Abstract
Objective
We aimed to investigate the associations between beta-blocker use and survival outcomes, including cancer-specific mortality, all-cause mortality, median overall survival, and median progression-free survival, in patients with colorectal cancer. The findings are intended to inform evidence-based strategies for optimizing adjuvant therapy in clinical practice.
Methods
We conducted a comprehensive search of PubMed, Embase, Web of Science, and the Cochrane Library to identify studies assessing the effect of beta-blockers on survival outcomes in patients with colorectal cancer. Studies reporting hazard ratios and 95% confidence intervals for cancer-specific mortality, all-cause mortality, median overall survival, or median progression-free survival were included. Adjusted hazard ratios were pooled using meta-analytic techniques. Subgroup and sensitivity analyses were performed to explore the potential sources of heterogeneity and assess result robustness.
Results
Overall, 13 high-quality cohort studies including >93,000 patients with colorectal cancer were included. Beta-blocker use was marginally associated with reduced cancer-specific mortality (hazard ratio = 0.90; 95% confidence interval: 0.81–1.00), with a more pronounced effect among patients who had not undergone surgery (hazard ratio = 0.86; 95% confidence interval: 0.75–0.98). Although no overall association was observed for all-cause mortality (hazard ratio = 0.76), a significant reduction was noted in the subgroup that underwent curative surgery (hazard ratio = 0.65; 95% confidence interval: 0.42–1.00). Among patients receiving bevacizumab, beta-blocker use was linked to improved median overall survival (hazard ratio = 0.70; 95% confidence interval: 0.56–0.87), whereas a nonsignificant trend toward improved median progression-free survival was observed (hazard ratio = 0.79; 95% confidence interval: 0.60–1.05). Sensitivity analyses supported the robustness and consistency of the pooled results.
Conclusions
This meta-analysis indicates that beta-blocker use is significantly associated with a reduced risk of cancer-specific mortality in patients with colorectal cancer, with the most pronounced benefit observed among those who did not undergo surgery. Additionally, patients undergoing radical resection or bevacizumab-based therapy may also experience improved survival with beta-blocker use. Given the well-established cardiovascular safety, affordability, and broad clinical availability, beta-blockers may serve as promising adjuncts for comprehensive colorectal cancer treatment. However, further randomized controlled trials are warranted to validate these findings and define optimal patient populations, timing, and combination strategies. This study was registered with PROSPERO (CRD420251079257).
Keywords
Introduction
Colorectal cancer (CRC) remains one of the most commonly diagnosed malignancies worldwide, ranking third in incidence and second in cancer-related mortality. According to the Global Cancer Observatory (GLOBOCAN), over 1.9 million new CRC cases and more than 900,000 related deaths occurred worldwide in 2020. 1 Although remarkable progress has been made in the fields of surgery, chemotherapy, targeted therapies, and immunotherapy, long-term outcomes for patients with locally advanced or metastatic CRC remain unsatisfactory. 2 This scenario highlights the urgent need for effective adjunctive strategies to enhance survival outcomes, particularly among high-risk populations.
Recent evidence has emphasized the role of neuroendocrine–immune interactions in cancer progression. Chronic psychological stress persistently activates the sympathetic nervous system, triggering the release of catecholamines such as norepinephrine and epinephrine. These neurotransmitters stimulate β-adrenergic receptor (β-AR) signaling, which promotes tumorigenesis via enhanced proliferation, angiogenesis, metastasis, and immune suppression.3–6 Based on these mechanisms, beta-blockers (BBs), which are widely used in cardiovascular disease treatment, have attracted interest for their ability to disrupt stress-mediated oncogenic pathways and exert potential antitumor effects.
