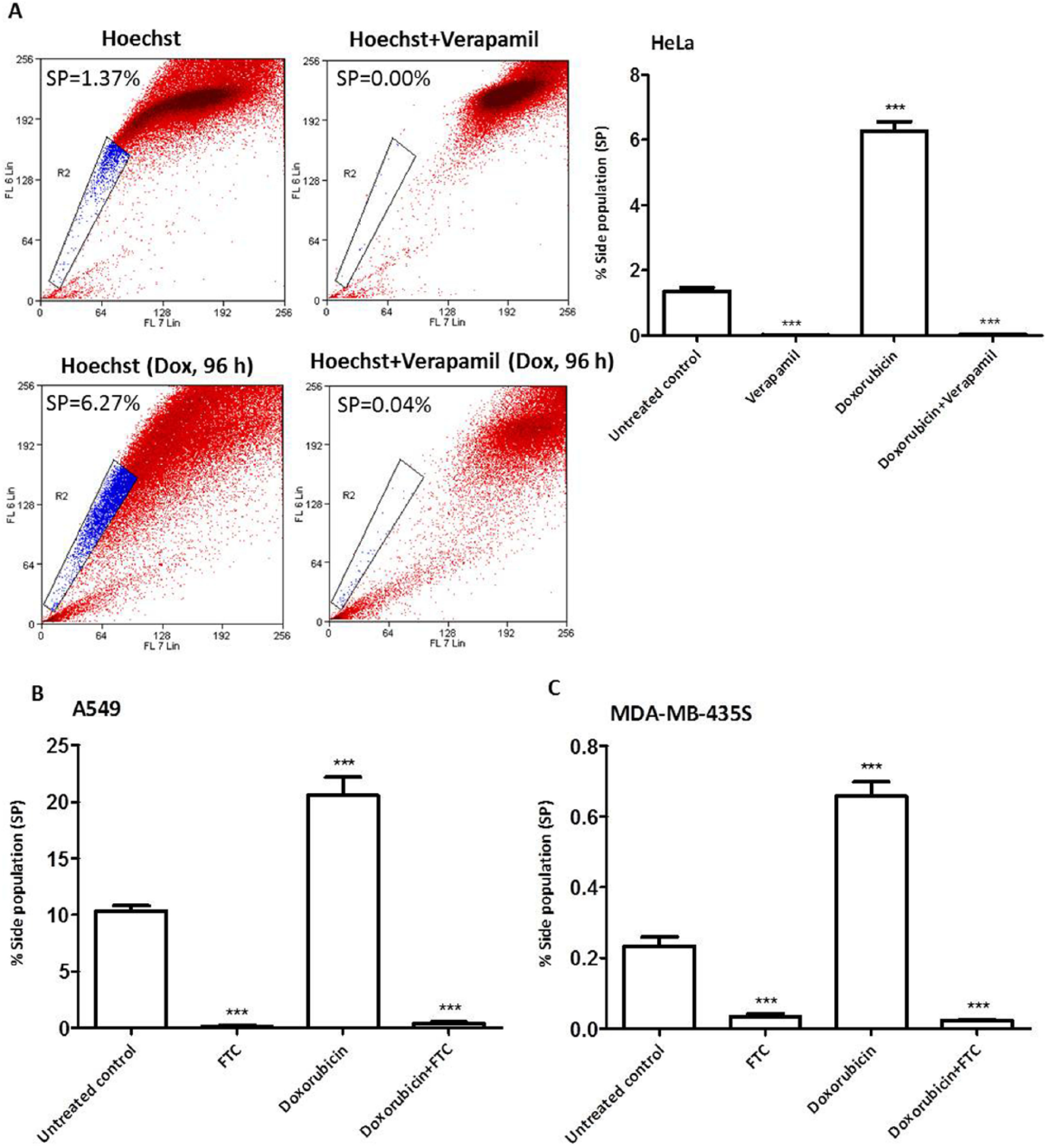
Effect of treatment with doxorubicin on side population (SP) content. HeLa (A), A549 (B), and MDA-MB-435S (C) cells were treated with doxorubicin (Dox, 0.5 µM) for 96 hours. At the end of the treatment period, cells were washed with Hank’s balanced salt solution and subjected to SP assay (90 minutes incubation with Hoechst 33342 at 37°C); verapamil (50 µM) or fumitremorgin C (FTC; 10 µM) were used for SP gating. Error bars represent ±standard error of the mean; n = 3, ***P < .001.
SP Inhibitory Effects of WS-ET and TC-ET
We have recently demonstrated that the ET but not the aqueous extracts of 2 Indian medicinal plants, WS and TC, possess anticancer activities. 28 In this study we investigated whether the ET extracts of WS (WS-ET) and TC-ET have any SP inhibitory activities on cultured human epithelial cancer cells. To this end, HeLa and MDA-MB-435S cells were treated with WS-ET (20 µg/mL) and TC-ET (50 µg/mL; see Methods section) for 96 hours. DOX (0.5 µM) was used as a standard anticancer drug, and DMSO was used as a vehicle control. At the end of the treatment period, SP assay was performed, and the ability of the extracts to reduce the SP, if any, was scored. As before, treatment with DOX led to an increase in SP in both the cell lines compared with vehicle-treated control cells (Figures 3A and 3B). In contrast, and interestingly, addition of TC-ET led to a >95-fold depletion of SP in both the cell types compared with vehicle control cells (Figure 3A and 3B). Treatment with WS-ET failed to cause a significant change in SP (Figure 3A and 3B). Thus, these data revealed that in contrast to conventionally used chemotherapeutic drugs like DOX, treatment with TC-ET led to a depletion of SP.
