Abstract
Hypotheses. Although there is evidence that psychosocial and behavioral interventions (PBIs) increase well-being, improve coping and adjustment, and reduce distress among cancer patients, findings regarding PBIs as a means for prolonging survival were not convincing. Conflicting findings resulted in tremendous controversies over the efficacy of PBIs. This study aims at estimating the pooled effects of PBIs on survival of cancer patients. Study Design and Methods. Randomized controlled trials (RCTs) testing the effects of any kind of PBIs on the survival of cancer patients included in MEDLINE, EMBASE, Cancer Lit, CINAHL, Cochrane Library, and reference lists of relevant articles were retrieved and reviewed by 2 independent researchers. Data items derived from the articles included time and duration of study, intervention types and doses, and numbers of patients dying and surviving 1, 2, 4, and 6 years after intervention. Estimation of the collective effects of the interventions used meta-analysis via Review Manager (version 5). Results. A total of 15 RCTs met inclusion criteria, involving 2041 subjects (1118 in intervention and 923 in control groups). Inclusive total mean Mantel-Haenszel risk ratios (RRs) ranged from 0.83 to 0.99, and 3 of these effect sizes were statistically nonsignificant. Yet when the RCTs with less than 30 hours of PBIs were excluded, all the RRs decreased to some extent, with the RR for the first 2 years being decreased to 0.69 (95% CI, 0.55-0.87) and 0.82 (95% CI, 0.71-0.95), respectively. Conclusion. PBIs with adequate intervention doses prolong survival at least for some cancer patients in the first 2 years after intervention, although longer term effects need to be determined via more studies.
Introduction
Cancer has become one of the most important threats to human life and health. Its incidence and mortality rates have increased steadily since the beginning of the 20th century. People diagnosed with and dying of the disease reached 10.1 million and 6.2 million, respectively, in 2000, 1 and these figures had increased to 12.67 and 7.56 million 8 years later. 2 It is estimated that by 2020, people living with cancer will reach 30 million, and annual new cases and deaths from the disease will rise to 15 million and 10 million, respectively. 3 Cancer and psychosocial and behavioral factors are closely intertwined. On one hand, cancer is almost universally experienced as stressful,4-6 and numerous studies have documented much higher risk of depression, anxiety, obsessive compulsion, hostility, bigotry, interpersonal impairments, and other disorders in cancer patients than in normal controls.7-9 On the other hand, there is compelling evidence that behavioral and psychosocial factors activate the neuroendocrine stress response and alter immune, angiogenic, and inflammatory pathways important in the development, progression, and control of cancer.10-15
Given the above evidence and beliefs, reports about various psychosocial and behavioral interventions (PBIs) incorporating cognitive-behavioral, mindfulness, supportive, and stress management approaches against cancer are emerging.16-19 However, these reports do not point to a conclusive finding. 20 Although there is evidence that PBIs increase well-being, improve coping and adjustment, and reduce distress among cancer patients,21-25 it is unclear whether PBIs prolong survival.26-28 While some randomized controlled trials (RCTs) have shown a survival effect,29-33 others have yielded no positive results.34-36 To estimate the pooled effects of PBIs on cancer patient survival, Chow et al 37 and Smedslund and Ringdal 38 performed meta-analyses separately in 2004. Both studies concluded that it was premature to draw a definite conclusion. Conflicting findings have resulted in tremendous controversies over PBIs. While some authors (eg, Andersen, Spiegel, Kraemer) are in favor of continued, even added, efforts exploring PBIs and survival, others (eg, Coyne, Palmer, Stefanek) have raised severe criticisms against published studies, especially the earlier randomized controlled studies with positive findings, and have even called for an end to funding for any studies in this area. 39 Since the publication of the 2 earlier meta-analyses, several additional RCTs on PBIs and survival have been published. This study aims to combine these old and new RCTs and perform more in-depth and powerful meta-analysis to shed new light on this long-disputed issue.