Experimental and clinical studies support the antineoplastic activity of BBs via multiple mechanisms, which include inhibition of the PI3K/AKT signaling pathway, downregulation of vascular endothelial growth factor (VEGF), suppression of tumor-associated macrophage (TAM) infiltration, and enhancement of cytotoxic T-cell responses.7–10 In addition, BBs may counteract the immunosuppressive effects induced by intraoperative stress, thereby reducing the risk of postoperative tumor recurrence and distant metastasis.11–13
An increasing number of clinical studies have reported associations between BB use and improved outcomes in patients with solid tumors, including breast, lung, ovarian, and liver tumors. These studies have demonstrated significant reductions in cancer-specific mortality (CSM) and all-cause mortality (ACM).14–17 Moreover, the perioperative administration of BBs has been associated with improved overall survival (OS) and recurrence-free survival (RFS) in patients with cancer. 18 However, in the context of CRC, evidence regarding the effects of BBs remain limited and inconsistent. Some observational studies have suggested potential survival benefits, whereas others have reported null associations.19–22 Furthermore, few investigations have assessed whether treatment-related variables, such as surgery, chemotherapy, or bevacizumab, modulate these effects.
Owing to such uncertainties, we aimed to systematically evaluate the existing clinical evidence on BB use in patients with CRC by conducting a comprehensive meta-analysis in this study. The primary objective was to examine the associations between BB exposure and key prognostic outcomes, including CSM, ACM, median overall survival (mOS), and median progression-free survival (mPFS). Subgroup analyses were conducted to assess potential effect modifications according to the treatment strategy. Additionally, sensitivity analyses were conducted to evaluate the robustness of the pooled estimates. This study aimed to inform individualized treatment decisions and guide future mechanistic research.
Methods
Information and methodology
We performed a systematic review and meta-analysis to evaluate the associations between BB use and survival outcomes in patients with CRC. The study protocol was rigorously designed and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines to ensure methodological transparency and reproducibility. 23 Additionally, the protocol was prospectively registered in the PROSPERO database (ID: CRD420251079257). Ethical approval and informed consent were not required as the analysis was based exclusively on publicly available studies and did not involve individual patient data or interventional procedures.
Screening and searching for literature
A comprehensive literature search was conducted across four major electronic databases, including PubMed, Embase, the Cochrane Library, and Web of Science, to identify all relevant studies published before 20 May 2025. No restrictions were placed on the date of publication or language. The search strategy incorporated a combination of standardized Medical Subject Headings (MeSH) and free-text terms, including “beta-blocker” and “colorectal cancer.” In addition, it was tailored to the syntax and structure of each individual database. We manually screened the reference lists of the included studies to identify any additional eligible publications to ensure completeness. Detailed search strings for each database were developed and are available upon reasonable request.
The study selection process was independently performed by two reviewers. In the initial stage, titles and abstracts were screened to exclude studies unrelated to the research topic. Full-text articles were subsequently reviewed to determine eligibility based on predefined inclusion criteria. Any disagreements between reviewers were resolved via discussion or consultation with a third investigator until a consensus was reached.
Eligibility criteria
Studies were considered to be eligible for inclusion if they met the following criteria: 1. The study population included patients diagnosed with CRC (confirmed either histologically or clinically); 2. The study compared survival-related endpoints (CSM, ACM, mOS, or mPFS) between individuals who received BBs and those who did not; 3. The study directly reported hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) or this information could be calculated from the available data; 4. The study adopted a cohort design (prospective or retrospective) or a nested case–control design; and 5. The article was a complete, peer-reviewed, original research publication available in full text.
The following types of studies were excluded: (a) reviews, editorials, conference abstracts, case series, animal experiments, and in vitro studies; (b) reports lacking sufficient data to extract or estimate HRs and corresponding 95% CIs; (c) reports with unclear definitions of BB exposure or those in which the effects of BB exposure could not be isolated from other concurrent interventions; (d) studies with duplicate or overlapping datasets, in such cases, the most recent or most comprehensive study was retained; and (e) articles for which the full text was not accessible.
Data extraction
Key data variables were independently extracted from each included study by two investigators. These variables included publication metadata (first author, year, and country/region), study characteristics (design type and follow-up duration), participant demographics (sample size, age, and definitions of BB exposure), outcome-related information (specified endpoints, number of events, and assessment methods), and covariates used in the multivariable adjustment models. Any discrepancies between investigators were resolved via discussion with a third investigator to ensure the accuracy and consistency of the dataset.
