Abstract
Aims. Relapsed high-grade gliomas (HGGs) have poor prognoses and there is no standard treatment. HGGs have ischemia/hypoxia associated and, as such, drugs and oxygen have low access, with increased resistance to chemotherapy and radiotherapy. Tumor hypoxia modification can improve outcomes and overall survival in some patients with these tumors. In previous works, we have described that cervical spinal cord stimulation can modify tumor microenvironment in HGG by increasing tumor blood flow, oxygenation, and metabolism. The aim of this current, preliminary, nonrandomized, study was to assess the clinical effect of spinal cord stimulation during brain reirradiation and chemotherapy deployed for the treatment of recurrent HGG; the hypothesis being that an improvement in oxygenated blood supply would facilitate enhanced delivery of the scheduled therapy. Materials and methods. Seven patients had spinal cord stimulation applied during the scheduled reirradiation and chemotherapy for the treatment of recurrent HGG (6 anaplastic gliomas and 1 glioblastoma). Median dose of previous irradiation was 60 Gy (range = 56-72 Gy) and median dose of reirradiation was 46 Gy (range = 40-46 Gy). Primary end point of the study was overall survival (OS) following confirmation of HGG relapse. Results. From the time of diagnosis of last tumor relapse before reirradiation, median OS was 39 months (95% CI = 0-93) for the overall study group: 39 months (95% CI = 9-69) for those with anaplastic gliomas and 16 months for the patient with glioblastoma. Posttreatment, doses of corticosteroids was significantly decreased (P = .026) and performance status significantly improved (P = .046). Conclusions. Spinal cord stimulation during reirradiation and chemotherapy is feasible and well tolerated. In our study, spinal cord stimulation was associated with clinical improvement and longer survival than previously reported in recurrent anaplastic gliomas. Spinal cord stimulation as adjuvant during chemotherapy and reirradiation in relapsed HGGs merits further research.
Keywords
Introduction
Despite multimodal approaches in the treatment of high-grade gliomas (HGGs), almost all patients present tumor recurrence. Patients with relapsed HGG have very poor prognoses and, to date, there is no standard treatment. 1 The most frequently evaluated option is chemotherapy alone (usually temozolomide), but with limited response rate and survival. Surgery, with or without implants of carmustine (Gliadel), is performed only in highly selected patients, and complete resection is rarely possible. The risk of brain radiation necrosis is increased with a de novo radiotherapy course; the tolerance dose of the healthy brain tissue being the limiting factor in the reirradiation dose.2,3 Thus, most studies have been conducted with stereotactic reirradiation of small volumes, with/without chemotherapy. 1
High-grade gliomas are characterized by ischemia/hypoxia and, consequently, drugs and oxygen have low access and the result is an increased resistance to chemotherapy and radiotherapy. This adverse microenvironment is worse on relapse due to vascular alterations resulting from previous surgery and radiotherapy. Tumor hypoxia modification can improve outcomes and overall survival in some tumors. 4 However, studies in brain tumors have not been conclusive.
We have previously described that cervical spinal cord stimulation (SCS) can modify tumor microenvironment in HGG by increasing tumor blood flow, oxygenation, and metabolism5,6 as well as by decreasing tumor hypoxia. 6 The current report shows our preliminary observations regarding the use of SCS during the deployment of chemotherapy, and reirradiation, for the treatment of recurrent HGG.
Methods and Materials
Patient Population
Between February 1999 and March 2008, 7 patients had SCS applied at our referral hospital in the course of scheduled reirradiation and chemotherapy for the treatment of recurrent HGG. Median age at the time of reirradiation was 38 years (range = 23-62 years). All patients had previously received external radiotherapy (median dose = 60 Gy; range = 56-72 Gy), with or without concurrent chemotherapy (Table 1).
Patients’ Characteristics.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; RT, radiotherapy; EQD2B, equivalent dose in 2-Gy fractions for normal brain; EQD2T, equivalent dose in 2-Gy fractions for brain tumor. OS, overall survival; PFS, progression-free survival; G, grade of tumor; HGG, high-grade glioma; PET, positron emission tomography; CT, chemotherapy; SCS, spinal cord stimulation.
First RT was planned by standard fractionation (2 Gy/fraction, 1 fraction/day) except in patients 3 and 4, who received hyperfractionation, that is, 1.2 Gy/fraction, 2 fractions/day.