Methods
Article Retrieval
MEDLINE, EMBASE, Cancer Lit, CINAHL, and Cochrane Library were used for searching eligible English-language publications. The main search algorithm was (Cancer OR tumor OR neoplasm) AND (behavior OR behavioral OR psychology OR psychological OR psychosocial OR psychiatric) AND (mortality OR survival). Reference lists of included articles were also searched manually for further relevant articles.
Inclusion Criteria
Each of the articles retrieved was screened for eligibility according to the following: (1) the article documents an RCT involving a psychosocial intervention among patients with cancer of any histology; (2) the intervention involved one or a combination of education, social support, psychotherapy, skills training, and relaxation; (3) the outcome variables included survival time or data or curves; (4) the patients in both arms received the same medical care; (5) the article allows calculation of relative risk ratios (RRs) between the intervention and control groups for survival with an associated variance.
Data Extraction
Data extracted from the selected articles consisted of name of the first author, year of publication, study location, number of patients randomized to the intervention and control groups, types of cancer, types and doses of intervention, duration of follow-up on survival, and survival rates at 1, 2, 4, and 6 years after intervention. Whenever possible, the raw numbers of survival rates were used; otherwise, the rates were estimated from published survival curves: that is, the percentage surviving was read and multiplied by the total number of patients randomized to the group to estimate the absolute number of survivors. The data extraction was performed independently by 2 of the authors, and any disagreement in extracted data was resolved by consensus with reference to the published article.
Statistical Analysis
Review Manager (version 5) was used to perform meta-analysis and estimate the pooled effects of the interventions. RRs with 95% confidence intervals (CIs) were calculated using the random effects model.
Risk of Biases
There existed biases both within and across individual studies due to (a) heterogeneous cancer sites and stages and intervention contents and dosage; (b) subject attrition; (c) too small sample size and chance finding; and (d) selective reporting and publication. Measures taken to assess the effect of these biases included (a) heterogeneity measurement using forest plot; (b) publication bias assessment by funnel plot; (c) selective analysis pooling only the studies with adequate intervention dosage; and (d) sensitivity analysis by excluding a few of the most flawed RCTs as indicated by a combined rating for each individual study generated by the 2 literature reviewers against a checklist adapted from the indicators recommended by the Cochrane Collaboration. 39
Results
Trials Included
Fifteen RCT articles were identified using the search strategy and inclusion criteria described above (Figure 1).18,30,31,33,34,36,39-47 They comprised a total of 2041 patients (1118 in intervention groups vs 923 in control groups). Nine of the RCTs involved only breast cancer patients with metastases (n = 1390). Three of the RCTs included patients with gastrointestinal cancer, cutaneous malignant melanoma, and colorectal cancer at different stages. The remaining trials had various cancer types and stages. Table 1 gives an overview of the designs and settings of included studies. These studies were conducted in only 5 countries; half of them were conducted in the United States. The interventions were diverse, consisting of education, support, skills training, and others. Duration of the interventions ranged from a few weeks to more than 1 year, and estimated intervention doses ranged from 12 hours to more than 90 hours.
