Abstract
Objective. Chemotherapy-induced oral mucositis (COM) is characterized by painful inflammation with prolonged damage that involves the pathological pain-evoking prostaglandin E2 (PGE2). We previously found that gargling with hangeshashinto (HST), a traditional Japanese medicine, was effective for the treatment of COM. However, little is known regarding the mechanisms. Our aim was to identify the active ingredients and clarify the characteristic effects of HST on the PGE2 system. Methods. Prostanoids produced by human oral keratinocytes (HOK) stimulated with IL-1β were measured by enzyme immunoassay. Active ingredients that regulate PGE2 production were identified and quantified by liquid chromatography–tandem mass spectrometry (LC-MS/MS) and a culture system of HOK cells. Results. Inducible PGE2, PGD2, and PGF2α, metabolites of cyclooxygenase (COX) pathways, were reduced by HST (10-300 µg/mL) without inducing cytotoxicity. The active ingredients of HST were quantified by LC-MS/MS, and [6]-shogaol, [6]-gingerol, wogonin, baicalein, baicalin, and berberine were shown to reduce PGE2 production. A mixture of these 6 ingredients at concentrations equal to 300 µg/mL of HST strongly suppressed PGE2 production to the same level as HST. [6]-Shogaol and [6]-gingerol did not decrease COX-2 mRNA expression and mostly inhibited PGE2 metabolic activity in an assay using intact HOK cells, suggesting that they regulate PGE2 synthesis at the posttranscriptional level. Wogonin, baicalin, and berberine inhibited expression of COX-2 mRNA without affecting PGE2 metabolic activity. Moreover, wogonin, but not [6]-shogaol, suppressed phosphorylation of mitogen-activated protein kinases (p38s and JNKs). Conclusions. These lines show that HST includes several PGE2-regulating ingredients that have different mechanisms and can function as a multicomponent and multitarget agent for treatment of COM, indicating that HST may be beneficial in a new medical strategy for COM treatment.
Introduction
Chemotherapy-induced oral mucositis (COM) is a complication that is frequently encountered in cancer patients and may delay the treatment plan in the case of severe morbidity.1,2 The oral mucosal injury involves chronic and/or intense pain, and affects nutritional intake and oral hygiene, as well as increases the risk for local and systemic infection, resulting in deterioration of the quality of life.1,3
Prostaglandin E2 (PGE2) is a critical mediator produced by cyclooxygenase (COX) and well established to instigate acute inflammation characterized by edema and hyperemia and to increase bradykinin-evoked pain. 4 Activation of the COX pathway is reported to be associated significantly with pain scores in COM. 5 Constitutive PGE2 is expressed in the normal mucosa of digestive organs; however, inducible PGE2 is produced largely by inflammation-evoking factors such as injury, infection, and mental stress. PGE2 produced largely and continuously in the oral cavity is surely involved in mucositis, followed by refractory stomatitis and hyperalgesia.
Hangeshashinto (HST) is a traditional Japanese medicine containing 7 herbal crude drugs.6,7 HST is prescribed in Japan to treat inflammatory diarrhea, gastritis, and stomatitis. We recently found that HST was effective as a gargle for the treatment of COM in a pilot clinical study 8 and a randomized, placebo-controlled, double-blind clinical trial (ESMO2012 meeting, abstract 1595P). HST was demonstrated to inhibit PGE2 production directly in human gingival fibroblasts 9 and reduce the PGE2 contents in the colons of several animal models of diarrhea using anticancer drug, cholera toxin, or castor oil, resulting in amelioration of inflammatory damage.10-12
It has been reported that some ingredients of HST inhibit PGE2 production and/or COX-2 expression.13-22 Phenylpropanoids like [6]-shogaol (6SG) and [6]-gingerol (6GG), flavonoids like wogonin (WGN), baicalein (BCN), and baicalin (BC), and isoquinoline alkaloids like berberine (BBR) are well established to have anti-PGE2 activity via their particular mechanisms. However, these studies demonstrated the effects and features of each as only a single compound, but not HST as a multicomponent agent. Moreover, effective concentrations and target molecules differ among reports and are controversial. Because it is unclear how much of these active ingredients HST contains, the mechanism by which HST inhibits PGE2 production is not accurately understood. Therefore, the aim of this study was to identify and quantify the active ingredients in HST that regulate PGE2 production by liquid chromatography–tandem mass spectrometry (LC-MS/MS) and a culture system of human oral keratinocytes, and to clarify the characteristics of the anti-PGE2 effect of HST as a preventive and/or therapeutic agent for COM.
Materials and Methods
Reagents
Hangeshashinto (Lot No. 2100014010) is an aqueous extract containing a mixture of 7 kinds of crude drugs: Pinellia tuber, Scutellaria root, processed ginger, glycyrrhiza, jujube, ginseng, and Coptis rhizome. The dried powdered extract form of HST was obtained from Tsumura and Co (Tokyo, Japan). HST was suspended in dimethyl sulfoxide (DMSO) at 100 mg/mL, diluted 100-fold with culture medium, and filtered through a 0.45-µm membrane. HST was added to cultures at final concentrations of 3, 10, 30, 100, or 300 µg/mL.