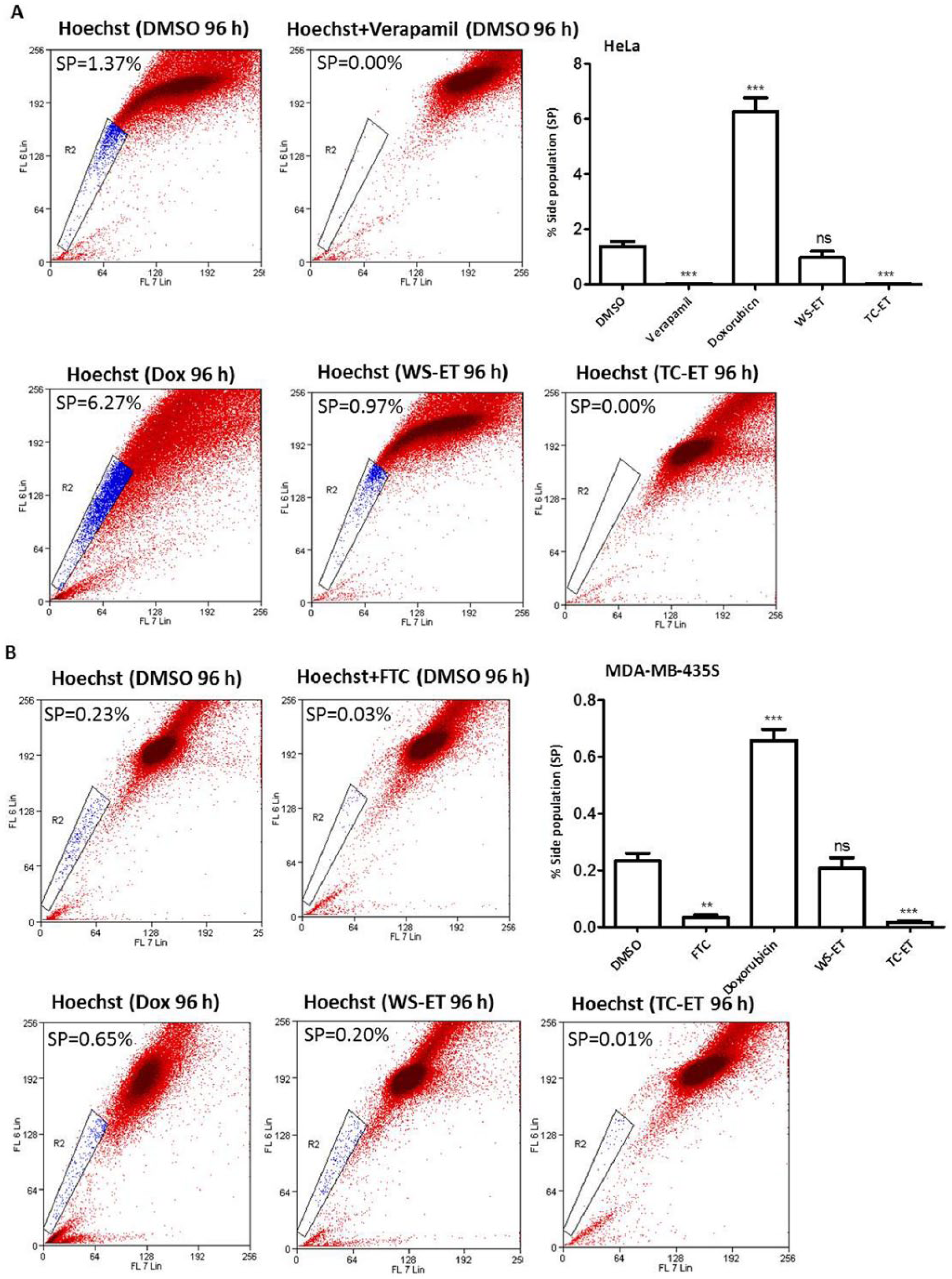
Effect of treatment with ethanolic extracts (ETs) of Withania somnifera (WS-ET) and Tinospora cordifolia (TC-ET) on side population (SP) content. HeLa (A) and MDA-MB-435S (B) cells were treated with WS-ET (20 µg/mL), TC-ET (50 µg/mL), and doxorubicin (Dox; 0.5 µM) for 96 hours; dimethyl sulfoxide (DMSO) served as vehicle control. At the end of the treatment period, cells were washed with Hank’s balanced salt solution and subjected to SP assay; verapamil (50 µM) or fumitremorgin C (FTC; 10 µM) were used for SP gating. Error bars represent ±standard error of the mean; n = 3, ***P < .001, **P < .01.
ABC Transporter Inhibitory Effects of TC-ET Fractions
We reasoned that one possible cause for the observed depletion of SP brought about by treatment with TC-ET may be a result of direct inhibition of ABC transporters that mediate the drug efflux (in a fashion similar to ABC transporter inhibitors like verapamil and FTC). To address this, we undertook a typical SP assay (involving 90 minutes incubation with Hoechst) in the presence of TC-ET, in parallel with the known ABC transporter inhibitors verapamil and FTC. We observed that similar to verapamil/FTC, treatment with TC-ET also led to the disappearance of SP in both HeLa and MDA MB 435S cells (Figures 4A and 4B), suggesting that the TC-ET may possess ABC transporter inhibitory functions.
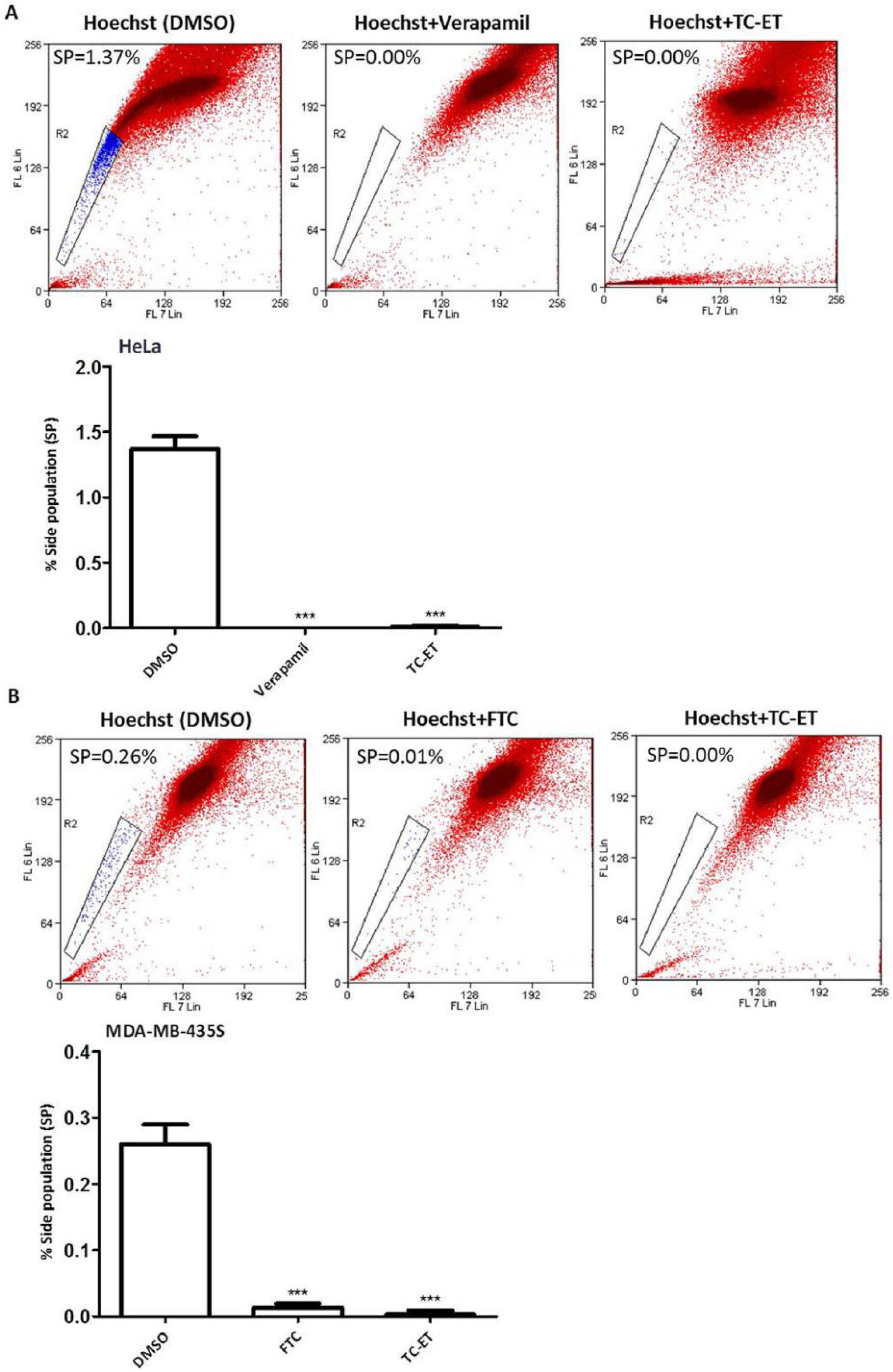
Effect of Tinospora cordifolia ethanolic extract (TC-ET) on Hoechst 33342 efflux. HeLa (A) and MDA-MB-435S (B) cells were subjected to side population (SP) assay (90 minutes incubation with Hoechst 33342 at 37°C) in the presence or absence of TC-ET (50 µg/mL); verapamil (50 µM) and fumitremorgin C (FTC; 10 µM) served as positive control for ABC transporter inhibitors, and dimethyl sulfoxide (DMSO) served as vehicle control. Error bars represent ±standard error of the mean; n = 3, ***P < .001.
