Abstract
Background: High-grade gliomas are the most common and invasive malignant brain tumors in adults, and they are almost universally fatal because of drug resistance and recurrence. In spite of the progress in adjuvant therapy (like temozolomide) and irradiation after surgery, no effective salvage therapy is currently available for relapsed patients. A Korean herbal recipe MSC500 has been reported to have beneficial therapeutic effects in patients with high-grade gliomas who are relapsed or refractory to conventional treatments. But the underlying molecular mechanisms remain unclear.
Introduction
According to the World Health Organization classification, high-grade gliomas include grade III astrocytomas (anaplastic astrocytoma) and grade IV glioblastoma multiforme (GBM). The high-grade gliomas are the most common intrinsic brain tumors in adults and almost universally fatal. GBMs are particularly invasive and aggressive. 1 Nearly all GBMs (>90%) are primary, whereas secondary GBMs develop from lower-grade astrocytomas. 2 Despite multimodality approaches to treatment, the prognosis remains poor. Recurrences are unavoidable even after tumor resection followed by irradiation and temozolomide. 3 In addition to the current treatment using temozolomide and radiotherapy, antiangiogenic therapy with bevacizumab has been introduced in clinical trials as salvage therapy for recurrent high-grade gliomas; however, no promising effects can be concluded as standard therapy. 3 As an alternative strategy, integrative cancer therapy with herbal medicine and standard treatment becomes the most popular choice for this approach. Sasang Constitutional Medicine is a traditional Korean form of medicine widely used in the clinical diagnosis and treatment of disease. It classifies individuals into 4 constitutional types termed as Tae-yangin, So-yangin, Tae-eumin, and So-eumin by the distinctive complex of external manifestations and innate natures. 4 According to Sasang Constitutional Medicine, a Korean herbal medicine recipe MSC500 (mainly composed of 8 herbs including Phellinus linteus, Gastrodia elata, and Mulberry leaf) has been found to be effective for the Tae-Eumin-type patients with refractory high-grade glioma. In the 4 cases receiving MSC500 treatment described below, most of the refractory high-grade glioma patients had not only symptomatic improvement but also promising response in the aspects of tumor shrinkage and prolongation of progression-free survival even up to more than 2 years (case no. 3). But the mechanisms responsible for such dramatic clinical benefit are still unknown. In this study, we intended to investigate the underlying molecular mechanism of this Korean herbal recipe with modern experimental technology.
Recent studies demonstrate the critical role of the cancer stem cell (CSC) in drug resistance, relapse, and metastasis of cancer. The existence of CSC in high-grade glioma has been demonstrated in many reports,5-7 and these CSCs are resistant to the current radiotherapy6,8 and chemotherapy.5,9 The notable effects of medicinal plants and natural products are to eliminate CSC-like cells.10-12 Because MSC500 may preferentially suppress the CSC-like cells in high-grade glioma, we first study the effects of MSC500 to eliminate CSC-like cells in human GBM cells.
CSCs exist as a small proportion in tumor population and typically overexpress ATP-binding cassette (ABC) transporters, which pump out drugs and protect CSCs from chemotherapeutic agents. 13 These overexpressed ABC transporters can also pump out fluorescent DNA binding dyes, such as Hoechst 33342, and thus facilitate detecting Hoechst 33342-excluding CSCs by UV laser-equipped flow cytometry. Due to Hoechst 33342 dye exclusion, the fluorescence signals of CSCs fell to the left-down “side” of the bulk positively stained cells in FACS (fluorescence-activated cell sorter) analysis plots, and they were therefore referred as “side population” (SP) cells. 14 Besides the SP technique, recent evidence suggests that enhanced aldehyde dehydrogenase (ALDH) activity is a hallmark of CSC measurable by the aldefluor assay. 15 These 2 methods have been used for studying human glioma stem cells.6,16 As GBM is the most aggressive and invasive type of glioma, 17 we isolated the SP cells from GBM8401 cells, a permanent human GBM cell line established from a 31-year-old ethnic Chinese female patient. 18 The CSC-like properties of these isolated SP cells have been characterized by higher expression of glioma stem markers such as CD133 5 and Nanog 7 in addition to ABCG2, the gene encoding the phenotype of SP cells. 19 The effects of MSC500 targeting on CSC-like cells have been evaluated by the decrease of both the SP and high ALDH populations in GBM8401 cells. To elucidate the underlying molecular mechanisms, we intended to examine the MSC500-induced suppression on gene expression related to drug resistance, glioma stem markers, as well as major stem signaling pathways 20 in GBM8401 SP cells.
Case Reports
Case 1
A female patient, aged 72 years, was found to have an unresectable right frontal-parietal brain tumor in October 2009. After an open biopsy, the pathology showed GBM. She received temozolomide and radiotherapy during November and December 2009. The tumor had regressed a little after completing 6 full courses of temozolomide. The tumor had regrown as shown in the magnetic resonance imaging (MRI) scan of October 12, 2010. But no active treatment was restarted. Her condition worsened slowly till April 28, 2011, as shown by MRI. After a 3-week oral treatment with MSC500, most of her neurologic deficits improved; the relapsed tumor continuously shrunk, lasting for 5 months as shown by MRI of September 16, 2011 (see figure for Case 1). Unfortunately, she expired in October 2011 due to incidental pneumonia and acute respiratory failure.

After MSC500 treatment, the relapsed tumor continuously shrunk, lasting for 5 months.
Case 2
A male patient, aged 39 years, was diagnosed with GBM of the left temporal lobe in November 2007. He received surgery followed by tomotherapy for a month and temozolomide for 1 year. An MRI in September 2009 showed multiple intracranial recurrences over the right parieto-occipital region and the periventricle area. He refused any major treatment except chi-gong exercise. The tumor was in progression; radiotherapy was reinitiated in March 2010, but his condition rapidly declined into semicoma after irradiation due to brain edema, which was not responsive to mannitol treatment. He received the shunt and then oral treatment with MSC500 in May 2010. Soon after taking the herbal medicine, his consciousness and verbal communication gradually improved. Two months later, he could walk and was even able to go mountain tracking by himself. The tumor was shrunk after a 7-month treatment, as seen in the MRI of November 25, 2010 (see figure for Case 2). Because of poor familial support and discontinuation of medicine in December 2010, the tumor regrew, as shown in the MRI scans 3 months later (February 13, 2011). He was lost to follow-up and expired in February 2012.

