Abstract
6-Shogaol from ginger has anti-inflammatory, anti-oxidation and anti-cancer effects.
Introduction
The second deadliest malignancy in both sexes is colorectal cancer (CRC). 1 With the improvement of living standards, unhealthy diet habits and obesity lead to an increase in the incidence of colorectal cancer. The incidence of colorectal cancer in transition countries is approximately 3 times higher than that in nontransition countries.2,3 In the past decade, the morbidity and mortality of CRC in China have been on the rise, and CRC is one of the major public medical burdens in China. 4 Although advances in medical technology have significantly improved the survival rate of patients with advanced cancer, the life quality of patients with colorectal cancer has been negatively affected by cancer metastasis. 5 In the past 10 years, among patients with obvious characteristics, metastasis accounted for 90% of cancer-related deaths. 6 Approximately 35% of colorectal cancer patients have metastatic disease at the time of diagnosis, and up to 50% of nonmetastatic colorectal cancer patients will eventually show metastatic disease. 7 For metastatic colon cancer, conventional surgery, radiotherapy, and chemotherapy have unsatisfactory results. Therefore, it is very important to explore new methods for the treatment of colorectal cancer, especially in inhibiting cancer cell metastasis.
Epithelial-mesenchymal transition (EMT) plays a key role in promoting the metastasis of quiescent tumor cells. 8 EMT is a procedure in which cells lose epithelial properties and gain their stromal properties. 9 Previous studies have found that the metastasis of colorectal cancer is related to EMT.10,11 Mesenchymal like fibroblasts will be formed in EMT process, and the formed fibroblasts can produce more MMPS. 12 Subsequently, cells with EMT features are often able to degrade and invade their basal extracellular matrix by expressing the matrix metalloproteinase family (MMPS). 13 The whole process enhances cell mobility, so it is not surprising that it has been linked to the spread of tumor cells. 14 MMP-2, as one of the gelatin proteases in matrix metalloproteinases, can degrade the extracellular matrix (ECM), which helps cells metastasize, significant changes in the adhesion phenotype of tumor cells have been demonstrated through the use of altered MMP-2 cell transfection systems. Therefore, MMP-2 can be used as a marker of cancer cell migration, and the increase of MMP-2 protein expression also indicates the increase of cell migration. 15 In general, the higher the expression of E-cadherin protein, the denser the cell, the more polarity, and the more difficult it is to separate. In contrast, the higher the expression of N-cadherin protein, the looser the cells, indicating a greater EMT process. Therefore, the expression of proteins related to the EMT process can be used as key information to judge the degree of cell metastasis.16,17 Therefore, inhibiting the expression of EMT process- and migration-related proteins is an effective method to inhibit cancer metastasis.
Ginger has been used as medicine and food in China for a long time. Ginger can warm the stomach and stop vomiting, help digestion and protect intestinal function.18,19 Studies have found ginger to help reduce acute and delayed nausea and vomiting in breast cancer patients after chemotherapy, and since there are no serious side effects, ginger is considered safe. 20 However, preclinical safety data do not rule out potential toxicity and should be monitored, especially after prolonged consumption of ginger. 21 An increasing number of studies have shown that ginger has a variety of active ingredients, and 6-Shogaol derived from ginger has obvious antioxidant, 22 anti-inflammatory, 23 obesity, diabetes, and anticancer effects. 24 6-Shogaol can regulate the balance of differentiation between colon epithelial absorption cell lines and secretory cell lines, and repair damaged colon mucosal tissues. 25 Moreover, 6-Shogaol has a protective effect against tumor necrosis factor-α (TNF-α) induced intestinal barrier dysfunction and chemotherapeutic intestinal epithelial cell injury in human intestinal cell models.26,27
6-Shogaol was found to inhibit NF-κB signaling pathway to reduce the expression of MMP-2 and MMP-9 to inhibit the invasion and metastasis of liver cancer. 28 6-Shogaol activated PPARγ in breast cancer MCF-7 cells and colon cancer HT29 cells and inhibited NF-κB activity in a PPARγ-dependent manner to inhibit the proliferation and promote apoptosis of breast and colon cancer cells. 29 It has also been suggested that 6-Shogaol can inhibit the phosphorylation of NF-κB p65 to reduce the nuclear displacement of NF-κB and reduce the occurrence of related tumors. 30 At the same time, ginger reduces the secretion of VEGF and interleukin-8 (IL-8) in ovarian cancer by inhibiting NF-κB activation. 31 In the EMT process, snail mRNA levels can be reversed by inhibiting NF-κB signaling. 32 However, the role and mechanism of 6-Shogaol in inhibiting the colon cancer EMT process through the NF-κB signaling pathway have not been clarified, and more in-depth research is needed.
