Abstract
Background. The anomalous fruits of Gleditsia sinensis Lam. (Leguminosae), a crude drug in China, have long been used in traditional Chinese medicine for the treatment of various diseases. The saponin fraction isolated from the fruits (SFGS) is considered as the active component for the antitumor activity of this crude drug. Objectives. The present study was performed to investigate the anti-angiogenesis activities and active constituents of SFGS. Methods. Human umbilical vein endothelial cells (HUVECs) were treated with SFGS in the presence or absence of basic fibroblast growth factor (bFGF) in vitro. The proliferation, migration, and tube formation were studied by MTT, Transwell, and 2D Matrigel assays, respectively. The cell cycle and apoptosis were analyzed by flow cytometry. Enzyme-linked immunosorbent assay for protein expression of vascular endothelial growth factor (VEGF) and western blot analysis for caspase-3, caspase-8, and caspase-9 as well as Fas were performed. In addition, the effects of 13 saponin compounds isolated from SFGS on the tube formation of HUVECs were screened, and the structure–activity relationships were discussed. Results. SFGS, at concentrations (1, 3, and 10 µg/mL) without significant cytotoxicity on endothelial cells, significantly inhibited the proliferation, migration, and tube formation of HUVECs induced by bFGF (10 ng/mL). It moderately arrested the cell cycle to G1 phase but greatly induced cell apoptosis and increased the expressions of caspases-3, caspase-8, and Fas but not caspase-9 in HUVECs. Moreover, SFGS did not affect the bFGF-induced autosecretion of VEGF from endothelial cells. Among the 13 saponin compounds tested, gleditsiosides B, I, J, O, and Q showed inhibition of the tube formation at a concentration of 3 µM, and only gleditsioside B exerted significant inhibition at 1 µM. Conclusion. SFGS is substantially able to prevent angiogenesis by interfering with multiple steps. The findings provide a new explanation for the antitumor effects of G sinensis fruits. Gleditsiosides B, I, J, O, and Q are probably the main active constituents of SFGS.
Introduction
Angiogenesis, the development of new blood vessels from preexisting ones, is a normal physiologic phenomenon associated with embryonic development, wound healing, and tissue regeneration. However, especially when unabatedly continuing for months or years, it contributes to many diseases such as solid and liquid (leukemia) tumors as well as rheumatoid arthritis. 1 Establishment of new blood vasculature is a key step for tumor growth and metastasis, and interference with tumor angiogenesis has long been considered as a promising therapeutic strategy. Moreover, synovial angiogenesis is essential for the maintenance and progression of chronic inflammation, hyperplasia, edema, and invasiveness in rheumatoid arthritis.2,3 More interestingly, stage-specific forms of anti-angiogenic therapy may provide a possibility that retards or blocks the growth of tumors early in their evolution before they acquire metastatic potential. 4
Gleditsia sinensis Lam. (Leguminosae), a perennial shrub, is widely distributed throughout China. “Zhu-Ya-Zao,” the anomalous fruits of G sinensis, has long been applied in traditional Chinese medicine for the treatment of various diseases. Previously, we demonstrated that the saponin fraction isolated from the anomalous fruits of G sinensis (SFGS) could ameliorate inflammation and joint destruction of collagen-induced arthritis in mice. 5 Other researchers showed that the ethanol extract and saponin compounds from the fruits had antitumor effects.6-10 These findings attract us to study the anti-angiogenesis potential, underlying mechanisms, and main active constituents of SFGS.
