Abstract
Lung cancer is the leading cause of cancer death worldwide, and most chemotherapeutic drugs have limited success in treating this disease. Furthermore, some drugs show undesirable side effects due to the enrichment of cancer stem cells (CSCs) that are present, leading to resistance to conventional chemotherapy and tumor relapse. CSCs possess self-renewal characteristics, aggressive tumor initiating activity, and ability to facilitate tumor metastasis. Therefore, development of nontoxic agents that can potentiate chemotherapy and eliminate CSCs would be highly desirable. In the present study, we investigated whether Sasa quelpaertensis leaf extracts (SQE) and cisplatin (CIS), individually or in combination, would exert anti-CSC and antimetastatic effect in H1299 and A549 human lung cancer cells. Following these treatments, cell growth, phosphorylation of phosphoinositide-3 kinase, and activation of the mammalian target of rapamycin were inhibited. Decreased serial sphere formation, clonogenicity, and expression of major stem cell markers, such as CD44 and SOX-2, in CD44+ cancer stem cells were also observed. In addition, inhibition of cell migration and invasion in both cell lines as well as inhibition of matrix metalloproteinase-2 activity and expression were detected. Importantly, the anticancer stemness and antimetastasis effects in each of these assays were greater for the combined treatment with SQE and CIS than with each treatment individually. In conclusion, the data suggest that SQE alone, or in combination with CIS, represents a promising therapeutic strategy for eliminating cancer stemness and cell invasion potential of CSCs, thereby treating and preventing metastatic lung cancer cells.
Introduction
Lung cancer is one of the leading causes of cancer death worldwide and has a 5-year survival rate of 13%. 1 Cases of lung cancer are categorized as small cell lung cancers (SCLCs) and non–small cell lung cancers (NSCLCs), depending on the incidence and lethality. The latter category includes adenocarcinoma, squamous cell carcinoma, and large cell carcinoma.2,3 Furthermore, NSCLCs account for the majority of lung cancer cases, and most NSCLC patients present with advanced disease at diagnosis. NSCLC is resistant to standard chemotherapy and radiotherapy regimens due to cell protective mechanisms and survival signaling that are induced in the cancer cells. 4 In particular, the phosphoinositide-3 kinase (PI3K)-AKT-mammalian target of rapamycin (mTOR) pathway is involved in cell proliferation, tumorigenesis, cell invasion, and drug resistance.5,6 Correspondingly, PI3K signaling represents a major cell survival pathway that is frequently activated in various cancers, including lung, cervical, ovarian, and esophageal cancers, and is associated with a poor clinical outcome. 7
An emerging body of evidence also indicates that a subpopulation of tumor cells possesses stem cell and progenitor characteristics, and these cells are resistant to chemotherapy and radiotherapy. These cancer stem cells (CSCs) exhibit normal stem cell-like traits, including differentiation and self-renewal potential.8,9 Furthermore, these CSCs exhibit tumor-initiating activity, clonal expansion as “oncospheres,” and differentiation into transiently amplified cells comprising the bulk tumors. 8 CSCs are also responsible for tumor growth, metastasis, and relapse of cancer after treatment and are relatively chemoresistant to cancer therapeutic agents compared with non–tumorigenic cells.9,10 Therefore, it is hypothesized that targeting of CSCs may represent a successful therapeutic strategy for the eradication of cancer and the inhibition of tumor relapse.
Moreover, although CSCs were initially discovered in human hematopoietic cancer, CSCs have been identified in cancers of the brain, colon, breast, ovary, prostate, and, most recently, lung.11 -15 In addition, the cell surface glycoprotein CD44 exhibits leukocyte homing receptor activity that mediates cellular adhesion to the extracellular matrix (ECM) and cell migration. 16 CD44 has been identified as a potential CSC marker in several tumor types, including in breast, ovarian, colon, pancreatic, and lung cancers.17 -19
Metastasis is the spread of cells from a primary neoplasm to distant sites, and the persistent growth of metastases can lead to death. 20 For 30% to 40% of patients with advanced lung cancers, metastasis to other organs, including bone, has been detected. 21 The invasion and metastasis of malignant cells is facilitated by degradation of the ECM, 22 and this process is initiated by proteinases. In particular, matrix metalloproteinases (MMPs) comprise a family of zinc-dependent endopeptidases. 23 Of the human MMPs that have been identified, MMP-2 has been shown to degrade type IV collagen and fibronectin and appears to play an important role in invasion and metastasis of tumor cells. 24
Cis-diammineplatinum (II) (CIS) is a platinum-based chemotherapeutic agent that has traditionally been used to treat NSCLC. Moreover, it remains the most widely used first-line chemotherapeutic agent for NSCLC. 25 However, continuous and repeated administrations of CIS are associated with toxic side effects, including myelosuppression, asthenia, nausea, emesis, renal failure, and neurotoxicity, due to the nonspecific cytotoxic effects of CIS on both cancer cells and normal cells.26,27 Hence, improving sensitivity to drugs is an important strategy for reducing chemotherapy doses, while maintaining treatment efficacy. One possibility is the use of multiple adjuvant agents for the treatment of NSCLC in order to reduce chemotherapy resistance, increase therapeutic efficiency, and reduce toxic side effects.
The genus Sasa (poaceae), known as bamboo grass, is broadly cultivated in Asian countries. 28 Although the nutritional content of Sasa leaves can be influenced by many environmental factors, Sasa leaves are commonly considered to be beneficial for the treatment of hypertension, cardiovascular disease, inflammation, and cancer.29 -31 Previously, the anticancer properties of Sasa albomarginata and Sasa senanensis extracts have been demonstrated.32,33 Moreover, lignin and polysaccharide preparations from Sasa kurilensis have been shown to inhibit the growth of sarcoma-180 tumor cells implanted in mice. 34 Sasa quelpaertensis Nakai is a bamboo grass native to Korea that only grows on Mt Halla on Jeju Island in South Korea. In the extract of Sasa quelpaertensis leaves, a mixture of polysaccharides, amino acids, and polyphenols have been detected. 35 It is hypothesized that these polyphenols mediate anticancer and antioxidative effects. However, the potential for Sasa quelpaertensis leaf extract (SQE), and/or the combination of SQE with CIS, to affect cancer stemness and cancer metastasis remains poorly understood. Therefore, in the present study, SQE and CIS were administered individually and in combination to investigate whether the antistemness effect of CIS can be enhanced, whether the self-renewal potential and stem cell markers expression by tumor cells can be suppressed, and whether the metastasis of human lung cancer cells can be inhibited with these agents.