Through this study, we hope to confirm that 6-Shogaol can weaken or inhibit the EMT process via the IKKβ/NF-κB/Snail signaling pathway, thereby inhibiting the migration of colon cancer cells, and prove that 6-Shogaol can also promote the apoptosis of colon cancer cells and inhibiting proliferation. We hope to provide more basic research evidence of anti-tumor metastasis for 6-Shogaol.
Materials and Methods
Reagents
6-Shogaol was obtained from Purechem-Standard (Cas:555-66-8, Chengdu), dissolved in dimethyl sulfoxide (DMSO), and stored at −40°C. Antibodies against E-cadherin (3195), MMP-2 (40994), N-cadherin (13116), Snail (3879), NF-κB p65 (8242), P-NF-κB p65 (3033), and GAPDH (8884) were purchased from Cell Signaling Technology (Massachusetts, USA). Anti-EG-VEGF antibody (ab150375) was purchased from Abcam Technology (Cambridge, MA). IKKβ (sc-34673) was purchased from Santa Cruz Biotechnology (Dallas, USA). Annexin V/PI staining dye was purchased from BD Biosciences (San Jose, CA, USA).
Cell Lines and Cell Culture
The human colon cancer lines Caco2 and HCT116 were purchased from Shanghai Fuheng Biology. 6-Shogaol was obtained from Purechem-Standard (Cas:555-66-8, Chengdu). The Caco2 cell lines were cultured in the RPMI-1640 medium, and the HCT116 cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM) and grown in monolayers. Both media supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin, FBS, and penicillin/streptomycin were purchased from Gibco and incubated in an incubator containing 5% CO2 at 37℃.
Cytotoxicity Assay
Cell viability was analyzed using an MTT assay. Approximately 5 × 103 Caco2 cells/well were plated in 96-well plates with RPMI 1640 (containing 10% FBS) medium overnight, and then 6-shogaol was configured to be used in different concentrations (0, 20, 40, 60, 80, 100 μM). Approximately 5 × 103 HCT116 cells/well were plated in 96-well plates with DMEM (containing 10% FBS) overnight and then treated with different concentrations of 6-Shogaol (0, 10, 20, 40, 60 μM). The effect of different concentrations of 6-Shogaol on HCT116 was not as obvious as that of Caco2 in the pre-experiment, and the selected concentration shows a better concentration-dependent trend, so 5 concentrations were used in Caco2 cells and 4 concentrations were used in HCT116 cells. After 48 hours of dosing treatment, add 10 µl MTT solution to each well of the 96-well plates, and put the plate back into the incubator for 4 hours. Then, 100 µl DMSO was added to dissolve the formazan crystals. Finally, the plates were measured at 490 nm using a Tecan microplate reader (Tecan US, Inc., Morrisville, NC, USA). All experiments were performed at least 3 times.
Clone Formation Assay
A colony formation assay can be used to test the effect of 6-shogal on the sensitivity of colon cancer cells. Approximately 1 × 103 Caco2 cells/well were seeded in 6-well plates and each well in 2 ml RPMI 1640 (containing 10% FBS) medium overnight. Approximately 1×103 HCT116 cells/well were seeded in 6-well plates and each well in 2 ml DMEM (containing 10% FBS) medium overnight. Then, the medium was changed to a fresh medium containing different doses of 6-Shogaol (0, 20, 40, 60, 80, 100 μM) and (0, 10, 20, 40, 60 μM). The determination concentration of colony formation is selected based on the MTT results, which showing a better concentration-dependent trend. The culture medium containing 6-Shogaol was removed and replaced with new medium. After 12 days of treatment, Caco2 cells and HCT116 cells were washed with phosphate-buffered saline (PBS) twice, fixed with 4% paraformaldehyde for 30 minutes, and stained with 0.1% crystal violet. All experiments were performed at least 3 times.
