Abstract
Hypotheses. Epithelial-mesenchymal transition (EMT) and invasion play a critical role in cancer progression and metastasis. We have shown that low E-cadherin and high Twist expression are significantly correlated with prognostic survival prediction in oral squamous cell carcinoma (OSCC). This study aimed to determine the anti-invasive effect of curcumin on the expression of matrix metalloproteinases (MMPs) and of EMT regulators in OSCC. Methods. SCC-25 cells were treated with curcumin, and cell proliferation, invasion, and expression of MMPs and EMT regulators were assessed for cell viability by trypan blue exclusion, for invasion by Matrigel invasion chamber, and for EMT regulators and MMP changes in the levels of proteins by immunoblotting. Results. Our data showed that curcumin treatment not only decreased the expression of MMP-2 and MMP-9 to inhibit invasiveness in oral cancer but also modulated the expression of EMT markers, such as Snail, Twist, and E-cadherin, and induced p53 expression that is crucial to EMT repression. Conclusion. Curcumin has the potential to become an adjunctive regimen for the prevention of cancer progression and metastasis in oral cancer.
Introduction
Oral cancer is the sixth most frequent cancer worldwide and has become a major concern, with the rising trend of incidence in young and middle-aged men.1,2 It is a leading cause of cancer-related death, ranked the fourth most prevalent carcinoma in Taiwan, and has the fastest increase in incidence among all cancers in adult male patients. 3 The most common subtype of oral cancer is oral squamous cell carcinoma (OSCC). 2 OSCC is a highly invasive malignant tumor with cervical lymph node metastasis.
Tumor invasion and metastasis represent a multistep process that depends on many factors associated with the proteolytic degradation of extracellular matrix components. Matrix metalloproteinase (MMPs), a family of zinc-dependent endopeptidases, also play a crucial role in extracellular matrix degradation associated with cancer cell invasion and metastasis. 4 Epithelial-mesenchymal transition (EMT) is a key step involved in the progression of primary tumors toward metastasis. Loss of E-cadherin expression is emerging as one of the most common indicators of EMT onset, and reduced expression of E-cadherin has been reported in various cancers, being associated with tumor progression and metastasis.5,6 The transcription factors Slug, Snail, and Twist have been described to be direct repressors of E-cadherin through an interaction of their COOH-terminal region with a 5′-CACCTG-3′ sequence in the E-cadherin promoter. 7 Emerging evidence shows that EMT regulators Slug, Snail, Twist, and E-cadherin play a crucial role in repressing cancer invasion and metastatic progression. Overexpression of Snail or Twist plays a distinct role in tumor progression by loss of E-cadherin expression and has also been identified in various kinds of epithelial tumors, including gastric cancer, 8 prostate cancer, 9 breast cancer, 10 and head and neck cancer. 11 Several studies have identified that reduced E-cadherin expression is associated with short overall survival in many epithelial tumors.12-16 Our previous study has shown that Snail and Twist are upregulated and E-cadherin is downregulated specifically in OSCC patients. Lower E-cadherin expression is an independent prognostic marker of OSCC patients. 17
Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione] is a natural polyphenolic compound derived from the root of Curcuma longa, which has been widely used for medical and other purposes. It has been already verified in many studies that curcumin has many medical characteristics, as can be demonstrated in animal experiments and molecular biological studies.18-21 Curcumin has anti-inflammatory properties through suppressing cyclo-oxygenase-2 (COX-2) and cytokines,22,23 prevents tumor metastasis by blocking matrix metalloproteinases (MMP-2 and MMP-9),24,25 and can regulate the activation of Bcl-2 and caspases, thus inducing cancer cells to an apoptotic program.26,27 In addition, curcumin can inhibit the transcriptional activation of nuclear factor κB and activating protein-1 to reduce inflammation and tumor hyperplasia.28,29 Recent studies showed that curcumin suppresses EMT/metastasis via FAK inhibition and E-cadherin upregulation in colorectal cancer30,31 and in liver cancer. 32 In this study, we aimed to determine the relationship between the anti-invasive activity of curcumin and the EMT markers Snail, Twist, and E-cadherin in human oral cancer. Furthermore, we found that curcumin regulates the expression level of EMT markers through induction of p53 expression.