Materials and Methods
Chemicals
M199 medium and enzyme-linked immunosorbent assay (ELISA) kit of vascular endothelial growth factor (VEGF) were purchased from Invitrogen (Carlsbad, CA), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) was purchased from Sunshine Biotechnology (Nanjing, China). Basic fibroblast growth factor (bFGF) was from PeproTech (London, England), and new born calf serum (NBCS) was from PAA (Linz, Austria). Dimethylsulfoxide (DMSO) and paraformaldehyde were from Jiangsu Yonghua Fine Chemical Limited (Nanjing, China). Matrigel was from BD (Franklin Lakes, NJ). Propidium iodide (PI) and RNaseA were from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Enhanced chemiluminescence (ECL) reagent and monoclonal antibodies against caspase-3 (D146), caspase-8 (T341), caspase-9 (G140), as well as Fas (L315) were obtained from Bioworld (Minneapolis, MN). Annexin V-FITC apoptosis detection kit and Hoechst 33258 detection kit were obtained from KeyGEN Bio Co Ltd (Nanjing, China). Other reagents were from Sinopharm Chemical Reagent Co Ltd (Nanjing, China).
Preparation of SFGS and Its Constituents
SFGS and 13 kinds of triterpenoid saponin compounds were prepared by the authors as described previously.5,11,12 These saponins, with purities >98% as determined by high-performance liquid chromatography analysis, included 9 oleanane-type ones (gleditsioside A, gleditsioside B, gleditsioside D, gleditsioside I, gleditsioside N, gleditsioside O, gleditsioside Z, pitheduloside C, and vitalboside A) and 4 echinocystic acid–type ones (pitheduloside A, pitheduloside E, gleditsioside J, and gleditsioside Q). Oleanolic acid, a sapogenin, was also used as a control. They were dissolved in DMSO just before use, and the terminal concentration of DMSO was below 1‰.
Chemical characteristics of SFGS were analyzed by selected ion monitoring chromatogram. SFGS (1 g) was obtained with 20 mL of methanol under ultrasonication for 30 minutes. The solution was filtered with membrane filter (0.45 µm) and then subjected to liquid chromatography–mass spectrometry (LC-MS) analysis using an Agilent 1100 series liquid chromatography (Agilent Technologies, Palo Alto, CA). A method was developed using a Shim-pack CLC-ODS column (150 × 4.6 mm id, 5 µm). The binary gradient elution system consisted of water (A) and acetonitrile (B), and separation was achieved using the following gradients: 0 to 10 minutes, 27% B; 10 to 20 minutes, 27% to 35% B; 20 to 26 minutes, 35% to 40% B; 26 to 33 minutes, 40% to 45% B; 33 to 38 minutes, 45% to 48% B; 38 to 40 minutes, 48% to 60% B. The column temperature was kept constant at 30°C, and the flow rate was 1 mL/min. An Agilent SL G1946D single quadruple mass spectrometer with an ESI source in negative mode was used as the detector and conditions of MS analysis were as follows: drying gas (N2) flow rate, 9 L/min; drying gas temperature, 320°C; nebulizing gas (N2) pressure, 16 psi; capillary voltage, 3500 V. The samples were analyzed in selective ion monitoring (SIM) mode by monitoring the molecular ions [M-H]−. Eight saponins (gleditsioside A, gleditsioside B, gleditsioside D, gleditsioside I, gleditsioside J, gleditsioside N, gleditsioside O, and pitheduloside C) with relatively higher content in SFGS were identified (Figure 1).

LC-MS chromatogram of SFGS.
Cell Culture
Human umbilical vein endothelial cells (HUVECs) were obtained as described previously with a slight modification. 13 Cells were routinely cultured in M199 medium supplemented with 20% NBCS (v/v), penicillin (60 U/mL), streptomycin (100 U/mL), and L-glutamine (2 mM) at 37°C in a humidified atmosphere containing 5% CO2. The cells were used between passages 3 and 6.
VEGF-Level Analysis
VEGF levels in the supernatants of HUVECs were measured using ELISA kits according to the manufacturer’s recommendations.