We further fractionated the TC-ET for the isolation of active phytochemicals for their SP inhibitory properties. To do so, TC-ET was fractionated successively with solvents of increasing polarity index, such as PET, DCM, and nBT to obtain the fractions TC-PET, TC-DCM, and TC-nBT. The effects of these fractions were tested in a standard SP assay. Interestingly, our data revealed that compared with vehicle control, TC-PET and TC-DCM fractions led to a more than 90% reduction in SP in HeLa and MDA MB 435S cells (Figures 5A and 5B); however, TC-nBT led to an enrichment of SP. Similar data were obtained with A549 cells (Figure 5C). Furthermore, dose-response studies revealed that TC-ET, TC-PET, and TC-DCM imparted dose-dependent inhibitory effects on SP (Figures 6A and 6B). Interestingly, TC-ET, TC-PET, and TC-DCM significantly depleted SP at lower concentrations (10 µg/mL), thus indicating potent SP-inhibitory effects (Figures 6A and 6B). TC fractions (TC-PET and TC-DCM) were found to be more potent than TC-ET at lower doses (25 and 10 µg/mL) (Figures 6A and 6B). Taken together, these data revealed that specific fractions of the medicinal plant TC depleted the SP, at least in part, by inhibiting ABC transporter activity.
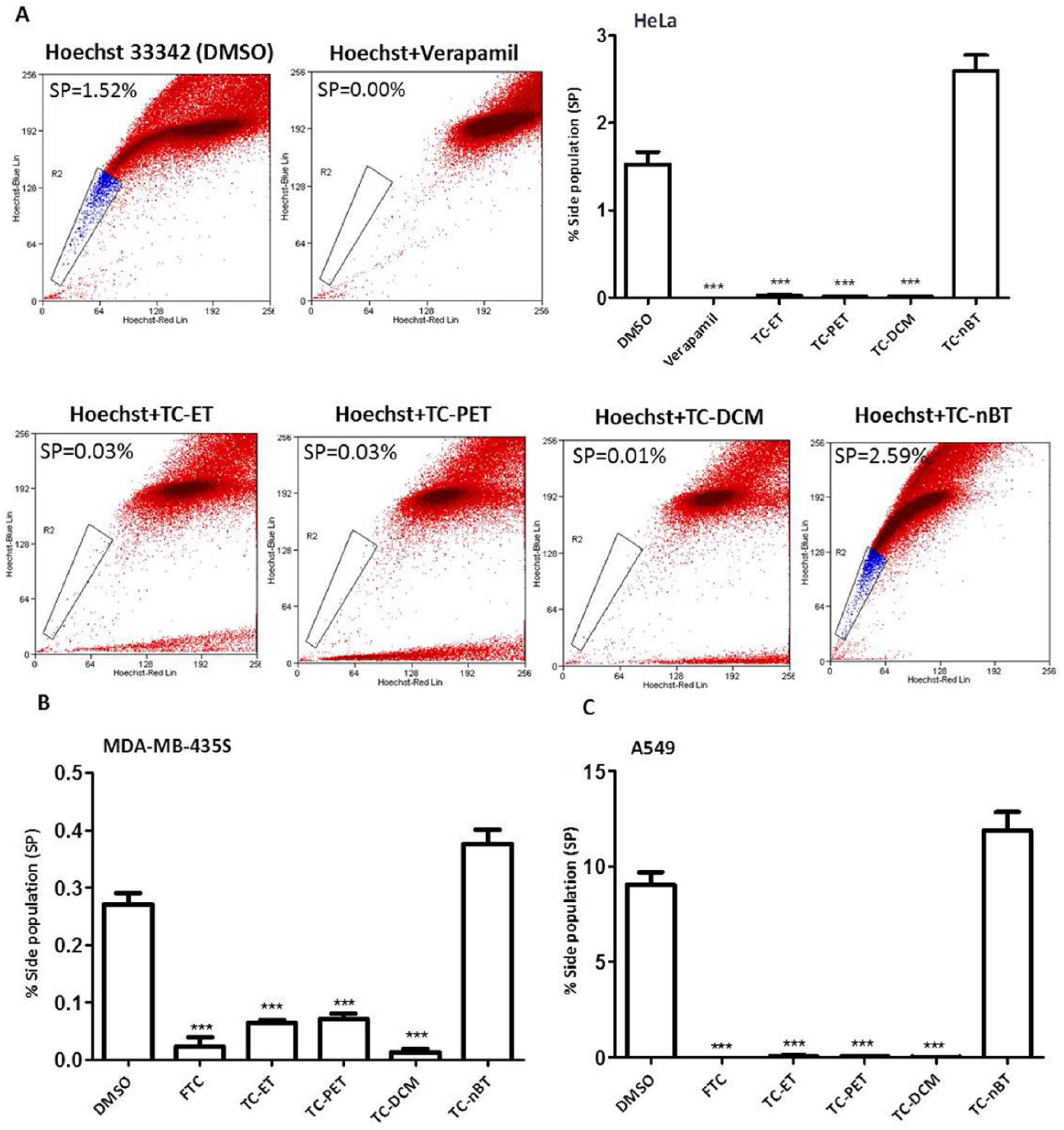
Effect of fractions of Tinospora cordifolia ethanolic extract (TC-ET) on Hoechst 33342 efflux. HeLa (A), MDA-MB-435S (B), and A549 (C) cells were subjected to side population (SP) assay (90 minutes incubation with Hoechst 33342 at 37°C) in the presence or absence of TC-ET and its fractions: petroleum ether fraction (TC-PET), dichloromethane fraction (TC-DCM), and n-butyl alcohol fraction (TC-nBT); verapamil and fumitremorgin C (FTC) served as positive control for ABC transporter inhibitors, and dimethyl sulfoxide (DMSO) served as vehicle control. Error bars represent ±standard error of the mean; n = 3, ***P < .001.