The primary outcomes of interest were CSM and ACM. The secondary outcomes were mOS and mPFS. Studies reporting adjusted HRs with corresponding 95% CIs were prioritized. In cases where HRs were not directly reported, estimations were reconstructed from the available raw data when feasible, and the original authors were contacted for additional information if necessary.
Quality assessment
The methodological quality of the included nonrandomized studies was assessed by the Newcastle–Ottawa Scale (NOS), which is a validated tool that evaluates research based on the following three domains: selection of study groups, comparability of cohorts, and ascertainment of exposure or outcome. The NOS contains nine items, with a maximum possible score of 9 points. Studies scoring ≥7 points were classified as high quality. The quality assessment was independently conducted by two reviewers. In cases of disagreement, a third reviewer intervened to resolve any discrepancies and reach a consensus. The final NOS scores are presented in the Supplementary Material and were considered in the sensitivity analyses and interpretation of the findings.
Statistical analysis
All analyses were performed using Cochrane Review Manager (RevMan) software, version 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration; Copenhagen, Denmark). For each included study, the adjusted HRs and their corresponding 95% CIs from multivariable models were extracted, and only studies reporting adjusted estimates were included in the meta-analysis. Heterogeneity was assessed using Cochran’s Q test and the I2 statistic. All pooled analyses were conducted using a random-effects model, which accounts for potential between-study heterogeneity. Subgroup and sensitivity analyses were conducted to identify the sources of heterogeneity and evaluate the robustness of the results. A leave-one-out method was utilized to assess the influence of individual studies on the pooled estimates.
Due to the limited number of included studies (≤10) per outcome, Egger’s and Begg’s tests were not performed in accordance with the methodological recommendations. Instead, publication bias was preliminarily assessed by visually inspecting funnel plot symmetry, in addition to the performance of sensitivity analyses to determine the extent to which such bias may have influenced the overall findings.
Results
Study selection and study characteristics
Overall, 351 potentially eligible studies were identified through systematic searches of four major databases, including PubMed (n = 125), Embase (n = 173), the Cochrane Library (n = 27), and Web of Science (n = 26). After removing duplicates, 290 unique studies remained. Title and abstract screening excluded 264 articles that were irrelevant to the study topic. The remaining 26 full-text articles were thoroughly assessed, of which 13 met the predefined inclusion and exclusion criteria and were ultimately included in the meta-analysis.19–22,24–32 The complete selection process is presented in Figure 1.

PRISMA flowchart of study selection. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
This meta-analysis included 13 cohort studies published between 2013 and 2025 involving prospective and retrospective designs. The participants were histologically or clinically diagnosed with CRC, and several studies also included patients with comorbid hypertension. Notably, these studies reflected a wide international scope, with research being conducted across 10 countries. Among them, Sweden contributed to the highest number of studies (three studies), followed by the United States (two studies). Additional data originated from single studies conducted in China, Türkiye, Lithuania, the United Kingdom, Canada, the Netherlands, Italy, and the Czech Republic. The study populations predominantly consisted of middle-aged and older individuals, with sample sizes ranging from 181 to 23,076. Definitions of BB exposure varied across studies, including recent initiation, use within the first year following diagnosis, or coadministration with chemotherapy. The follow-up duration of the studies ranged from 1 to 8.6 years. The assessed outcomes included CSM, ACM, mOS, and mPFS. Moreover, majority of the studies reported HRs derived from multivariable models, which were commonly adjusted for age, sex, tumor stage, comorbid conditions, and treatment regimens. A detailed summary of the included studies is presented in Table 1.
Characteristics of the included studies.
