Abstract
Total alkaloids is an active ingredient of the natural plant
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignancies with a high mortality rate, especially in China, due to the prevalence of hepatitis B virus infection. 1 Although surgery is the most effective and widely performed treatment for HCC, it is still unsuitable for many patients with large tumor size, severe hepatic cirrhosis, or inoperable anatomical location of the tumor.2,3 Therefore, drugs such as doxorubicin, cisplatin, and gemcitabine are the most frequently administered chemotherapeutic drugs for treating HCC.4-7 Unfortunately, the patient response to these regimens is less than 20% due primarily to drug resistance. 8 Thus, approaching treatment of HCC through alternative means and natural products may provide less toxic and effective options and, importantly, new hope for afflicted patients. No matter what the course of treatment is, targeting support mechanisms for tumorigenesis and growth will yield the most effective results.
Angiogenesis, the formation of microvasculature, is important in metabolic function and solid tumor growth, facilitating tumor invasion and metastasis.9,10 Tumor angiogenesis is controlled by angiogenic factors directly induced from tumor cells, as well as by factors derived indirectly from surrounding stromal tissues. Several growth factors have angiogenic activity, and one of the best known is vascular endothelial growth factor (VEGF), a highly specific factor for endothelium and vascular permeability. VEGF-A is highly expressed in and secreted from a wide variety of human cancers and is associated with cancer progression, invasion and metastasis, and poor patient prognosis.11-13 VEGF-A primarily interacts with specific receptors on the surface of vascular endothelial cells,11,14 thus stimulating tyrosine kinase signaling and subsequent endothelial cell proliferation, migration, survival, sprouting, and eventually tube formation. As angiogenesis may play a key role in the progression of solid tumor growth, blocking this process has become a major focus of anticancer drug development.
Materials and Methods
Materials and Reagents
Roswell Park Memorial Institute Medium 1640 (RPMI 1640), Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), penicillin–streptomycin, trypsin-EDTA, and Trizol Reagent were purchased from Life Technologies (Carlsbad, CA). Cell cycle assay kit was purchased from BD Biosciences (San Jose, CA). SuperScript II reverse transcriptase was obtained from Promega (Madison, WI). An in vitro angiogenesis assay kit was purchased from Millipore (Billerica, MA). Human VEGF-A ELISA kit was obtained from R&D Systems (Minneapolis, MN). All other chemicals, unless otherwise stated, were obtained from Sigma Chemicals (St Louis, MO). The roots of
Preparation and Content of Total Alkaloids of Rubus alceifolius Poir
TARAP was prepared according to previously described methods.
18
On identification and authentication, alkaloids were extracted and purified as reported.
18
Briefly, the roots of
Cell Culture
Human umbilical vein endothelial cells (HUVECs) and human hepatocellular carcinoma HepG2 cells were obtained from American Type Culture Collection (ATCC, Manassas, VA). HUVECs or HepG2 cells were grown in RPMI 1640 or DMEM, respectively. Both RPMI 1640 and DMEM were supplemented with 10% (v/v) FBS and 100 Units/mL penicillin and 100 µg/mL streptomycin. All cell lines were cultured at 37°C in 5% CO2 under humidified environment.
Chick Chorioallantoic Membrane (CAM) Assay
A CAM assay was performed to determine the anti-angiogenic activity of TARAP in vivo. Briefly, 10 µL of TARAP (100 µg/µL) was loaded onto a 0.5 cm diameter Whatman filter paper. The filter was then applied to the CAM of a 7-day embryo. After 72-hour incubation at 37°C, angiogenesis around the filter was photographed with a digital camera. The number of blood vessels located within a circular perimeter surrounding the implants (distance of 0.25 cm from the edge of the filter) were manually quantified. Assays were performed twice, containing a total of 10 eggs for each data point. The animal studies were approved by the Fujian Institute of Traditional Chinese Medicine Animal Ethics Committee (Fuzhou, China). The experimental procedures were carried out in accordance with the Guidelines for Animal Experimentation of Fujian University of Traditional Chinese Medicine (Fuzhou, China).