Cell Proliferation Assay
The cell proliferation was evaluated by MTT assay. 14 In brief, HUVECs were plated into 96-well plates (8 × 103 cells/well) for 24 hours and were synchronized and exposed to SFGS in the presence or absence of bFGF (10 ng/mL) for 72 hours. Aliquots (20 µL) of MTT (5 mg/mL) were added to each well, followed by incubation for an additional 4 hours at 37°C. The supernatant was carefully removed, and DMSO (150 µL) was added into each well to dissolve the formazan crystals. The absorbance at 570 nm was read using Model 1500 Multiskan spectrum microplate reader (Thermo, Waltham, MA).
Cell Cycle Analysis
HUVECs were allowed to grow to 90% confluence in cell culture flasks. Following treatment with SFGS for 24 hours, cells were exposed to bFGF (10 ng/mL) for another 24 hours. Cells were trypsinized, and aliquots of 1 × 106 cells were fixed overnight in 70% ethanol at 4°C. Fixed cells were washed and suspended in 1 mL phosphate-buffered saline (PBS; containing 50 mg PI and 1 mg RNaseA) and then incubated for 30 minutes in the dark at room temperature. Cell cycle was analyzed using a FACSCalibur flow cytometer (BD, Franklin Lakes, NJ).
Apoptosis Detection
Hoechst 33258 staining was performed using in situ cell death detection kit according to the manufacturer’s instructions. 15 Briefly, after treatment with various concentrations of SFGS for 24 hours, HUVECs were stimulated with bFGF (10 ng/mL) for 24 hours. The cells were washed with PBS and fixed in 4% paraformaldehyde, followed by Hoechst 33258 staining for 15 minutes in the dark. The fluorescence staining was subsequently monitored at a magnification of 200× with an Olympus IX51 inverted microscope (Olympus Corporation, Tokyo, Japan).
Apoptosis rate was measured using an Annexin V-FITC apoptosis detection kit according to the manufacturer’s recommendation. HUVECs were harvested, washed twice with PBS, and resuspended in 0.5 mL of binding buffer. Then, they were incubated with Annexin V-FITC (5 µL) and PI (5 µL) at 25°C for 15 minutes in the dark and were analyzed by flow cytometry.
Western Blot Analysis
As described previously, 16 after exposure to SFGS for 24 hours, HUVECs were treated with bFGF (10 ng/mL) for 9 hours. Subsequently, cells were washed and lysed, followed by centrifugation at 15 000g for 5 minutes. Samples (30-50 µg of protein) were separated in 10% sodium dodecyl sulfate-polyacrylamide gel (SDS-PAGE) and transferred to nitrocellulose membranes (Millipore, Billerica, MA). The membranes were blocked with 10% nonfat milk in PBS–Tween (0.1%; PBST) for 2 hours and then incubated with monoclonal antibodies against human caspase-3, caspase-8, caspase-9, and Fas in PBST containing 5% bovine serum albumin for 2 hours. After washing 3 times with PBST, the membrane was hybridized with secondary antibody conjugated to horseradish peroxidase for 1 hour, and again washed with PBST for 3 times. The membrane was incubated with ECL reagent for about 2 minutes and exposed to X-ray film. The bands were semiquantified using Image-Pro Plus software (Media Cybernetics, Bethesda, MD).
Cell Migration Assay
Cell migration assay was performed in 24-well Transwell plates (Millipore, Billerica, MA) as previously described with a slight modification. 17 HUVECs were pretreated with indicated concentrations of SFGS for 24 hours and then detached and resuspended in M199 medium (containing 2% NBCS). An aliquot (200 µL) of cells was plated into the upper chamber of the Transwell, while the lower chamber was filled with 600 µL of M199 medium with 2% NBCS in the presence or absence of bFGF (10 ng/mL) as a chemoattractant. After being incubated for 6 hours at 37°C, the nonmigrated cells on the upper surface of the membrane were removed by PBS-soaked cotton swab, and the migrated cells on the bottom face of the membrane were fixed and stained by crystal violet staining solutions. Then, 5 fields per filter were selected randomly at a magnification of 200× with an Olympus IX51 inverted microscope.