Materials and Methods
Cell Culture and Treatment
OSCC cells (SCC25) were cultured in RPMI 1640 containing 2000 mg/L NaHCO3, supplemented with 10% fetal bovine serum (FBS), 20 units/mL penicillin, and 20 µg/mL streptomycin in an atmosphere of 5% carbon dioxide and 95% air at 37°C. 33 At about 90% confluence, the cells were trypsinized by treatment with trypsin-ethylenediaminetetraacetic acid (EDTA) and seeded in 6-well plates, grown to confluence, and rested in medium without FBS overnight.
siRNA Transfection
SCC25 cells were seeded (104 cells/well) in 6-well plates. After 48 hours of incubation, they reached 80% confluence and were transfected with siRNA oligos (p53-siRNA was purchased from Sigma-Aldrich, St Louis, MO; GAAAUUUGCGUGUGG AGUA[dT][dT] and UACUCCACACGCAAAUUUC[dT][dT]) and control siRNA in serum-free medium using LipofectAMINE 2000 (Invitrogen, Carlsbad, CA).
Cell Viability Assay
SCC25 cells were plated at 1 × 105 cells in 60-mm tissue culture dishes. After 24 hours of culture, cells were treated with curcumin (0, 2.5, 5, 10, 15 or 30 µM) as indicated then incubated at 37°C for 24 hours. Cells were collected by trypsinization and stained with trypan, blue and suspensions were counted in duplicate using a hemocytometer. Data were taken from averages of 3 independent experiments.
Invasion Assay
Invasion assays were carried out by methods based on others described previously, in modified Boyden’s chambers constructed with Falcon multiwell cell culture plates and Falcon cell culture inserts. Cell culture inserts were converted into invasion chambers by applying a layer of basement membrane onto the surface of microporous filters present in each unit. Briefly, 6.4-mm-diameter filters (8-µm pore) of Falcon cell culture inserts were coated with 25 µg/filter of reconstituted basement membrane Matrigel (Collaborative Biomedical Products, Bedford, MA). Matrigel was diluted to the desired concentration with ice-cold distilled water, applied to filters, dried overnight at room temperature, and reconstituted with RPMI for 90 minutes at room temperature. Uniformity of the coating was checked by Coomassie Blue staining and low-power microscopic observation. Exponentially growing SCC25 cells were harvested with 5 mM Na-EDTA in PBS (pH = 7.4); washed in RPMI + 10% FBS by centrifugation; resuspended in RPMI supplemented with 10% FBS, penicillin (20 units/mL), and streptomycin (20 µg/mL); and passed repeatedly through a 25-gauge needle to produce a single-cell suspension. After determination of the cell count and viability in a hemocytometer by the trypan blue exclusion test, the cells were added to the upper compartment of the modified Boyden’s chamber (5 × 104 cells/chamber). Fibronectin (16 µg/chamber) was placed in the lower compartment as a chemoattractant. At the beginning of the assay, curcumin (Sigma Chemical Co.), in portions of 0, 2.5, 5, 10, 15/µM, was added to the upper compartment. After treated cells had been incubated at 37°C for 5 days, cells that had invaded through the basement membrane into the lower chamber were fixed, stained with hematoxylin and eosin, and counted in 10 randomly selected microscopic fields (400×). Experiments were performed 3 times with 3 dishes, for each experimental condition each time.
Immunoblotting
The changes in intracellular protein molecules in the treated SCC25 cells were assessed by the methods described in a previous study. 34 Briefly, cells were washed with ice-cold phosphate-buffered saline and lysed in homogenization buffer (10 mM Tris–HCl at pH 7A4, 2 mM EDTA, 1 mM Ethylene glycol-bis(2-aminoethylether)-N,N,N,N’-tetraacetic acid (EGTA), 50 mM NaCl, 1% Triton X-100, 50 mM NaF, 20 mM sodium pyrophosphate, 1 mM sodium orthovanadate, and 1:100 proteinase inhibitor cocktail) on ice for 30 minutes. After centrifugation for 30 minutes at 12 000g at 4°C to remove insoluble materials, the lysate was determined for protein concentration by BCA protein assay kit, and the proteins were separated on SDS-PAGE. The resolved bands were electrotransferred to polyvinylidene fluoride (PVDF) membranes using a semidry blot apparatus (Bio-Rad) as in a previous report. 35 Immunoblotting was performed by incubating PVDF membranes with 5% nonfat milk in Tris-buffered saline supplemented with Tween 20 (Tri-buffered saline, with Tween 20, pH7.5(TSBT), 10 mM Tris, pH 7.4, 150 mM NaCl, 0A2% Tween 20) for 1 hour at room temperature for blocking the residue-free protein binding sites on PVDF. The membrane was incubated with different primary antibodies (β-actin, mouse anti-β-actin, monoclonal, Biovision; E-cadherin, mouse anti-E-cadherin, Sigma-Aldrich; Twist, mouse anti-Twist, monoclonal, Sigma-Aldrich; Snail, rabbit anti-snail, Sigma-Aldrich; MMP-2, goat anti-MMP-2, polyclonal, R & D; and MMP-9, rabbit anti-MMP-9, monoclonal, Epitomics) in 3% nonfat milk in TBST at 4°C for 18 hours. After repeating washing with TBST, the membrane was incubated with secondary antibodies (anti-mouse IgG, anti-Goat IgG, anti-Rabbit IgG, R & D) conjugated with Horseradish Peroxidase (HRP).