Evaluation of Cell Viability
Cell viability was assessed by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) colorimetric assay. HUVECs cells were seeded into 96-well plates at a density of 1 × 104 cells/well in 0.1 mL medium. The cells were treated with various concentrations of TARAP for 24 hours, 48 hours, and 72 hours. At the end of treatment, 10 µL MTT (5 mg/mL in phosphate buffered saline [PBS]) were added to each well, and samples were incubated for an additional 4 hours at 37°C. The purple-blue MTT formazan precipitate was dissolved in 100 µL DMSO and absorbance was measured at 570 nm with an ELISA reader (BioTek, Model ELX800). Sorafenib was used as a positive control. HUVECs cells were seeded into 96-well plates at a density of 1 × 104 cells/well in 0.1 mL medium. The cells were treated with various concentrations of sorafenib for 24 hours. At the end of treatment, 10 µL MTT (5 mg/mL in PBS) was added to each well, and samples were incubated for an additional 4 hours at 37°C. The purple-blue MTT formazan precipitate was dissolved in 100 µL DMSO, and absorbance was measured at 570 nm with an ELISA reader (BioTek, Model ELX800) and the IC50 calculated.
Cell Cycle Analysis
HUVECs were harvested and adjusted to a concentration of 1 × 106 cells/mL following incubation with various concentrations of TARAP for 24 hours. Cell cycle progression of HUVECs was determined by flow cytometry analysis with a propidium iodide (PI) staining cell cycle assay kit (BD Biosciences). Staining was performed according to the manufacturer’s instructions. The fluorescent signal was detected through the FL2 channel, and the proportion of DNA in different phases of the cell cycle was determined using ModfitLT Version 3.0 (Verity Software House, Topsham, ME).
Wound-Healing Cell Migration Assay
HUVECs were seeded into 12-well plates at a density of 2 × 105 cells/well in 1 mL medium. After 24 hours of incubation, cells were scraped away vertically in each well using a P100 pipette tip. Three randomly selected views along the scraped line were photographed in each well using a phase-contrast inverted microscope at a magnification of 100×. Cells were then treated with indicated concentrations of TARAP for 24 hours, and another set of images were taken. A reduction in the scraped area indicated cellular migration.
Capillary-Like Tube Formation Assay
HUVEC tube formation was examined using the ECMatrix assay kit (Millipore), following the manufacturer’s instructions. Briefly, HUVECs were incubated with indicated concentrations of TARAP for 24 hours, and the cells were harvested and diluted (1 × 104 cells) in 50 µL of medium. Next, the harvested cells were seeded onto 1:1 ECMatrix gel (v/v) coated 24-well plates and incubated for 9 hours at 37°C. Cellular morphology and capillary tube network development were evaluated using a phase-contrast inverted microscope. Photographs were taken at a magnification of 100×. The extent of tube formation was quantified by calculating the length of tubes in 3 randomly chosen fields from each well.
RNA Extraction and RT-PCR Analysis
A total of 2 × 105 HUVECs or HepG2 cells were seeded into 6-well plates in 2 mL medium and treated with indicated concentrations of TARAP for 24 hours. Total RNA was isolated with Trizol Reagent (Invitrogen). Oligo(dT)-primed RNA (1 µg) was reverse-transcribed with SuperScript II reverse transcriptase (Promega) according to the manufacturer’s instructions. The obtained cDNA was used to determine the amount of VEGF-A mRNA by polymerase chain reaction (PCR). GAPDH was used as an internal control.
Measurement of VEGF-A Secretion by ELISA
A total of 2 × 105 cells were seeded into 6-well plates in 2 mL medium and treated with various concentrations of TARAP for 24 hours. Cell culture media were collected from cultures of both HUVECs and HepG2 cells to measure the secretion level of VEGF-A. Measurement was performed using Quantikine ELISA kits (R&D) according to the manufacturer’s instructions.
Statistical Analysis
All data are presented as the means of 3 determinations. Dr Zhiyun Cao performed statistical analysis for this study. The data were analyzed using the SPSS software package for Windows (Version 11.5). Statistical analysis was performed with 2-sample Student’s
Results
TARAP Inhibited Angiogenesis in a CAM Model
We tested the anti-angiogenic effect of TARAP in vivo using a classic chick chorioallantoic membrane (CAM) model. We observed that TARAP treatment significantly reduced the total number of blood vessels in the chicken embryos as compared with untreated controls (Figure 1A and B), indicating that TARAP suppressed angiogenesis in vivo (Supplemental Figure 1; available online at http://ict.sagepub.com/supplemental).

Effect of TARAP on the angiogenesis of chick chorioallantoic membrane (CAM). (A) TARAP treatment significantly reduced the total number of blood vessels in the chicken embryos. Assays were performed twice, containing a total 10 eggs for each data point. Images are representative photographs. (B) Quantification of CAM assay.