Wound Healing Assay
Caco2 cells were plated at densities of 2.5 × 105/well, and HCT116 cells were plated at densities of 3.0 × 105/well into 6-well plates until the well bottom was 85%−95% covered. Then, the medium was removed using a 200 µl pipette tip to create a cell-free gap in the well bottom. After rinsing with PBS twice, different concentrations of 6-Shogaol were added to the plate and cultured for 24 and 48 hours at a constant temperature incubator at 37℃. Finally, 4 fields were randomly selected under the light microscope (×40, inverted microscope, Olympus, CKX53 IPC) to observe cell migration, and the migration distance was evaluated using ImageJ software. Quantitative results are expressed as the means ±SEMs of 3 independent experiments.
Transwell Migration Assay
The migration of Caco2 cells and HCT116 cells were evaluated using Transwell assays. Caco2 cells and HCT116 cells were suspended in 200 µl serum-free medium at densities of 2 × 105/well and were plated in the upper chamber of Transwell (6.5 mm diameter inserts; 8.0 µm PET membrane; Corning Incorporated), Subsequently, 750 µl 1640 medium (containing 10% FBS) and DMEM medium (containing 10% FBS) were added to the lower chamber overnight in a constant temperature incubator. Then, the culture medium was removed, and different concentrations of culture medium containing 6-Shogaol were added to the upper chamber for 48 hours after incubation. The migrated cells were passed through the membrane and fixed with 4% paraformaldehyde for 30 minutes, washed twice with PBS, and then stained with 0.1% crystal violet for 15 minutes. Finally, images of the migrated cells were obtained with a microscope (×40 and ×100, inverted microscope, Olympus, CKX53 IPC) and counted using ImageJ software. Quantitative results were expressed as the means ±SEMs of 3 independent experiments.
Western Blot Analysis
After preparing whole cell protein lysates, the protein sample was denatured with a mixture of 5 × sample loading buffer of 1:4 volume and boiled at 100℃ for 10 minutes. SDS polyacrylamide gel electrophoresis (SDS-PAGE) was used to separate equal amounts of proteins and transfer them to nitrocellulose membranes (Millipore, Billerica, MA, USA). The nitrocellulose membranes were then blocked with 5% nonfat milk in TBST (TBST containing 0.1% Tween-20) for 1 hour at room temperature.
The following primary antibodies were used in the current study: E-cadherin (1:1000), N-cadherin (1:1000), Snail (1:1000), MMP-2 (1:1000), VEGF (1:1000), IKKβ (1:1000), NF-κB (1:1000), p-NF-κB (1:1000), GAPDH (1:1000). The primary antibody was incubated overnight on a shaker at 4°C. After being washed 5 times (5 minutes/wash), the secondary antibody with HRP-conjugated anti-rabbit IgG was diluted with 1:10000 and then put into the membrane and incubated for 2 hours at room temperature, and then visualized by chemiluminescence (4A Biotech, BJ, China) and detected by Amersham ImageQuant 800 (GE Healthcare, CT, UAS). Actin or GAPDH was used as a control for normalization treatment, and ImageJ software was used to quantify the densities of the bands.
Apoptosis Detection
The Caco2 cell line and HCT116 cell line were plated on a 6-well plate of about 2.5 ×105 cells/well and placed in an incubator overnight. The cells were then treated with different concentrations of 6-Shogaol for 48 hours. After treatment, the cells were digested with trypsin and collected by centrifugation. Then, the cells were washed twice with sterilized ice-cold 1x PBS and stained with 100 µl/well 1× Annexin-binding buffer (BD Biosciences) that contained 2 μl of 1 μl Annexin V and PI (1 mg/ml) at room temperature in the dark for 15 minutes. The percentage of apoptotic cells was quantitatively determined using a Beckman Coulter flow cytometer 4 Lasers 13 Colors (Beckman Coulter CytoFLEX).
Statistical Analysis
All data are expressed as the means ±SEMs of 3 individual experiments. Statistical analysis was performed using the t-test in GraphPad Prism software (version 8.0, GraphPad Software, San Diego, CA). P < .05 indicates a significant difference.
Results
6-Shogaol Inhibits the Proliferation of Human Colon Cancer Cells
The cytotoxicity was detected by MTT with different concentrations of 6-Shogaol in Caco2 cell lines (0, 20, 40, 60, 80, and 100 µM) and HCT116 cell lines (0, 10, 20, 40, 60 µM) for 48 hours. As shown in Figure 1A, 6-Shogaol showed inhibitory effects on Caco2 cells in a dose-dependent manner, and the half-maximal inhibitory concentration (IC50) of Caco2 was 86.63 µM, and the LogIC50 is 1.938. In Figure 2A, 6-Shogaol showed inhibitory effects on HCT116 cells in a dose-dependent manner, and the IC50 of HCT116 was 45.25 µM, and the LogIC50 is 1.596. At the same time, the colony formation experiment showed that 6-Shogaol inhibited the proliferation of Caco2 cells and HCT116 cells with increasing concentrations (Figures 1C and 2C).