Gelatin Zymography Analysis
In the gelatin zymography assay for matrix metalloproteinase (MMP)-2, 9 activities were performed as described previously with a slight modification. 18 After exposure to SFGS for 24 hours, HUVECs were pretreated with bFGF (10 ng/mL) for another 24 hours. Supernatant solution of each group were separated in 10% SDS-PAGE copolymerized with 1% gelatin. Then, the gels were rinsed twice and incubated at 37°C for 48 hours in 150 mM NaCl, 10 mM CaCl2, 0.02% NaN3, and 50 mM Tris–HCl buffer (pH 7.5). The gels were stained with 0.25% Coomassie blue R250, and then destained in destaining solution (30% methanol, 10% acetic acid). Gelatinolytic enzymes were detected as transparent bands on the gel. Gelatinolytic activities were detected as unstained bands against background of Coomassie blue–stained gelatin by densitometry assay using Image-Pro Plus software (Media Cybernetics).
Tube Formation Assay
The capillary tube formation assay was performed as described previously. 19 Matrigel (150 µL/well) was added to 48-well plates and allowed to polymerize for 1 hour. HUVECs were suspended and added into each well (8 × 104 cells/well) together with indicated concentrations of SFGS or saponin compounds in the absence or presence of bFGF (10 ng/mL). The capillary tube formations were visualized after 24 hours. The images were captured by an Olympus IX51 inverted microscope using a 100× objective lens. The tube formation was defined by measuring the branch length of the formed tubes.
Statistical Analysis
All data were presented as means ± standard deviations (SD). Statistical significance was evaluated by 1-way analysis of variance (ANOVA) followed by post hoc Tukey’s test. It was considered to be statistically significant when P value was <.05.
Results
The proliferation, differentiation, migration, and tube formation of endothelial cells are key steps of angiogenesis. In this study, we evaluated the in vitro anti-angiogenesis effects of SFGS in HUVECs using bFGF as a stimulator. Figure 2 showed the effects of SFGS on the proliferation of HUVECs. At concentrations of 1, 3, and 10 µg/mL, without evident cytotoxicity on normal cells, SFGS inhibited the proliferation of bFGF-stimulated cells in a concentration-dependent manner, and the inhibitory percentages were 10.0%, 23.1%, and 60.0%, respectively.

Effect of SFGS on the proliferation of HUVECs.
To recognize the routes by which SFGS exerted antiproliferative activity in HUVECs, the effects on cell cycle and apoptosis were studied. SFGS (10 µg/mL) only moderately arrested the cell cycle to the G1/S transition in bFGF-stimulated HUVECs (Figure 3). But it could greatly promote the apoptosis of HUVECs as demonstrated by Hoechst 33258 staining (Figure 4) and flow cytometry analysis (Figure 5). The apoptotic rates in SFGS (1, 3, 10 µg/mL)-treated groups reached 42.3%, 52.6%, and 53.4%, respectively, as compared with that of the control group (12.2%). Furthermore, some apoptosis regulatory proteins were detected, and the results are presented in Figure 6. The bFGF stimulation only slightly increased the expressions of caspase-3, caspase-8, and Fas but not caspase-9. SFGS (1, 3, 10 µg/mL) was able to promote the expressions of caspase-3, caspase-8, and Fas in bFGF-stimulated HUVECs in a concentration-dependent manner, but it did not affect the expression of caspase-9.

Effect of SFGS on the cell cycles of HUVECs.

Effect of SFGS on morphological change of HUVECs.

Effect of SFGS on the apoptosis of HUVECs.

Effect of SFGS on the expressions of apoptosis regulatory proteins in HUVECs.
With regard to the migration of endothelial cells, Transwell assay was performed. bFGF stimulation resulted in an increased migration of HUVECs (Figure 7). SFGS (1, 3, 10 µg/mL) inhibited the migration by 21.5%, 48.4%, and 72.0%, respectively. It is well known that cell migration is closely related to matrix degradation, a process dependent on the activities of degradation enzymes such as matrix metalloproteinases (MMPs). Figure 8 shows that SFGS (1, 3, 10 µg/mL) remarkably decreased the activity of MMP-2 but not MMP-9 in bFGF-stimulated HUVECs. The findings suggested that SFGS might exert inhibitory effects on the migration of endothelial cells by decreasing the activity of MMP-2.