TARAP Inhibited HUVEC Proliferation
To explore potential influence of TARAP on HUVEC proliferation, an MTT assay and cell morphology analysis were performed. As shown in Figure 2, treatment with 0.25 to 1 mg/mL of TARAP for 24, 48, or 72 hours reduced cell viability by 6% to 61%, 30% to 70%, or 63% to 85%, respectively, compared to untreated control cells (

TARAP decreased HUVEC viability. An MTT assay indicated that TARAP treatment significantly reduced HUVEC viability in both dose- and time-dependent manners. Data are means with SD (error bars) from at least 3 independent experiments. *, #, &

TARAP’s effects on HUVEC morphology. HUVECs were treated with various concentrations of TARAP for 24 hours and morphological changes were observed using phase-contrast microscopy. Photographs were taken at a magnification of 100×. Images are representative of 3 independent experiments.
TARAP Blocked Cell Cycle Progression of HUVECs
The effect of TARAP on G1 to S progression of the cell cycle in HUVECs was examined via PI staining followed by FACS analysis. As shown in Figure 4, the percentage proportion of S-phase cells following treatment with 0, 0.25, 0.5, 0.75, and 1.0 mg/mL of TARAP was 40.59 ± 4.96%, 32.04 ± 5.26%, 28.96 ± 4.49%, 23.13 ± 3.51%, and 20.03 ± 3.09%, respectively (Figure 4B;
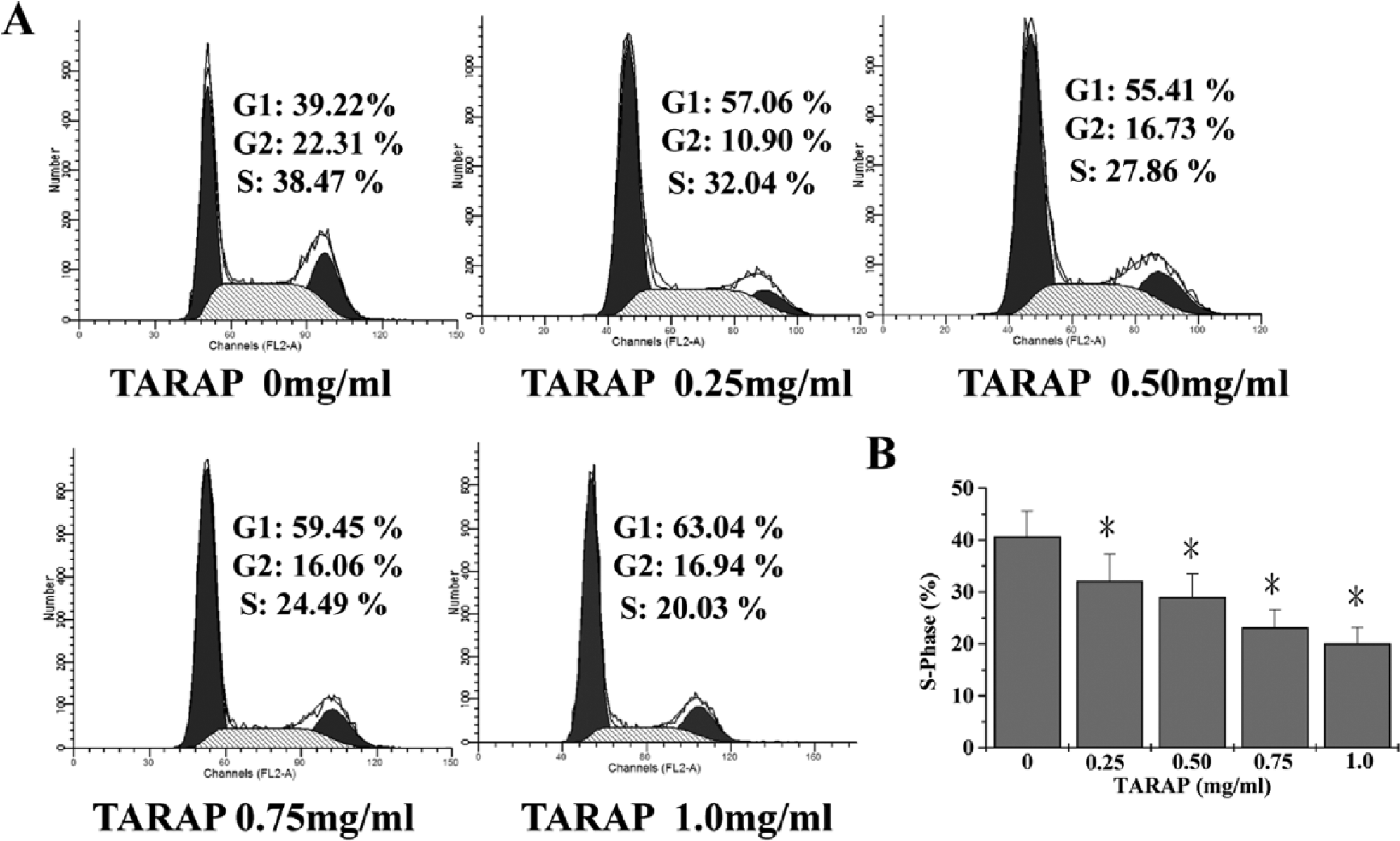
TARAP downregulated cell cycle progression of HUVECs. (A) FACS analysis with PI staining showed that TARAP treatment dose-dependently reduced the percentage proportion of S-phase cells. (B) Quantification of FACS analysis. Data shown are means with SD (error bars) from 3 independent experiments.