Reirradiated at second or third tumor relapse.
Multifocal relapse.
Individual patient–relevant clinical comments: Patient 1: Initial parietal G-II astrocytoma, first surgery. First tumor relapse as G-II 9 years later; second surgery and first RT. Seconds relapse as multifocal HGG by PET. Patient 2: Initial frontoparietal G-I oligodendroglioma: biopsy and first RT. Tumor relapse as HGG, subtotal resection. Patient 3: Initial parietal G-III astrocytoma: surgery and first RT + CT + SCS. First tumor relapse 7 years later, second surgery with further lung thromboembolism. Second tumor relapse as multifocal HGG by PET. RT reached 60 Gy in previously nonirradiated tumor areas. Patient 4: Initial G-III oligodendroglioma: biopsy and first RT + CT + SCS. Tumor relapse as HGG by PET. Patient 5: Initial temporal G-II astrocytoma: first surgery and first RT. Tumor relapse as HGG by PET 21 months later, subtotal resection. Patient 6: Initial temporal G-II astrocytoma: first surgery. First tumor relapse as G-II 6 years later, second surgery. Second tumor relapse as G-III 16 months later, third surgery + Gliadel and first RT + CT + SCS. Third tumor relapse as HGG by PET. Patient 7: Initial occipital G-IV: first surgery and first RT + CT. Tumor relapse as G-IV, macroscopic resection + Gliadel.
The study of SCS to improve tumor ischemia/hypoxia was approved by the hospital’s ethics committee. Written informed consent was obtained from all patients. Before the first radiotherapy, all patients had histologically confirmed glioma: 1 glioblastoma multiform (GBM), 3 anaplastic gliomas (AGs), and 3 low-grade gliomas, which progressed to secondary AG at relapse. Tumor resection before reirradiation was performed in 3 patients: complete tumor resection in the patient with GBM, and subtotal tumor resection in 2 patients with previous low-grade glioma and progression to AG. In the remaining 4 patients, the diagnosis of HGG relapse was by 18F-fluorodeoxyglucose positron emission tomography (FDG-PET), 3 of whom were reirradiated at second or third tumor relapse. Further details are shown in Table 1.
Retreatment
Reirradiation was by linear accelerator in all patients except patient 1 who was treated with a Co60 unit in our old hospital facilities. The planning target volume (PTV) for recurrence consisted of the GTV with 1.5 to 1 cm margin for AG and 2 to 1.5 cm margin for the GBM.
The median time lapse between first irradiation conclusion and second irradiation commencement was 45 months (range = 25-109 months). A median physical reirradiation dose of 46 Gy (range = 40-46 Gy) was prescribed to the 100% isodose line, fractionation dose was 2 Gy/d. Median cumulative radiotherapy dose (initial + reirradiation) was 106 Gy (range = 96-117 Gy).
Linear quadratic model–based radiobiological calculations were performed to enable comparison of the reirradiation treatment regimen 7 , that is, the biological dose on normal brain tissue 8 as well as on brain tumor. 9 Since the time interval between fractions was generally 24 hours, and at least 6 hours for the hyperfractionation schedules, repair between radiation fractions was assumed to be completed. No correction was made for tumor cell repopulation. The EQD2 represents the total equivalent dose if applied in fractions of 2 Gy, according to the formula: EQD2 = D(d + α/β)/(2 + α/β) [Gy] with d = fraction size [Gy], n = number of fractions, D = d × n = total physical dose [Gy] and the α/β ratio [Gy]. 7 An α/β ratio of 2 Gy was selected for the late responding normal brain tissue 8 and 10 Gy for brain tumor. 9 The cumulative EQD2 is defined as the sum of the EQD2 of the initial irradiation course and the EQD2 of the reirradiation course (EQD2cumulative = EQD2initial + EQD2reirradiation). EQD2 values calculated for the group of patients are presented in Table 1.
Chemotherapy was with temozolomide (concurrent and postradiotherapy) in all patients except patient 1 who received lomustine over the first year until temozolomide became authorized for use in Europe. In the last 2 patients, bevacizumab was administered during adjuvant temozolomide.
Spinal Cord Stimulation
Spinal cord stimulation was performed using a Medtronic system (Medtronic Neurological, Minneapolis, MN) with a 1.27-mm diameter tetrapolar electrode percutaneously inserted on the posterior surface of the spinal cord at C2-C4, in the epidural space. The electrode was connected to an impulse generator device. The parameters of the stimulator were set at 1 to 3 V, a pulse width of 200 seconds, and a rate of 80 to 100 Hz.