Materials and Methods
Preparation of Sasa quelpaertensis Leaf Extracts
Sasa quelpaertensis is a bamboo grass native to Jeju Island in South Korea. It grows only on Mt Halla on Jeju Island and is the only bamboo grass (genus Sasa [Poaceae]) present on the island. A voucher specimen was collected and stored at the Research Institute for Hallasan in the Jeju Special Self-Governing Province. Dr Hyun Cheol Kim, a botanist, assisted with the identification of the species. Sasa quelpaertensis leaves were collected during October and November from Mt Halla on Jeju Island. Harvested leaves were cleaned, dried at 60°C, and pulverized to 100 to 200 mesh. Dried leaves (1 kg) were then extracted with 70% ethanol at room temperature (RT) for 48 hours. After being filtered through a membrane, SQE was concentrated at 40°C using a vacuum rotary evaporator, freeze-dried, and then ground into a powder. The SQE yield was approximately 11.4%, and these extracts were stored at −20°C.
Cell Culture and Materials
Human NSCLC cell lines, A549 and H1299, were purchased from American Type Culture Collection (ATCC; Manassas, VA). Cells were cultured in RPMI-1640 medium (Gibco, Rockville, MD) containing 10% fetal bovine serum (v/v) (FBS; Hyclone, Logan, UT) and 1% penicillin–streptomycin (100 U/mL and 100 µg/mL, respectively; Invitrogen, Carlsbad, CA) at 37°C in a humidified atmosphere of 5% CO2.
Cis-diammineplatinum (II) dichloride (CIS; Sigma-Aldrich, St Louis, MO) was dissolved in diemthyl sulfoxide (DMSO) for a final concentration of 10 mg/mL. This stock solution was diluted into fresh medium to provide concentrations of CIS ranging from 0.25 to 8 µg/mL.
Cell Viability Assays
Cell viability was assessed using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT; Sigma-Aldrich, St Louis, MO) assays. 36 Briefly, A549 or H1299 cells were seeded into 96-well plates (5 × 103 cells/well). After 24 hours at 37°C and 5% CO2, cells were treated with SQE, CIS, or a combination of SQE and CIS. After 48 hours, medium was replaced with culture medium containing 500 µg/mL MTT. Cells were incubated at 37°C for another 3 hours before supernatant was removed. The formazan crystals were dissolved by adding DMSO, and absorbance values were measured at 560 nm using a microplate reader (Molecular Device, Sunnyvale, CA). Concentrations that induced a 50% reduction in cell viability (IC50) were determined from dose–response curves.
Isolation of CD44+ CSC Using Flow Cytometry
Expression of the CSC marker, CD44, was evaluated using flow cytometry. Briefly, H1299 cells were treated with 0.05% trypsin and washed with phosphate-buffered saline (PBS). Cells were then resuspended in 90 µL PBS containing 0.5% bovine serum albumin for 10 minutes at RT. Alexa Flour 488 fluorescent dye conjugated to CD44 monoclonal antibody (10 µL; Cell Signaling, Danvers, MA) was incubated with the cells for 30 minutes in the dark at 4°C. The samples were then washed and analyzed using a FACS DiVa (BD, San Jose, CA). Twenty-five percent of the cells with the highest levels of fluorescence were designated positive cells. Conversely, 20% of the cells with the lowest levels of fluorescence were selected as the negative population.
Sphere Formation Assays
To measure sphere formation, 10% poly(2-hydroxyethy methacrylate) (polyHEMA; Sigma-Aldrich, St Louis, MO) stock solution (12% poly HEMA in 95% ethanol) was added to each well of 6-well plates. 37 Serum-free stem cell medium was prepared with DMEM-F12 (1:1; Gibco, Rockville, MD) supplemented with 10 µg/mL insulin, 100 µg/mL epidermal growth factor (EGF), and 40 µg/mL basic fibroblast growth factor (bFGF; Sigma-Aldrich, St Louis, MO). Primary cells were plated in polyHEMA-coated 6-well plates at 1 × 105 cells/well, while subsequent passages were plated at 5 × 104 cells/well. Various concentrations of SQE, CIS, or a combination of SQE and CIS were applied to primary cells, whereas subsequent cell passages were grown in the absence of these treatments. After incubation for 6 days, the number of spheres present were counted and photographed.
Clonogenic Assays
Clonogenic assays were performed as previously described. 38 Briefly, H1299 cells were plated in 6-well plates (5 × 102 cells/well) for 24 hours, then were treated with SQE, CIS, or SQE plus CIS for 6 days. Colonies were fixed with 0.9% NaCl and stained with crystal violet (Sigma-Aldrich, St Louis, MO). Stained colonies were counted and plating efficiency was calculated as follows: Plating efficiency = (colony number/total cell number) × 100%. Experiments were performed in triplicate.
Wound Healing and Transwell Invasion Assays
Cell motility was assessed using wound healing assays as described previously. 39 Briefly, A549 or H1299 cells were grown to confluency in 6-well plates. The cell monolayers were then wounded using a sterile 200 µL pipette tip and then washed twice with PBS buffer to remove detached cells. Cells were treated with SQE, CIS, or SQE plus CIS and were then incubated at 37°C for 48 hours (A549) or 24 hours (H1299). Cells were monitored using a light microscope, and 5 randomly chosen fields were photographed.
Invasion assays were performed using 24-well BD Biocoat Matrigel invasion chambers containing 8-µm polycarbonated membranes (Becton Dickinson, Bedford, MA) according to the manufacturer’s instructions. Briefly, A549 cells were seeded into the upper chambers (1 × 105 cells/chamber) and were treated with SQE, CIS, or SQE plus CIS. The lower chamber contained culture media with 5% FBS as a chemoattractant. After the cells were incubated at 37°C for 24 hours, the cells present on the underside of the upper chamber were fixed with methanol and stained with crystal violet. Five fields from each membrane were randomly selected and the numbers of stained cells present were counted. These experiments were performed in triplicate.
Gelatin Zymography Assays
Forty-eight-well plates containing 4 × 104 cells/well and serum-free medium were treated with SQE, CIS, or SQE plus CIS for 48 hours (A549) or 24 hours (H1299). Supernatant from each well was then collected and subjected to 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) in the presence of 0.1% gelatin (Sigma, St Louis, MO). Gels were washed twice with 2.5% Triton X-100 to remove the SDS, and then were incubated with developing buffer (50 mM Tris–HCl [pH 7.6], 10 mM CaCl2, 51 mM NaCl, 0.2 µM ZnCl2) at 37°C overnight. The gels were subsequently stained with 0.5% Coomassie Brilliant Blue R-250 (Amresco, Solon, OH) for 1 hour and were then destained with a 40% methanol/10% acetate solution. The enzymatic activity of MMP-2 was detected as a clear band against a blue background.