Effect of SFGS on the migration of HUVECs.

Effect of SFGS on the activities of MMP-2 and MMP-9 in HUVECs.
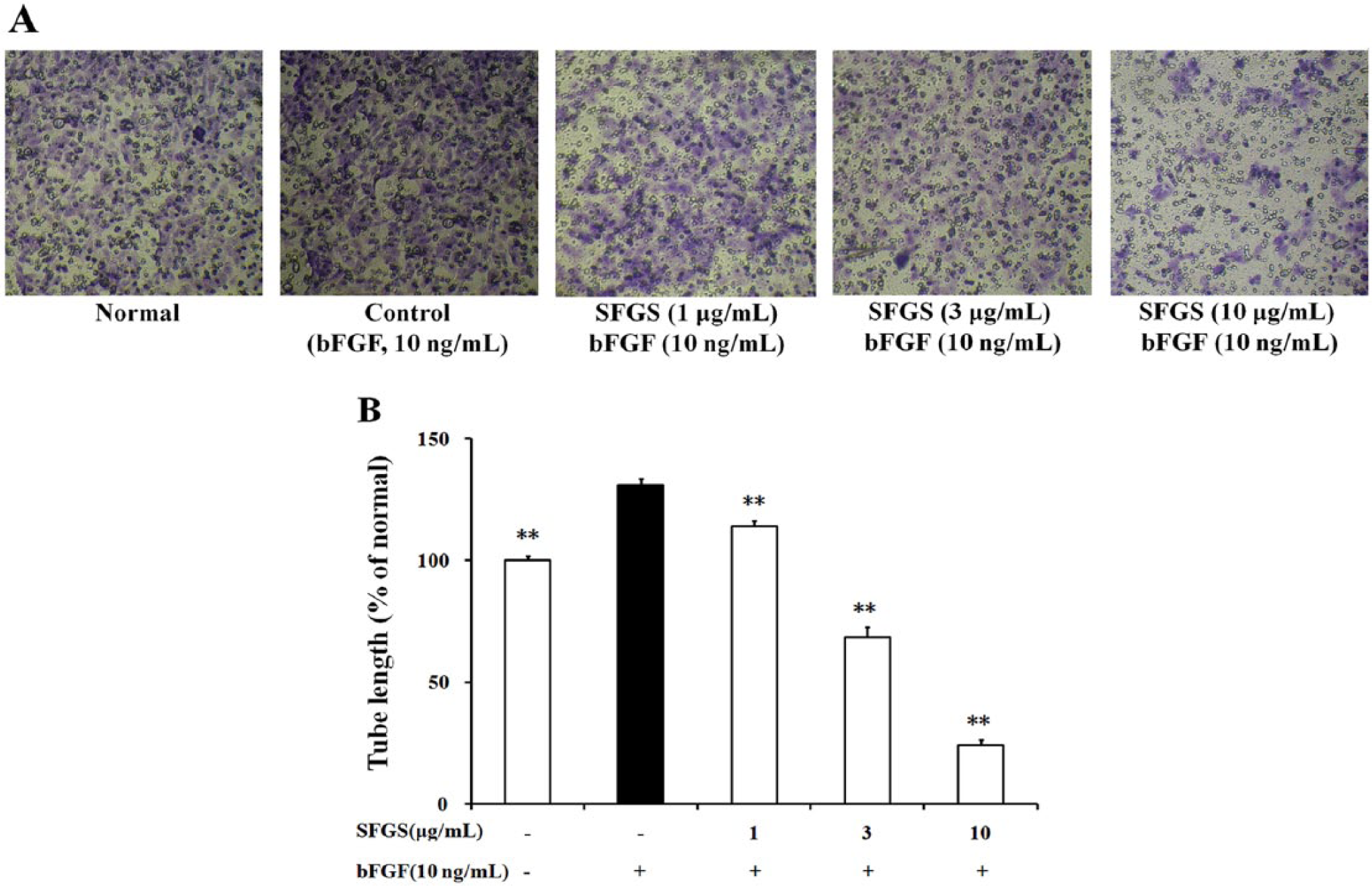
The tube formation of HUVECs was observed on 2D Matrigel. SFGS (1, 3, 10 µg/mL) pretreatment dramatically reduced the tube formation induced by bFGF (Figure 9). However, it was unable to affect the preexisting tubes (data not shown) at concentrations mentioned above, suggesting that it did not belong to vascular disrupting agents.