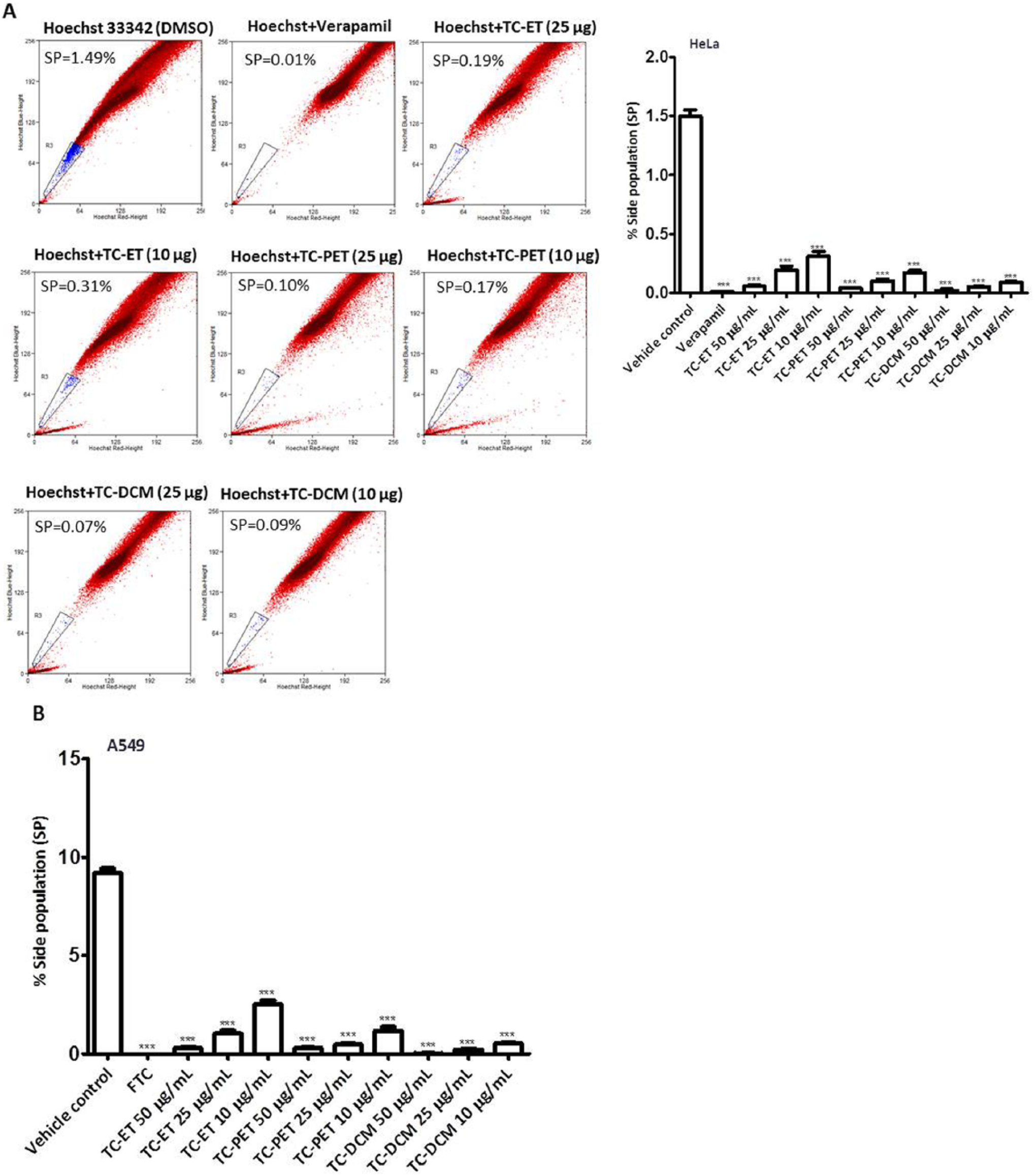
Dose-dependent inhibitory effects of fractions of Tinospora cordifolia ethanolic extract (TC-ET) on side population (SP). HeLa (A) and A549 (B) cells were subjected to SP assay (90 minutes incubation with Hoechst 33342 at 37°C) in the presence of varying concentrations (10, 25, and 50 µg/mL) of TC ethanolic extract (TC-ET), TC petroleum ether fraction (TC-PET), and TC dichloromethane fraction (TC-DCM); verapamil and fumitremorgin C (FTC) served as positive control for ABC transporter inhibitors; dimethyl sulfoxide (DMSO) served as vehicle control. Error bars represent ±standard error of the mean; n = 3, ***P < .001.