Western Blot Assays
Cells were harvested, washed with cold PBS, and lysed with buffer (50 mM Tris–HCl [pH 7.6], 150 mM NaCl, 1% Nonidet P-40, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS), 1 mM PMSF, 1 mM Na3VO4, and 1 mM NaF). Total protein was determined using a Bio-Rad Protein Assay Kit (Bio-Rad, Hercules, CA). Denatured cell lysates were separated by 10% SDS-PAGE and transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA). Membranes were then blocked with 5% non–fat milk for 1 hour and incubated with primary antibodies against MMP-2 (Santa Cruz, Santa Cruz, CA), phospho-PI3K (p85, Tyr458), phospho-mTORSer2448, CD44 (Cell Signaling, Danvers, MA), SOX-2 (Millipore, Billerica, MA), or β-actin (Sigma-Aldrich, St Louis, MO). After rinsing membrane twice with Tris-buffered saline containing Tween-20 (TBS-T), membranes were incubated with the appropriate secondary antibodies conjugated with horseradish peroxidase for 1 hour at RT. Bound antibody was visualized using an enhanced chemiluminescence (ECL) detection system (Animal Genetics, Inc, Suwon, Kyonggi-do, Korea). β-actin was used as a loading control.
Statistical Analysis
The data presented are the standard error of the mean (SEM) from at least 3 independent experiments. Data were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test with GraphPad Prism, version 3.0 (GraphPad Software, Inc, San Diego, CA). P values less than .05 were considered statistically significant.
Results
Combination of SQE and CIS Inhibits the Growth of A549 and H1299 Lung Cancer Cells via the PI3K/mTOR Signaling Pathway
Growth assays were performed to assess the effect of SQE in combination with CIS on the proliferation of lung cancer cell lines, A549 and H1299 (Figure 1A-F). For comparison, cells were treated with various doses of CIS and SQE individually for 48 hours. Cell growth was measured using MTT assays, and IC50 values were determined. For A549 cells, the IC50 values were ~1.0 µg/mL for CIS and ~600 µg/mLfor SQE (Figure 1A and B). For H1299 cells, the IC50 values were ~2.0 µg/mL for CIS and ~400 µg/mL for SQE (Figure 1D and E). Growth assay data further demonstrated that treatment with CIS and SQE at IC50 doses decreased A549 cell growth by 45.48% and 43.64%, respectively, while SQE plus CIS inhibited cell growth by more than 66.85% (Figure 1C). For the same treatments, the growth of H1299 cells decreased by 52.72%, 46.58%, and 75.85%, respectively (Figure 1F). When the inhibitory effect of SQE and CIS on lung carcinoma cell growth and proliferation was further investigated using western blotting assays, both CIS and SQE were found to weakly inhibit PI3K and mTOR phosphorylation, while the combination of SQE and CIS resulted in a greater inhibition of PI3K and mTOR phosphorylation in both cell lines. Taken together, these data suggest that SQE and CIS decrease cell growth via inhibition of the PI3K/mTOR signaling pathway in NSCLC.
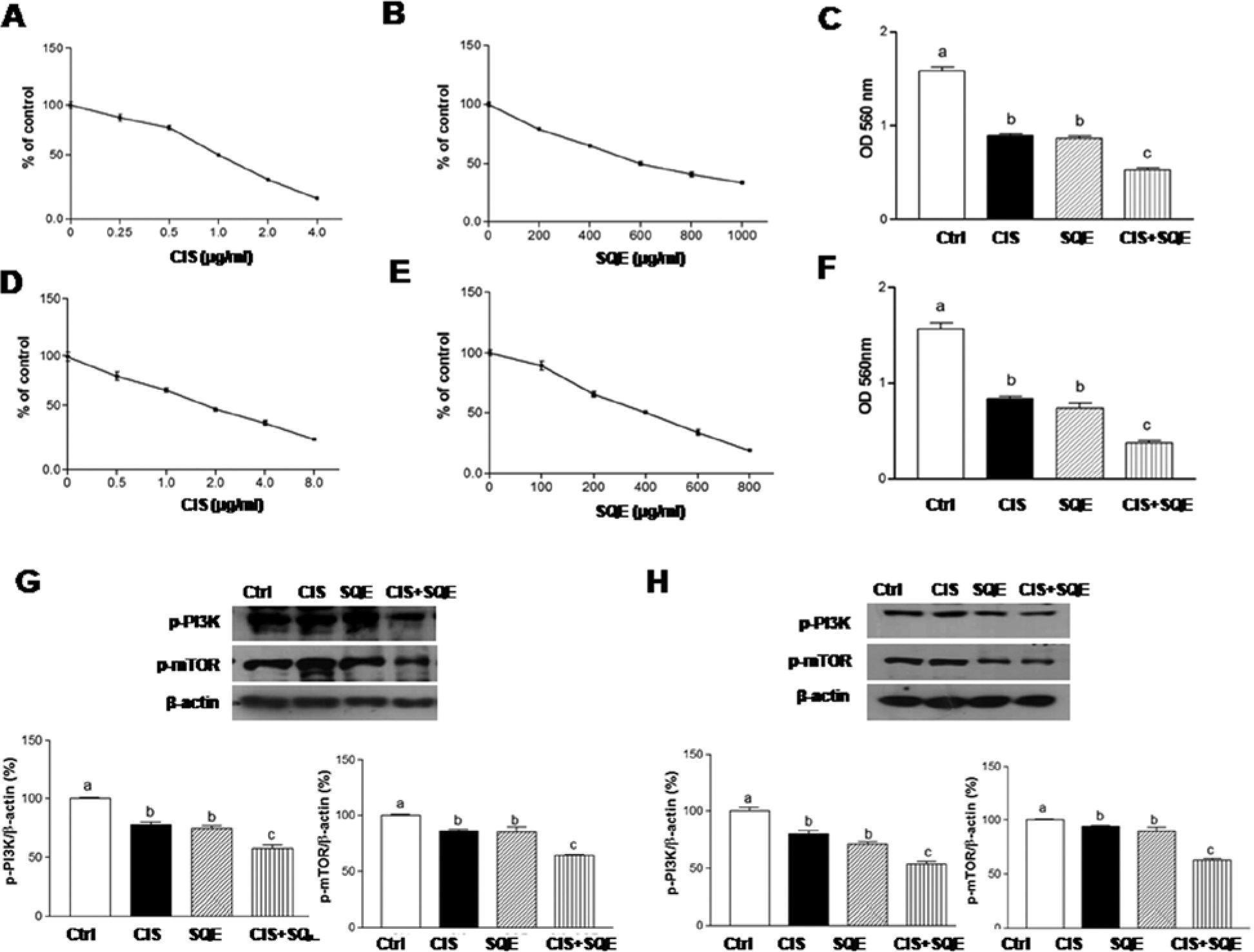
Combination of SQE and CIS inhibits the growth of A549 and H1299 lung cancer cells via the PI3K/mTOR signaling pathway.