Effect of SFGS on the tube formation of HUVECs.
Considering that bFGF might exert proangiogenic action through the autosecretion of VEGF from endothelial cells, we explored the effect of SFGS on bFGF-induced secretion of VEGF. The data showed that bFGF stimulation did enhance the secretion of VEGF from HUVECs as compared with normal groups (298.3 ± 4.0 pg/mL vs 121.2 ± 4.8 pg/mL). In the SFGS (10 µg/mL)-treated group, the VEGF level in the cell supernatants was 300.0 ± 7.2 pg/mL, suggesting that SFGS did not affect bFGF-induced autosecretion of VEGF.
Finally, to recognize the active constituents accounting for the anti-angiogenesis action of SFGS, 13 saponin compounds isolated from it together with the sapogenin oleanolic acid (Figure 10A) were screened by tube formation assay in bFGF-stimulated HUVECs. At the concentration of 3 µM, gleditsiosides B, I, J, O, and Q significantly inhibited the tube formation, and other compounds except for pitheduloside C and oleanolic acid showed inhibitory tendencies (Figure 10B). Nevertheless, at the lower concentration (1 µM), only gleditsioside B exhibited significant inhibition, and it could be considered to be most potent among the 5 active saponins mentioned above.

Chemical structures and effects on the tube formation of the saponin compounds.
Discussion
Angiogenesis is a complex process that includes proliferation, migration, capillary tube formation of endothelial cells, and deposition of basement membrane, as well as the proliferation and migration of pericytes and smooth muscle cells. 20 It substantially contributes to various pathological conditions, such as cancer, psoriasis, diabetic retinopathy, and chronic inflammatory diseases in joints or lungs. 21 A range of growth factors, cytokines, and chemokines are capable of influencing angiogenesis. Among these, bFGF belongs to the heparin-binding growth factors. It can act directly through relative receptors, including FGF receptor-1 (FGFR1), FGFR2, FGFR3, and FGFR4, or act indirectly by upregulating the expression of VEGF. 16 The observations in the present study demonstrated that SFGS was unable to affect bFGF-induced autosecretion of VEGF from HUVECs, suggesting that SFGS might function via directly preventing the binding of bFGF to its receptors or the downstream signal pathways.
At concentrations below 10 µg/mL, SFGS was absent of evident cytotoxicity against resting HUVECs. However, its pretreatment resulted in concentration-dependent inhibition of the proliferation, migration, and tube formation in bFGF-activated HUVECs. It also apparently induced apoptosis and decreased the activity of MMP-2 in endothelial cells, but only slightly arrested the cell cycle to the G1/S transition. Moreover, distinct from the vascular disrupting agents such as combretastatin A4 phosphate and TZT-1027, 22 SFGS was unable to affect the preexisting tubes at the concentrations tested. The findings indicated the SFGS exerted anti-angiogenesis activities through multiple pathways, such as proapoptotic action on endothelial cells and prevention of extracellular matrix degradation.
