Abstract
Objective. The use of complementary or alternative medicine has increased greatly over the last decade. This study describes a cross-sectional survey of women with breast cancer to describe their use of herbs and supplements that might have placed them at elevated risk for bleeding at the time of their primary treatment surgery for breast cancer. Methods. We present cross-sectional survey results from a cohort of 316 women with breast cancer. The participants included a convenience sample of 98 women who received integrative oncology treatment from local providers and a larger group of women recruited from the local cancer registry who were matched on their similarity to the integrative oncology patients’ demographic characteristics and stage of cancer at time of diagnosis. Results. Almost 16% of women with breast cancer report using one or more herbs or supplements thought to potentially increase their risk for adverse bleeding-related outcomes at the time of their primary surgical treatment. This does not include the 22% who used fish and flaxseed oils, which were at one time thought to increase risk for bleeding but for which there is now evidence to suggest that they are safe. Conclusion. Further research is needed to better understand the risks associated with use of a variety of herbs and supplements among women approaching surgery.
Introduction
The use of complementary or alternative medicine (CAM) has increased substantially over the past decade.1,2 While most CAM treatments, particularly mind–body and energetic-therapy forms of CAM, are presumably safe (although of unknown efficacy), there are several ingestible CAM substances, including herbal remedies, teas, and supplements, which are considered contraindicated for use by patients undergoing surgery, because of their potential to increase the risk of adverse surgical outcomes.3-5 Substances of concern include herbs and vitamins with antiplatelet activity that have the potential to prolong bleeding time, including garlic (Allium sativum), gingko (Gingko biloba), cranberry (Vaccinium macrocarpon), vitamin E, ginger (Zingiber officinale), and ginseng (Panax ginseng).6-10 Although most of the evidence suggesting a need for caution in the use of these comes from in vitro studies, the case for these herbs’ having significant anti-platelet activity is fairly strong. There have been case reports of bleeding adverse events associated with use of ginkgo11,12 and with garlic.6,7 Case reports have suggested that ginger and cranberry can dramatically increase bleeding problems in association with the use of warfarin. In the case of cranberry the mechanism for this interaction is incompletely understood. 13 However, there has been no systematic study of the effects of these substances on bleeding in vivo. These concerns, and concerns about the possible risks associated with fish and flaxseed oil use have been described in depth in several review articles.3-5 Review articles discussing the concerns associated with these substances generally advise that for safety reasons, these CAM substances be avoided for a period of 1 to 2 weeks prior to the surgery, after which time they can be resumed. In the case of fish and flaxseed oils, research has since been pursued to examine the effects on surgical outcomes of supplement use and those studies have found that in spite of the evidence for fish and flaxseed oils ability to decrease platelet aggregation, they neither increase bleeding complications in cardiac patients taking aspirin and clopidogrel, 14 nor do they increase adverse surgical outcomes during coronary artery bypass graft surgery 15 or spinal decompression surgery, 16 suggesting that they are safe for use by cardiac patients and by extension likely safe for cancer patients undergoing surgery. We could find no similar studies examining the effects of any of the other supplements on patients undergoing cancer-related surgeries.
The presence of in vitro evidence suggesting a need for concern and an absence of evidence that use of these CAM supplements are associated with significantly elevated risks for adverse outcomes in most patients puts physician in a quandary. There are significant barriers for physicians associated with compliance with conservative guidelines promoting the discontinuation of these CAM supplements by breast cancer patients. These include the need to take time from other important issues to be discussed at a critical point in treatment, and the potential for unfortunate effects on the patient–physician relationship. This study sought to describe how frequently women with breast cancer use the CAM substances of concern during the week immediately prior to their initial surgery for their cancer in order to better understand the extent of the problem.