The tumor was shrunk after a 7-month MSC500 treatment, and the tumor regrew due to discontinuation of regular treatment.
Case 3
A male patient, aged 36 years, was found to have a right brain tumor after an acute seizure attack in December 2005. He received partial resection of right insular and temporal lobe due to deep tumor location: the tumor size of 6.0 × 6.2 × 6.6 cm was reduced to 3.4 × 5.3 × 6.6 cm. The pathology was infiltrative malignant glioma. Radiotherapy (RT), a total of 5040 cGy in 28 fractions, had been delivered between January and February 2006. He received MSC500 concurrently since January 2006. His seizures were under control soon after the initiation of oral treatment with MSC500. The residual tumor was markedly reduced after April 2006, and he was asymptomatic lasting for 26 months until April 2008, even as the tumor was starting to regrow slowly since August 2007. The findings of serial MRI studies are shown in the figure for Case 3. He was lost to follow-up. We learned that he received gamma knife and chemotherapy as palliative treatments in another hospital at the end of 2008, and that he expired 1 year later.
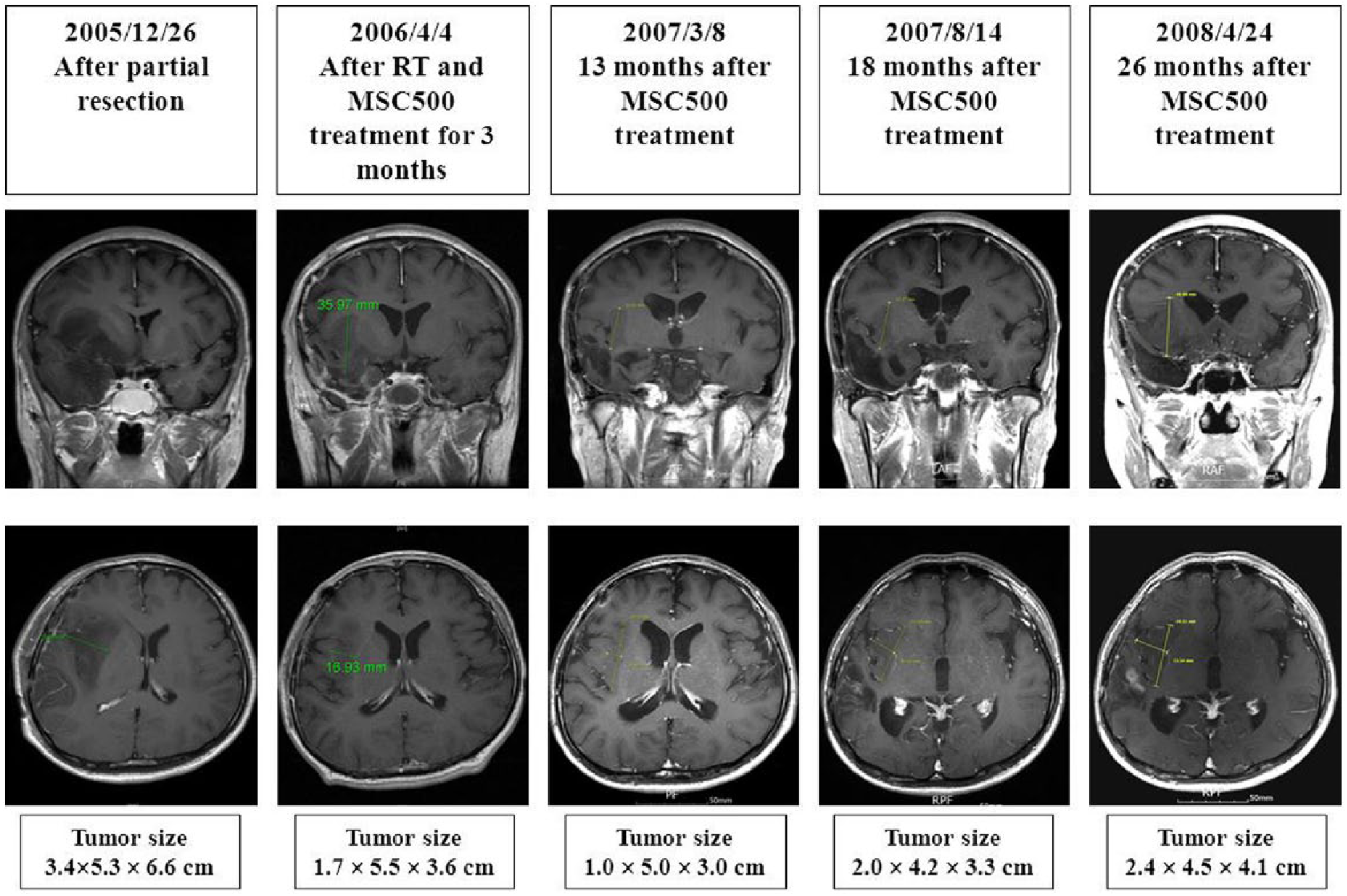
After the initiation of MSC500 treatment, the residual tumor was markedly reduced and was asymptomatic lasting for 26 months till April 2008, and the tumor was found to regrow slowly since August 2007.
Case 4
A 10-year-old boy patient presented with double vision, problems of chewing and gait, and slurred speech since August 2012. He was found to have brain stem glioma and received craniotomy on September 25, 2012. The pathological finding revealed anaplastic astrocytoma.
After partial resection, the patient received 28 fractions of irradiation followed by oral treatment with MSC500 since late November 2012.
After a 1-month MSC500 treatment, the volume of residual mass (December 28, 2012) with central necrosis was further reduced from 23.7 mL to 6.6 mL (see the figure for Case 4). Because of the great improvement of his previous symptoms, he could go back to school in February 2013.