To get insight into the mechanisms responsible for the proapoptosis action of SFGS, its effects on apoptosis-related proteins were studied. It is generally accepted that cells undergo apoptosis primarily by 2 pathways. The extrinsic one is evoked through death receptors (Fas, tumor necrosis factor, and Trail) and a variety of G-protein-coupled receptors. The activation of the receptors leads to the recruitment of Fas-associated death domain and caspase-8 as well as the formation of the death-inducing complex, followed by activation of caspase-3. In contrast, the intrinsic pathway may be evoked by a variety of cytotoxic stimuli or environmental stressors. The formation of an apoptosome complex (Apaf1/cytochrome-c/caspase-9) leads to the activation of caspase-3. In the current study, the protein expressions of caspase-3, caspase-8, caspase-9, and Fas in HUVECs were detected by western blot. Of interest, SFGS was shown to increase the expressions of caspase-3, caspase-8, and Fas but not caspase-9, and it was simply postulated to exert proapoptosis action in HUVECs through the extrinsic death receptor signaling pathway rather than the intrinsic pathway. However, it should be noted that activated caspase-8 is also able to activate caspase-3 indirectly via the cleavage of the Bid protein, and thereby connecting the extrinsic and intrinsic apoptotic pathways.23,24 To rule out the participation of intrinsic pathway in SFGS-induced apoptosis, further studies are needed.
For the purpose of identifying the possible active constituents that contribute to the anti-angiogenic activity of SFGS, 13 saponin compounds together with the sapogenin oleanolic acid were screened by tube formation assay of HUVECs. It was documented that oleanolic acid was ineffective, and gleditsiosides B, I, J, O, and Q, 5 compounds that were simultaneously glycosylated at both hydroxy of C-3 and carboxyl of C-28, showed remarkable inhibition against the tube formation at the concentration of 3 µM. Four compounds without glucosylation at C-28 (pitheduloside A, pitheduloside C, pitheduloside E, and vitalboside A) showed no significant inhibition against tube formation. It was therefore inferred that the oligosaccharide chain at C-28 was necessary for the inhibitory activity. On the other hand, gleditsioside Z with a single sugar chain at C-3 showed inferior activity than gleditsioside A with a trisaccharide chain. In comparison with gleditsioside I, gleditsioside B was more effective, suggesting that the monoterpene unit on C-28 of the oligosaccharide chain might be beneficial for the activity. Nevertheless, linking with 2 monoterpene units (gleditsioside N) resulted in a dramatic decrease of the activity, suggesting that 1 monoterpene unit on C-28 of oligosaccharide chain was enough. Furthermore, the replacement of 2-hydroxymethyl group (gleditsioside B) at the monoterpene unit by a methyl group (gleditsioside A) should greatly decrease the activities. Gleditsioside Q, with a hydroxyl group at C-16, had weaker activity compared with gleditsioside B.
Based on the above statements, we could draw conclusions about the structure–activity relationship of the saponin compounds in SFGS as follows: (a) glucosylation of the carboxyl at C-28 of an aglycon is essential, and both structures and numbers of monoterpene groups on the sugar chain affect the action potency; (b) the sugar chain length on the hydroxy of C-3 is also important, and compounds that contain a trisaccharide chain at C-3 have stronger activities; (c) hydroxyl chain at C-16 will reduce the activity of compounds. Of interest, the structure–activity relationships mentioned above are well consistent with those drawn from in vitro antitumor tests, 25 which provide useful information for designing and synthesizing the analogs of these saponin compounds with anti-angiogenic or antitumor activities.
In conclusion, SFGS is able to prevent angiogenesis by interfering with multiple steps, which provides a new explanation for the antitumor activities. Gleditsiosides B, I, J, O, and Q are probably the main active constituents of it.
Footnotes
Authors’ Note
Authors Dan Lu and YuFeng Xia contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Science & Technology Major Project “Key New Drug Creation and Manufacturing Program” of China (2009ZX09103-392), a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions, and Innovation and Practice Training Program for Undergraduate Students of China Pharmaceutical University.