Methods
Study methods and questionnaires were approved by the institutional review boards of the Fred Hutchinson Cancer Research Center and Bastyr University in Kenmore, Washington. The authors have no conflicts of interest to declare regarding the work described. This article reports on 316 women with breast cancer who responded to items about supplement use at the time of their initial surgery as part of a questionnaire completed during their enrolment in a larger longitudinal study of complementary and alternative medicine use by breast cancer patients. The participants included a convenience sample of 98 women who received integrative oncology (IO) treatment from local providers as a supplement to their conventional care and a larger group of women recruited from the local cancer registry that were matched on their similarity to the IO patients demographic characteristics and stage of cancer at time of diagnosis. Breast cancer patient/survivors were eligible for this analysis and the larger longitudinal study if they spoke English fluently enough to complete surveys, were older than 21 years, and were diagnosed with breast cancer less than 2 years prior to their visit to the IO office, or were selected from the registry based on their similarity to an enrolled IO clinic patient. Most of the IO clinic women in this sample were recruited at a single clinic and were approached in person during either their first or in some cases second appointment with their IO provider. These women were given a packet containing: the informed consent form, a medical records release, and the enrolment questionnaire. In most cases, they filled out all study documents and informed consent was administered at the clinic appointment. In a few cases, they were allowed to take the packet home with them to review and complete the forms. In these cases, informed consent was administered during a follow-up telephone call and the recruitment documents were returned in the self-addressed stamped envelope provided.
Women identified through the cancer registry were approached by mail first by the registry to determine their interest in research participation. Three weeks following the initial approach, women who did not decline research participation, were sent a packet with the forms given the women recruited via the IO clinics—a cover letter, 2 copies of the informed consent, 2 copies of the medical records release form, the enrollment questionnaire, and a stamped return envelope. Approximately 3 weeks following the mailing, reminder calls were made to women who did not respond. Of 754 women approached to participate, 4 were recently deceased, 64 (8%) could not be reached at the address recorded with the registry, and 284 (41.4% of those initially approached) returned the forms, including the completed questionnaire.
Overview of Study Measures
The study questionnaire included sections assessing patient demographics (ie, age, education, income, and race/ethnicity), health status, breast cancer diagnosis, conventional treatment, and CAM use both before diagnosis and at the time of primary surgery as well as items assessing if and with whom patients consulted about their use of various CAM supplements at the time of their surgery.
Assessment of Supplement Use
One section of the questionnaire specifically assessed the use of herbs and supplements of specific concern around the time of surgery, and asked women explicitly about the week prior to surgery for the treatment of their breast cancer. This section assessed women’s use of garlic, ginkgo, fish oil, flaxseed oil, cranberry, vitamin E, and ginseng. Women were asked about their use of each substance individually by name, and the substances were not labeled as CAM or categorized in any other way. Where questions asked about supplements and substances that are also ingested as foods (eg, garlic, ginger, and cranberry) patients were asked to indicate that they had used a substance only if they used it in quantities greater than those associated with cooking, or if they had consumed it in concentrated, dried, encapsulated, or powdered forms. Other than this request that women tell us only about substances they used in quantities greater than that of common cooking, we did not assess doses for the CAM substances. Women responded to the question “During the week prior to any surgery for the treatment of your cancer did you use any of the following herbs, foods, or supplements? Please check the box that describes your use.” Then women were presented with a table that included the substances as rows and which asked, “Did you use the substance? With response options including “Yes,” “No,” and “Don’t know” for each row. In the remaining columns of the table women were asked if they had discussed their use of each of the CAM substances with a CAM provider (“Yes,” “No,” or “Don’t know”) or a conventional physician/provider involved in preparing you for surgery (“Yes,” “No,” or “Don’t know”).