Cytotoxicity detection for 6-Shogaol on Caco2 cells. (A) The colon cancer cell line Caco2 was cultured and treated with different concentrations of 6-Shogaol (0, 20, 40, 60, 80, 100 µM) for 48 hours. (B) Quantifications of (C). (C) Proliferation detection for 6-Shogaol, the colon cancer cell line Caco2 was cultured and treated with different concentrations of 6-Shogaol (0, 20, 40, 60, 80, 100 µM) for 48 hours until the clumps of cells were clearly visible to the naked eye. *** P < .001. ****P < .0001 versus the control group (0 µM).

Cytotoxicity detection for 6-Shogaol on HCT116 cells. (A) The colon cancer cell line HCT116 was cultured and treated with different concentrations of 6-Shogaol (0, 10, 20, 40, 60 µM) for 48 hours. (B) Quantifications of (C). (C) Proliferation detection for 6-Shogaol, the colon cancer cell line HCT116 was cultured and treated with different concentrations of 6-Shogaol (0, 10, 20, 40, 60 µM) for 48 hours until the clumps of cells were clearly visible to the naked eye.** P < .01. ****P < .0001 versus the control group (0 µM).
Effect of 6-Shogaol on Colon Cancer Cell Apoptosis
The Annex V/PI staining test was used to detect the effect of 6-Shogaol on the apoptosis of colon cancer cells at 48 hours. Flow cytometry result showed that apoptosis increased with the increase of 6-Shogaol concentration, and apoptosis was evident in 80 μM Caco2 cells and 40 μM HCT116 cells (Figure 3A and C).

Apoptosis of 6-Shogaol on Caco2 in colon cancer cells. (A) Flow cytometry analysis of 6-Shogaol (0, 40, and 80 µM)-induced Caco2 cell apoptosis. (B) Quantification of (A). (C) Flow cytometry analysis of 6-Shogaol (0, 20, and 40 µM)-induced HCT116 cell apoptosis. (D) Quantification of (C). The upper and lower right quadrants show the percentage of late or early apoptotic cells. V/PI staining showed that the percentage of apoptosis after 48 hours of the 6-Shogaol treatment was significantly higher than that after 0 hour of the Caco2 and HCT116 treatment. *P < .05. **P < .01, versus the control group (0 µM).
Inhibitory Effect of 6-Shogaol on Colon Cancer Cell Migration
In order to eliminate the interference of proliferation inhibition on the experiment, Caco2 cell experiment selects 40 and 80 µM as the drug concentration for migration experiment, HCT116 cell experiment selects 20 and 40 µM the drug concentration used as the migration test. Wound healing assays and Transwell migration assays showed that 6-Shogaol inhibited the migration of Caco2 cells and HCT116 cells in a dose-dependent manner (Figures 4 and 5). By detecting the expression of the proteins N-cadherin, E-cadherin, MMP-2, and VEGF, which are related to the EMT process, the mechanism of 6-Shogaol on the migration of colon cancer cells was explored. The protein expression of MMP-2, VEGF and N-cadherin was lower than that of the control group in a concentration-dependent manner. The protein expression of E-cadherin did not change significantly in Caco2 cells but even decreased in HCT116 cells (Figures 6 and 7).

Inhibitory effect of 6-Shogaol on colon cancer cell migration. (A) A wound-healing experiment was used to detect the effects of different concentrations of 6-Shogaol on Caco2 cell migration. (B) Quantification of (A). (C) A wound-healing experiment was used to detect the effects of different concentrations of 6-Shogaol on HCT116 cell migration. (D) Quantification of (C), and all experiments were repeated at least 3 times.