Spinal cord stimulation was connected from 60 minutes pre- to 60 minutes postradiotherapy session. After reirradiation, the SCS was turned “on” during the days of adjuvant chemotherapy intake (while the patient was awake) and turned “off” at night or on the days without chemotherapy.
Before reirradiation, a basal metabolic assessment using FDG-PET study was performed in all patients. Additionally, in 5 patients, a second FDG-PET study was performed before reirradiation, and with the SCS device turned “on.”
Clinical Evaluation
Follow-up was planned every 4 months (or earlier if clinical worsening occurred) using computed tomography (CT) and FDG-PET, or single-photon emission computed tomography (SPECT). Selected magnetic resonance imaging (MRI) was also performed in 4 patients (in 3 of them when the SCS device had been withdrawn).
Primary end-point of the analysis was overall survival (OS), which was calculated from the diagnosis of last tumor relapse until death from any cause. Secondary end-points included doses of corticosteroids required, performance status on the ECOG (Eastern Cooperative Oncology Group) scale, and progression-free survival (PFS; calculated from reirradiation commencement until last imaging study without tumor progression, or onset of related clinical impairment; whichever occurred first).
Statistical Analysis
The SPSS (version 15.0) for Windows was used throughout. Comparisons between groups before/after reirradiation were with the paired t test (mean ± standard deviation) or Wilcoxon test (median and 25% to 75% quartiles). Comparisons between FDG-PET studies “without” versus “with” SCS were with the Wilcoxon test (median and 25% to 75% quartiles). Survival analyses were based on Kaplan–Meier estimates. Two-tailed P values <.05 were considered statistically significant.
Results
The maximum standardized uptake values (SUVmax) in FDG-PET studies were 6.5 (range = 3.7-7.9) pre-SCS and 7.7 (range = 4.7-9.8) post-SCS; P = .043.
Mean EQD2 values for the initial and reirradiation courses, as well as their cumulative doses, are presented in Table 1. The EQDinitial on the tumor amounted to 59.3 ± 5.8 Gy, which is the generally prescribed dose. The EQD2reirradiation on the tumor was 43.1 ± 2.9 Gy. The applied EQD2cumulative on the tumor was slightly larger than the normal brain tissue EQD2 cumulative: 102.4 ± 5.6 Gy versus 99.5 ± 3.5 Gy, respectively.
Reirradiation with chemotherapy + SCS was well tolerated. No neurological toxicities higher than CTC grade 2 occurred. Six patients needed corticosteroids before reirradiation. Following reirradiation, the corticosteroid dose requirement was significantly decreased in 5 patients (from 8.7 ± 2.4 to 2.6 ± 1.7 mg/d; P = .022). Performance status (ECOG scale) also showed a significant (P = .046) improvement from 1 (range = 1-2) to 1 (range = 0-2), pre- versus posttreatment. Patients were receiving temozolomide treatment for 11 ± 3 months (range = 4-24 months). Hematological toxicity was grade 4 in 1 patient, and grade 1 in 2 patients.
The median OS (mOS) following confirmation of HGG relapse was 39 months (95% CI = 0-93) for the overall study group: 39 months (95% CI = 9-69) for those with AG and 16 months for the patient with GBM (Figure 1).