Combination of SQE and CIS Reduces the CD44+ Cell Population and Expression of CSC Markers
To evaluate whether SQE may potentiate the cytotoxicity of chemotherapy against CSCs, the CD44+ cell population was characterized. CD44 is a marker that has been found to be expressed by lung cancer cells exhibiting stem cell properties such as self-renewal, high levels of proliferation, and a tumorigenic and chemoresistant phenotype. 17 Previously, it was reported that A549 cells do not express CD34, CD44, and CD133. 17 Therefore, only H1299 cells were used to analyze the effects of SQE and CIS on CD44+ CSCs. Over 80.9% of untreated H1299 cells were found to be CD44+. In contrast, 5.1% of untreated H1299 cells were CD44− (Figure 2A). When H1299 cells were treated with SQE or CIS, the CD44+ population decreased. Treatment with SQE plus CIS further suppressed the CD44+ population detected (Figure 2B). The effects of SQE and CIS on expression of CD44 were analyzed for both unsorted H1299 cells (Figure 2C) and sorted H1299 cells (Figure 2D). For both cell populations, treatment with SQE or CIS led to a decrease in CD44 expression. However, the combination of SQE plus CIS led to a greater decrease for both cell groups. Similar results were obtained for another CSC marker, SOX-2. Thus, the combined treatment of SQE with CIS was found to be effective in suppressing CSC-markers in H1299 cells.
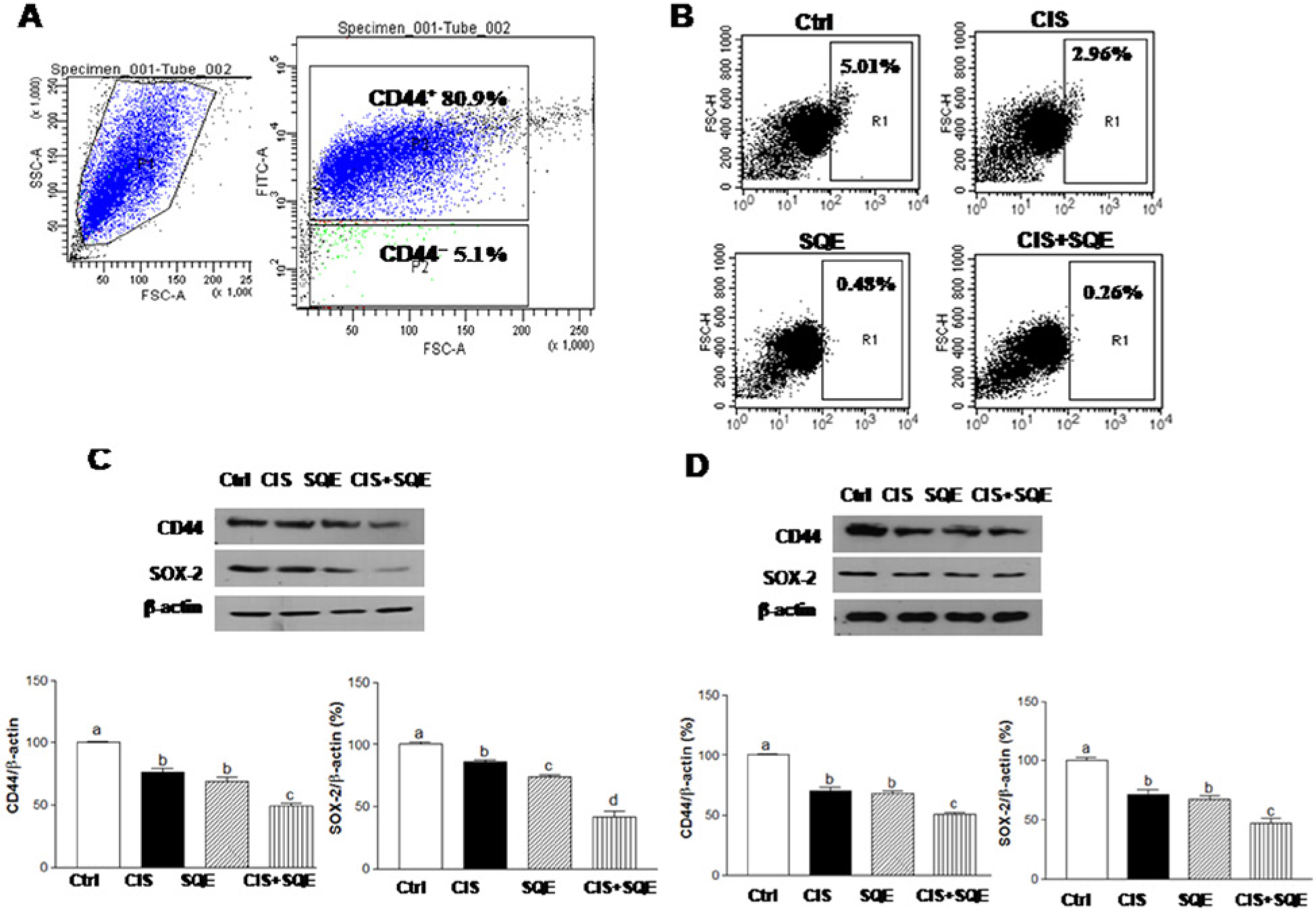
Combination of SQE and CIS reduces the CD44+ cell population and expression of CSC markers.
Combination of SQE and CIS Effectively Inhibits the Self-Renewal Characteristics of CSCs
The ability to grow as nonadherent spheroids in serum-free medium has been widely used to evaluate the self-renewal capacity of CSCs. 40 Therefore, H1299 spheres cultured in polyHEMA-coated plates were exposed to various concentrations of SQE or CIS. After 6 days, the number and size of the spheres observed were found to be attenuated in the presence of SQE or CIS in a dose-dependent manner (Figure 3A). Furthermore, the IC50 values were ~0.3 µg/mL for CIS and ~100 µg/mL for SQE, respectively (Figure 3B).