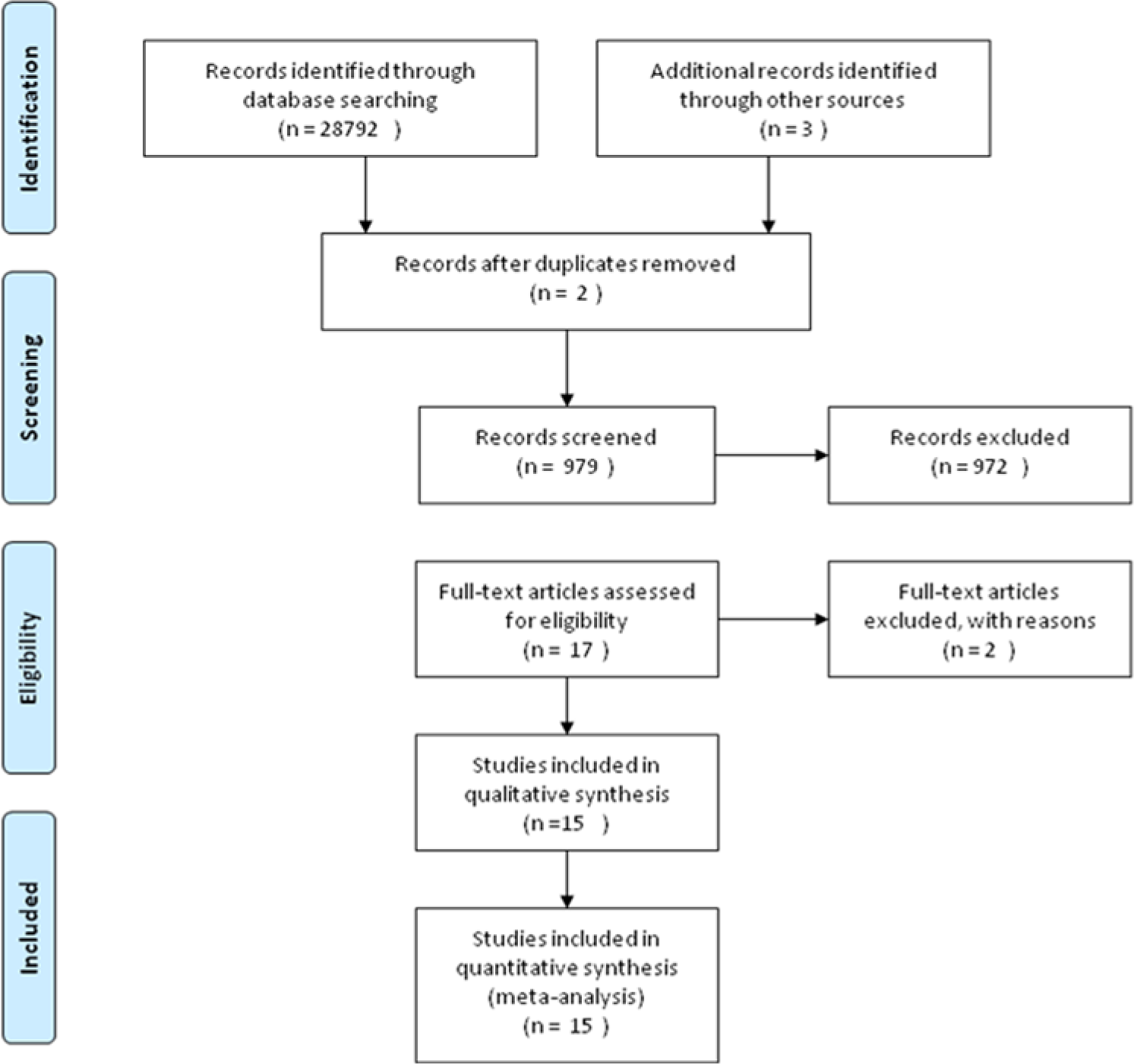
Article retrieval and selection.
Profile of Randomized Controlled Trials Selected.

Abbreviation: NS, not significant.
Risk Ratios
The 1-, 2-, 4-, and 6-year survival rates were available in 12, 14, 13, and 9 trials, respectively. The total mean Mantel-Haenszel risk ratios ranged from 0.83 to 0.99, and 3 of these 4 effect sizes were statistically nonsignificant (Figure 2). The heterogeneity ranged from 27% to 70%, and all the nonsignificant effect sizes were accompanied by more than 50% of heterogeneity.

Meta-analysis of all randomized controlled trials identified (sorted by estimated intervention dose).
When the RCTs with less than 30 hours of PBIs were excluded, the RRs for all 4 follow-up years decreased to some extent (Figure 3). The most substantial changes occurred in the RRs for years 1 and 2, from 0.86 (95% CI, 0.69-1.06) and 0.85 (95% CI, 0.75-0.96) to 0.69 (95% CI, 0.55-0.87) and 0.82 (95% CI, 0.71-0.95), respectively. The RRs showed an increasing trend from year 1 to year 6. The heterogeneity then again showed a similar feature: That is, nonsignificant effect sizes were accompanied by more than 50% of heterogeneity. Exclusion of 1, 2, and 3 RCTs with the lowest quality scores mentioned above did not change the statistical significance of these pooled RRs.