After a 1-month MSC500 treatment and radiotherapy, the volume of residual mass was reduced from 23.7 mL to 6.6 mL. Four months after the herbal treatment, the tumor volume was further reduced in size to 5.4 mL.
Four months after the MSC500 treatment, the MRI (March 27, 2013) showed slightly enhanced mass with central necrosis, and the tumor volume was further reduced in size to 5.4 mL. The patient was continuously under herbal therapy. His condition recovered without problems of walking and chewing. At school, he did not have any problem with studying.
Materials and Methods
Chemicals and Reagents
MSC500 was provided by Banronginsu Oriental Medicine Clinic, Seoul, Korea. Hoechst 33342, reserpine, trichloroacetic acid, temozolomide, and sulforhodamine B were purchased from Sigma (St Louis, MO). The MSC500 stock solution was diluted by double distilled water and filtrated through 0.22-µm-pore membrane filter and then collected in a sterile container. It was further diluted in sterile culture medium immediately prior to use. For maintaining the SP cells, the HEScGro medium was purchased from Millipore (Billerica, MA), and human recombinant bFGF and EGF were from Invitrogen (Carlsbad, CA).
Cell Culture
The human glioblastoma multiforme cell line GBM8401 (BCRC 60163) was purchased from the BCRC (Bioresource Collection and Research Center, Hsin Chu, Taiwan). GBM8401 cells were maintained as monolayers in Dulbecco’s modification Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (Gibco), 1 × penicillin-streptomycin-glutamine (PSG, Gibco), and 1 × nonessential amino acids (Gibco). Cells were cultured at 37°C in a water-jacketed 5% CO2 incubator.
Side Population Analysis and Purification Using Flow Cytometry
The SP cells were analyzed and sorted by Hoechst 33342 staining and FACSAria III cell sorter (Becton Dickinson, BD). GBM8401 cells were detached from the dishes with Trypsin-EDTA (Invitrogen) and suspended at 1 × 106 cells/mL in Hank’s balanced salt solution (HBSS) supplemented with 3% fetal calf serum and 10 mM HEPES. These cells were then incubated at 37°C for 90 minutes with 2.5 µg/mL Hoechst 33342, either alone or in the presence of 100 µM reserpine (Sigma), a potent functional inhibitor of several ATP-binding cassette (ABC) transporters. The diminishment of SP cells in the presence of reserpine was used to define the flow cytometry gate for sorting SP cells. After 90-minute incubation, the cells were centrifuged immediately for 5 minutes at 300 × g, 4°C, and resuspended in ice-cold HBSS. The cells were kept on the ice to inhibit efflux of Hoechst dye and 1 µg/mL propidium iodide (BD) was then added to discriminate dead cells. Finally, these cells were filtered through a 40-µm cell strainer (BD) to obtain single suspension cells for the analysis and sorting in FACSAria III flow cytometer.
Culture of SP Cells
After sorting, GBM8401 SP cells were seeded in a 10-cm dish and maintained in media containing HEScGro medium (Millipore), EGF (10 ng/mL), and bFGF (6 ng/mL) but without serum. After culture for 10 days, spheroidal SP cells were harvested for the subsequent assays of SP cells.
Identification of ALDH-Expressing CSC-Like Cells
The CSC-like population in GBM8401 cells was also identified and analyzed by using Aldeflour reagent–based flow cytometry method according to the manufacturer’s instructions (Stem Cell Technologies, Vancouver, Canada). This method relies on the increased ALDH activity of CSCs. Briefly, the cells were trypsinized and washed with phosphate buffered saline (PBS) and then suspended (1 × 106 cell/mL) in the media provided in the kit. The Aldeflour reagent was then added to the cell suspension followed by 40 minutes of incubation at 37°C. The negative control comprising cells was treated with 15 µM of diethylaminobenzaldehyde (DEAB), a specific ALDH inhibitor. Cells were recovered by centrifugation and washed twice with PBS to remove unbound reagent and finally suspended in ice-cold cell suspension media. Analysis was performed using a FACSAria III flow cytometer (BD). Cells treated with DEAB (negative control) were used to establish the basal fluorescence level in the flow cytometry dot plot and then the gate region for high ALDH activity cells could be defined. The results were quantified calculating the percentage of cells that displayed high ALDH activity.
Assessment of Cell Viability
At harvest, cells were fixed with trichloroacetic acid (TCA) by gently adding it to each well to a final concentration of 10% with subsequent incubation for 1 hour at 4°C. The plates were then washed 5 times with tap water and air dried. The dried plates were stained with 100 µL of 0.4% (w/v) Sulforhodamine B (SRB) prepared in 1% (v/v) acetic acid for 10 minutes at room temperature. The plates were rinsed quickly 4 times with 1% acetic acid to remove unbound dye, and then air dried until no moisture was visible. The bound dye was solubilized in 20 mmol/L Tris base (100 µL/well) for 5 minutes on a shaker. Optical densities were read on a microplate reader (BioTek Instrument) at 570 nm. The optical density is directly proportional to the cell number over a wide range.
Treatment of GBM8401 SP and Parental Cells
For cell viability test, the GBM8401 SP and parental cells were seeded into 96-well plates in DMEM medium supplemented with 2% fetal bovine serum (Gibco), 1 × penicillin-streptomycin-glutamine (PSG, Gibco), and 1 × nonessential amino acids (Gibco) at a density of 1.5 × 103 cells/well. After 24 hour, various doses of MSC500 or temozolomide were added into the medium and the cells were incubated for another 48 hours. For mRNA expression and the CSC-like populations (SP and high ALDH activity cells) assay, the GBM8401 SP and parental cells were seeded in a 6-cm dish at a density of 2 × 105 cells/dish in medium as described in the cell viability test. After 24 hours, various doses of MSC500 were added into the medium and the cells were incubated for another 48 hours. All the control groups were treated with the same volume of PBS.