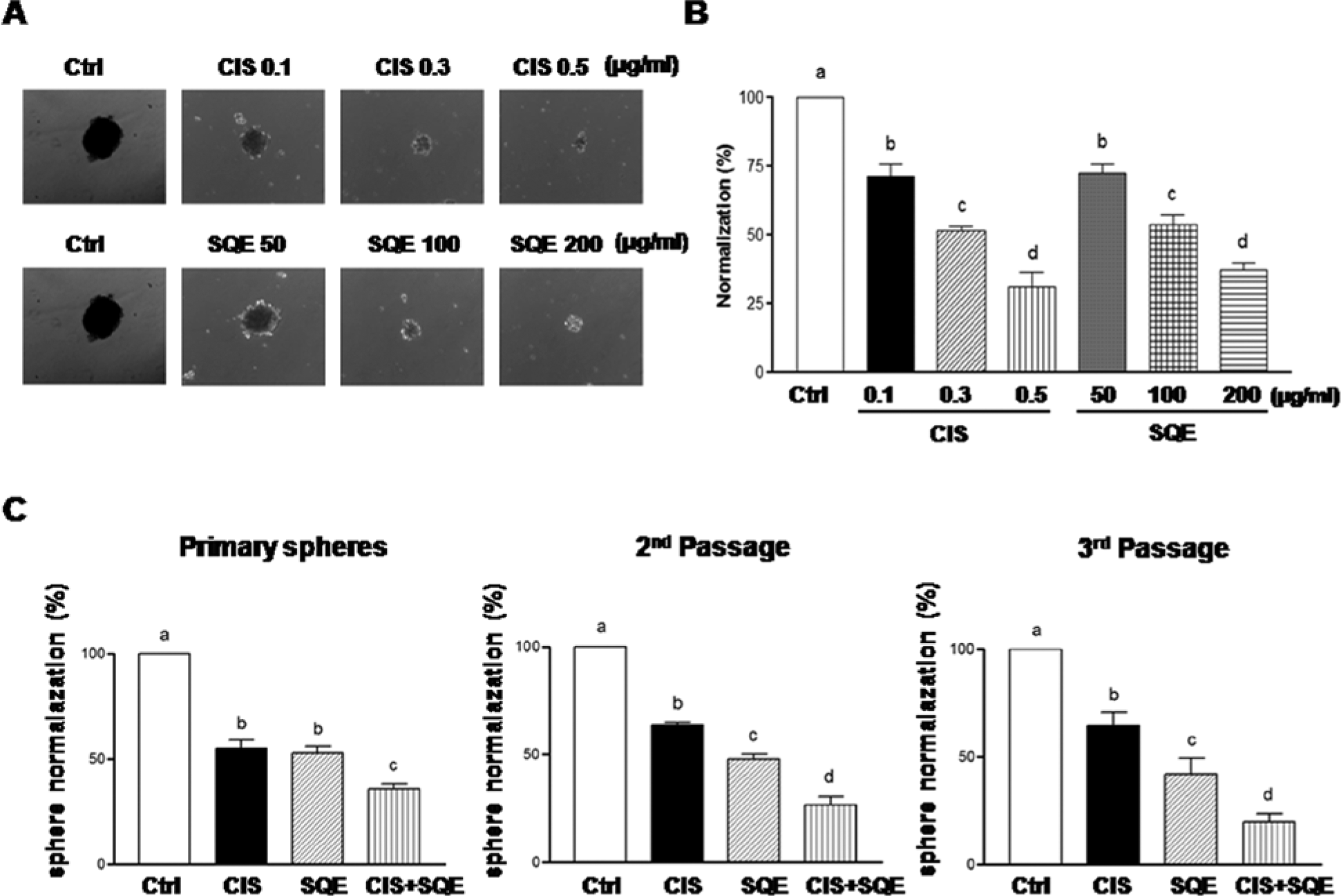
Combination of SQE and CIS effectively decreases nonadherent sphere formation.
Spheres are capable of yielding secondary spheres along multiple lineages. 41 Therefore, serial dilutions of spheres were performed to isolate and expand the lung stem/progenitor cells that constituted the nonadherent cells. An IC50 treatment with SQE (100 µg/mL) or CIS (0.3 µg/mL) was found to inhibit the formation of primary spheres. In addition, a significant decrease in the number of sphere-forming cells during 2 subsequent passages was also observed in the absence of treatment, indicating that the self-renewal capacity of pretreated stem/progenitor cells was reduced (Figure 3C). Furthermore, the combination of SQE plus CIS was associated with an additive inhibitory effect on sphere formation in subsequent passages.
Clonogenic assays are another widely used method for monitoring the reproductive integrity and self-renewal potential of CSCs in vitro. 37 When H1299 cells were treated with CIS (0.3 µg/mL), SQE (100 µg/mL), or a combination of both reagents for 6 days, the clonogenicity of these cells was significantly inhibited by 43.3%, 42.8%, and 96%, respectively (Figure 4). Taken together, these results indicated that the combination of these treatments was more effective in inhibiting colony formation than each treatment alone.