Drug Efflux Inhibitory Activities of TC-ET Fractions
ABC-B1 and ABC-G2 are 2 key regulators of the SP phenotype and eventually contribute to MDR in various cancers. 20 Because TC-ET fractions led to the disappearance of SP in all the cell types studied, we further confirmed their ABC-B1- and ABC-G2-specific transporter inhibitory functions by flow cytometry–based drug efflux assay. Rho 123 and MXR (fluorescent substrates of ABC-B1 and ABC-G2, respectively) efflux assays have been effectively used as measures of ABC-B1 and ABC-G2 activities.31,32 Accordingly, we set up flow cytometry based Rho 123 and MXR efflux assays in the presence of TC-ET and its fractions. Verapamil and FTC were used as standard inhibitors of ABC-B1 and ABC-G2, respectively and served as positive controls; DMSO was used as vehicle control. Briefly, in HeLa cells, the MFI of Rho 123 in the efflux phase (with vehicle control) was 89.70. However, in the presence of verapamil, the MFI increased to 348.10, indicating the increased intracellular accumulation of Rho 123 owing to the inhibition of ABC-B1-mediated efflux. The addition of TC-ET, TC-PET, and TC-DCM also increased the MFI to 177.80, 215.40, and 217.90, respectively (Figure 7A), suggesting that TC fractions also enabled increased intracellular accumulation of Rho 123, further suggesting that they may have a role in inhibiting ABC-B1. Compared with verapamil, the TC-ET, TC-PET, and TC-DCM fractions showed 51.10%, 61.90%, and 62.60% inhibition of ABC-B1 activity, respectively.
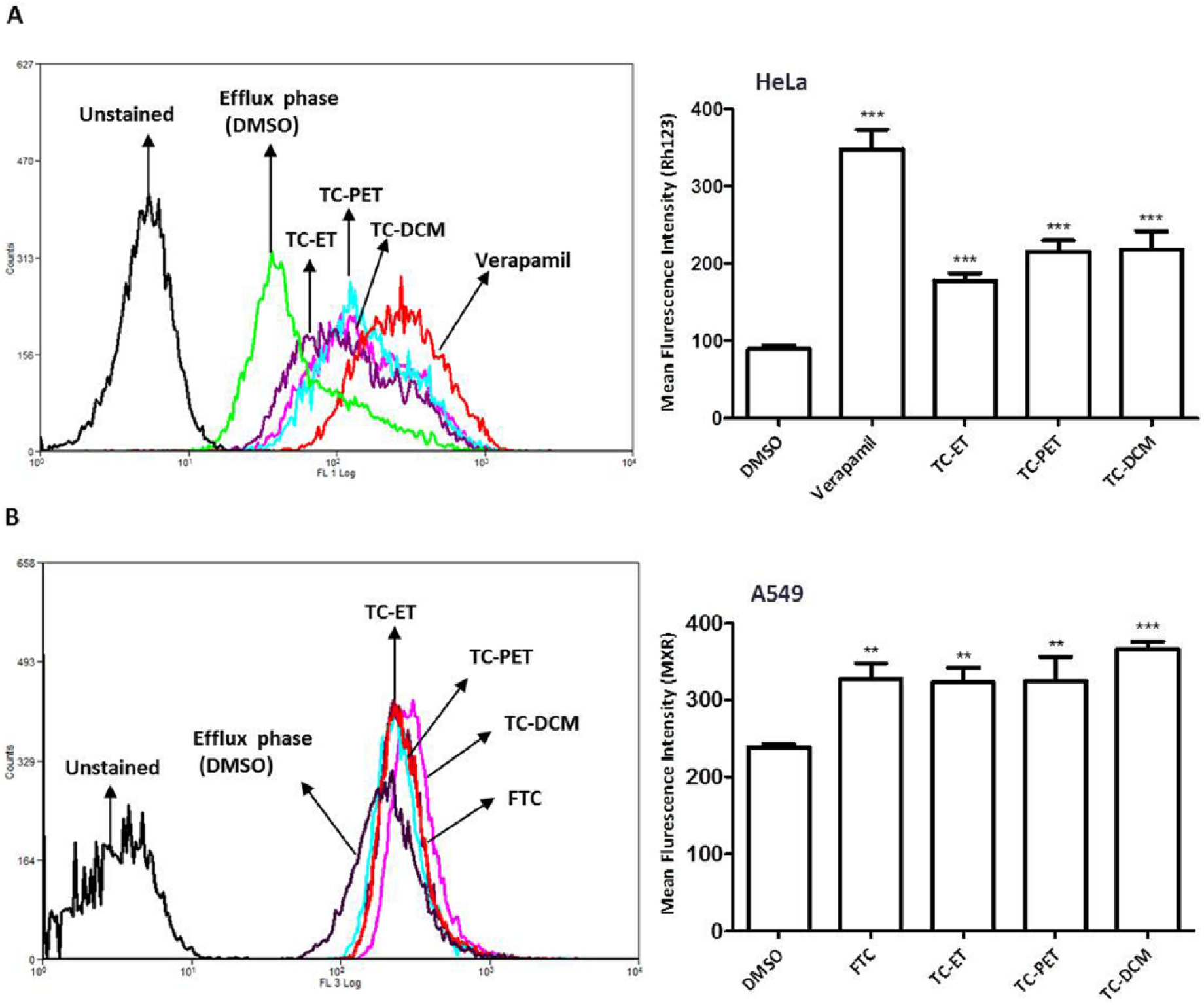
Effect of fractions of Tinospora cordifolia ethanolic extract (TC-ET) on ABC-B1 and ABC-G2 activities. (A) Rhodamine (Rho) 123 efflux assay for the measurement of ABC-B1 activity. HeLa cells were incubated with Rho 123 (0.5 µg/mL) containing media for 30 minutes at 37°C (accumulation phase), after which excess dye was washed and cells were again incubated in media for 60 minutes at 37°C (efflux phase) in the presence or absence of verapamil (50 µM), TC ethanolic extract (TC-ET), TC petroleum ether fraction (TC-PET), and TC dichloromethane fraction (TC-DCM); DMSO served as vehicle control. The mean cellular Rho123 fluorescence was analyzed by flow cytometry. (B) Mitoxantrone (MXR) efflux assay for the measurement of ABC-G2 activity. A549 cells were incubated with MXR (10 µM) containing media for 30 minutes at 37°C (accumulation phase); excess dye was washed off, and they were then allowed to incubate in media for 60 minutes at 37°C (efflux phase) in the presence or absence of fumitremorgin C (FTC; 10 µM), TC-ET, TC-PET, and TC-DCM; DMSO served as vehicle control. The mean cellular MXR fluorescence was analyzed by flow cytometry. Error bars represent ±standard error of the mean; n = 3, ***P < .001, **P < .01.