RT-PCR Analysis of Stemness Gene Expression
The Thermo Scientific GeneJET RNA Purification Kit (Thermo) was used to extract the mRNAs from the GBM8401 SP and parental cells according to the manufacturer’s recommended protocol. Total RNA (1 µg) was reverse transcribed using the Thermo Scientific RevertAid H Minus First Strand cDNA Synthesis Kit (Thermo Scientific, Logan, UT). The cDNA was amplified by polymesars chain reactions (PCR) containing primers at a concentration of 0.5 µM. PCR amplification was performed at 94°C/30 seconds, 55°C/30 seconds, and 72°C/1 minute for 30 cycles. The amplified PCR products were stained with EZ-Vision DNA Dyes (Amresco, Solon, OH) and run on 1.5% agarose gels for identification. The human glyceraldehyde 3-phosphate dehydrogenase (GAPDH) or 18S ribosomal RNA (18S) gene was used as internal control. Primers used are listed below:
Nanog: 5′-GCTTGCCTTGCTTTGAAGCA-3′ and 5′-TTCTTGACTGGGACCTTGTC-3′
CD133: 5′-TCGGAGGACGTGTACGATGATG-3′ and 5′-CACTACCCAGAGACCAATGGTG-3′
ABCG2: 5′-CTGAGATCCTGAGCCTTTGG-3′ and 5′-TGCCCATCACAACATCATCT-3′
GAPDH: 5′-ACCACAGTCCATGCCATCAC-3′ and 5′-TCCACCACCCTGTTGCTGTA-3′
E-Cadherin: 5′-TTTGTACAGATGGGGTCTTGC-3′ and 5′-CAAGCCCACTTTTCATAGTTCC-3′
ABCB5: 5′-CATCGCCTATGGTGACAACAGC-3′ and 5′-TGAGTGACCACTAGGCATGTCC-3′
Oct-4: 5′-CAGTGCCCGAAACCCACAC-3′ and 5′-GGAGACCCAGCAGCCTCAAA-3′
Sox-2: 5′-GCACAACTCGGAGATCAGCAAG-3′ and 5′-CAGCCGTTCATGTGCGCGTAAC-3′
β-Catenin: 5′-ACTGGCAGCAACAGTCTTACC-3′ and 5′-TTTGAAGGCAGTCTGTCGTAAT-3′
Cyclin D1: 5′-GGATGCTGGAGGTCTGCGAGGAAC-3′ and 5′-GAGAGGAAGCGTGTGAGGCGGTAG-3′
Gli-1: 5′-CAGGGAGGAAAGCAGACTGACT-3′ and 5′-AGTTGATGTAGCTGGTCCAG-3′
Notch-1: 5′-ATCGGGCACCTGAACGTGGCG-3′ and 5′-CACGTCTGCCTGGCTCGGCTC-3′
18S: 5′-CGACGACCCATTCGAACGTCT-3′ and 5′-GCTATTGGAGCTGGAATTACCG-3′
Immunofluorescence
GBM8401 SP and parental cells were plated on chamber slide (Millicell EZ SLIDE, Cat. No. PEZGS0816) and glass coverslips, respectively, and allowed to adhere for a minimum of 24 hours. Cells were fixed with 4% paraformaldehyde at room temperature for 15 minutes, and then washed twice with PBS. After fixation, cells were permeabilized with 0.3% Triton X-100 for 15 minutes at room temperature, then washed again with PBS before blocking and applying rabbit anti-E-cadherin primary antibody (#3195, Cell Signaling). Secondary antibody was goat anti-rabbit DyLight 488 (Jackson Cat. No. 111-485-003). The slides were mounted by adding DAPI-fluoromount G (Cat. No. 0100-20, SouthernBiotech, Birmingham, AL) and examined in an Olympus BX51 System microscope using a 40× objective lens. Images were acquired with Olympus DP71 microscope digital camera. The green fluorescence indicates the expression of E-cadherin.
Photograph of the Cells
The images of cultured SP and parental cells were photographed by a digital microscope camera (MIS/PAXcam2+) adapted to an inverted microscope (Olympus/CKX31) under 10× objective lens.
Results
SP Cells Isolated From GBM8401 Human GBM Cells Possess Some Properties of Cancer Stem Cells
To investigate the effects of MSC500 on CSC-like cells, we isolated the SP cells from GBM8401 human GBM cells by FACSAria III Cell Sorter and characterized their properties of CSC-like cells. As shown in Figure 1A, a small percentage (1.3%) of SP could be detected and are shown in the left-down side of the flow cytometry histogram, which could be diminished in the presence of reserpine (100 µM). After 3 days of culture in special media described in Materials and Methods, the SP cells started to display spheroidal morphology in contrast to the flatten-shaped parental cells (Figure 1B). Consistent with the lowered Hoechst fluorescent intensity, the mRNA level of the molecular determinant of SP phenotype, ABCG2 (ATP-binding cassette subfamily G member 2), 19 was much higher in the SP cells after growing in stem cell medium without serum for 10 days (Figure 1C). As expected, the SP cells also expressed markedly higher glioma stem markers CD133 5 and Nanog 7 in comparison with the unsorted parental cells (Figure 1C). In addition, we found the SP cells expressed higher level of E-cadherin mRNA compared to the parental cells (Figure 1D). Immunofluorescence staining also showed the expression of E-cadherin in SP cells was obviously higher than that in parental cells (Figure 1E). The green fluorescence of E-cadherin in parental cells was even lower in the cell–cell contact area when compared and contrasted with the image of cellular area listed left. In contrast, the green fluorescence of E-cadherin in SP cells was higher in cell–cell contact and dispersed along the plasma membrane rather than only restricted in the cell–cell contact area (Figure 1e). E-cadherin is usually recognized as a tumor suppressor in epithelial cells and its expression is rare in GBM. 21 However, recent reports by Lewis-Tuffin et al 21 and by Rani et al 22 had shown that E-cadherin expression was positively correlated to the aggressiveness of GBM cells. Therefore, the markedly higher E-cadherin level implies the crucial role of SP cells in the progression of GBM.

GBM8401 SP cells possess the properties of CSCs.
It is reported that the CD133 (+) CSCs in glioblastoma were more resistant to temozolomide in comparison to the autologous CD133 (−) cells. 5 To further confirm the CSC-like property of these SP cells, we compared the sensitivities of the SP and parental GBM8401 cells to temozolomide. As expected, the SP cells were much more resistant to temozolomide than the parental cells. At dose of 25 µM for 48 hours, temozolomide suppressed 61.2% of parental GBM8401 cells in contrast to 6.1% of SP cells (Figure 2).