In a separate question, women were asked about the use of these CAM substances prior to the breast cancer diagnosis and asked whether the herb or supplement was originally prescribed or recommended by a “Conventional Health Care Provider” and/or a “Complementary/Alternative Health Care Provider” and/or “Self / Friend”, and/or “Not sure/Don’t remember.” This was in a separate table with a check all that apply format. These questions have been used in a prior study.17,18
Results
Analyses examining the women recruited from both sources revealed no differences between the groups in age, stage, use of chemotherapy, frequency of supplement use, or in Health-Related Quality of Life. Based on these findings, the 2 samples were combined for all remaining study analyses.
Patient characteristics are shown in Table 1, of the 316 participating patient survivors, 167 (52.9%) were less than 2 years postdiagnosis, 148 (46.8%) were 2 to 5 years postdiagnosis, and 1 (0.3%) was more than 5 years postdiagnosis. Responding patients were distributed throughout the stage spectrum, with 38 (12%) reporting that their breast cancer was originally diagnosed at stage III or IV, and 24 (7.8%) of women reporting a diagnosis of stage 0, 98 (31.0%) stage I disease, and 116 (36.7%) stage II disease.
Patient Characteristics.
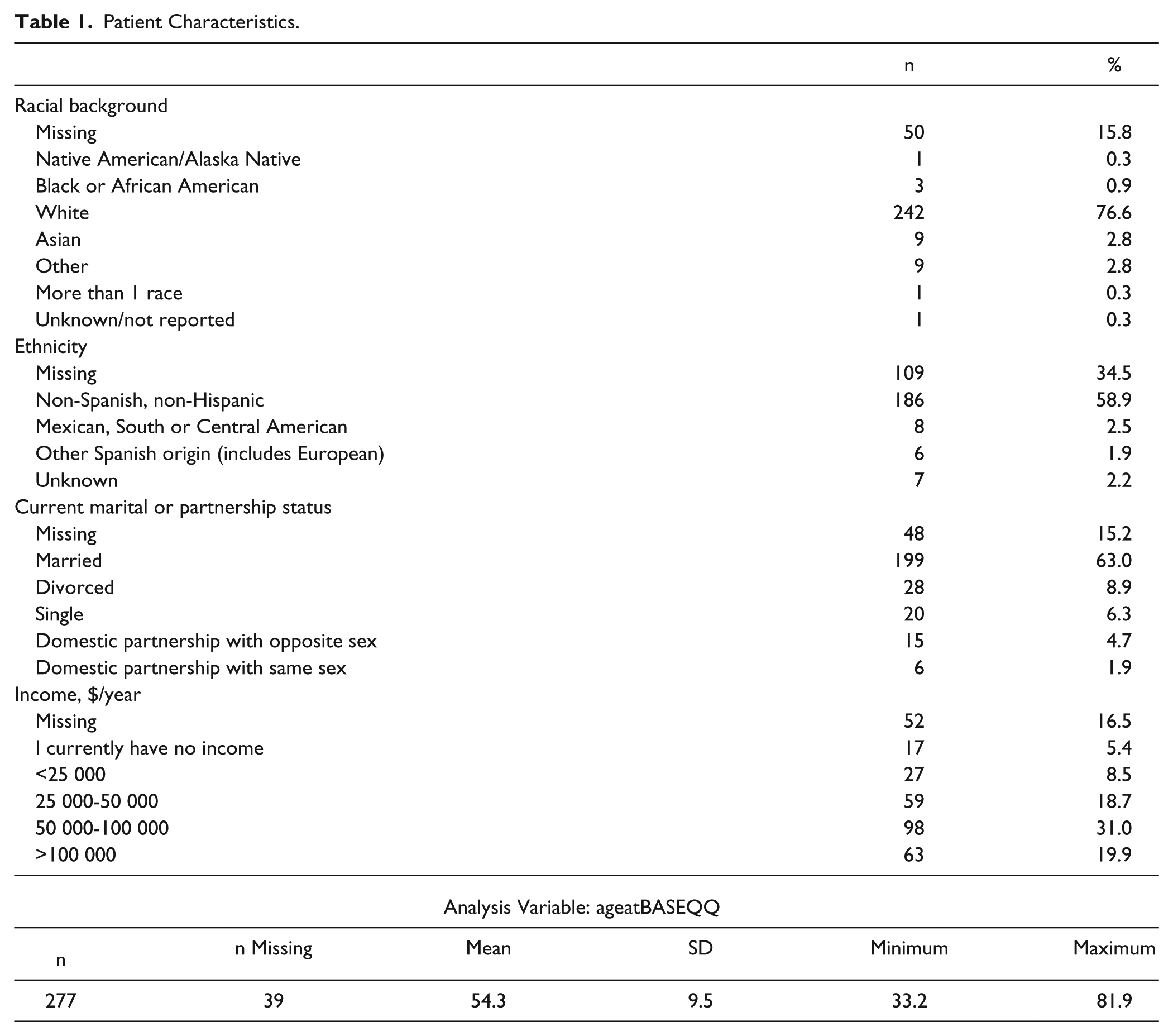
Table 2 summarizes the number of women who reported both an initial surgery for their cancer and who also reported using garlic, ginseng, cranberry, gingko, vitamin E, and fish or flaxseed oil, at the time of their initial surgery. In total, 29.4% (n = 93) of women reported taking one or more of these substances at the time of their surgery, 49 (15.5%) when fish and flaxseed oils were removed from consideration. Omega-3 supplements, including fish oil, used by 69 women (22% of all participants), were the most commonly used supplement, followed by flaxseed oil (n = 25; 7.9%). Among the supplements of more concern the most commonly