[6]-Shogaol, 6GG, WGN, BC, BBR, [10]-gingerol, and indomethacin were purchased from Wako Pure Chemical Industries, Ltd (Osaka, Japan). [8]-Shogaol, [10]-shogaol, [8]-gingerol, and BCN with purities high enough to be evaluated in biological tests were obtained from Tsumura and Co JNK inhibitor I, and SB202190 were purchased from Calbiochem (San Diego, CA).
Primary human oral keratinocytes (HOK, Lot No. 5854) were purchased from ScienCell Research Laboratories (Carlsbad, CA), and maintained in poly-
Measurement of Prostanoids
Human oral keratinocyte cells (5000 cells/well) seeded in 96-well plates were incubated in medium containing 2% FBS overnight. After replacement of culture fluids with the same fresh medium, the cells were preincubated with test samples for 20 to 30 minutes, followed by an additional culture for 6 hours with or without 10 ng/mL of human interleukin-1β (IL-1β; PeproTech, Inc, Rocky Hill, NJ). PGE2, PGD2, PGF2α, and leukotriene B4 (LTB4) in the culture supernatants were determined by enzyme immunoassay (EIA) according to the manufacturer’s instructions (Cayman Chemical, Ann Arbor, MI). PGD2 was measured after conversion of PGD2 to a stable methoxime derivative.
THP-1 cells (25 000 cells/well) were treated for 2 days with 100 nmol/L of phorbol 12-myristate 13-acetate (PMA; Sigma-Aldrich, St Louis, MO) in 96-well microplates to differentiate them into macrophage-like cells. 23 The culture fluids were replaced with fresh medium without PMA, and cells were allowed to rest for 1 day. Then, the cells were preincubated with test sample for 20 minutes, following by additional culture for 24 hours with or without 1 µg/mL of Escherichia coli–derived lipopolysaccharide (LPS; Sigma-Aldrich). PGE2 concentrations were measured as previously described.
Cell Viability
A lactate dehydrogenase (LDH) release assay was performed using a Cytotoxicity Detection LDH kit from Kyokuto Pharmaceutical Industrial (Tokyo, Japan). Cell viability was calculated following the formula as relative LDH activity in comparison with the maximum value generated by 0.2% (v/v) Triton X-100: viability (%) = 100 × ((c − a)/(c − b)), where a = HST, b = cells alone, and c = Triton-X.
Human oral keratinocyte (OK) cells (5000 cells/well) seeded in 96-well plates were incubated in medium containing 2% FBS overnight. Then, the cells were further cultured for 6, 12, or 24 hours after replacement of culture fluids with fresh medium containing 10% FBS and the test sample. Cell metabolic activities were measured using an XTT reduction assay kit (Biological Industries, Beit Haemek, Israel) according to the manufacturer’s instructions.
Measurement of Active Ingredients by LC-MS/MS
Hangeshashinto powder (500 mg) was suspended in 12.5 mL of methanol/purified water (75:25, v/v). The suspension was shaken for 5 minutes and sonicated for 15 minutes. After centrifugation at 3000 rpm for 5 min at 4°C, the supernatant was collected as the first extract solution. Then, 12.5 mL of methanol/purified water (50:50, v/v) was added to the residue, and the second extraction solution obtained by the procedure described above was mixed with the first extract solution. The mixture was diluted 1000-fold with the elution buffer, and was applied to the LC-MS/MS analysis. Ingredients of HST were measured by a QTRAP 5500 system (AB SCIEX, Tokyo, Japan) equipped with an Agilent 1260 HPLC system (binary pump, online degasser, auto plate-sampler and column oven; Agilent Technologies, Tokyo, Japan). Analytical conditions are summarized in the supplemental material (Tables S1 and S2). The lower limits for quantification were 0.01 ng/mL for 6GG, [8]-gingerol, and [10]-gingerol; 0.1 ng/mL for 6SG, [8]-shogaol, [10]-shogaol, WGN, and BBR; and 1 ng/mL for BCN and BC.
Gene Expression Analysis
To measure COX-2 mRNA, we used real-time qRT-PCR with TaqMan technology (Applied Biosystems, Warrington, UK). HOK cells were stimulated with IL-1β for 2 hours in the presence of test sample and lysed in RLT solution (Qiagen, Valencia, CA) containing 1% 2-ME, and total RNA was isolated using an RNeasy kit (Qiagen) according to the manufacturer’s recommendations. The respective cDNA were prepared using a high-capacity RT kit (Applied Biosystems). PCR was performed using the default parameters of the ABI Prism 7900 sequence detector. Sample-to-sample variation in RNA loading was controlled by comparison with β-actin.