Meta-analysis of randomized controlled trials with more than 30 hours of intervention (sorted by intervention dose).
Funnel Plot
A funnel plot was produced to detect the presence of publication bias and revealed a relatively moderate degree of asymmetry (Figure 4). More specifically, most of the studies with a lower standard error of mean (SE of log [RR]) were concentrated together and beside the centerline (the overall effect sizes); some studies (about 7) were dispersed quite far away from the centerline (either below 0.5 or above 5); and the RR of one study was even plotted to the left of the 0.1 vertical line.

Funnel plot of the effect sizes against the logarithm standard error of the individual studies.
Discussion
Although PBIs have gained widespread recognition and there is growing evidence that PBIs improve the well-being of cancer patients,47,51 it remains controversial whether addition of PBIs to routine medical care can prolong survival.42,50 Our present study partly reflects this situation. Due to the time difference, we were able to double both the numbers of eligible RCTs and the patients involved compared with previous similar studies. 52 Yet the findings of our inclusive meta-analysis (ie, 3 of the 4 estimated effect sizes were statistically nonsignificant) are still discouraging. Inadequate intervention dosage and inappropriate intervention focus may be the most important causes of this phenomenon. Of the 15 RCTs included, accumulated time spent in performing PBI sessions ranged from 9 to 87 hours. Compared with the total follow-up period, the time of PBI sessions accounted for only 0.010% to 0.497%. If we take the study by Spiegel et al 18 as an example, the estimated accumulated Supportive-Expressive Group Therapy time was 81 hours (1.5 hours per session, once a week, 54 weeks in total), and the follow-up measurement of survival at the end of the intervention was 122 640 hours starting from the baseline, so the length of intervention sessions assumed only 0.066% (81/122 640) of total follow-up duration. If the Supportive-Expressive Group Therapy was 100% effective during intervention sessions and did not exert any additional effect outside the sessions, the intervention only reduced 0.066% of stress; such a small proportion of effects are hardly detectable in a small sample RCT. Given this huge time discrepancy, it is obvious that “out-of-session” intervention is much more important than “in-session” intervention, while most published trials neglected this point. Researchers generally took it for granted that interventions practiced during in-session time would automatically be repeated by patients in out-of-session time and gave little attention to implementing effective measures to promote and monitor patients’ out-of-session practices. Of course, readers are reminded about the subtle differences between doses of PBI and those of pharmacologic interventions. Pharmacologic agents are cleared from the body by the liver and kidneys in a relatively short period of time, whereas PBIs may remain in the patients’ minds for a longer time. Thus, our in-session versus out-of-session dose comparison may be inappropriate in a strict sense. Yet it points to the importance of out-of-session focus: That is, future studies should pay ample attention to PBIs that have enduring effects. In addition, out-of-session focus does not necessarily mean long-term continuous intervention for a patient’s entire survival period, which would be neither realistic nor cost-effective. Instead, it emphasizes reaping extended effects in out-of-session time from limited in-session activities by means of careful intervention redesign. Another factor contributing to the inconsistent findings of the included studies may be study heterogeneity. The statistics given in Figure 2 show that 3 of the 4 heterogeneity values were greater than 50%, yet meta-analysis applies well only if the heterogeneity is less than 50%. Researchers have pointed out a variety of possible sources, including discrepancies in types of cancer, stages of diseases, demographics of patients, contents and settings of PBIs, accompanying medical treatment, and compliance with treatment and interventions. 53 All these factors may have played a role in the studies. In addition, subject sizes were generally too small: The largest sample size was 375 and the smallest was only 66; most of the studies recruited fewer than 300 participants. This may have made higher or lower effects prone to be detected because of chance findings.