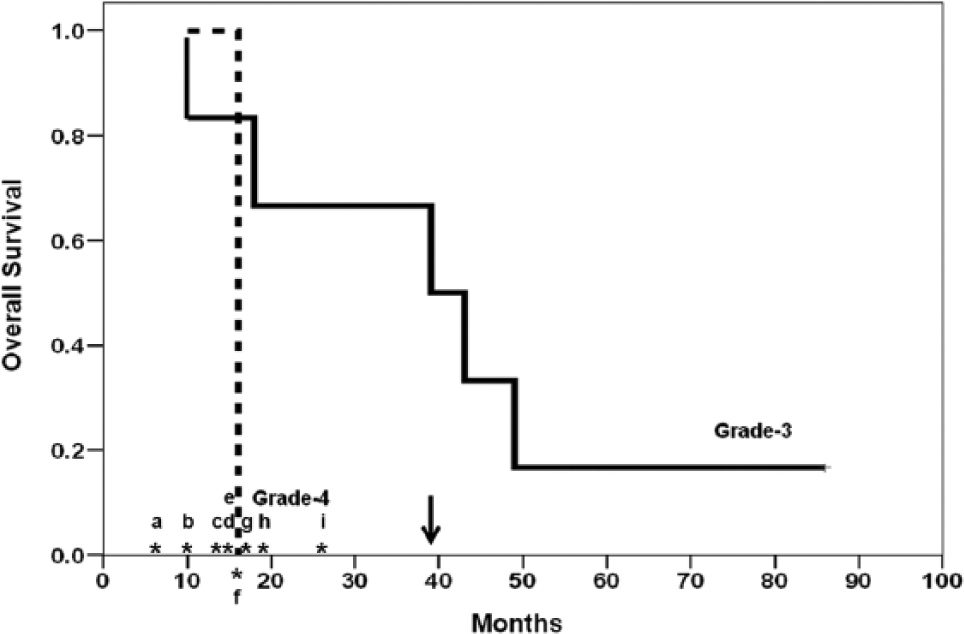
Overall survival following diagnosis of the last tumor relapse before initiating reirradiation course.
The median PFS (mPFS) after initiating reirradiation was 19 months (95% CI = 0-50 months) for the overall study group: 19 months (95% CI = 2-36) for those with AG and 5 months for the patient with GBM. Six-month PFS was 71% for the overall study group and 83% for those with AG.
Discussion
Overview of Previous Studies
Most studies of reirradiation or retreatment of AG have included <50 patients, and our mOS (measured from the time of the last relapse) compared favorably with the values reported by others. Treatment with temozolomide was for 14.6 months (28 anaplastic astrocytomas) to 18 months (24 anaplastic oligoastrocytomas). 10 Similar results were obtained with continuous, dose-intense, temozolomide; mOS of 14.6 months for 28 AG. 11 Other chemotherapies with/without targeted therapies showed comparable outcomes of mOS of 6 to 14 months. 1 Fractionated stereotactic radiotherapy (SRT) of 16 months (42 AGs) 12 ; hypofractionated SRT with/without chemotherapy or surgery of 10 months (42 AGs). 13 Hypofractionated SRT + bevacizumab was between 14 months (4 AGs) 14 and 16.5 months (5 AGs). 15 Stereotactic radiosurgery had mOS of 26 months with mPFS of 8.6 months for 49 AGs (albeit the mOS after the first diagnosis was only 37.5 months). 16
The study most similar to our own is that of Kohshi et al 17 using fractionated SRT combined with hyperbaric oxygen therapy to modify tumor hypoxia in 14 patients with recurrent anaplastic astrocytoma (mOS = 19 months). However, a relatively high incidence of radio-necrosis (28%) was observed, which was likely to be due to a radiosensitizing effect of oxygen on nervous tissue.
Spinal Cord Stimulation During Reirradiation and Chemotherapy
In our study, the OS of 16 months postrelapse in the single patient with glioblastoma is not representative, but the mOS of 39 months after diagnosis of AG relapse in 6 patients is more representative, and encouraging. It is of note that none of our AG patients had complete tumor resection; 50% were in second or third recurrence, and 33% presented as multifocal tumors. However, many of the aforementioned studies had included a high percentage of patients with previous complete tumor resection after relapse, and reirradiation using stereotactic radiosurgery, or hypofractionated SRT, applied to smaller tumor volumes.2,3
Using color Doppler measurements, transcranial Doppler, and SPECT studies, we had described that SCS can increase blood flow in common carotid arteries, middle cerebral arteries, and brain tumor tissue. 5 Using the polarographic probe technique to directly measure brain tissue pO2 we had also described that SCS can increase tumor oxygenation and decrease tumor hypoxia. 6 We propose that the encouraging results of the present study could be related to the modification of those parameters by SCS; an increase in tumor blood flow could improve chemotherapy delivery and tumor oxygenation and, hence, decrease tumor hypoxia. These effects might be even more relevant if reirradiation is administered at higher doses per fraction, that is, more sensitive to the hypoxia-mediated radioresistance, as occurs for stereotactic radiosurgery or hypofractionated SRT. 18 We had described that SCS can increase glucose metabolism around 35% to 40% in brain tumor and peritumor bed metabolism.6,19 In the current report in which patients who had received previous irradiation, SCS was also able to increase glucose uptake in HGG. We propose that this effect on tumor metabolism could recruit quiescent tumor cells toward phases of the cell cycle which are more sensitive to chemotherapy and radiotherapy, 20 as supported by the reported correlation between higher pretreatment metabolic rate of glucose and better response to temozolomide. 21 Further details regarding potential effects, and consequences, of SCS on brain tumors have been described in our previous works.5,6,19 A clinical trial using PET with 18F-fluoromisonidazol (FMISO-PET) in patients with HGG is ongoing to further evaluate the effects of SCS on tumor hypoxia (trial: NCT01868906).