used were vitamin E (n = 24; 7.6%), garlic (n = 17; 5.4%), and cranberry (n = 13; 4.1%). In total, 77 women (24.4%) of all participants reported having consulted with the conventional physician who was involved in preparing them for surgery about their use of any of the CAM substances. Some women reported consultation about a supplement that they simultaneously reported not using in the week prior to their surgery. This was most common with gingko, ginseng, and vitamin E where in all cases more women discussed the substance with a provider preparing them for surgery than in fact reported using the substance at the time of their surgery.
Use of Complementary and Alternative Medicine (CAM) Substances in the Week Prior to Surgery Among Women Who Reported Having Surgery to Treat Their Breast Cancer (n = 316).
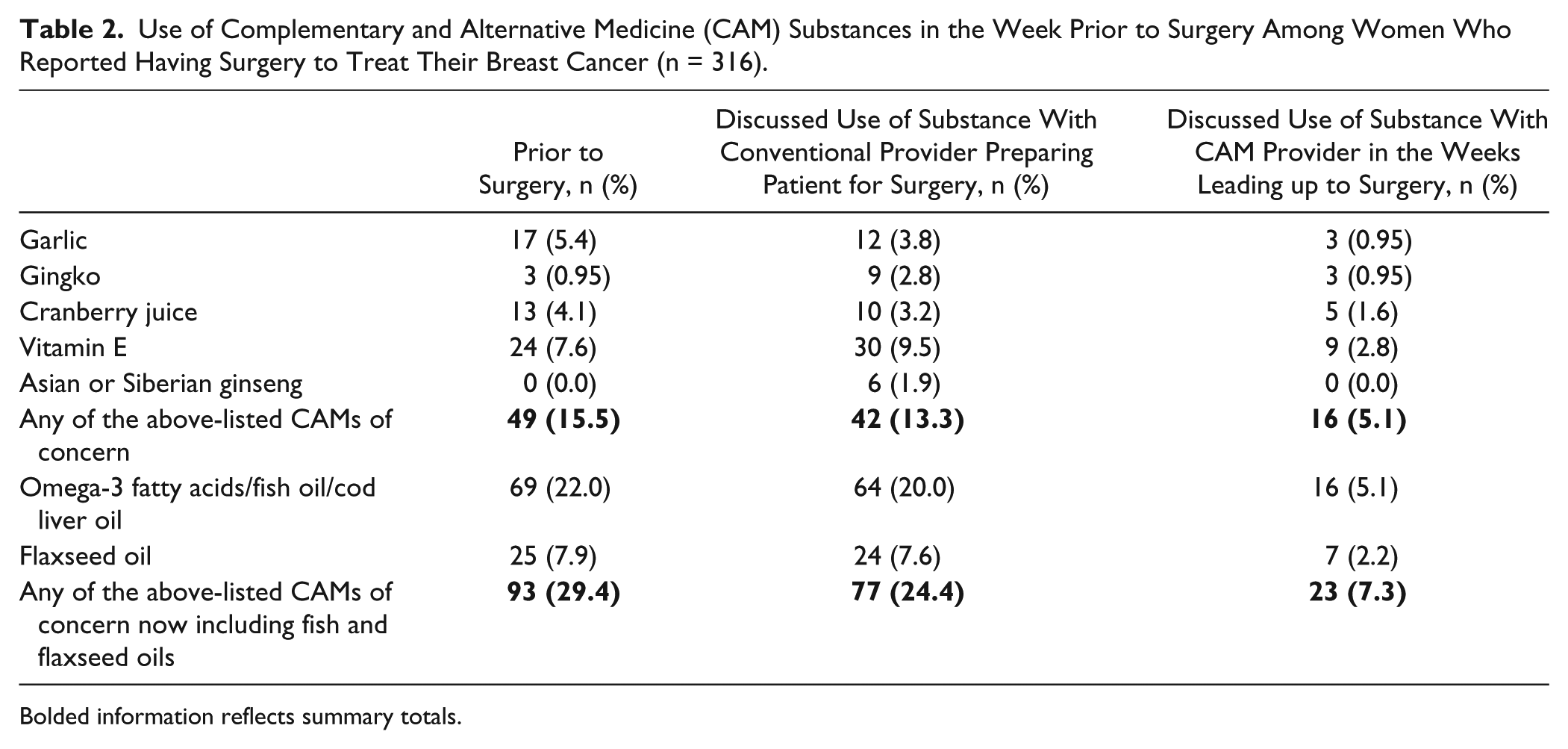
Bolded information reflects summary totals.
Consultation about fish and flaxseed oils also appears to be frequent, but these were also the most commonly used supplements. Twenty-three (7.3%) of the women reported consulting with a CAM provider about at least one substance though again some reported consultations in the weeks prior to surgery and not taking the substance about which they consulted at the time of their surgery. As the survey was designed to explicitly ask about consultations only of women who reported using a CAM substance at the time of their surgery, it is unclear if there were more women who consulted with physicians or CAM providers shortly prior to surgery and after consultations did not use substances of concern because of advice they received at the consultation.
We also examined women’s rates of use of these CAM substances of concern prior to their cancer diagnosis. Overall, 40.2% of our participants used 1 or more of our 7 substances of interest prior to diagnosis (Table 3). In all cases, more women had used the substance prior to their diagnosis than used it at the time of their surgery, but