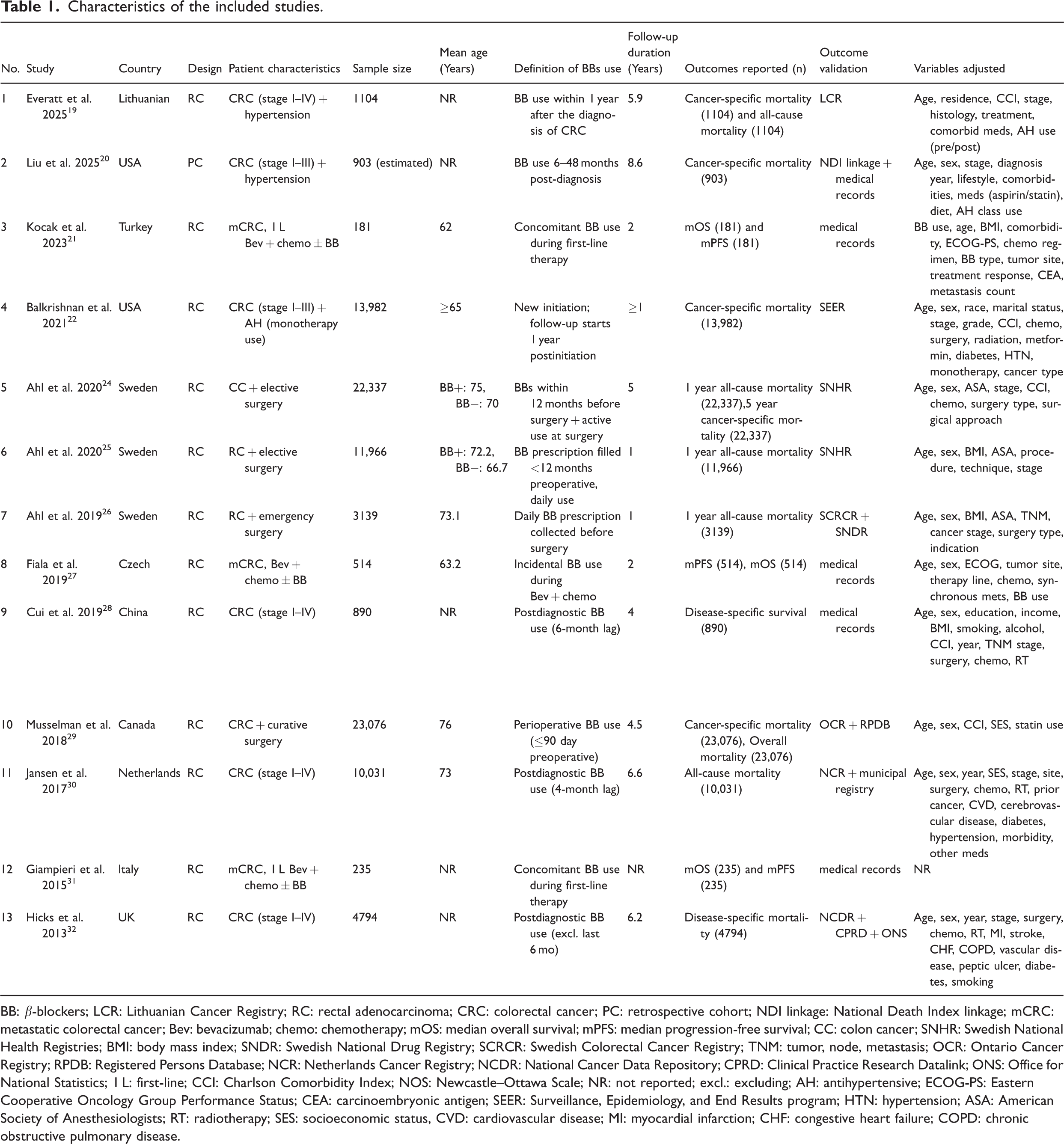
BB: β-blockers; LCR: Lithuanian Cancer Registry; RC: rectal adenocarcinoma; CRC: colorectal cancer; PC: retrospective cohort; NDI linkage: National Death Index linkage; mCRC: metastatic colorectal cancer; Bev: bevacizumab; chemo: chemotherapy; mOS: median overall survival; mPFS: median progression-free survival; CC: colon cancer; SNHR: Swedish National Health Registries; BMI: body mass index; SNDR: Swedish National Drug Registry; SCRCR: Swedish Colorectal Cancer Registry; TNM: tumor, node, metastasis; OCR: Ontario Cancer Registry; RPDB: Registered Persons Database; NCR: Netherlands Cancer Registry; NCDR: National Cancer Data Repository; CPRD: Clinical Practice Research Datalink; ONS: Office for National Statistics; 1 L: first-line; CCI: Charlson Comorbidity Index; NOS: Newcastle–Ottawa Scale; NR: not reported; excl.: excluding; AH: antihypertensive; ECOG-PS: Eastern Cooperative Oncology Group Performance Status; CEA: carcinoembryonic antigen; SEER: Surveillance, Epidemiology, and End Results program; HTN: hypertension; ASA: American Society of Anesthesiologists; RT: radiotherapy; SES: socioeconomic status, CVD: cardiovascular disease; MI: myocardial infarction; CHF: congestive heart failure; COPD: chronic obstructive pulmonary disease.