HUVEC Migration and Tube Formation Was Inhibited by TARAP
Endothelial cell migration was evaluated in a scratch wound-healing assay. As demonstrated in Figure 5, after wounding for 24 hours, untreated HUVECs migrated into the wounded (cleared) area of the cell monolayer, whereas TARAP treatment dose-dependently inhibited this migration. To assess whether TARAP affected endothelial capillary tube formation, HUVECs were grown on a solid gel containing mouse basement proteins in which cultured endothelial cells can rapidly align and form hollow tube-like structures. As shown in Figure 6A and B, untreated HUVECs formed elongated tube-like structures, whereas TARAP treatment significantly decreased capillary tube formation in a dose-dependent fashion.

TARAP reduced HUVEC migration. Data from a wound-healing assay showed that TARAP treatment dose-dependently inhibited HUVEC migration. The photographs were taken at a magnification of 100×. Images are representative of 3 independent experiments.

Tube formation was prohibited by TARAP treatment. (A) TARAP treatment significantly decreased capillary tube formation of HUVECs in a dose-dependent fashion. (B) Quantification of tube formation analysis. Data shown are means with SD (error bars) from 3 independent experiments.
TARAP Suppressed Expression of VEGF-A in Both HepG2 Cells and HUVECs
VEGF-A mRNA and protein expression were determined by RT-PCR and ELISA analysis, respectively, in both HepG2 human hepatoma cells and HUVECs. RT-PCR results showed that TARAP profoundly reduced VEGF-A mRNA expression in both cell types, with an apparent dose-dependent downregulation (Figure 7). Data from the ELISA assay showed that VEGF-A protein expression was significantly reduced by TARAP at all doses compared to untreated controls (Figure 8).
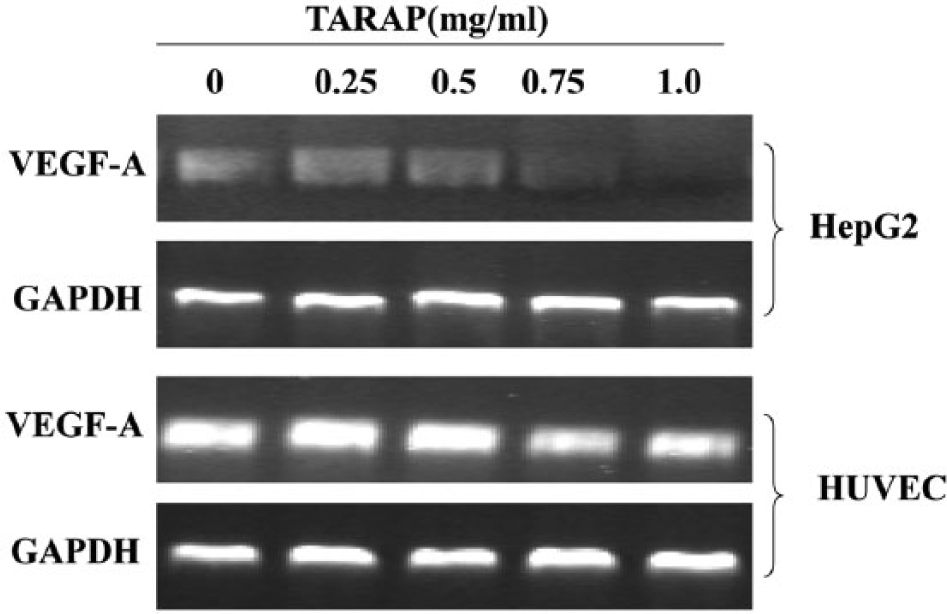
TARAP reduced VEGF-A mRNA expression. RT-PCR analysis indicated that TARAP treatment profoundly reduced VEGF-A mRNA expression in both HepG2 human hepatocellular carcinoma cells and HUVECs, with a dose-dependent manner. GAPDH was used as an internal control. Data are representative of 3 independent experiments.