Bearing in mind the assumption that differences in intervention doses and quality may be among the most important reasons causing the huge effect size heterogeneity, we performed selective meta-analysis by excluding the 8 RCTs with less than 30 hours of PBIs. Not surprisingly, our selective meta-analysis yielded significantly improved statistics compared with the aforementioned inclusive analysis. Just as the dose of a chemotherapy regime contributes to its treatment effects, the dose of a PBI should play a key role in determining its intervention efficacy if the PBI were effective. Due to limited information about interventions and number of RCTs, we did not take account of intervention quality. Given this and the poor compliance reported, 41 it should be viewed as encouraging that some 30 to 90 hours of PBIs resulted in an RR of 0.69 in the first year. However, readers are cautioned about a few issues in interpreting this finding. First, several of the studies included in our analysis have been criticized heavily for methodological reasons, including inappropriate intervention content and time point,54,55 flawed research design and data interpretation, 56 subject loss, 57 and small scale. 40 The limited number of RCTs did not allow us to produce a quality weighted meta-analysis. Yet when we excluded the few most heavily criticized RCTs (eg, Andersen 2008, 30 Spiegel 1989, 42 Spiegel 2007, 18 Kuchler 2007 31 ), the statistically significant effects of PBIs on survival remained unchanged. Second, as mentioned earlier, various cancer sites and stages were contained in this meta-analysis and the pooled overall effect may not be representative of the actual total cancer population since we were not able to perform site- and stage-adjusted analysis given the information available. Even so, we can still argue that PBIs have positive effects on survival at least for patients with similar composition of cancer sites and stages as those in the studies included. Third, the relatively moderate degree of asymmetry shown by the funnel plot (Figure 4) indicates some extent of publication bias, although it is not quite significant. In addition, most of the precautions advised by Chow et al 37 apply here as well.
Our meta-analysis also indicates a “tapering off” trend in the effects of PBIs year after year. This may be attributable to various reasons. First, almost all the PBIs ended within 1 year, yet some of the intervention effects may be dependent on ongoing practices. For example, relief of recurring stress may need continuing relaxation exercises. Second, due to the PBIs implemented, more patients in the intervention group than in the control groups survived the earlier years, and this may have resulted in the intervention groups containing more severely diseased patients in the later years. Third, in addition to using the PBIs provided by the researchers, the patients may have tried to seek PBIs from other sources on their own. And thus as time passed by, more patients in the control groups may have been involved in some sort of PBI, whereas patients in the intervention groups may have been less motivated to do so since they had already been involved in PBIs.
Finally, our meta-analysis suggests that more rigorous research is needed regarding PBIs and cancer patient survival. More than 30 years have passed since publication of the first article on this issue, 41 yet our systematic search found only 15 eligible RCTs. And, as mentioned above, most of them suffered from small sample size. As well as attending to more investment and larger scale, future RCT plans and reports should pay added attention to (1) appropriate intervention focus; (2) disease and health status, for example, types and stages of cancer and complications; (3) PBI features, such as content, quality, duration, doses, sustainability, and generalization; (4) background pharmaceutical treatments and PBIs used during and after the intervention period in both trial and control groups; (5) compliance with both PBIs and pharmaceutical treatments in both groups; and (6) patients’ characteristics such as age, gender, and education.
Conclusion
Our selective meta-analysis revealed that PBIs with adequate intervention doses prolong cancer patients’ survival at least for some patients in the first 2 years after intervention, although the longer term effects need to be determined via more studies. The effect sizes may taper off to some extent as time passes after the PBIs have terminated.
Footnotes
Contributors
Yi Xia and Guixian Tong contributed equally in retrieving and processing the RCT articles, performing statistical analysis, and drafting the manuscript. Rui Feng, Jing Chai, and Jing Cheng participated in study design and manuscript revision and refinement. Debin Wang, corresponding author for this paper, conceived, funded, and facilitated the whole study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was co-supported by the Natural Science Foundation of China (grant number 81172201) and Anhui Provincial Fund for Elite Youth (grant number 2011SQRL060).