Several mechanisms of action has been proposed in explaining the vascular effect of SCS on brain tissue, including a functional reversible sympathicolytic effect,22-24 a segmental liberation of vasoactive substances as substance P, vasoactive intestinal polypeptide, cacitonin gene–related peptide, 25 and the activation of vasomotor centers in the brainstem in a competitive manner between SCS and CO2 on the mechanisms of cerebral blood flow regulation. 26 Further details are described in an updated review by Visocchi et al. 27
Spinal cord stimulation was not associated with increased toxicity to the scheduled radiotherapy, 6 temozolomide, or bevacizumab. Indeed, there was a significant decrease in the patient’s corticosteroid requirement, and an increase in performance status. No symptoms of radionecrosis were observed in our group of patients probably because SCS secondarily improved cerebral blood flow and tissue oxygenation, but without increasing arterial pO2 above physiological levels,5,28 as happens with the use of hyperbaric chambers. These potential benefits are further supported by our previously reported beneficial effects of SCS in patients with hypometabolic brain areas secondary to radiation-induced brain injury 29 or in several experimental models and clinical observations on stroke, subarachnoid hemorrhage and cerebral vasospasm.27,30 Good tolerance could be related to the interval between first and second irradiation in our study (median 45 months), which was higher than usually described. However, in 2 overviews from reirradiation studies, no effect was noticed with respect to the time interval between the initial and reirradiation exposure.2,3
The main limitations of our study are the following: (a) Small sample size, although series of reirradiation in patients with AG are generally small. (b) O-6-methylguanine-DNA-methyltransferase status was not assessed. (c) 3 patients had low-grade gliomas at the first irradiation, with further progression to AG. It is not clear if those could have had better prognosis. Despite these shortcomings, our results compare favorably with mOS of 23 months for 63 low-grade gliomas reirradiated with fractionated SRT. 31 (d) There are concerns regarding the use of MRI in patients carrying standard implantable devices, and MRI was not the primary technique for follow-up. Conversely, standard MRI has limitations in discriminating between tumor relapse and treatment alterations secondary to treatment. In patients with HGG who have had inconclusive MRI posttherapy, the accuracy of FDG-PET seems to be higher than additional MRI spectroscopy for diagnosis of tumor progression posttreatment. 32 As such, we considered primary follow-up with CT scan and metabolic assessment by FDG-PET or SPECT more appropriate, with MRI being employed in selected circumstances only. Currently, new SCS devices have obtained Food and Drug Administration approval for MRI brain scans, which should resolve this issue.
Conclusions
Spinal cord stimulation during scheduled brain chemotherapy and reirradiation in patient with relapsed HGG, is feasible and well tolerated. This approach was associated with clinical improvement and longer survival than previously reported in reirradiated patients with recurrent AGs. Cerebral blood flow modulation by SCS during radiotherapy and/or chemotherapy in recurrent HGGs merits further research, and a related clinical trial is in progress.
Footnotes
Acknowledgements
We thank the staff and nurses from the departments involved in the study for their invaluable collaboration. Dr D. Fiuza (Research Unit, Dr Negrin University Hospital, Las Palmas, Spain) supervised the statistical analyses. Editorial assistance was by Dr Peter R. Turner of t-SciMed (Reus, Spain).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the past, Dr F. Robaina received financial support for his research activities, and has been an external scientific consultant for Medtronic Ibérica (Madrid, Spain). None of the other coauthors has any conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by a grant (FUNCIS 03/09) from the Health and Research Foundation of the Autonomous Government of the Canary Islands (Spain), a grant (ISCiii, RTICCC C03/10) from the Canary Islands Institute for Cancer Research (Las Palmas, Spain), and a grant (ISCiii 12/02940) from the Carlos-III Health Institute (Madrid, Spain).