Statistical Analysis
All data are expressed as mean ± standard deviation, unless stated otherwise. The differences between groups were calculated using the Student’s unpaired t test. A P < .05 was regarded as statistically significant.
Results
Curcumin Inhibits the Proliferation of OSCC Cells
Curcumin has shown an inhibitory effect on proliferative activity in many cancers, including head and neck cancer. 36 SCC-25 cells were treated with different concentrations of curcumin. Indeed, the cell viability was decreased in a dose-dependent manner after curcumin treatment for 24 hours. At a concentration >15 µM, curcumin caused more than a 50% decrease in cell viability after the treatment (P < .05) when compared with untreated cells (Figure 1).

The dose-dependent response to curcumin in squamous cell carcinoma (SCC-25). Cells were treated with increasing concentrations of curcumin, as indicated, then incubated at 37°C for 24 hours. The viable cells were stained with trypan blue and counted under a microscope. The cell viability was expressed as a percentage of untreated cells. Data are taken from the averages of 3 independent experiments and expressed as means ± standard deviation.
Curcumin Suppresses the Invasive Ability of OSCC Cells
Tumor cell invasion and EMT are important to evaluate cancer aggressive and metastatic potential. Therefore, the ability of curcumin to suppress the invasion ability of OSCC cells was investigated. Curcumin (0-15 µM) was used for a standard invasion assay by being added to the upper compartment of the invasion chamber. SCC-25 cells penetrating into the lower chamber were significantly decreased after the treatment of curcumin in a dose-dependent manner; 10 µM curcumin led to a reduction in the invasiveness of SCC-25 cells by up to 95% (Figure 2). In addition, we assessed the expression of MMP-9 and MMP-2 in the curcumin-treated SCC-25 cells. Immunoblotting analysis showed that the levels of MMP-2 and MMP-9 decreased in a dose-dependent manner when the cells were treated with curcumin (Figure 3). These results indicated that curcumin inhibited the invasiveness of OSCC cells, which may be through a decrease in the level of MMP-9 and MMP-2.
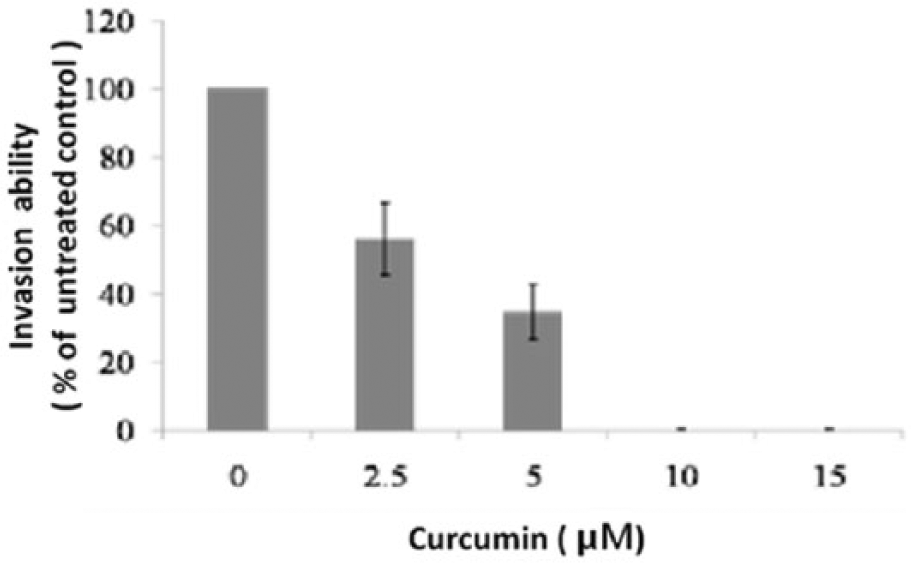
Effect of curcumin on invasive ability in squamous cell carcinoma (SCC-25): 5 × 104 cells/well were incubated at 37°C for 24 hours with or without curcumin (2.5-15 µM). The cells penetrating into the lower chamber were stained and counted under a microscope. Data are expressed as the percentage of the control response (mean ± standard error).