Similarly, in A549 cells, the MFI of MXR in the efflux phase (with vehicle control) was 238.90. However, in the presence of FTC, the MFI increased to 328.50, indicating an enhanced intracellular accumulation of MXR because of the inhibition of ABC-G2-mediated efflux. Similar to FTC, the addition of TC-ET, TC-PET, and TC-DCM also enhanced the MFI to 322.90, 324.50, and 366.20, respectively (Figure 7 B), suggesting that similar to FTC, they may also function to inhibit ABC-G2 (Figure 7B). Compared with FTC, the TC-ET, TC-PET, and TC-DCM fractions showed 98.29%, 98.78%, and 111.47% inhibition of ABC-G2 activity, respectively. Thus, these data suggested that the ETs and fractions of TC may possess ABC-B1 (MDR1) and ABC-G2 (BCRP) transporter inhibitory activities.
Sensitization of Cancer Cells Toward Chemotherapeutic Drugs by TC-ET and Its Fractions
Because treatment with TC-ET and its fractions led to an accumulation of Rho 123 and MXR in the efflux assays, we assessed whether TC-ET and its fractions could also sensitize cancer cells to chemotherapeutic drugs. To address this, we first determined the dose-response cell viabilities of TC-ET, TC-PET, and TC-DCM in HeLa and A549 cells using MTT-based cytotoxicity assay. The TC-ET, TC-PET, and TC-DCM showed dose-dependent reduction in the percentage cell viability (Figure S2). The IC50 values of TC-ET, TC-PET, and TC-DCM in HeLa were found to be 145.70, 124.30, and 104.70 µg/mL, respectively (Figure S2). Similarly, IC50 values of TC-ET, TC-PET, and TC-DCM in A549 were 138.50, 132.40, and 126.00 µg/mL, respectively (Figure S2).
To gauge if TC-ET and its fractions sensitized cancer cells to chemotherapeutic agents, HeLa and A549 cells were treated with 50 µg/mL TC-ET, TC-PET, and TC-DCM (less than their IC50 values) in the presence or absence of DOX (1.00 µM) or MXR (0.50 µM) for 48 hours, and percentage cell viability was measured using the MTT assay. Treatment with verapamil (in HeLa) and FTC (in A549) served as positive controls, whereas DMSO served as vehicle control. Data revealed that as expected,6,33 combinatorial treatment of verapamil/FTC with DOX/MXR significantly decreased percentage cell viability as compared with DOX/MXR alone (Figures 8A and 8B). Similar to this, combinatorial treatments of TC-ET, TC-PET, or TC-DCM with DOX/MXR also reduced percentage cell viability. Thus, these data indicated that TC-ET and its fractions TC-PET and TC-DCM sensitized HeLa and A549 cells to standard chemotherapeutic drugs.
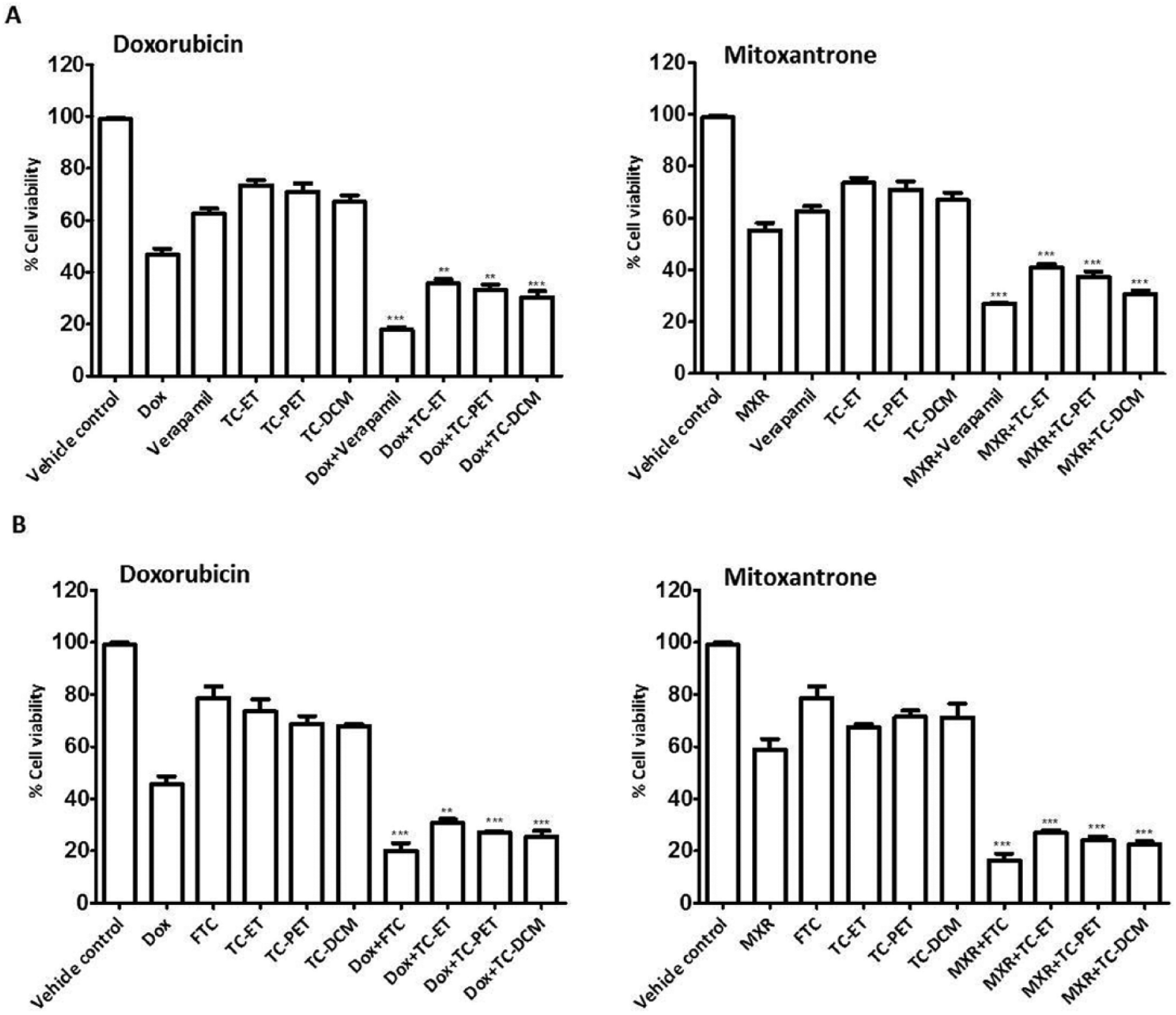
Effects of TC-ET and its fractions on chemotherapeutic drug-induced cell death: HeLa (A) and A549 (B) cells were treated with 50 µg/mL of TC ethanolic extract (TC-ET), TC petroleum ether fraction (TC-PET), and TC dichloromethane fraction (TC-DCM) in the presence or absence of doxorubicin (Dox, 1.00 µM) or mitoxantrone (MXR, 0.50 µM) for 48 hours; verapamil and fumitremorgin C (FTC) served as positive control, and dimethyl sulfoxide (DMSO) served as vehicle control. At the end of the treatment period, the percentage of cell viability was determined by MTT assay. Error bars represent ±standard error of the mean; n = 3, ***P < .001, **P < .01 compared with Dox/MXR alone.