Study quality
The methodological quality of all the included nonrandomized controlled studies was assessed using NOS, a validated tool that evaluates three domains (participant selection, group comparability, and outcome ascertainment) with a maximum possible score of 9. A total score of ≥7 was considered indicative of high methodological quality.
Quality assessment was independently conducted by two reviewers. In cases of discrepancy, a third evaluator was consulted to reach a consensus. Among the 13 included studies, 10 achieved a maximum score of 9, whereas the remaining three studies achieved a score of 8. These results suggested consistently high methodological quality across the studies and a low overall risk of bias. The detailed NOS scores for each study are summarized in Table 2.
NOS quality assessment of the included studies.

NOS: Newcastle–Ottawa Scale.
Efficacy outcomes
CSM
Seven observational studies were included to evaluate the association between BB use and CSM in patients with CRC.19,20,22–24,28,29,32 The pooled results indicated a marginally significant reduction in CSM risk among BB users (HR = 0.90; 95% CI: 0.81–1.00; Z = 1.95; P = 0.05). However, significant heterogeneity was observed across studies (I2 = 75%; P < 0.001).
A subgroup analysis based on treatment strategy revealed that BB use was associated with a statistically significant decrease in CSM (HR = 0.86; 95% CI: 0.75–0.98; Z = 2.33; P = 0.02) in patients who did not undergo surgical resection,19,20,22,28,32 with moderate heterogeneity observed (I2 = 62%; P = 0.03). In contrast, the pooled data from two studies involving patients who underwent surgery demonstrated no significant association (HR = 0.99; 95% CI: 0.76–1.29; Z = 0.07; P = 0.94)24,29 and exhibited high heterogeneity (I2 = 90%; P = 0.002).
A subgroup comparison revealed no statistically significant difference in the effect of BBs between surgical and nonsurgical cohorts (Χ2 = 0.91; df = 1; P = 0.34; I2 = 0%), thereby indicating that the survival benefit of BBs may be consistent regardless of surgical status. The complete results are illustrated in Figure 2.

Forest plot for cancer-specific mortality (stratified by treatment strategy).
ACM
Six cohort studies were included to evaluate the association between BB use and ACM in patients with CRC.19,24–26,29,30 The pooled analysis suggested a nonsignificant trend toward reduced mortality among BB users (HR = 0.76; 95% CI: 0.56–1.04; Z = 1.72; P = 0.09)19,30 along with substantial heterogeneity (I2 = 98%; P < 0.00001).
A subgroup analysis revealed no significant association between BB use and ACM in patients who did not undergo surgical intervention (HR = 1.08; 95% CI: 0.98–1.20; Z = 1.55; P = 0.12),24–26,29 with negligible heterogeneity observed (I2 = 0%). In contrast, among patients who underwent surgical treatment, BB exposure was associated with a borderline significant reduction in ACM (HR = 0.65; 95% CI: 0.42–1.00; Z = 1.95; P = 0.05), although the heterogeneity within this subgroup remained high (I2 = 98%, P < 0.00001).
The difference between the surgical and nonsurgical subgroups was statistically significant (Χ2 = 5.06; df = 1; P = 0.02; I2 = 80.2%), thus suggesting that the effect of BBs on survival outcomes may vary depending on surgical status. The detailed results are presented in Figure 3.