Effect of curcumin on the expression of matrix metalloproteinase (MMP)-9 and MMP-2 in squamous cell carcinoma (SCC-25) cells: SCC-25 cells (1 × 106) were treated with curcumin (0-15 µM) then cultured for 24 hours. Cells were then harvested and the proteins separated by SDS-PAGE, followed by immunoblotting with anti-MMP-9 and MMP-2 antibodies, respectively. The images are representative of the results of 3 independent experiments.
Curcumin Inhibits the EMT of OSCC Cells by Affecting the Expression of Snail, Twist, and E-cadherin
To understand whether curcumin inhibits the EMT of OSCC cells, we aimed to determine the expression level of EMT markers, Snail, Twist, and E-cadherin, in SCC-25 cells under curcumin treatment. Treated with different concentrations of curcumin, the expression amounts of Snail and Twist was decreased, whereas that of E-cadherin, an epithelial marker, prominently increased in a dose-dependent manner in SCC-25 cells (Figure 4). In addition, we found that the amount of p53 protein that responded to curcumin treatment showed the same trend as E-cadherin: both increased in a dose-dependent manner (Figure 4). This observation is consistent with our previous findings that p53 expression positively correlates with E-cadherin expression. This correlation could be a valuable marker for predicting OSCC patient outcome. 16

Effect of curcumin on the expression of p53, Snail, Twist, and E-cadherin in squamous cell carcinoma (SCC-25) cells. SCC-25 cells (1 × 106) were treated with curcumin (0-15 µM), then cultured for 24 hours. Cells were then harvested and the proteins separated by SDS-PAGE, followed by immunoblotting with anti-P53, E-cadherin, Snail, and Twist antibodies, respectively. The images are representative of the results of 3 independent experiments.
p53 Is Required for the Inhibition of EMT by Curcumin in OSCC Cells
To investigate whether curcumin-induced p53 expression is important in regulating the expression level of EMT markers, we first inhibited endogenous p53 expression by a siRNA method to check their expression. E-cadherin expression was decreased, and Snail was increased when SCC25 cells were treated with p53-siRNA (Figure 5), suggesting that p53 plays an important role in the regulation of the EMT process. Consistently, we found that the inhibition of p53 inhibits the expression of E-cadherin and increases that of Snail in curcumin-treated SCC-25 cells (Figure 5). These data indicate that p53 is important in the inhibition of the EMT process, and EMT repression by curcumin is partly through a p53-dependent pathway.
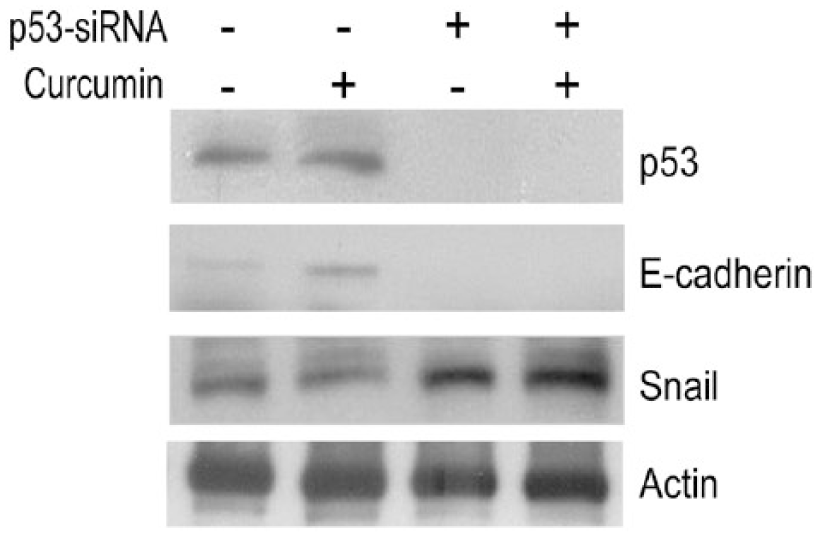
Effect of p53 inhibition on the expression of E-cadherin and Snail in squamous cell carcinoma (SCC)-25 cells. SCC-25 cells (1 × 106) were treated with p53 siRNA (30 nM) or p53 siRNA combination with curcumin (10 µM), as indicated. After 24 hours of transfection with siRNA, SCC-25 cells were treated with curcumin for 24 hours. SCC-25 cells without treatment with p53 siRNA were also treated with negative control siRNA. Cells were then harvested and the proteins separated by SDS-PAGE, followed by immunoblotting with anti-p53, E-cadherin, and Snail antibodies, respectively. The images are representative of the results of 3 independent experiments.