Measurement of PGE2 Metabolic Activity Using Intact Cells
Enzymatic activity relating to PGE2 synthesis in intact IL-1β-stimulated HOK cells was determined by measuring the accumulation of PGE2 in the culture fluids containing arachidonic acid. Briefly, HOK cells (5000 cells/well) cultured overnight in 96-well plates, were further incubated for 24 hours after replacement of medium with fresh medium containing 2% FBS and 10 ng/mL IL-1β. The next day, the culture fluids were removed, rinsed once, and replaced with fresh medium containing 2% FBS, 100 µmol/L cycloheximide (Wako Pure Chemical Industries, Ltd), and a test sample. Twenty minutes later, arachidonic acid was added to the culture at a final concentration of 10 µmol/L and further incubated for 20 minutes. PGE2 concentrations were measured as described above.
MAPK Signaling Assay
Human oral keratinocyte cells (8000 cells/well) cultured overnight in 24-well plates were incubated with test sample for 20 minutes, followed by additional incubation for 30 minutes with or without 10 ng/mL IL-1β. Cells were washed twice with cold phosphate-buffered saline, and lysed in the lysis buffer from a Proteome Profiler Human Phospho-MAPK Array Kit (R&D Systems, Minneapolis, MN). After stirring for 30 minutes at 4°C and centrifuging at 10 000 × g for 5 minutes at 4°C, cell lysis samples were collected. The protein concentration was measured using a Protein Assay kit (Bio-Rad Laboratories, Hercules, CA). The Proteome Profiler Human Phospho-MAPK Array Kit was used for detecting signal transduction focusing on phosphorylation of ERK1, ERK2, JNK1, JNK2, JNK3, JNK pan, MSK2, p38α, p38β, p38γ, p38δ, RSK1, RSK2, p70 S6K, Akt1, Akt2, Akt3, Akt pan, GSK-3α/β, GSK-3β, and HSP27. To detect the signals, membranes treated with HRP-labeled second antibodies (GE Healthcare, Piscataway, NJ) were exposed to ECL Plus solution (Thermo Scientific Pierce, Rockford, IL), followed by chemiluminescence visualization using a Typhoon 9410 (GE Healthcare) at Ex 457 nm/Em 520 nm.
Statistical Analysis
All values are expressed as the mean ± standard error of the mean. Statistical significance was evaluated by 1- or 2-way analysis of variance, and a probability of less than .05 was considered significant at the Dunnett’s test or Student’s t test.
Results
Effects of HST on Prostanoid Production
The PGE2 concentration in the culture fluids of HOK cells was increased 29.7 times at 6 hours after the addition of IL-1β (Figure 1). HST showed concentration-dependent inhibition of inducible PGE2 production at 10 to 300 µg/mL, exhibiting strong activity with more than 90% inhibition at 100 and 300 µg/mL, whereas HST had little effect on constitutive PGE2 production without IL-1β. Indomethacin as a positive control intensely inhibited PGE2 production as expected. As shown in Figure 2, HST significantly reduced LPS-induced PGE2 production by macrophage-like cells, PMA-activated THP-1 cells, at concentrations of 30 and 100 µg/mL.

Hangeshashinto (HST) inhibited prostaglandin E2 (PGE2) production by IL-1β stimulated human oral keratinocyte (HOK) cells. HOK cells were incubated with the indicated concentrations of HST or 100 µmol/L of indomethacin (IND) for 30 minutes, and then stimulated by 10 ng/mL of human IL-1β or vehicle for 6 hours. The PGE2 in the culture fluid was determined by PGE2-specific enzyme immunoassay. N = 3. *Significant at Dunnett’s test, †Significant at Student’s t test.

Hangeshashinto (HST) inhibited prostaglandin E2 (PGE2) production by lipopolysaccharide (LPS)-stimulated macrophage-like cell. Cells of the human monocytic leukemia line THP-1 were differentiated to macrophage-like cells by exposure to 100 nmol/L phorbol 12-myristate 13-acetate (PMA) for 2 days. After resting for 1 day, macrophage-like cells were incubated with the indicated concentrations of HST for 20 minutes, and then stimulated by 1 µg/mL Escherichia coli–derived LPS or vehicle for 24 hours. PGE2 in the culture fluid was determined by enzyme immunoassay (EIA). N = 3 (ND, not detected). *Significant at Dunnett’s test.
We next investigated the selectivity of the HST effect by measuring prostanoids other than PGE2. Namely, PGE2, PGD2, PGF2α, and LTB4 produced by IL-1β-stimulated HOK cells were measured using the same culture fluid samples (Figure 3). PGE2, PGD2, and PGF2α were increased 19.1, 20.3, and 4.8 times, respectively, by IL-1β, and HST inhibited these prostanoids in the same manner. In contrast, the LTB4 concentration was not changed by addition of either IL-1β or HST. To examine the possibility of cytotoxic activity by HST in HOK cells, LDH concentrations in the fluids were measured after 6-hour culture (Figure 4A). HST exhibited no cytotoxic activity even at the highest concentration of 300 µg/mL, whereas HST evidently inhibited PGE2 production. We further examined the effect of HST on the metabolic activity of HOK cells by an XTT reduction assay. HST did not have a significant effect during 6- and 12-hour cultures, but culture for 24 hours with the maximal concentration of 300 µg/mL HST reduced metabolic activity (Figure 4B).