Forest plot for ACM (stratified by treatment strategy). ACM: all-cause mortality.
mOS
Three cohort studies were included to assess the effect of BB use on mOS in patients with CRC treated with combination chemotherapy and bevacizumab.21,27,31 The meta-analysis revealed a statistically significant improvement in mOS among BB users, with a pooled HR of 0.70 (95% CI: 0.56–0.87; Z = 3.16; P = 0.002).
Heterogeneity testing indicated no significant variability across studies (I2 = 0%; P = 0.56), thereby suggesting a high degree of consistency in the effect estimates.
Among the included studies, Fiala et al. 27 contributed the highest weightage (70.1%) and reported a significant reduction in mortality (HR = 0.73; 95% CI: 0.56–0.95). Kocak et al. 21 contributed the second highest weightage (22.9%) and demonstrated the most substantial mortality reduction (HR = 0.57), indicating a 43% decrease in risk. Although the study by Giampieri et al. 31 also suggested a favorable trend (HR = 0.89), the association did not demonstrate statistical significance.
Collectively, these results suggest that BBs may offer a survival advantage for patients with CRC receiving anti-VEGF–based therapies. The detailed findings are illustrated in Figure 4.

Forest plot for mOS in patients with CRC receiving Bev + Chemo. mOS: median overall survival; CRC: colorectal cancer; Bev: bevacizumab; Chemo: chemotherapy.
mPFS
This meta-analysis synthesized data from three studies examining the association between BB use and mPFS in patients with CRC.21,27,31 Although the pooled HR indicated a trend toward improved mPFS among BB users (HR = 0.79; 95% CI: 0.60–1.05; Z = 1.64; P = 0.10), the association did not demonstrate statistical significance. Furthermore, moderate heterogeneity was observed (I2 = 44%; P = 0.17), thereby suggesting reasonable consistency across the included studies.
With respect to individual evaluation, the studies by Kocak et al. 21 and Fiala et al. 27 reported favorable associations between BB use and prolonged mPFS, with HRs of 0.66 and 0.76, respectively. In contrast, Giampieri et al. 31 reported no significant effect (HR = 1.35; 95% CI: 0.68–2.53), which may be attributed to the smaller sample size that limited the statistical power.
Collectively, these results suggest a potential benefit of BBs in delaying disease progression among patients with CRC, warranting further validation through large-scale prospective trials. The detailed outcomes are presented in Figure 5.

Forest plot for mPFS in patients with CRC. mPFS: median progression-free survival; CRC: colorectal cancer.
Publication bias and sensitivity analysis
Publication bias
Funnel plots were generated for CSM (Figure 6) and ACM (Figure 7) outcomes to evaluate the possibility of publication bias. The distributions appeared to be generally symmetrical, and no substantial evidence of small-study effects or publication asymmetry was observed. Given that <10 studies were included for each outcome, formal statistical tests for evaluating the publication bias (Egger’s and Begg’s tests) were not conducted, which is consistent with established methodological recommendations.

Funnel plot for publication bias-CSM. CSM: cancer-specific mortality.

Funnel plot for publication bias-ACM. ACM: all-cause mortality.
Sensitivity analysis
We conducted leave-one-out sensitivity analyses for both primary outcomes (CSM (Figure 8) and ACM (Figure 9)) to assess the robustness of our meta-analytic findings. In this approach, each included study was sequentially excluded to evaluate its influence on the pooled HRs and corresponding 95% CIs. The analyses revealed that the exclusion of any single study did not substantially alter the direction or magnitude of the overall effect estimates. This consistency across iterations supports the stability of the results and suggests that the observed associations are not driven by any outlier study, thereby reinforcing the internal validity of our conclusions.

Leave-one-out sensitivity analysis-CSM. CSM: cancer-specific mortality.

Leave-one-out sensitivity analysis-ACM. ACM: all-cause mortality.