Discussion
In this study, we investigated the anti-invasive effect of curcumin on the expression of EMT markers and MMP-2 and MMP-9 in OSCC cells. Consistently, we also observed that curcumin has antiproliferation and anti-invasion effects on SCC-25 cells. Our results indicate that curcumin inhibits invasion by a decrease of Snail, Twist, MMP-2, and MMP-9 and an increase of E-cadherin in SCC-25 cells. The mechanism underlying the effect of curcumin on EMT was further investigated. We found that curcumin induces p53 expression, which is important in regulating the expression levels of EMT markers, such as Snail, Twist, and E-cadherin.
Substantial evidence has shown that curcumin inhibits proliferation, migration invasion, and metastasis in multiple malignancies, including head and neck cancer.36,37 Our results showed that curcumin inhibits the invasion by decreasing the level of MMP-2 and MMP-9, which is in agreement with the previous studies showing that curcumin inhibits metastasis by targeting MMP-2 and MMP-9.38,39 In addition, we and other groups40,41 observed that curcumin induces the expression level of E-cadherin, an important EMT marker. EMT, a process characterized by loss of epithelial cell-cell adhesions, has been frequently extended to the phenotypical changes of increased motility and invasiveness of cancer cells.6,42 Twist and Snail are considered the major transcription factors modulating EMT in various cancer types by repressing E-cadherin transcription.7,43 We further showed that curcumin inhibits the expression of Snail and Twist in SCC-25 cells, suggesting that curcumin shows its anti-invasive effect through modulating the EMT process.
We then found that curcumin induces both expression of E-cadherin and p53 with the same trend. Our previous study showed that p53 expression positively correlates with E-cadherin expression in tissue from OSCC patients. 17 The emerging evidence showed that p53 plays a critical role in repressing cancer invasion and metastasis.34,44 These data suggest that p53 could be important in modulating the EMT process. Indeed, curcumin rescues the expression of p53 and modulates p53-dependent expression of Snail and Twist in SCC-25 cells. p53 Has the ability to bind with Snail and induces Snail degradation through MDM2-mediated ubiquitination 45 or increasing microRNA-34 expression. 46 Ectopic expression of wild-type p53 upregulated the expression of E-cadherin and repressed the expression of vimentin and ZEB2 in cancer cells.47,48 Moreover, p53 suppressed EMT in lung cancer by inducing the MDM2-mediated degradation of Slug, a member of the Snail family.48,49 It is interesting to note that we found that p53 plays an important role in Twist degradation in SCC-25 cells. We hypothesized that p53 increases the expression of E-cadherin by targeting, in part, Twist degradation in OSCC-25 cells. This evidence supports the involvement of p53 in the modulation of the EMT pathway. We showed that curcumin inhibits the EMT pathway and modulates p53-dependent degradation of Snail and Twist in SCC-25 cells. These results indicate that curcumin may inhibit EMT via p53-dependent signaling in SCC-25 cell lines. The mechanism underlying the effect of curcumin on the p53–E-cadherin pathway in EMT warrants further investigation.
Conclusion
Curcumin treatment not only decreases MMP-2 and MMP-9 expression to inhibit invasiveness but also rescues p53 expression and decreases the expression of Twist and Snail to prevent EMT of oral cancer cells. Curcumin has the potential to become an adjunctive regimen for the prevention of metastasis in oral cancer.
Footnotes
Authors’ Note
To date, the results of this research have not been reported or presented elsewhere.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Ministry of Science and Technology (MOST103-2320-B-400-022-MY3; MOST102-2627-M-400-001), Ministry of Health and Welfare (MOHW103-TDU-212-114005), and National Health Research Institutes (103A1-CA-PP-07) in Taiwan to A Y-L Lee. This work was also supported by grants from Mackay Memorial Hospital and Yuanpei University (102-COMP-05), Taiwan, to T-Y Kao.