individual cross-tabulations examining use substance by substance revealed significant differences in patterns of use for different substances. For example, in the cases of garlic, cranberry, and vitamin E use, of the 33, 30, or 41 (respectively) users of these substances prior to diagnosis most (73%, 70%, and 73%, again respectively) did not use these substances at the time of their surgery having discontinued their use at least temporarily. However, in each case between one third (cranberry) and half (garlic and vitamin E) of the women using the substance at the time of their surgery did not report use prior to diagnosis (“new use”). In contrast, a smaller percentage (41%) of the women using fish or cod liver oils as omega-3 supplements, and (53%) of those using flaxseed oils prior to diagnosis had discontinued use of these oils at the time of their breast cancer surgery. Rates of new use of oils at the time of surgery (one third for fish oils and two fifths for flaxseed) were within the range seen for the other supplements. In contrast, approximately 88% of the users of gingko prior to diagnosis discontinued prior to surgery, but 66% of the users of gingko at the time of surgery were not users prior to diagnosis (note however that these estimates are based on a very small number of users of gingko in this study). We can only speculate that while most of the women taking CAM substances at the time of their surgery do so for reasons that predate their diagnosis with breast cancer, women’s use of specific substances varies considerably over time. We found uptake and discontinuation of supplements between our prior to diagnosis and primary surgery time points. It is unclear how physician recommendations influence these transitions, but it is clear that a substantial proportion of women are willing to have these conversations with their providers and that they can and do result in discontinuation of use of some supplements notably garlic and ginkgo prior to surgery.