Discussion
In this meta-analysis, we synthesized data from 13 high-quality cohort studies to explore the relationship between BB use and survival outcomes in patients with CRC. The pooled analysis revealed a potential inverse association between BB use and CSM, particularly among patients who did not undergo surgical resection. Although the overall association between BB use and ACM was not statistically significant, a survival benefit was evident in the subgroup of patients who underwent surgical treatment. Notably, prior pan-cancer meta-analyses yielded inconclusive findings for CRC, often limited by insufficient subgroup analyses.33,34 This highlights the relevance of stratified evaluation in future research.
It is hypothesized that the observed reduction in CSM among BB users may be attributable to the ability of BBs to inhibit tumor-promoting signaling cascades triggered by the activation of the sympathetic nervous system. Within the tumor microenvironment, sympathetic stimulation is known to upregulate oncogenic mediators such as VEGF, interleukin-6 (IL-6), and matrix metalloproteinase-9 (MMP-9) via β-AR pathways. This mechanism results in enhanced angiogenesis, increased cellular invasiveness, and greater metastatic potential, all of which are suppressed by BB-mediated β-AR blockade.35–38
Furthermore, β-AR stimulation has been implicated in promoting tumor immune evasion by suppressing the cytotoxic activity of CD8+ T cells and natural killer (NK) cells. Additionally, β-AR stimulation drives macrophage polarization from the proinflammatory M1 phenotype to the immunosuppressive M2 subtype and facilitates the accumulation of regulatory T cells and myeloid-derived suppressor cells (MDSCs), thereby fostering an immune-tolerant tumor microenvironment.39–41 By antagonizing these neuroimmune regulatory pathways, BBs may attenuate neural-driven tumor progression and provide an immunomodulatory mechanism for improving long-term oncologic outcomes. 42
Evidence suggests that BBs may offer a greater reduction in ACM among patients with CRC who undergo surgical treatment, which is likely due to their ability to modulate critical physiological responses during the perioperative period. Sympathetic nervous system activation during this time period has been observed to impair cardiovascular homeostasis and suppress immune function, thereby increasing the risk of postoperative complications and noncancer-related mortality. BBs may mitigate these risks by stabilizing hemodynamics, attenuating surgical stress responses, and enhancing immune recovery. This dual mechanism not only supports the direct antitumor activity of BBs but also underscores their role in promoting systemic resilience and postoperative survival outcomes.43,44
A subgroup analysis revealed that the administration of BBs to patients with CRC receiving bevacizumab-containing regimens was significantly associated with improved mOS. Although a favorable trend was also observed for mPFS, the difference did not demonstrate statistical significance. These findings suggest that BBs may have a more pronounced effect on long-term clinical outcomes (delaying tumor relapse, enhancing responsiveness to subsequent treatment lines, and modulating the tumor immune microenvironment), whereas their impact on short-term disease control (as reflected by mPFS) may be limited. Furthermore, the heterogeneity in PFS definitions, short follow-up durations, and variability in radiological assessment protocols across studies may have diminished the reliability of the comparative analyses.
Under anti-VEGF therapeutic conditions, BBs may potentiate the antiangiogenic activity of bevacizumab by downregulating the expression of VEGF induced by sympathetic nervous system activation, thereby increasing treatment efficacy.45,46 Additionally, BBs have been reported to improve tumor oxygenation, enhance intratumoral drug delivery, and attenuate inflammatory signaling, which collectively may contribute to improved clinical responses to immunotherapy and chemotherapy.47–49 These multifaceted effects position BBs as promising adjunctive agents alongside current molecular-targeted and immune-based therapies, thereby offering substantial translational value in the context of comprehensive cancer care.