Prostaglandin E2 (PGE2), prostaglandin D2 (PGD2), and prostaglanding F2α (PGF2α), but not leukotriene B4 (LTB4), production by IL-1β-stimulated human oral keratinocyte (HOK) cells was inhibited by exposure to hangeshashinto (HST). HOK cells were incubated with indicated concentrations of HST for 30 minutes, and then stimulated by IL-1β or vehicle for 6 hours. The concentrations of PGE2, PGD2, PGF2α, and LTB4 in the culture fluid were determined using the respective enzyme immunoassay (EIA) assay kits. N = 3 (none, no IL-1β). *Significant at Dunnett’s test.

Hangeshashinto (HST) selectively inhibited prostaglandin E2 (PGE2) production without cell damage. (A) Human oral keratinocyte (HOK) cells were preincubated with the indicated concentrations of HST, and then stimulated by IL-1β or vehicle for 6 hours. PGE2 in the culture fluid was determined by enzyme immunoassay (EIA). N = 3. Lactate dehydrogenase (LDH) activity was measured using a cytotoxicity detection LDH kit, and cell viability was calculated. (B) HOK cells were cultured for 6, 12, or 24 hours with the indicated concentrations of HST. Metabolic activity of HOK cells was measured using an XTT reduction assay kit. (N = 3-4). *Significant at Dunnett’s test.
Measurement of Active Ingredients in HST by LC-MS/MS
Which ingredients of HST inhibit PGE2 production by HOK cells cannot be discussed unless concentrations of the active ingredients of HST are accurately known. Therefore, LC-MS/MS was performed to measure active ingredients that have been reported to inhibit PGE2 production and/or COX-2 expression; 6SG, [8]-shogaol, [10]-shogaol, 6GG, [8]-gingerol, [10]-gingerol, WGN, BCN, BC, and BBR were evaluated in this study. As shown in Table 1, all ingredients were detectable, and BC and BBR were more sufficient compared with the others.
Quantitative Analysis of Representative Ingredients in HST Was Performed by LC-MS/MS. a

Abbreviations: HST, hangeshashinto; LC-MS/MS, liquid chromatography–tandem mass spectrometry.
Methanol/water extracts of dried HST (Lot No. 2100014010) were analyzed by LC-MS/MS.
Screening for Active Ingredients That Inhibit PGE2 Production
We tried to identify the active ingredients that inhibit PGE2 production in HOK cells. The 10 ingredients shown in Table 1 were tested at their respective concentrations equal to 300 µg/mL of HST, and simultaneously at 3 times higher and one third. As shown in Figure 5A, BC, WGN, and 6SG actively inhibited PGE2 production at concentrations equal to 300 µg/mL of HST, and suppressed it with more than 90% inhibition at three times higher concentration. Furthermore, 6GG, BCN, and BBR reduced PGE2 production to 34%, 40%, and 43%, respectively, at the middle concentration compared with the control, although their activities were weak. Next, we combined the 3 most active ingredients, BC, WGN, and 6SG, at concentrations equal to 300 µg/mL of HST (Mix-3), and tested the activity of the mixture. As shown in Figure 5B, Mix-3 suppressed PGE2 production by more than 95%, but there was a significant difference between Mix-3 and HST. Additionally, we evaluated a mixture of the 6 most active ingredients (Mix-6), which were BC, WGN, 6SG, 6GG, BCN, and BBR, and showed that Mix-6 strongly suppressed PGE2 production at the same level as HST.

Active ingredients that inhibit prostaglandin E2 (PGE2) production were identified. Human oral keratinocyte (HOK) cells were stimulated for 6 hours with IL-1β, and PGE2 in the culture fluids was measured by enzyme immunoassay (EIA). N = 4 (none, no IL-1β). (A) Concentrations (black frame/gray) equal to 300 µg/mL hangeshashinto (HST) are shown below, and concentrations of 3 times (deep gray) and one third (pale gray) were evaluated as well. Baicalin, 20.26; berberine, 3.35; baicalein, 0.95; wogonin, 0.60; [6]-gingerol, 0.39; [6]-shogaol, 0.17; [8]-gingerol, 0.040; [10]-gingerol, 0.027; [8]-shogaol, 0.018; [10]-shogaol, 0.016. Unit shown is µmol/L. (B) Also, 300 µg/mL of HST, Mix-3, a mixture of wogonin, baicalin, and [6]-shogaol, and Mix-6, a mixture of wogonin, baicalein, baicalin, berberine, [6]-shogaol, and [6]-gingerol, were added to the culture at concentrations equal to 300 µg/mL HST after pooling them in dimethyl sulfoxide (DMSO). *Significant at Dunnett’s test.