Discussion
The ABC family of transporters is normally expressed by several cell types and serves to protect them from cytotoxins and toxic chemicals. More recently, several types of stem cells have been shown to have elevated levels of ABC transporter expression, which perhaps serves to protect them from cellular toxins throughout their long lifespan. 7 Cancer cells seem to exploit the body’s own natural defense system to evade chemotherapeutic drugs. Indeed, ABC transporters have been associated with cancer drug resistance for the past 20 years. 20 Several ABC transporter inhibitors have been identified to target ABC-B1 (MDR1), ABC-G2 (BCRP), and ABC-C1 (MRP-1), the 3 main transporters shown to be associated with MDR, but their clinical effectiveness in combating cancer drug resistance remains unsatisfactory. Some possible reasons for this could be their side effects and cytotoxicity or specific targeting of one type of receptor, whereas the resistant cells express another class. Furthermore, most studies have focused on inhibiting a single ABC transporter, but our recent study on the expression of ABC transporters revealed that cancer cells express a wide variety of these transporters 34 and hence one may have to target several of these simultaneously. The recent identification of the side population (SP) phenotype and the observation that increased expression of ABC transporters may contribute to the innate drug resistance of cancer stem-like cells has rekindled the interest in identifying inhibitors of these transporters. Indeed, SP cells from a variety of cancer cell lines and cells derived from primary tumors (leukemia, neuroblastoma cells, and gastrointestinal tumors) have increased expression of ABC-B1 and ABC-G2 in comparison to non-SP cells8,19 revealing the MDR characteristics of SP. In this study, we have used an SP assay–based bioactivity-guided fractionation to screen anticancer medicinal plants for their SP inhibitory activity. We report here the identification of medicinal plant extracts and fractions that deplete the SP in multiple epithelial cancer cell lines, at least in partly because of their ABC transporter inhibitory activity.
Our data confirmed the existence of SP in extensively cultured cancer cell lines,11,19 revealing the existence of 1.43%, 9.08%, and 0.31% of SP in HeLa, A549, and MDA-MB 435S cells, respectively. Although some articles have reported the detection of SP in T47D and MDA-MB- 231 cells,35,36 we failed to detect SP in these cell types. This may be a result of differences in the source of cells, the SP protocol followed, or flow cytometer configuration. Similar discrepancies related to detection of SP have been documented in the literature previously. For example, a few reports have shown the absence of SP in T47D and MDA-MB-231 cells.19,37 When SP assay was performed in the presence of ABC transporter inhibitors, the SP in HeLa was inhibited by verapamil (ABC-B1 inhibitor), whereas SP in both A549 and MDA-MB-435S cells was inhibited by FTC (ABC-G2 inhibitor). Because verapamil is an ABC-B1 (MDR1) inhibitor, whereas FTC is an ABC-G2 (BCRP) inhibitor, our data suggest that SP in HeLa may be mainly mediated by ABC-B1 (MDR1), whereas SP in A549 and MDA-MB-435S cells may be predominantly mediated by ABC-G2 (BCRP) transporters. These results corroborated the results in existing literature that has documented ABC-B1 and ABC-G2 as the 2 major mediators of the SP phenotype.8,19,20 The SP cells, by virtue of their increased MDR transporter expression, which enables them to pump out lipophilic agents, including anticancer drugs, 20 are highly drug resistant. Indeed, it has been demonstrated that treatment of cancer cells with chemotherapeutic drugs (such as MXR, temozolamide, and DOX) leads to an increase in the SP.3,8,19,21,22 We confirmed this by testing the effect of DOX, an anthracycline antibiotic and a routinely used chemotherapeutic drug for treatment of various types of epithelial cancers. Consistent with the literature,3,19,22,30 our data confirmed an enrichment of SP after treatment with DOX in human epithelial cancer cells. The enrichment of SP compartment in the presence of DOX treatment also suggested that DOX might be excluded by the SP cells by the same mechanism as the rapid efflux of Hoechst 33342. This increased drug efflux capacity of SP cells and enrichment of SP on treatment with DOX might influence the outcome of current chemotherapy for the treatment of cancer.
Medicinal plant-derived natural products have been shown to be effective in depleting cancer stem-like cells. For example, curcumin, a polyphenol present in turmeric, a spice widely consumed in India, depletes SP in the rat C6 glioma cell line 38 by inhibiting the activity of ABC-G2 and facilitating the accumulation of DOX, thereby rendering the cells sensitive to the chemotherapeutic drugs. 39 Berberine (a plant-derived alkaloid) and emodin (plant-derived resin) eliminate the SP and reduce ABC-G2 transporter expression in MCF-7 breast cancer cells and gallbladder carcinoma.40,41 The Chinese herbal mixture Tien-Hsien Liquid eliminates SP cells isolated from human hepatoma cells. 42 Furthermore, phytochemicals, such as parthenolide, piperine, sulforaphane, cyclopamine, and resveratrol, were effective against cancer stem-like cells. 23 Because SP cells harbor increased expression of ABC transporters and cancer stem-like properties, we wanted to identify and characterize the ETs of WS and TC that reduce the SP phenotype, in contrast to DOX that enriches it. Interestingly, we found that TC-ET, but not WS-ET, significantly depleted SP in cancer cells. TC-ET depleted SP at a concentration of 50 µg/mL, which indicates a potent anti-SP activity at the extract level. Because TC-ET depleted SP, it was further fractionated using solvents of increasing order of the polarity index to obtain TC-PET, TC-DCM, and TC-nBT and tested against cancer SP. We found that SP was significantly inhibited after treatment with TC-PET and TC-DCM. In contrast, treatment with TC-nBT led to an increase in SP. TC-nBT is a relatively polar fraction and may harbor compounds similar in action to DOX, which also leads to increase in SP. The dose-response studies revealed that TC-ET, TC-PET, and TC-DCM possessed dose-dependent inhibitory effects on SP at lower concentrations (10 µg/mL), revealing potent SP inhibitory effects. TC fractions (TC-PET and TC-DCM) were found to be relatively more potent than TC-ET at lower doses, indicating that fractionation may have enhanced the bioactivity of TC-ET fractions. The anti-SP activity of the TC-PET and TC-DCM fractions may be a result of the elimination of cancer stem-like cells; inhibition of MDR transporters, which mediate SP phenotype; or inhibition of specific pathways such as the Notch, Wnt, and Sonic-Hedgehog that are associated with the self-renewal of cancer stem-like cells. Our preliminary data reveal inhibition of MDR transporters in the presence of these fractions.