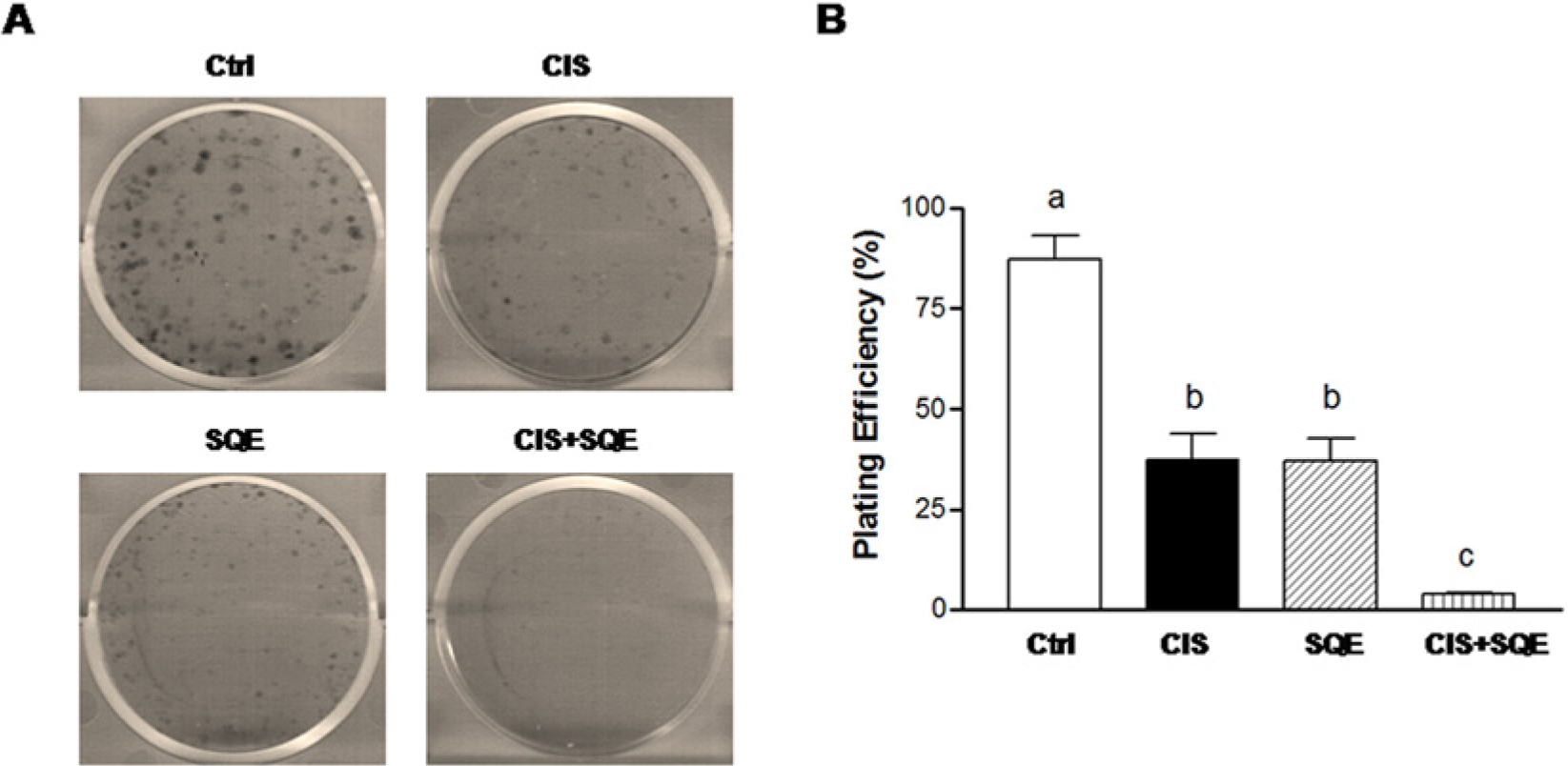
Combination of SQE and CIS inhibits colony formation.
Combination of SQE and CIS Suppresses the Metastatic Potential of Lung Cancer Cells
To investigate the regulatory effect of SQE and CIS on tumor metastatic potential, wound-healing assays and invasion assays were performed. Trypan blue exclusion assay was performed in parallel to rule out a cytotoxic effect of SQE and CIS on A549 and H1299 cells under these experimental conditions. Compared to controls, over 95% of cells survived treatment with SQE and/or CIS (data not shown), indicating that SQE and CIS are nontoxic to A549 and H1299 cells. In the wound healing assay, control cells were observed to readily migrate toward the opposite front of the wound, and wound healing was nearly achieved within 48 hours for both cell lines. However, cells treated with CIS (1.0 µg/mL for A549 cells, 2.0 µg/mL for H1299 cells) or SQE (600 µg/mL for A549 cells, 400 µg/mL for H1299 cells) were observed to migrate slower at both the 24-hour and 48-hour time points (Figure 5A and B). Furthermore, migration was even slower when cells were treated with SQE plus CIS. In transwell invasion assays, treatment with either CIS or SQE significantly decreased the invasion of cells by 39.5% and 52.9%, respectively, compared to control cells. Treatment with SQE plus CIS further reduced cell invasion by 83.13% compared to control cells (Figure 5C and D). These results suggest that SQE or CIS may inhibit lung cancer cell migration and invasion in vitro, and the combination of SQE and CIS has the potential to inhibit the metastasis of lung cancer cells to a greater extent than treatment with SQE or CIS alone.
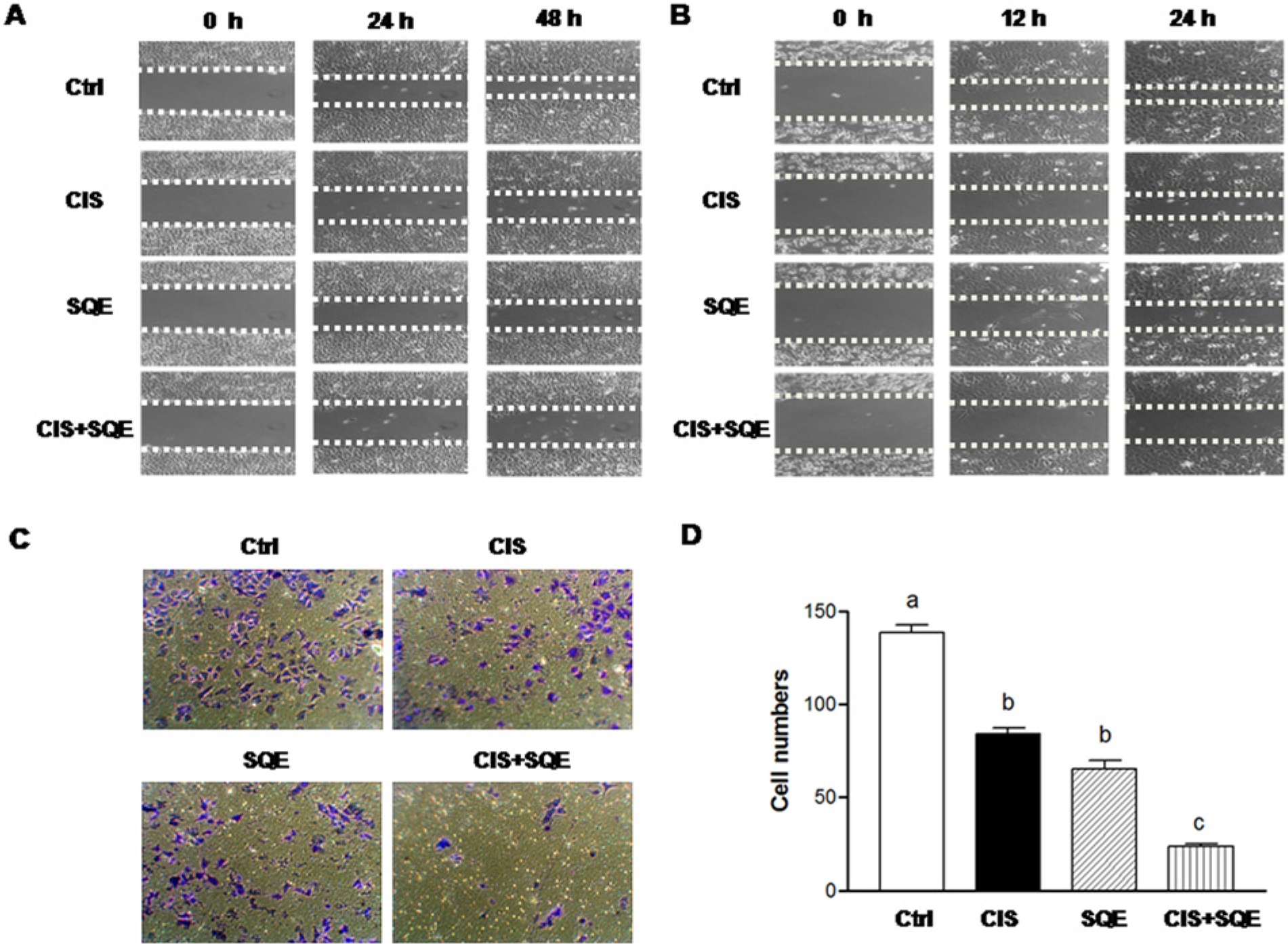
Combination of SQE and CIS inhibits cell migration and invasion.