Moreover, 16 ingredients, BC, WGN, 6SG, 6GG, BCN, BBR, corymboside, coptisine, homogentisic acid, liquiritin, glycyrrhizic acid, acteoside, ginsenoside Rg1, ginsenoside Rb1, cyclic AMP, and panaxynol were tested at 1 and 10 µmol/L. BC, WGN, 6SG, 6GG, BCN, and BBR reduced PGE2 production at 1 and/or 10 µmol/L, while corymboside, homogentisic acid, liquiritin, glycyrrhizic acid, acteoside, ginsenoside Rg1, ginsenoside Rb1, and cyclic AMP did not affect it significantly (supplemental material: Figure S1).
Different Mechanisms Among Active Ingredients to Regulate PGE2 Production
To address the mechanism by which HST inhibits PGE2 production and compare the pharmacological profiles of the six active ingredients (BC, WGN, 6SG, 6GG, BCN, and BBR), we first examined the effects of HST (300 µg/mL) and the 6 active ingredients at concentrations equal to 300 µg/mL HST on COX-2 mRNA expressions (Table 2). While IL-1β stimulation resulted in a great increase (27 times) of COX-2 expression, HST inhibited it to 81%. BC, WGN, and BBR exerted significant inhibition at 76%, 78%, and 64%, respectively, while 6SG, 6GG, and BCN did not decrease it. Next, we investigated enzymatic activities involved in PGE2 synthesis. HST, 6SG, and 6GG significantly inhibited PGE2 synthesis (Table 2), and 6SG especially showed strong and reproducible activity in every test. In contrast, BC, WGN, BCN, and BBR showed no effect at the indicated concentrations. Indomethacin suppressed PGE2 synthesis completely (data not shown).
Enzymatic Activity and Expression of COX-2 Were Suppressed by Exposures to HST and Active Ingredients.

Abbreviations: COX-2, cyclooxygenase-2; HST, hangeshashinto; HOK, human oral keratinocyte.
HOK cells were cultured overnight, and medium was replaced with fresh medium containing the indicated concentrations of [6]-shogaol, [6]-gingerol, baicalein, baicalin, wogonin, berberine, HST (300 µg/mL), or vehicle, and incubated for 20 minutes. Two hours after adding IL-1β (10 ng/mL), cell samples were harvested real-time polymerase chain reaction (RT-PCR) using RLT buffer containing 1% 2-mercaptoethanol. Gene expressions of COX-2 were measured by real-time PCR. N = 3.
HOK cells cultured with IL-1β for 24 hours were pretreated with the same test samples for 20 minutes and further incubated with arachidonic acid (AA, 10 μmol/L) for 20 minutes. Prostaglandin E2 (PGE2) in the culture fluids was measured by enzyme immunoassay (EIA). N = 4 (none, no IL-1β or no AA).
Significant at Dunnett’s test.
Significant at Student’s t test.
Wogonin But Not 6SG Regulates Activation of MAPK Cascade
Baicalin, WGN, and 6SG are undoubtedly the representative active ingredients of HST. BC and WGN inhibited COX-2 expression with no effect on enzymatic activity, while 6SG exhibited the inverse effect, followed by PGE2 reduction. Because the specific activity of WGN on inhibition of PGE2 production is stronger than that of BC, we focused on WGN and 6SG, and tried to identify their target molecules in cell signal transduction. The 26 kinds of phosphorylation signals of the MAPK cascade in IL-1β-stimulated HOK cells were examined using a phospho-MAPK array kit. Focusing on signals with more than 2-fold elevation due to IL-1β stimulation, phosphorylations were upregulated remarkably in HSP27, JNK1, JNK2, JNK pan, p38α, and p38γ. WGN weakened the upregulation signals of HSP27 (57% inhibition), JNK1 (75%), JNK2 (52%), JNK pan (30%), p38α (73%), and p38γ (20%), while 6SG did not affect these changes in any signal (Figure 6). These results imply that JNKs and p38s may be key targets in regulating PGE2 production in IL-1β-stimulated HOK cells, and their selective inhibitors, JNK inhibitor I and SB202190, were therefore evaluated, showing that both inhibitors significantly inhibited PGE2 production (Table 3).

Wogonin, but not [6]-shogaol, suppressed phosphorylation of JNKs and p38s in IL-1β-stimulated human oral keratinocyte (HOK) cells. HOK cells were incubated with 1 µmol/L of wogonin, [6]-shogaol, or vehicle for 20 minutes and further incubated with IL-1β (10 ng/mL). Thirty minutes later, HOK cells were lysed using a lysing buffer. The phosphorylation status of the 26 kinds of MAPK molecules were evaluated using a phospho-MAPK array kit and measured by image scanner instruments after treatment with ECL plus Western blotting detection reagents. The clipped images of JNK and p38 sets showing some changes are arranged in order.