Use of Complementary and Alternative Medicine (CAM) Substances of Concern Prior to Diagnosis (n = 215).
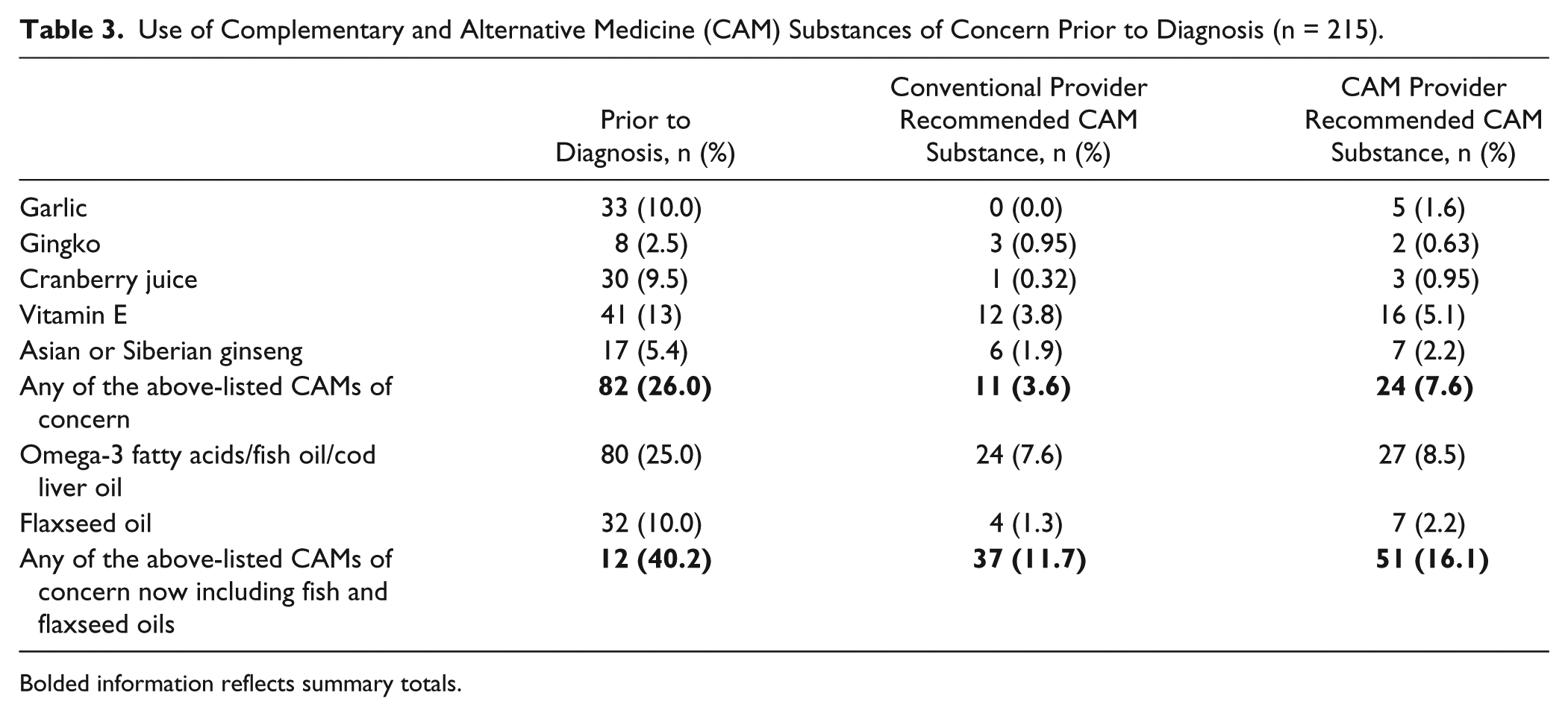
Bolded information reflects summary totals.