The inhibitory effects of SQE and CIS on MMP-2 expression was also examined, since MMP-2 has been shown to play a major role in the promotion of tumor invasion and metastasis. 42 The enzymatic activity and expression of MMP-2 were examined using gelatin zymography and western blotting assays, respectively (Figure 6). When both cell lines were treated with either SQE or CIS, lower levels of MMP-2 enzymatic activity were detected, along with reduced levels of MMP-2 expression. Furthermore, cells treated with combination of SQE and CIS exhibited a greater reduction in MMP-2 activity and expression compared to each treatment alone. Taken together, these data suggest that SQE enhances the antimetastatic effects of CIS in lung cancer cells by inhibiting the activity and expression of MMP-2.
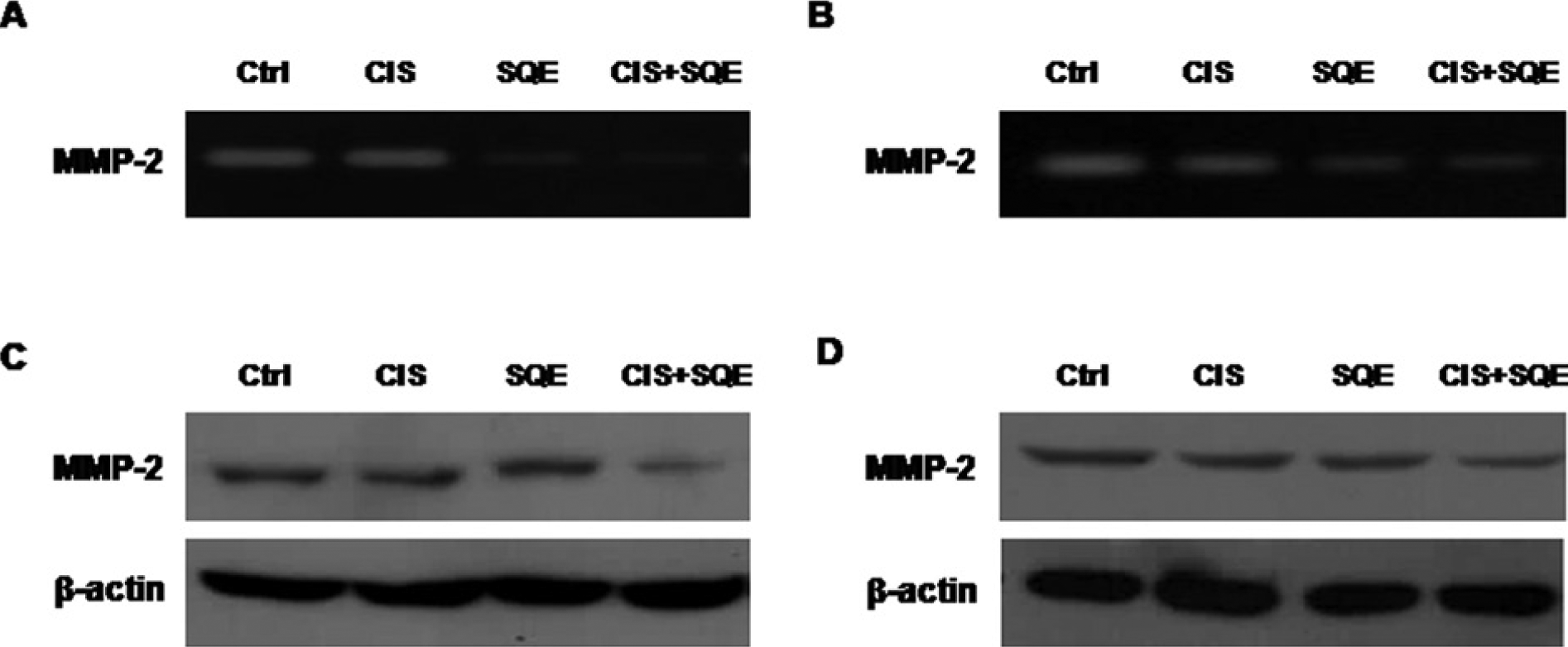
Combination of SQE and CIS suppresses the enzyme activity and expression of MMP-2.
Discussion
Lung cancer is the most common cause of cancer-related mortality, and most chemotherapeutic drugs have had limited success treating this disease. One reason for the drug resistance and/or tumor regeneration observed following chemotherapy is the enrichment of CSCs. 43 This small subpopulation of cells exhibiting a self-renewing and stem-like phenotype may be responsible for tumor initiation, maintenance, progression, and resistance to therapy.8,9 It has also been hypothesized that CSCs have an enhanced metastatic phenotype. 9 Therefore, therapeutic strategies aimed at eradicating CSCs may improve the clinical outcome of cancer patients.