GBM8401 SP cells are much more resistant to temozolomide than parental cells.
MSC500 Suppresses the Cancer-Stem-Like Population in GBM8401 Human GBM Cells
In contrast to temozolomide, MSC500 preferentially inhibited the SP cells rather than the bulk parental cells. As shown in Figure 3, after a 48-hour treatment, MSC500 decreased 46.2% of the SP cells survival at dose of 250 µg/mL; however, there was no major effect on the cell viability of the bulk parental cells. When the dose was increased to 500 µg/mL, MSC500 almost diminished 90% of SP cells, while the parental cells were only slightly affected. Next, we examined the proportion of SP cells after treatment with MSC500. After a 48-hour treatment, the percentage of SP cells in GBM8401 cells was dose-dependently decreased from 1.6% to 0.6% and 0.3% by MSC500 at dose of 125 µg/mL and 250 µg/mL, respectively (Figure 4A).
CSCs have higher expression of ALDH, and therefore, flow cytometry analysis of cellular ALDH activity was recognized as a recent method to detect the CSCs.23-27 By using DEAB, an ALDH inhibitor, a gate region could be identified to measure the proportion of high ALDH activity cells. The effect of MSC500 on the suppression of high ALDH activity population in GBM8401 cells was also examined. In Figure 4B, approximately 0.9% of high