Selective Inhibitors of JNK and p38 Phosphorylation Diminished PGE2 Production by IL-1β-Stimulated HOK Cells. a
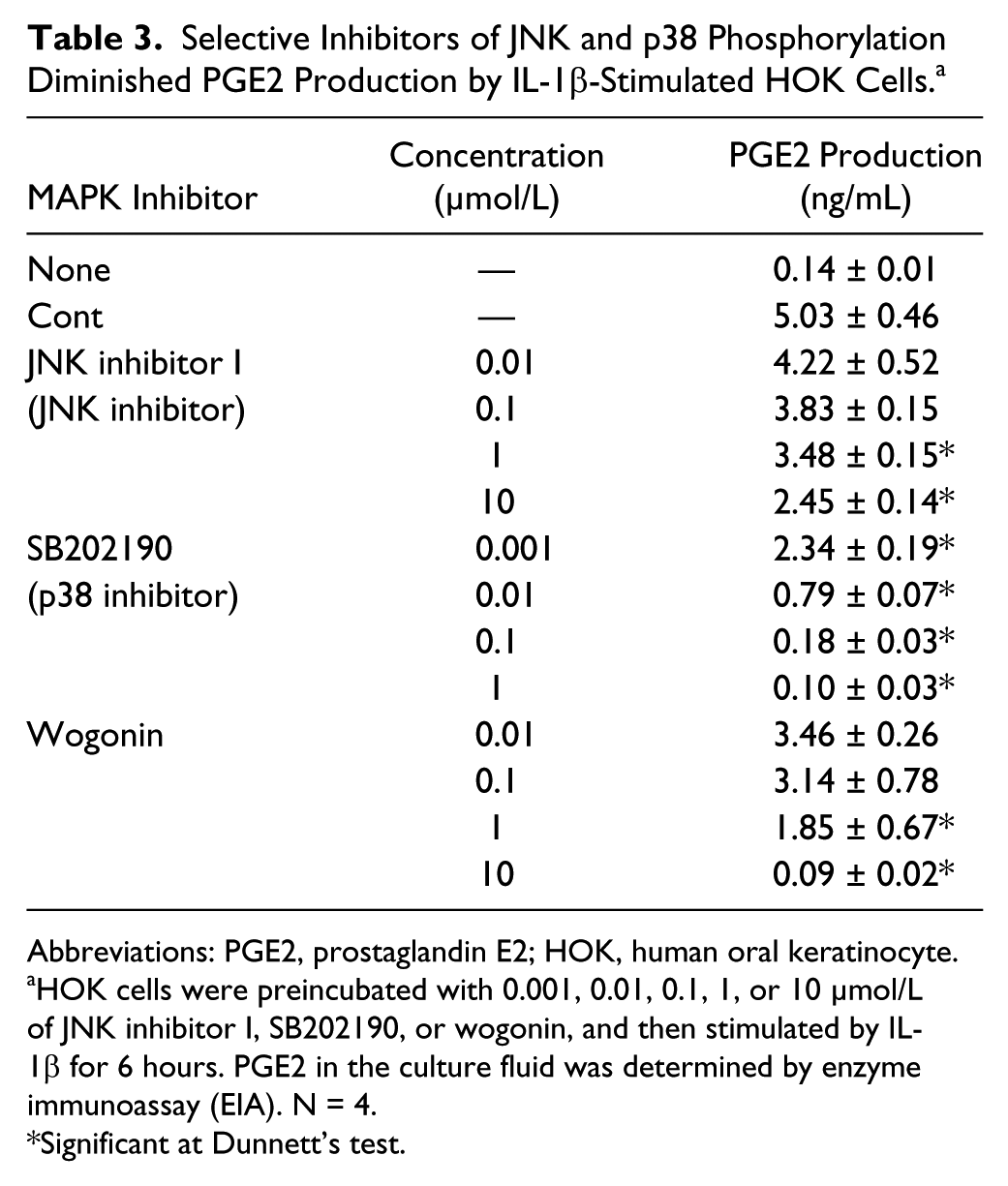
Abbreviations: PGE2, prostaglandin E2; HOK, human oral keratinocyte.
HOK cells were preincubated with 0.001, 0.01, 0.1, 1, or 10 µmol/L of JNK inhibitor I, SB202190, or wogonin, and then stimulated by IL-1β for 6 hours. PGE2 in the culture fluid was determined by enzyme immunoassay (EIA). N = 4.
Significant at Dunnett’s test.