The chemotherapy and radiation therapies currently applied are usually associated with serious side effects and toxicity. Consequently, dietary and botanical natural extract compounds have been increasingly investigated for their anticancer properties. 44 Although CIS-based chemotherapy is considered a standard treatment for advanced NSCLC, CIS has been consistently associated with marked toxicity in several epidemiological studies.45,46 In contrast, phenolic compounds present in the human diet have been used to inhibit, delay, or reverse carcinogenesis as a result of their chemopreventive properties, minimal toxicity, and fewer side effects.47,48 Several bioactive compounds including sulforaphane, curcumin, and epigallocatechin-3-gallate, have also shown the capacity to affect the self-renewal potential of CSCs by restricting sphere formation.49 -51 When combination of natural compounds with chemotherapy reagents have been studied, including combinations such as sulforaphane with CIS, or gemcitabine and curcumin with 5-FU, additive effects of their anticancer properties have been observed against colon, prostate, and pancreatic cancers.52,53 Ideally, therapeutic combinations that prevent unfavorable side effects, have low toxicity, and ultimately improve clinical outcome are preferred.
PI3K/AKT/mTOR signaling is involved in various cellular functions, including cell growth, proliferation, migration, survival, and cell cycle regulation.5,6 Correspondingly, a recent study has shown that inhibition of PI3K/AKT/mTOR signaling is consistently associated with suppressed proliferation by cancer cells. 54 Similarly, Lee at al have reported that a traditional herbal medicine, Andrographis paniculate, contributes to an inhibition of migration and invasion by NSCLC via downregulation of the PI3K/Akt signaling pathway. 39 On the other hand, several apoptosis markers were examined, including Bax, caspase 3, and PARP, and the SQE treatment had no observable effect on cell death/apoptosis in our experimental conditions. In the present study, it is demonstrated for the first time that treatment with SQE and CIS inhibits PI3K and mTOR signaling in human NSCLC cells, and this is associated with lower levels of cell proliferation.
In the present study, several techniques were used to isolate and characterize lung cancer CSCs in vitro in order to analyze the inhibitory effect of SQE on cancer stemness. First, sphere formation assays were conducted to analyze the self-renewal capacity of cancer cells. 40 Using the method of Dontu et al, the ability of spheroids to develop from a subpopulation of cells that can undergo self-renewal and proliferation in serum-free media was evaluated.55,56 In contrast, differentiated cells are unable to survive under these conditions. Sphere formation was observed over 2 passages of H1299 cancer cells, indicating that stem cell populations are present in these cell lines. Furthermore, treatment of these cells with SQE significantly suppressed sphere formation. The ability to target the self-renewal potential of CSCs is not unique to SQE, since other natural products, including curcumin, piperine, and sulforaphane, have been shown to target the self-renewal potential of breast, pancreas, and prostate CSCs.49,53 Second, colony formation capacity, another characteristic of CSCs, was investigated using clonogenic assays. The number of colonies formed by H1299 cells was found to decrease following treatment with SQE and CIS, both individually and in combination. Third, stem cell markers were used to distinguish CSCs from differentiated, non-CSCs in the H1299 cells lines. There are several CSC markers that have been identified in many types of cancer, including CD133, CD34, ALDH, and CD44.15,17 Although CD133 is the most frequently used CSC marker for liver, brain, colon, and lung cancers, biochemical studies have demonstrated that CD133 plays a role in cell cycle regulation and proliferation but not in tumor initiation. 57 In another flow cytometry analysis of NSCLCs, expression of cell surface CD44 was detected in 81.58% of cells, while levels of CD44 in serum were also identified as an indicator of NSCLC in clinical patients.17,58 In particular, it has been shown that an enrichment of CD44+ cells is associated with tumor propagating phenotype, and CD44 is a potential CSC marker of NSCLC cell lines. In vivo, CD44+ cells were able to establish a lung tumor that resembles the parental tumor, while CD44− cells did not. 59 Consequently, the high tumorigenic capacity of CD44+ cells was demonstrated. In the present study, treatment of CD44+ expressing H1299 cells with SQE and CIS, individually or in combination, resulted in lower levels of expression of certain CSC markers.
Metastasis involves the spread of tumor cells from a primary site to a distant secondary site, and this can be driven by a small population of CSCs. 60 Therefore, the effect of SQE and CIS on cell metastasis was investigated. Treatment with SQE was found to suppress not only cancer stemness but also cancer cell migration and invasion. These effects were further enhanced when SQE was applied in combination with CIS. MMPs are a family of zinc-dependent endopeptidases that play a major role in metastasis by degrading the ECM, thereby allowing cancer cells to invade the blood or lymphatic system and spread to other tissues. 61 MMP-2 expression has been detected in several lung cancer cell lines and surgical specimens and has been shown to modulate cell invasion and angiogenesis in lung cancer. 62 In the present study, treatment with SQE was associated with a decrease in the activity and expression of MMP-2, while the combination of SQE and CIS exhibited an additive effect for the suppression of MMP-2 in both lung cancer cell lines.
The health benefits of Sasa quelpaertensis leaves have been recognized, leading to its use as an alternative medicine. 63 Other extracts containing polysaccharides, lignin, chlorophyllin, and flavonoids have also been shown to exhibit strong antitumor activity and to provide protective effects for spontaneous mammary tumorigenesis.32,64 The bioactive compounds in SQE include polyphenol, p-courmaric acid, and tricin, and these are recognized by xenobiotic metabolizing enzymes to alter the metabolic activation of potential carcinogens.35,65 In a recent study, the anticancer activity of SQE was applied to the human colon cancer cell line, HCT116. It was hypothesized that the phenolics of SQE reduced the amount of cellular protein and the mitotic index during cell proliferation. 66 In the present study, the anticancer potential of SQE may be due to the combination of several bioactive compounds that are present in the extract. Therefore, further studies are needed to identify the activities of the individual bioactive components that affect cancer cell stemness and metastasis.
Conclusion
The present study has provided evidence that SQE exerts a novel and broad spectrum of anticancer effects by inhibiting cancer cell stemness and the invasive potential of NSCLCs. Furthermore, the combination of SQE and CIS enhanced these anticancer effects, and this may be due to an enhanced sensitivity of SQE-treated cells to cytotoxic agents. Fewer side effects induced by SQE may also play a role. Thus, SQE is identified as a potential tumor suppressive factor and cancer preventive food supplementation that requires further study to confirm these roles.
Footnotes
Acknowledgements
We thank Dr Hyun Cheol Kim of the Research Institute for Hallasan in the Jeju Special Self-Governing Province for his assistance with identifying the Sasa quelpaertensis species.
Authors’ Note
Mina Kim and Yoo-Sun Kim have equal contribution as the first author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