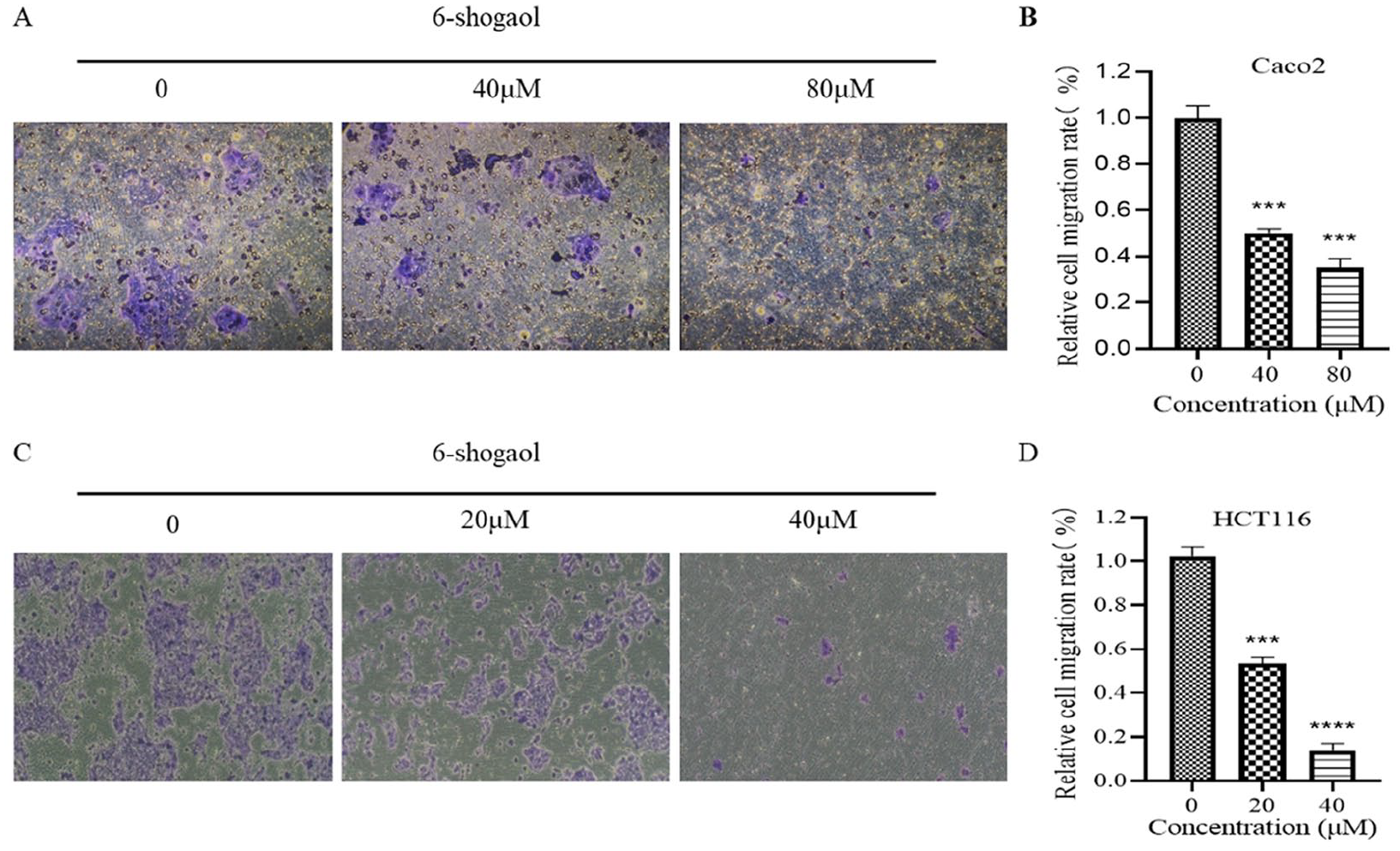
Inhibitory effect of 6-Shogaol on the migration of Caco2 cells. (A) A Transwell migration assay was used to determine the effects of different concentrations of 6-Shogaol on the migration of Caco2 cells. (B) Quantification of (A). (C) A Transwell migration assay was used to determine the effects of different concentrations of 6-Shogaol on the migration of HCT116 cells. (D) Quantification of (C). *** P < .001. ****P < .0001 versus the control group (0 µM).

6-Shogaol inhibited cell migration by inhibiting EMT in Caco2 cells. The colon cancer cell line Caco2 was treated with different concentrations of 6-Shogaol (0, 20, 40, 60, 80, and 100 µM). (A) the expression of MMP-2, VEGF, N-cadherin, and E-cadherin in Caco2. (B), (C), and (D) Quantification of (A). *P < .05. ** P < .01. *** P < .001 versus the control group (0 µM).