Discussion
Our study is the first to measure and identify the ingredients in HST that inhibit PGE2 production, although the effects of HST have been demonstrated in a culture system of human gingival fibroblasts and in animal models of diarrhea.9-12 LC-MS/MS showed that HST contains at least 10 active ingredients, and that there was no large difference among several production lots of HST (data not shown). Mix-6, including BC, WGN, 6SG, 6GG, BCN, and BBR, showed the same level of activity as HST in assays considering their proportions in HST, evidently indicating that the anti-PGE2 effect of HST is caused by these 6 ingredients. Three ingredients, BC, WGN, and 6SG, were especially critical because Mix-3 reproduced most of the strong activity of Mix-6 and HST. It allowed us to understand that HST has 2 targets to suppress PGE2 production in HOK cells, as shown in Figure 7. One is blockage of the MAPK cascade, followed by accelerations of COX-2 expression and the arachidonic acid metabolic pathway. This blocking is caused by WGN and probably BC. The other target is direct inhibition of the PGE2 metabolic pathway by active ingredients like 6SG. Furthermore, these lines are almost entirely consistent with the previous report that HST exerted anti-PGE2 effects by inhibition of the MAPK pathway and COX-2 enzyme reaction. 9
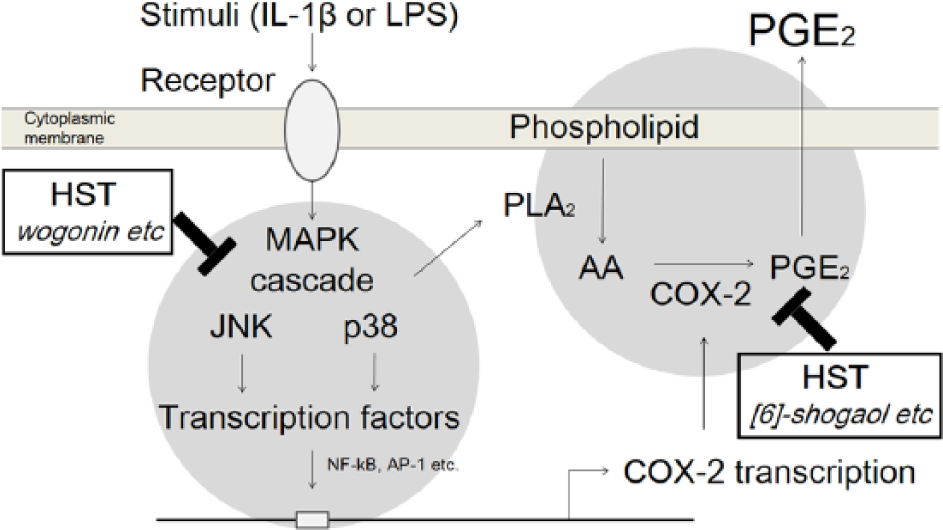
Hangeshashinto (HST) acted on multitargets relative to inducible prostaglandin E2 (PGE2). HST inhibited PGE2 production through suppression of the MAPK cascade, followed by acceleration of cyclooxygenase-2 (COX-2) expression and the arachidonic acid (AA) metabolic pathway by active ingredients like wogonin, and direct inhibition of PGE2 metabolic activity by active ingredients like [6]-shogaol.
Accumulating evidence demonstrates that WGN suppresses COX-2 expression in various inflammatory models, via inhibition of the MAPK cascade and transcription factor.24-27 The exposure of HOK cells to WGN at 0.6 µmol/L, equal to 300 µg/mL HST, evidently reduced expression of COX-2 mRNA. The results of the MAPK array showed that WGN lowered phosphorylations of JNKs and p38s critical to the activation of transcriptional factors like AP-1 and NF-κB, which are well-established to control COX-2 gene expression. In fact, JNK inhibitor I and SR11302 significantly inhibited PGE2 production as well as WGN did. These lines imply that WGN inhibited COX-2 gene expression by blocking activation of JNK and p38 signals, followed by downregulation of PGE2 production. In this study, BC and BBR also inhibited COX-2 expression at the same level as WGN. Because BC is a flavonoid analogue to WGN, 28 BC may use the same mechanism. Several studies showed that BBR modifies the intracellular signal pathway. For instance, BBR was demonstrated to suppress MAPK signals, followed by inhibition of COX-2 expression.20-22
[6]-Shogaol did not affect IL-1β-induced COX-2 expression at 0.177 µmol/L, equivalent to 300 µg/mL HST. Focusing on the results of the MAPK array, the phosphorylation of p38s and AKTs tended rather to increase due to the addition of 6SG, and therefore the effect of 6SG was quite different from that of WGN. The targets of 6SG on PGE2 inhibition must be in the post-transcriptional events of COX-2, and it is certain that the action mechanism of 6SG is the direct effect of enzymatic reactions to synthesize PGE2. One study demonstrated that 6SG inhibited PGE2 synthesis in a test targeting COX-2 using intact cells. 29 In addition, ginger-derived phenylpropanoids such as shogaols and gingerols were reported to inhibit the COX-2 reaction in an assay using purified COX-2 enzyme. 16 Therefore, 6SG and 6GG are thought to act directly on the PGE2 metabolic pathway. In addition, it should be noted that flavonoids have the potential to inhibit PGE2 synthesis at high concentrations. 19 In our study, WGN, BC, and BCN actually exerted the enzymatic activity at higher concentrations than those equivalent to 300 µg/mL HST.