TARAP decreased VEGF-A protein expression. ELISA assay showed that TARAP treatment dose-dependently suppressed the protein expression of VEGF-A in both HepG2 cells and HUVECs. Data are means with SD (error bars) from at least 3 independent experiments. *
Discussion
Angiogenesis from preexisting vasculature occurs through complex processes and extensive interplay between cells, soluble factors, and the extracellular matrix, leading to proliferation and migration of endothelial cells for vessel formation. Although angiogenesis is essential for normal physiological processes, it also plays an important role in the development of cancer. 19 As such, potential anti-angiogenic therapies may be valuable for cancer treatment; however, blocking angiogenesis for anticancer therapy is complicated as many inhibitors are given systemically and target more than vasculature of tumors. For example, administration of angiogenesis inhibitors can cause cardiovascular complications due to their intrinsic cytotoxicity against non-tumor-associated endothelial cells. Furthermore, multiple signaling pathways are involved in tumor angiogenesis, and inhibitors targeting a single pathway may be insufficient and promote resistance. Traditional Chinese medicine (TCM) formulations, including natural plants and extracts, involve a complex combination of many natural products, each of which contains numerous chemical compounds. Therefore, TCM formulas and natural plants therapies are considered as multicomponent and multitarget treatments exerting a more holistic therapeutic function. Importantly, such treatments often have few side effects and minimal toxicity.
In this study, we have shown for the first time that TARAP inhibits both blood vessel formation in an in vivo CAM model and inhibits the proliferation of HUVECs in vitro, thus demonstrating its potential as an anti-angiogenic therapy. Solid tumors induce neovascularization, and the resultant angiogenesis stimulates tumor growth and metastasis.19-21 Angiogenesis typically proceeds through key endothelial cell activities including proliferation, migration, and capillary tube formation. Here we showed TARAP could inhibit CAM angiogenesis, and inhibited the proliferation of HUVECs in both dose- and time-dependent manners. Generally, HUVECs only express low level of VEGF-A, though in this study, the content of HUVEC-A was 1775.7 pg/mL. Therefore, maybe the HUVEC cells were cultured for too many generations and some unknown influence stimulated the cells to secrete higher levels of VEGF.
Eukaryotic cell proliferation is primarily regulated by the cell cycle, and inhibition of the cell cycle could control the growth of HUVECs and further inhibit the formation of tumor vasculature. The G1/S transition is 1 of the 2 main checkpoints in the regulation of cell cycle progression and, thus, cell proliferation. PI staining and FACS analysis demonstrated that TARAP treatment dose-dependently blocked HUVEC G1 to S progression, suggesting its anti-proliferative effects likely result from cell cycle arrest. A wounding-healing assay and ECMatrix culture system revealed that TARAP prohibited migration and capillary tube formation of HUVECs in a dose-dependent fashion. During tumor growth, interaction between tumor cells and vascular endothelial cells leads to considerable increase in tumor vascularization, which promotes further tumor growth through paracrine action.22,23 VEGF is a multifunctional cytokine commonly expressed in a wide variety of human tumor cells and is associated with angiogenesis, tumor growth, and metastasis. 24 In the present study, TARAP downregulated expression of VEGF-A in both HepG2 human HCC cells and HUVECs, suggesting its potential involvement in TARAP’s anti-angiogenic mechanism. Further research is necessary to clarify VEGF’s role in TARAP-mediated blockade of angiogenesis and other details of this natural therapeutic action.
In summary, this study demonstrated the ability for TARAP to inhibit tumor angiogenesis via suppressing microvessel formation, HUVEC cell replication, and reduce VEGF expression. These results improve our understanding of the molecular basis of TARAP in hepatoma growth and angiogenesis, which will be useful for rational design for future cancer prevention and therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