6-Shogaol inhibited cell migration by inhibiting EMT in HCT116 cells. The colon cancer cell line HCT116 was treated with different concentrations of 6-Shogaol (0, 10, 20,40 and 60 µM). (A) the expression of MMP-2, VEGF, N-cadherin, and E-cadherin in Caco2. (B), (C), and (D) Quantification of (A). *P < .05. ** P < .01 versus the control group (0 µM).
6-Shogaol Inhibited Colon Cancer Cell Migration via IKKβ/NF-κB/Snail Signaling
To explore the mechanism by which 6-Shogaol inhibits IKKβ/NF-κB/Snail signaling in colon cancer, we tested the expression of IKKβ/NF-κB/Snail-related proteins. Western blot results showed that compared with the control group, the protein expression of NF-κB did not change significantly, and P-NF-κB, Snail, and IKKβ concentrations were significantly reduced between 80 and 100 µM after 6-Shogaol treatment in the Caco2 cell line (Figure 8); the protein expression of NF-κB did not change significantly, and P-NF-κB, Snail, and IKKβ concentrations were significantly reduced between 40 and 60 µM after 6-Shogaol treatment in HCT116 cell line (Figure 9).

6-Shogaol can inhibit the IKKβ/NF-κB/Snail signaling pathway. The colon cancer cell line Caco2 was treated with different concentrations of 6-Shogaol (0, 20, 40, 60, 80, and 100 µM) for 48 hours. Western blot detection. (A) IKKβ, NF-κB, P-NF-κB, and Snail expression in Caco2 cell. (B), (C), and (D) Quantification of (A). *P < .05. ** P < .01. *** P < .001 versus the control group (0 µM).

6-Shogaol can inhibit the IKKβ/NF-κB/Snail signaling pathway. The colon cancer cell line HCT116 was treated with different concentrations of 6-Shogaol (0, 10, 20, 40 and 60 µM) for 48 hours. Western blot detection. (A) IKKβ, NF-κB, P-NF-κB, and Snail expression in HCT116 cell. (B), (C), and (D) Quantification of (A). *P < .05. * P < .01. *** P < .001, **** P < .0001 versus the control group (0 µM).
Discussion
Although 6-Shogaol has been shown to inhibit the metastasis of some cancer cells, 33 its effect on colon cancer cell migration remains unclear and its mechanism remains to be explored. The purpose of this study was to investigate the effects and possible mechanisms of 6-Shogaol on the migration of Caco2 and HCT116 colon cancer cells, and to verify the effects of 6-Shogaol on the growth and apoptosis of these 2 cell lines. Through wound healing and transwell migration assays, we found that 6-Shogaol can significantly inhibit colon cancer Caco2 and HCT116 cell migration. In addition, MTT assay confirmed that 6-Shogaol had inhibitory effect on colon cancer Caco2 and HCT116 cells. Annexin V/PI staining confirmed that 6-Shogaol can significantly promote Caco2 cell apoptosis at 80 μM and HCT116 cell apoptosis at 40 μM, and focus on late apoptosis, which possible reason is that NF-κB p65 from cytoplasmic heterotopia to nucleus can inhibit DNA degradation at later stage of cell and inhibit apoptosis at later stage. However, 6-Shogaol can inhibit the expression of NF-κB p65, so we think it may be due to this reason, which leads to more obvious late apoptosis. 34 These results are consistent with previous studies,6-Shogaol can inhibit the growth of colon cancer cells and promote apoptosis.35,36 All these results indicate that 6-Shogaol is a promising anticancer agent.
Colon cancer metastasis is closely related to the EMT process.10,11 The typical characteristics of EMT are E-cadherin downregulation and N-cadherin upregulation. Snail is an important transcription factor that promotes EMT, and its upregulated expression can be used as a common biomarker of type 3 EMT. 37 For tumor occurrence and metastasis, angiogenesis is very important for tumor metastasis and cancer growth. Tumor growth requires substances in blood vessels, and vascular endothelial factor (VEGF) regulates angiogenesis. 38 In summary, MMP-2, N-cadherin, E-cadherin, snail, and VEGF are often used as markers of colon cancer migration. Therefore, the increased expression of MMP-2, Snail, and VEGF can also promote the EMT process to a certain extent. In this study, the protein expressions of N-cadherin, MMP-2 and VEGF were significantly decreased in HCT116 cell lines; the protein expression of VEGF in Caco2 cells were reduced, although it did not reach statistical significance, it still had important reference value, N-cadherin and MMP-2 were significantly reduced. However, the expression of E-cadherin in Caco2 cells and HCT116 cells was different from that in general. The expected results showed that the expression of E-cadherin should be increased after administration, but there was no significant change in Caco 2 cell line, and the expression was inhibited in HCT116 cell line. This is interesting and worth discussing separately.