Although this investigation demonstrates substantial clinical relevance and biological plausibility, several limitations should be considered. First, the included studies were cohort studies with an observational design, which inevitably introduces confounding by indication (e.g. hypertension or cardiovascular disease prompting BB prescriptions) that may affect oncologic outcomes. Although adjusted HRs were reported, residual confounding cannot be excluded; therefore, our findings should be regarded as hypothesis-generating rather than causal. Second, substantial heterogeneity was observed for ACM, CSM, and mPFS, reducing the reliability. This likely reflects differences in patient stages, treatment regimens, and study designs. Variability in BB type, dosage, duration, and adherence may further limit consistency. Subgroup analyses partly addressed this issue; however, residual heterogeneity was observed to remain. Third, inconsistencies in defining CSM and mPFS as well as potential misclassifications of the causes of death, could introduce bias into the effect estimates. Additionally, nonselective BBs were not differentiated from selective BBs in most studies, precluding reliable subtype analyses; therefore, any apparent overall benefit might have been driven by a specific subtype. Future studies should examine the differential effects of selective and nonselective BBs. Moreover, substantial heterogeneity was observed in the analysis of ACM (I2 = 98%) and certain subgroups, which may reflect variations in the study design, population characteristics, definitions of BB exposure, concomitant treatments, and follow-up durations. Although subgroup and sensitivity analyses were implemented to explore potential sources of heterogeneity, residual heterogeneity may have influenced the pooled estimates. Finally, several subgroup analyses were based on small sample sizes with limited statistical power, thereby warranting a cautious interpretation of these results.
In conclusion, BBs, which are widely available, cost-effective, and generally well tolerated, may offer survival benefits in patients with CRC, particularly in the perioperative setting and in combination with anti-VEGF regimens. Given the observational nature of the included studies, the causal relationship between BB use and improved survival cannot be definitively established. Therefore, future large-scale, multicenter, randomized controlled trials are warranted to validate these findings; clarify causality; and determine optimal patient selection, treatment timing, and therapeutic combinations. Moreover, our findings provide an important theoretical groundwork for the design and implementation of future randomized controlled trials exploring the oncologic potential of BBs in CRC.
Conclusion
Through this comprehensive meta-analysis, we revealed that BBs may be associated with improved survival outcomes in patients diagnosed with CRC. Notably, beneficial effects were observed across the key survival endpoints (CSM, ACM, and mOS), particularly pronounced in patients undergoing radical resection or receiving bevacizumab-based anti-VEGF therapy. These findings suggest that BBs may contribute to sustained disease control by attenuating neurohumoral stress signaling, enhancing antitumor immunity, and promoting physiological stability during perioperative care. However, as the included studies were observational in nature, these associations should not be interpreted as causal, nor do they justify the initiation of BB therapy solely for anticancer purposes. Existing evidence supports the continuation of BBs in patients already using them for cardiovascular indications, whereas future confirmation from large, well-designed randomized controlled trials is required. Although improvements in mPFS did not achieve statistical significance, BBs may still exert delayed and cumulative benefits via immunomodulation, suppression of metastatic progression, and enhanced responsiveness to subsequent treatment lines.
In the future, rigorously designed prospective clinical trials will be essential to elucidate the underlying mechanisms of BB therapy; identify appropriate patient subgroups; and determine the optimal timing, dosage, and synergistic combinations of BBs with immunotherapies, targeted agents, and perioperative interventions. Given their low cost, established cardiovascular safety, and broad clinical availability, BBs exhibit considerable promise as adjunctive agents in individualized CRC management. These results provide a foundation for hypothesis generation and prospective validation via randomized controlled trials.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251397839 - Supplemental material for Beta-blocker use and survival outcomes in colorectal cancer patients: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605251397839 for Beta-blocker use and survival outcomes in colorectal cancer patients: A systematic review and meta-analysis by Rui Li, Jie Chen, Yingkai Chen, Kai Jin, Yong Chen, Changyu Deng, Xuefen Liu and Yue Li in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank the staff of the Department of Oncology and the Department of Cardiology, The People’s Hospital of Rongchang District, for their assistance in data collection.
Authors’ contributions
Rui Li, Jie Chen, Xuefen Liu, and Yue Li contributed to study conception and design. Rui Li, Jie Chen, and Yingkai Chen performed data collection and statistical analysis. Kai Jin, Yong Chen, and Changyu Deng critically revised the manuscript. Xuefen Liu and Yue Li supervised the project and approved the final version. All authors read and approved the final manuscript.
Data availability statement
All data used for conducting this meta-analysis are publicly available online from previously published studies cited in the reference list.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