ALDH activity cells were detected in GBM8401 cells, which was down to 0% in the presence of DEAB (15 µM). After a 48-hour treatment, MSC500 dose-dependently diminished the high ALDH cells from 0.9% to 0.5%, 0.4%, and 0.1% at dose of 50, 125, and 500 µg/mL, respectively.
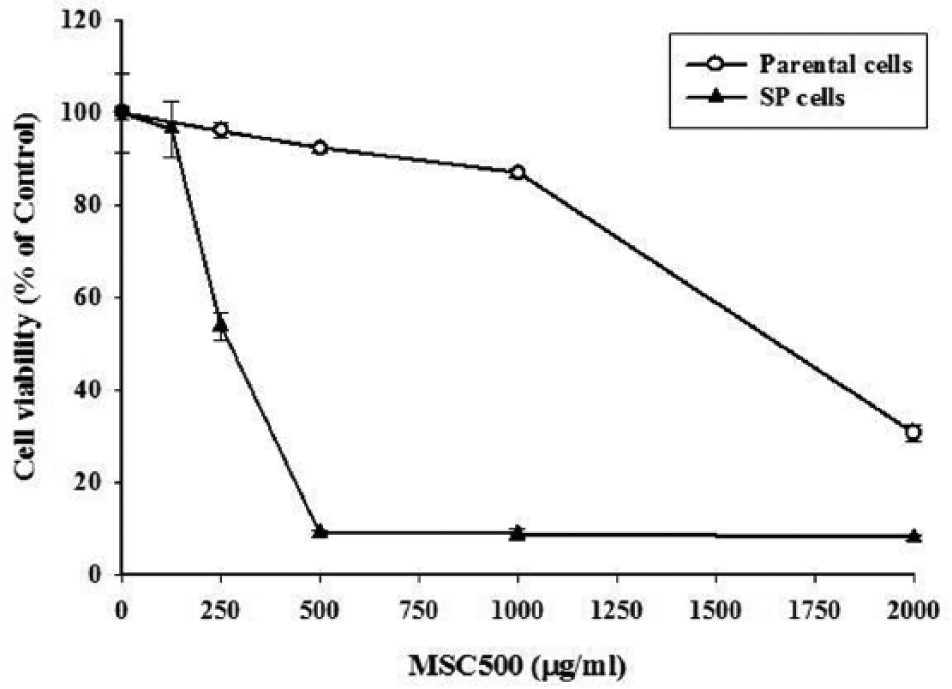
GBM8401 SP cells are much more sensitive to MSC500 than parental cells.
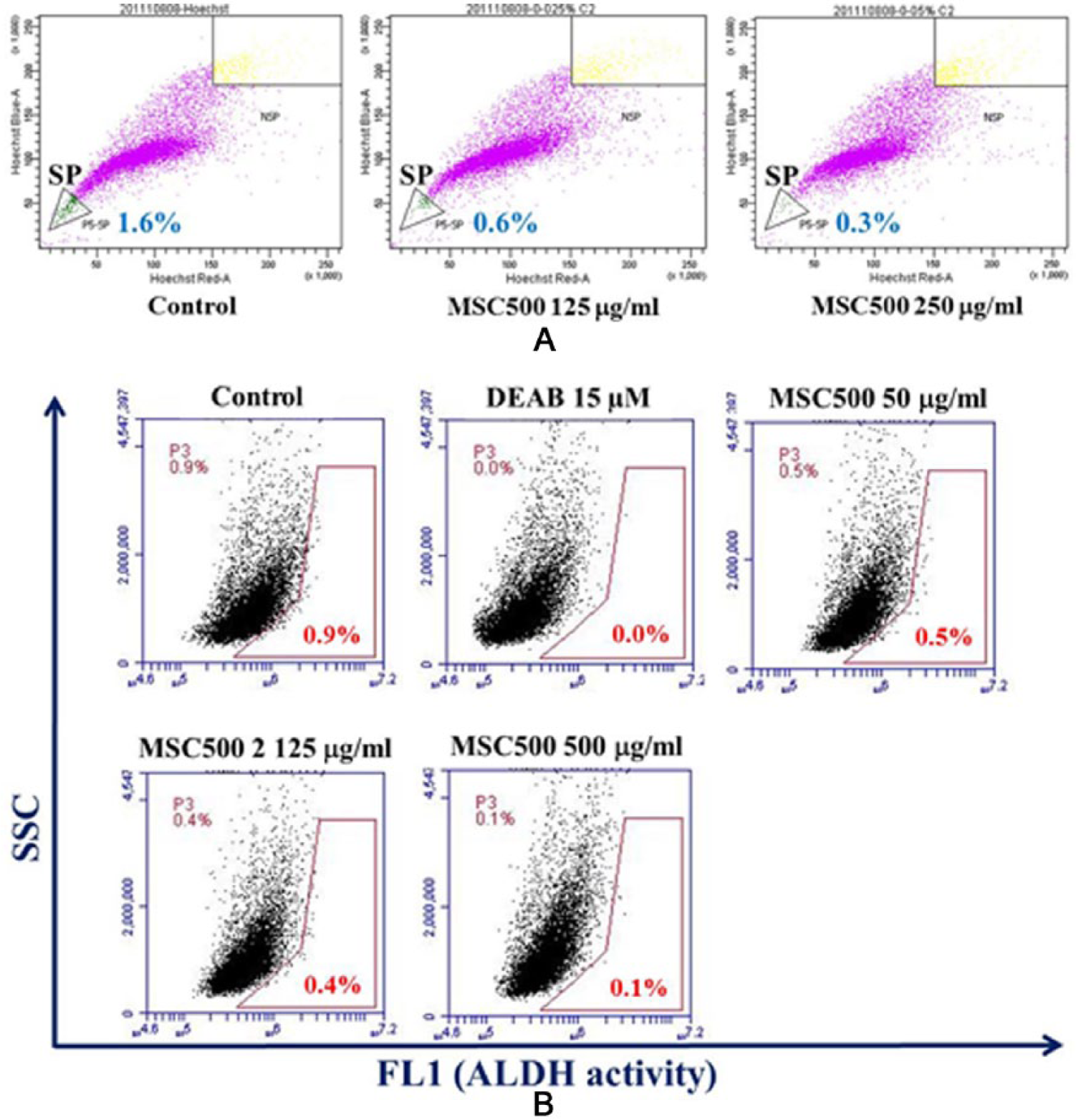
MSC500 significantly reduces both the CSC-like populations marked by Hoechst 33342 dye excretion (SP) and high ALDH (aldehyde dehydrogenase) activity in GBM8401 cells.
MSC500 Suppresses the Drug Resistance and CSC Marker Genes in GBM8401 SP Cells
Regarding the effects of MSC500 on the elimination of cancer stem–like populations shown in Figure 4, we examined the mRNA levels of drug resistance and CSC markers genes in MSC500-treated SP cells. After a 48-hour treatment, MSC500 dose-dependently suppressed the expression of ABCG2. Besides, another ATP-binding cassette gene ABCB5, which had been suggested to mediate chemotherapeutic drug resistance in cancer stem-like subpopulation 28 was inhibited as well by MSC500 (Figure 5A).
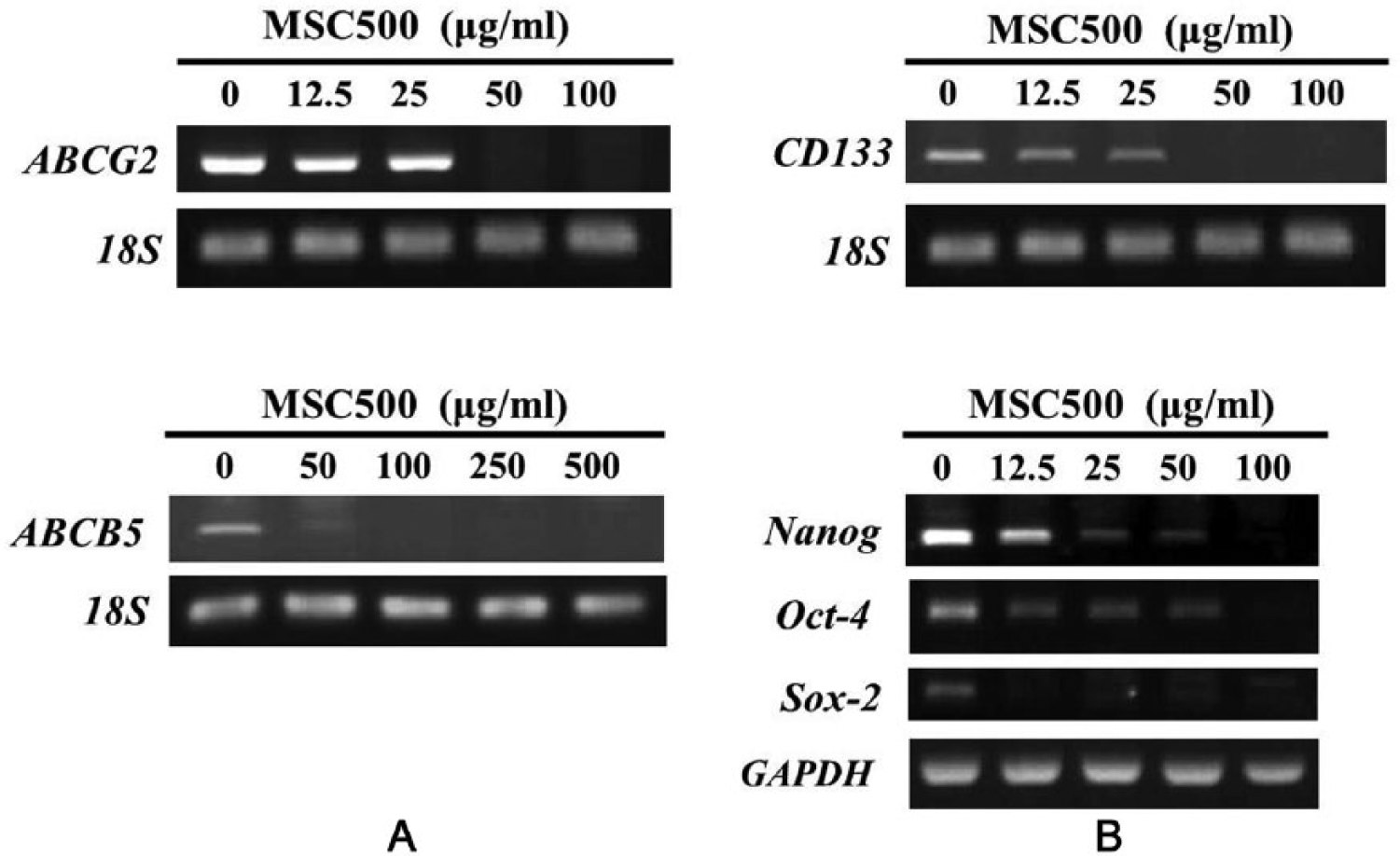
MSC500 downregulates the genes of ABC transporter and glioma stem marker in GBM8401 SP cells after 48 hours of treatment.