Overcoming drug resistance by inhibiting the ABC transporters is one of the key strategies to avoid drug resistance. 6 Natural products show great promise as anti-MDR agents. Several plant-derived natural products, such as curcumin, epigallocatechingallate, quercetin, silymarin, resveratrol, naringenin, and hesperetin, have been reported to have inhibitory functions against ABC-B1 and ABC-G2. 23 Considering the strong link between cancer stem-like cells and MDR, the phytochemicals that are effective against SP may also have the ability to inhibit ABC transporters (or MDR reversal agents). Hence, we designed experiments to evaluate the ABC transporter (ABC-B1 and ABC-G2) inhibitory functions of the SP-active TC-ET and its fractions. In this direction, functional assays for the direct inhibition of ABC-B1 and ABC-G2 were carried out using flow cytometry–based drug efflux assays. Because SP in HeLa was sensitive to verapamil (inhibitor of ABC-B1) and SP in A549 was sensitive to FTC (inhibitor of ABC-G2), we investigated the ABC-B1 and ABC-G2 inhibitory functions of SP-active TC-ET and its fractions in HeLa and A549 cells. The potency of TC-ET and its fractions to inhibit ABC-B1 and ABC-G2 activity was studied using Rhodamine 123 (Rho) efflux assay and MXR efflux assay, respectively.31,32 Fluorescent substrates (Rho 123 for ABC-B1 and MXR for ABC-G2) and inhibitors of ABC transporters (verapamil for ABC-B1 and FTC for ABC-G2) were used to quantitatively assess ABC transporter activity. When a fluorescence substrate diffuses into the cell, ABC-B1 or ABC-G2 actively pumps out the fluorochrome (efflux phase). If an inhibitor of ABC-B1 or ABC-G2 is present at the same time along with the fluorescence substrate, the fluorescence marker accumulates in the cell, thus resulting in a higher intensity of fluorescence. Therefore, increased MFI in the presence of a specific fluorescent substrate and inhibitor pair for a specific ABC transporter measures the activity of that particular ABC transporter. Our data revealed that TC-ET, TC-PET, and TC-DCM increased the intracellular accumulation of Rho 123 in HeLa cells and MXR in A549 cells, indicating the inhibition of ABC-B1 and ABC-G2 mediated drug transport, respectively. Interestingly, the TC extracts/fractions were almost as potent as verapamil in completely diminishing the SP in HeLa cells. However, the TC extracts/fractions were markedly less effective than verapamil in the Rho 123 efflux assay. One possible explanation for this discrepancy could be that in the SP assay, only a rare population of cells (~1.5%) contributes to the SP phenotype, suggesting that the SP phenotype in HeLa cells may be contributed by a subpopulation of ABC-B1-expressing cells, which might additionally have other properties (such as expression of other ABC transporters or additional stemness properties). In contrast, all cells expressing ABC-B1 are likely to contribute in the Rho 123 assay, which is a specific assay for the function/expression of ABC-B1 transporter alone. 31 Furthermore, it is possible that the TC fractions are less efficient in inhibiting ABC-B1 transporter but contain a mix of chemicals that inhibit multiple ABC transporters or other stemness properties that contribute to the SP phenotype. Indeed, this is also supported by the differential shifts in the main population of HeLa in the SP assay in the presence of verapamil versus the TC fractions.
The inhibition of MDR transporters might be most beneficial when combined with an anticancer agent for the effective treatment of drug-resistant cancer. In fact, clinical studies have attempted to overcome drug resistance through combination therapies in which a cytotoxic drug was given along with ABC-transporter inhibitors. However, the results with various known MDR modulators, including verapamil and FTC, were largely negative. 20 We show that combinatorial treatments of fractions of TC-ET with chemotherapeutic agents (DOX and MXR) resulted in the sensitization of cancer cells to DOX/MXR-induced cell death, thus indicating that fractions of TC-ET may enhance cancer therapeutics. This may also be a result of modifying the activity of key cell proliferation and survival pathways, such as those controlled by Akt, nuclear factor-κB, and cyclooxygenase-2. 33
To conclude, in this study, we investigated the effects of indigenous medicinal plants with known anticancer properties—WS and TC—on the SP and ABC transporter functions in human epithelial cancer cells. Interestingly, we found that TC-ET and its fractions, TC-PET and TC-DCM, significantly depleted SP, indicating that they may target cancer stem-like cells. Moreover, these TC fractions inhibited ABC-B1 (MDR1) and ABC-G2 (BCRP) mediated drug transport in human epithelial cancer cells and sensitized cancer cells to chemotherapeutic agents, revealing their potential as MDR reversing agents. The bioactive TC fractions, TC-PET and TC-DCM, may contain diverse classes of phytochemicals that are responsible for this biological activity. To identify bioactive molecules, a bioactivity-guided fractionation strategy (based on anti-SP activity) using chromatographic methods can be used. Thus, the isolation of bioactive phytochemicals from TC-PET and TC-DCM fractions and their physiochemical characterization may result in the development of novel molecules that can be useful in the targeting of cancer stem-like cells and in overcoming drug resistance, leading to a more effective treatment of cancer.
Footnotes
Acknowledgements
Support from Indian Institute of Science (IISc) and Department of Science and Technology-FIST programme, Government of India, to the Department of MRDG is acknowledged. We thank D. Alaguraj and Dr Williams R. Surin (Central FACS Facility, IISc) and Dr Paresh Jain (Scientific Advisor, BD Biosciences, India) for help with flow cytometry. AR is a Wellcome-DBT India Alliance Senior Research Fellow; SAB is a recipient of the Prime Minister’s Fellowship, Government of India.
Authors’ Note
Asmy Appadath Beeran and Sai A. Balaji contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was mainly funded by a grant from the Department of Biotechnology, Ministry of Science and Technology, Government of India (BT/PR14135/PBD/17/695/2010 to NU and AR). Support from Indian Institute of Science, and Department of Science and Technology-FIST programme, Government of India, to the Department of MRDG is acknowledged.