We previously performed a preliminary study in cancer patients with COM during mFOLFOX6 or FOLFIRI treatment, and reported the efficacy of topical application of HST. 8 In that clinical study, HST was administered to patients with grade 1 or worse COM according to the Common Terminology Criteria for Adverse Events version 4. HST was suspended at 30 mg/mL in water and gargled for 10 seconds after meals. Additionally, HST was topically applied to oral ulcers using a cotton ball. Thirteen of the 14 patients (92.8%) responded to the HST therapy. Grade 3/4 mucositis was significantly reduced from 3.2 to 1.8 (P = .02), as well as grade 1/2 mucositis from 1.6 to 0.6 (P = .034), after 2 weeks of HST treatment. Moreover, we recently found that HST was effective using the same protocol for the treatment of COM, and it reduced ulcer healing time by half in patients with grade 2 or higher mucositis in a randomized, placebo-controlled, double-blind clinical trial (ESMO2012 meeting, abstract 1595P). Although there is no report that clarifies the mechanism by which HST affects pain evoked in the oral cavity, we often observed that oral pain was alleviated several minutes after topical administration of HST.
Dysregulated PGE2 is known to promote cancer progression, in part by increasing cell proliferation and angiogenesis and inhibiting apoptosis.30-33 PGE2 exerts its biological functions by activating 4 receptors, namely EP1, 2, 3, and 4. 34 In particular, signals through EP2 and EP4 receptors increase cAMP levels via activation of adenylate cyclase, followed by activation of the cAMP/PKA/CREB pathway. This signal suppresses differentiation and proliferation of Th1 cells, which are important for immune defense against viral/bacterial infection and cancer.35-37 That is why COX-2 inhibitors like celecoxib have been evaluated for their anti-cancer effect in clinical studies. Recently, nonsteroidal anti-inflammatory drugs (NSAIDs) and selective COX-2 inhibitors have proven to be promising and attractive candidates for clinical cancer chemoprevention. The preventive efficacy of these agents is supported by a large number of animal and epidemiological studies.38,39 HST with a potential to inhibit PGE2 production by a multitargeted mechanism may be also beneficial from the perspective of enhancing immunosurveillance.
Monocytes/macrophages are activated by microbe-derived substances such as LPS or inflamed tissue–derived proinflammatory cytokines, and evoke a subsequent immune response. Activated macrophages are well known to produce a large amount of PGE2, and to be key cells in the control of morbidity in oral mucositis. It can be speculated that mucosal barrier functions are downregulated in patients receiving chemotherapy and/or radiotherapy, allowing invasion by microbes, and that chronic activation of macrophages occurs in oral mucositis.1,2 Our study demonstrated that HST inhibited LPS-induced PGE2 production (Figure 2) and A23187-induced IL-1β production (supplemental material: Figure S2) by PMA-activated THP-1 cells. Moreover, HST is reported to suppress nitric oxide production by LPS-stimulated macrophage-like cells, RAW 264.7. 40 Taken together, HST is expected to regulate macrophage functions in the oral cavity.
Bacterial infection in the oral cavity is one of the factors aggravating stomatitis. Dental health care prior to medical treatment can help decrease oral complications during and after treatment.41,42 It is demonstrated that BBR has an antibiotic effect against some kinds of microbes including Porphyromonas gingivalis.43-45
The inhibitory effect of HST on PGE2 metabolic activity is due mainly to 6SG, 6GG, and partially to flavonoids. In addition, BC and WGN inhibit COX-2 expression. According to the literature, ginger-derived phenylpropanoids, including shogaols and gingerols suppress cPLA2 enzymatic activity. 15 These lines show that HST is quite different from NSAIDs and COX-2 selective inhibitors. It should be noted that shogaols and gingerols are hardly detected in human plasma in their respective unchanged forms after they are administered orally because they are easily metabolized by β-glucuronidase and sulfatase in the small intestine and liver. 46 Therefore, unlike NSAIDs and COX-2 selective inhibitor, HST does not exert a serious risk of cardiovascular complications.
Conclusions
Liquid chromatography–tandem mass spectrometry and PGE2 production screening assays identified BC, WGN, 6SG, 6GG, BCN, and BBR as the anti-PGE2 ingredients of HST. HST is a multicomponent anti-PGE2 agent with multitargeting effects, at least dual suppression of COX-2 expression and PGE2 metabolic activity. As demonstrated by clinical evidence, HST may be a promising agent for the treatment of COM.
Footnotes
Acknowledgements
We thank Drs Yuji Omiya, Masahiro Yamamoto, and Yoshio Kase for helpful discussions, and Tatsuro Shigenobu for technical assistance.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AK, CM, CM, KO, and YM are employees of Tsumura and Co, which funded this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Tsumura and Co. TK, KM, and YU received grant support from Tsumura and Co.