In addition to CD133, 5 the core transcription factors of embryonic stem cells (Nanog, Oct-4, and Sox-2) could be primarily detected in highly malignant gliomas 29 and correlated with increased malignancy. 30 It was proposed that these transcription factors (Nanog, Oct-4, and Sox-2), which regulated the self-renewal and pluripotency of stem cells, also functioned similarly in gliomas. 30 Examining the expression of these transcription factors genes in MSC500-treated SP cells, we found Nanog, Oct-4, and Sox-2 expression were all markedly inhibited (Figure 5B).
MSC500 Suppresses the Gene Expression of Notch, Hedgehog, and Wnt/β-Catenin Signaling Pathways in GBM8401 SP Cells
The major signaling pathways in normal stem cells such as Notch, Hedgehog, and Wnt/β-catenin also control the self-renewal, differentiation, proliferation, and tumorigenicity of CSCs. 20 To further delineate the MSC500-mediated elimination of GBM8401 SP cells, we investigated its effects on these signaling pathways.
The key component of the Wnt pathway β-catenin is commonly overexpressed in GBM. 31 At dose of 50 µg/mL, MSC500 markedly diminished the mRNA levels of β-catenin and its downstream gene cyclin D1 after a 48-hour treatment (Figure 6A). It has been shown that GBMs contain stem-like cells with higher Notch activity, 32 and targeting the key component of this pathway, Notch-1, decreases the stem cell proliferation and tumor growth of glioma. 33 MSC500 also markedly decreased the Notch-1 mRNA level at dose of 50 µg/mL (Figure 6B). In Hedgehog pathway, the GLI-1 (Glioma-associated oncogene homologue 1) gene was first identified as an amplified gene in a human GBM. 34 The Hedgehog-GLI-1 signaling is frequently activated in malignant gliomas and is considered to regulate their tumorigenicity and cancer stem cell self-renewal. 35 As shown in Figure 6C, a dramatic diminishment of the GLI-1 mRNA was observed in the GBM8401 SP cells treated with 50 µg/mL of MSC500 for 48 hours.

MSC500 downregulates the genes of Wnt/β-catenin, Notch, and Hedgehog signaling pathways in GBM8401 SP cells after 48 hours of treatment.
MSC500 Suppresses the E-cadherin Gene, Which Is Much More Highly Expressed in SP Cells
Unlike in epithelial tissues, E-cadherin expression in GBM has been proposed to correlate with aggressiveness21,22 and an unfavorable clinical outcome. 21 As mentioned in Figure 1D and E, we found that the GBM8401 SP cells expressed much higher E-cadherin than the parental cells. Consistent with its suppressing effects in these SP cells, MSC500 also dose-dependently decreased the E-cadherin mRNA level at dose range of 12.5 to 100 µg/mL (Figure 7). Although it seems to conflict with the conventional tumor suppressor role of E-cadherin, this suppressing effect on E-cadherin by MSC500 could further support its clinical benefits observed in those cases presented above. The regulatory role of E-cadherin in glioma stem cells is worth to be further investigated.
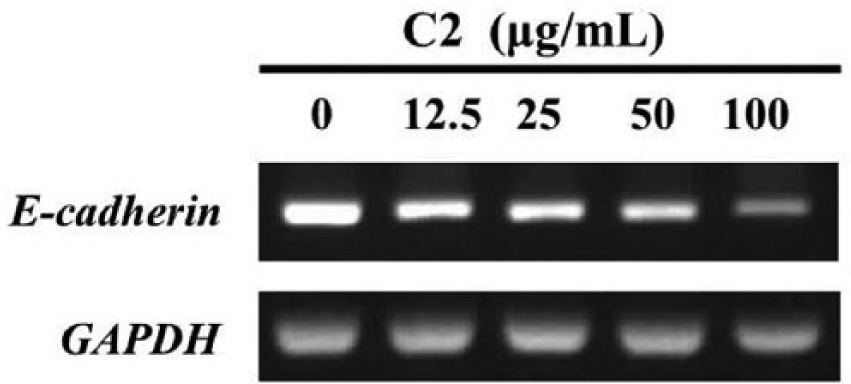
MSC500 downregulates the mRNA level of E-cadherin in GBM8401 SP cells.
Discussion
The fluorescent Hoechst 33342 dye excluding SP determined by high expression of ABC transporter Bcrp1/ABCG2 becomes a useful method for isolating stem cells from various sources. 19 Being consistent with this hypothesis, SP technique has been used to define a small population of stem-like cells in glioblastoma cell lines.6,16 The GBM8401 SP cells used in this study expressed much higher level of ABCG2 relative to the bulk parental cells. Nevertheless, the study by Patrawala et al showed that in contrast to the tumorigenic differences between the SP and non-SP cells, the ABCG2(+) and ABCG2(−) cancer cells are similarly tumorigenic in mice. 36 The CSC-like property of GBM8401 SP cells should be confirmed by higher expression of other stemness genes in addition to the ABCG2. Among the stemness markers, CD133 is essential for glioblastoma stem cell maintenance and could be used as a therapeutic target in GBMs. 37 Nanog is preferentially required in CD133 (+) glioma stem cells and is essential for glioma tumourigenicity in vivo. 7 The CSC-like property of GBM8401 SP cells is thus confirmed by higher expression of these 2 well-documented glioma stemness genes (CD133, Nanog) in comparison to the unsorted parental cells.
E-cadherin expression is limited in normal brain and rare in GBM tumors. 21 In addition to the recent reports, which showed the positive correlation between E-cadherin expression and the aggressiveness of GBM cells,21,22 we found that the GBM8401 SP cells expressed much higher levels of E-cadherin than the unsorted parental cells, indicating the regulatory role of E-cadherin in glioma stem cells.