Discussion
Herbs and supplements are readily available without prescription in health food, drug, and grocery stores, and may be used by women in the general population, including those with breast cancer and those soon to be diagnosed with breast cancer without discussion with any form of medical provider. Some foods with biological activity beyond their nutritive value (garlic, ginger, and cranberry) are also widely available over the counter in concentrated or dried forms, often encapsulated or available as tablets. In this sample of breast cancer patient survivors in western Washington state, we found that 15.5% of women reported use of one or more of a short list of CAM substances that could be considered potentially contraindicated at the time of their initial surgical treatment.
In most cases, a majority of women who used a CAM substance of concern prior to diagnosis reported having discontinued use by the time of their surgery. It is unclear, however, how many discontinued use because of the surgery and how many did so at the time of their diagnosis or even before. It also appears that a considerable percentage of women report consulting with their conventional physician about their use of at least one of our CAM substances of concern prior to their surgery, and that at least a few appear to have discontinued use because of this consultation. How often this occurs is not clear from this analysis as numbers are small. It is also not clear that the women using our CAM substances of concern at the time of their surgery are suffering ill effects because of their use. Recommendations that women avoid the use of these herbs and supplements, like those that recommend against the use of fish and flaxseed oils, are based on a limited body of evidence, mostly in vitro evidence, while in vivo studies have generally not been done and could—like those done for fish and flaxseed oils—result in reduced concern about use of CAM supplements at time of surgery. There is a clear need for more research regarding the extent to which most of these CAM substances actually increase the rates of risk of adverse bleeding-related outcomes in doses commonly taken by patients. Unfortunately, we did not determine whether adverse outcomes associated with excessive bleeding occurred among those women using CAM substances at the time of their surgery in this study as the study was retrospective preventing us from using an objective assessment of bleeding at the time of surgery. A larger study where limitations associated with retrospective reporting of CAM use over long periods, and actual rates of bleeding-related adverse events are recorded, would be helpful.
When patients did report consulting with a provider about the CAM substances they used at the time of their surgery, the reported rates of consultation with conventional providers were generally as high as or higher than their rates of consultation with CAM providers. Because 26% of breast cancer patients in this study reported the use of potentially contraindicated substances prior to diagnosis and almost 16% reported use at the time of their surgery, there is clearly a need to better understand the risks associated with the use of herbs and supplements among women undergoing surgery.
A clear limitation of this study is the use of retrospective self-reports of supplement use. Although women appear to have little difficulty answering questions about their use of herbs, foods and supplements in the past and at the time of their surgery (few indicate “Don’t know” when given an option), there have been few studies of accuracy of such self-reports. In addition, while our findings are suggestive, a serious examination of the rates at which women discontinue use of CAM supplements either temporarily or permanently based on the advice of conventional or complementary providers with whom they consult at the time of their cancer surgery would require a larger study ideally of different design.
Footnotes
Authors’ Note
The data used for this report were collected as part of an ongoing National Center for Complementary and Alternative Medicine–funded research project. The data will be retained as a registry by the researchers for at least 5 years after the completion of the original research project. Access to the registry data can be arranged for researchers with institutional review board–approved research plans, on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Center for Complementary and Alternative Medicine of the National Institutes of Health under Award Number R01AT005873.