E-cadherin is usually considered to be a tumor suppressor. However, studies have shown that E-cadherin can also promote the survival of colon cancer cells, and E-cadherin-positive short-term cultured colon cancer cells are more resistant to chemotherapy drugs, 39 which challenges the classical process of EMT. Combined with the analysis of relevant research results, this may be because E-cadherin forms a complex with the polar component DLG1 and the cell death regulator FAS, which inhibits apoptosis. 40 Studies also show that the lack of E-cadherin increases the invasiveness of breast cancer cells but reduces the proliferation and viability of cancer cells at the same time. 41 These results indicate that the survival and migration of E-cadherin in cancer cells are 2-sided. In this experiment, the role of E-cadherin in the EMT process has great uncertainty, but this does not affect the fact that 6-Shogaol can inhibit the EMT process.
The NF-κB signaling pathway plays a key role in tumor proliferation, apoptosis, and cell migration. NF-κB can promote vascular endothelial growth factors and lead to the expression of matrix metalloenzymes. 42 NF-κB can regulate snails, while snails, as nuclear gene proteins, can regulate the expression of related proteins (including N-cadherin, MMP, vimentin, etc.) during EMT. 43 That is, activation of the NF-κB/Snail signaling pathway can increase snail expression and promote EMT, which can accelerate tumor metastasis and invasion. 44 Previous studies have shown that apigenin inhibits the EMT process by inhibiting the NF-κB/Snail pathway, thus inhibiting the metastasis of colon cancer. 45 Our experimental data showed that the protein expression of IKKβ, P-NF-κB, and Snail was significantly reduced after the effect of 6-Shogaol on Caco 2 cells and HCT116 cells. This finding may explain that 6-Shogaol may inhibit the key role of EMT in colon cancer migration by inhibiting NF-kB/Snail signaling pathway (Figure 10). However, how the IKKβ/NF-κB/Snail signaling pathway affects EMT at the gene level needs to be further studied.

Mechanism diagram of 6-Shogaol on the growth and migration of colon cancer Caco2 cells. 6-Shogaol decreased the expression of N-cadherin, MMP-2, and VEGF by inhibiting the expression of IKKβ and P-NF-κB Snail, but E-cadherin expression was not affected. The reason may be that E-cadherin forms a complex with the polar component DLG1 and the cell death regulator FAS to inhibit apoptosis. MMP-2, N-cadherin, E-cadherin, and VEGF are important factors affecting the EMT process. These results indicate that 6-Shogaol can inhibit EMT through the IKKβ/NF-κB/Snail signaling pathway.
Conclusions
This study proposes and confirms that 6-Shogaol can significantly inhibit the migration of colon cancer Caco2 and HCT116 cells, and inhibit the expression of IKKβ/NF-κB/Snail signaling pathway and EMT-related proteins. it’s speculated that the mechanism may be produced by inhibiting EMT through IKKβ/NF-κB/Snail signaling pathway. It was also confirmed that 6-Shogaol inhibited the proliferation and promoted apoptosis of colon Caco2 and HCT116 cells. This indicates that 6- Shogaol may be a potential anti-tumor drug, which is worth further study.
Footnotes
Author Contributions
M. C, L. Z, and L. X designed the research study. C. T performed the research. Z. Z and Q. H provided help and advice on the experiment. Q. W analyzed the data and revised the manuscript. M. C and C. T wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Science and Technology Development Fund, Macau SAR (file No.:0114/2022/A, 0010/2019/A and 0098/2021/A2); Scientific Research Project of Guangdong Provincial Bureau of Traditional Chinese Medicine (20201324); Science and Technology Fund Project of Guizhou Provincial Health Commission (gzwkj2022-023); Special Project of Academic New Seedling Cultivation and Innovation Exploration of Zunyi Medical University for 2017 (file No.: Talents of Guizhou Science and Technology Cooperation Platform [2017]5733-080); The Project of Guizhou Provincial Natural Science Foundation, QKH-J [2020]1Y378.
Ethics Approval and Consent to Participate
Not applicable.