E-cadherin is an epithelial marker that is well known. The link between epithelial–mesenchymal transition (EMT) and CSC phenotype has been reported.38-40 But evidences for the function of E-cadherin in regulating pluripotency and self-renewal signaling pathways in embryonic stem and induced pluripotent stem (iPS) cells have emerged recently.41 The crucial role of E-cadherin in CSC properties has also been demonstrated in prostate cancer cells by Bae et al.42,43 The E-cadherin (+) prostate CSC-like cells have self-renewal capacity and are highly tumorigenic and invasive in comparison to the nontumorigenic E-cadherin (−) cells.42,43 According to their findings, Bae et al suggested that E-cadherin can serve as a distinct surface marker to isolate prostate CSCs.42,43 Being consistent with this notion, we found the high E-cadherin expression GBM8401 SP cells had much higher levels of stemness genes (ABCG2, CD133, and Nanog) than the unsorted parental cells, which possessed much lower level of E-cadherin (Figure 1D and E). Our results imply the potential of E-cadherin as a marker of GBM stem cells.
Besides the stemness gene expression, the relatively high resistance to temozolomide practically represents the CSC-like property of GBM8401 SP cells. The diminished SP proportion by MSC500 was accompanied with marked repression of drug-resistant genes such as ABCG2 and ABCB5. In brain tumor, ABC transporters have been suggested to be a possible target to eliminate CSCs. 44 Although temozolomide is not a substrate of ABC transporters,45,46 the ABCB5-expressing cells are enriched after a temozolomide treatment in melanoma xenograft model, being considered as potential therapeutic targets for enhancing the therapeutic effects of temozolomide. 47 Thus, the repression of ABCB5 by MSC500 may inhibit the temozolomide-resistant SP cells.
Beyond inhibiting ABC transporters, other mechanisms may contribute to the higher sensitivity of GBM8401 SP cells to MSC500 compared to parental cells. Ulasov et al reported that CD133 (+) glioma stem cells overexpressed genes involved in Notch and Sonic Hedgehog pathways and that inhibition of both pathways can enhance the therapeutic efficacy of temozolomide in these glioma stem cells. 9 In agreement with this report, the temozolomide-resistant GBM8401 SP cells expressed much higher level of CD133 and MSC500 markedly suppressed their Notch-1 and Gli-1, the key components of Notch and Sonic Hedgehog pathways, respectively, in glioma.33-35 Inhibiting these 2 signaling pathways may underlie the higher sensitivity of GBM8401 SP cells to MSC500.
Among the effects of MSC500 on the components of signaling pathways (Notch, Wnt, and Hedgehog) in GBM8401 SP cells, the suppression is most pronounced in the level of Gli-1 (Hedgehog-Gli-1 pathway). Inhibiting Gli-1 may be important in eliminating GBM8401 SP cells by MSC500, as the Hedgehog-Gli-1 signaling forms a positive loop with Nanog, to regulate stem cell self-renewal and glioma cell proliferation. 7 Notably, Nanog, Oct-4, and Sox-2 have been shown to reprogram differentiated somatic cells to induce pluripotent stem (iPS) cells.7,48 Gliomas express this core embryonic stem signature including Nanog, Oct-4, and Sox-2, and inhibition of Hedgehog-Gli-1 signaling by natural product such as cyclopamine suppresses this signature and glioma stem cells. 35 In agreement with these reports mentioned above, MSC500 markedly suppressed Nanog, Oct-4, and Sox-2 levels in GBM8401 SP cells concomitantly with the suppression on Gli-1. Inhibition of this key signaling pathway in association with suppressing core glioma stem signature may essentially contribute to MSC500-mediated elimination of GBM8401 SP cells.
Besides the chemotherapy agent temozolomide, the glioma stem cells are also resistant to radiotherapy. 8 The CSC signaling pathways such as Hedgehog and Notch had been shown to promote radioresistance of glioma stem cells and inhibition of these pathways rendered these glioma stem cells more sensitive to radiation.6,8 Regarding the marked inhibition of Notch-1 and Gli-1 by MSC500 in GBM8401 SP cells, further study is worthwhile investigating the radiosensitizing effects of MSC500 in glioma stem cells.
Our results demonstrate the effects of MSC500 in eliminating temozolomide-resistant GBM CSC-like cells. To broaden the study of MSC500 in GBM CSCs isolated with techniques other than SP sorting and investigate its effect on other phenotype of CSC, more studies are warranted. For example, as reported by Weber et al, 49 these GBM SP cells have limited migration activity. On the contrary, GBM CSCs isolated by neurosphere formation are highly invasive and resistant to multiple chemotherapeutic agents. 50 It may be a suitable model to analyze the effects of MSC500 on the E-cadherin level and the aggressiveness of GBM CSCs in future studies.
Conclusion
MSC500 was proposed to preferentially suppress the small subpopulation of CSC-like cells, which cause the relapse and poor prognosis of high-grade gliomas, owing to its clinical effects observed in the high-grade glioma patients presented in the Introduction. Being consistent with this hypothesis, both the CSC-like populations marked by Hoechst 33342 dye excretion (SP) and ALDH activity in GBM8401 cells were significantly reduced or eliminated by MSC500. The underlying molecular mechanisms were associated with the marked inhibition on the genes of ABC transporters, glioma stem markers, and signaling pathways as well as the decrease of E-cadherin, a novel potential target for glioma stem cells elimination. Our results suggest the potential use of MSC500 to eliminate CSC-like cells for high-grade gliomas, and those findings might explain its clinical benefits observed in the relapsed patients as well as those treated as adjuvant setting after chemotherapy and radiotherapy.
Footnotes
Acknowledgements
We thank Mr. Il-Young Han for his kind help in this collaboration between Wan Fang Hospital, Taiwan and Banronginsu Oriental Medicine Clinic, Korea.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Wan Fang Hospital, Taipei Medical University, Taiwan and Banronginsu Oriental Medicine Clinic, Korea (Grant W212); National Health Research Institutes, Taiwan (Grant 02A1 CAPP37-014); and Ministry of Health and Welfare, Taiwan (Grant MOHW103-TD-B-111-01).
