Abstract
Objectives
To investigate the effects of γ-secretase inhibitor (GSI), a Notch signalling inhibitor, on the proliferation of multiple myeloma cells in vitro and its molecular mechanism of action.
Methods
RPMI 8226 cells were treated with increasing concentrations of GSI (0–20 µmol/l) for 24–72 h. Proliferation was measured using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) assay. Cell-cycle analysis was performed on RPMI 8226 cells treated with 0–10 µmol/l GSI for 48 h using flow cytometry. Expression of Notch signalling proteins (Notch1, Jagged 1 and Jagged 2), Bcl-2 and phosphorylated Akt (p-Akt) was determined using Western blotting in RPMI 8226 cells treated with various concentrations of GSI for various time periods.
Results
GSI inhibited proliferation of RPMI 8226 cells in a concentration- and time-dependent manner by inducing G0/G1 cell-cycle arrest. GSI-mediated antiproliferative effects were associated with significant reductions in the expression of Notch1, Jagged1, Jagged2, p-Akt and Bcl-2.
Conclusion
Inhibition of the Notch signalling pathway by GSI may be a promising therapeutic approach for the treatment of multiple myeloma.
Keywords
Introduction
Multiple myeloma (MM), a haematological malignancy that primarily affects the elderly, is characterized by clonal proliferation of plasma cells. Chemotherapy is the standard treatment for MM. Although the emergence of new drugs, including proteasome inhibitors and antiangiogenesis agents, has greatly improved the prognosis of patients with MM, it remains an incurable disease. The bone marrow microenvironment consists of bone marrow stromal cells, vascular endothelial cells, osteoblasts, osteoclasts, lymphocytes, extracellular matrix proteins, and a variety of secreted cytokines such as interleukin-6, vascular endothelial growth factor, insulin-like growth factor-1 and tumour necrosis factor-α.1,2 The interaction between MM cells and the bone marrow microenvironment enhances the proliferation of MM cells and induces drug resistance to chemotherapy. 3 Therefore, the identification of new targeted therapies for this disease remains an active area of research.4,5
The Notch signalling pathway is widespread in animals and has been highly conserved during evolution. Notch signalling is initiated by binding of the extracellular domain of Notch to a Notch ligand. 6 At present, two Notch ligand families – Delta and Jagged – have been identified. 7 The Notch receptors include four family members (Notch1 to Notch4); each member is a large single heterodimeric protein comprised of extracellular, transmembrane and intracellular subunits. 8
Notch signalling influences multiple processes that govern normal morphogenesis, programmed cell death, cellular proliferation and differentiation.7,9,10 Therefore, Notch signalling dysfunction may inhibit cell differentiation, eventually leading to the malignant transformation of undifferentiated cells and tumourigenesis. 11 There is ample evidence linking the Notch signalling pathway with cancer. Overexpression of wild-type Notch receptors, ligands and targets has been observed in an increasing number of solid tumours. 12 Similar defects have been found in haematological malignancies, including non-Hodgkin’s lymphoma 13 and acute myeloid leukaemia. 14 Inhibition of Notch expression by antisense retrovirus or pharmacological blocking of γ-secretase activity induces antineoplastic effects in Notch-expressing transformed cells in vitro.15–17 Moreover, inhibition of Notch signalling has been shown to induce apoptosis and to increase sensitivity to chemotherapy in MM cells. 18 These findings indicate that inhibition of Notch signalling may represent a new therapeutic approach in the treatment of haematological malignancies. In the present study, the effects of γ-secretase inhibitor (GSI), a Notch signalling inhibitor, on the proliferation of MM cells in vitro and its molecular mechanism of action were investigated.
Materials and methods
Cell culture
The human RPMI 8226 MM cell line (Shanghai Cell Bank, Shanghai, China) was cultured in RPMI 1640 medium containing 10% fetal bovine serum in a humidified incubator at 37℃ in 5% CO2. The culture medium was changed every 2–3 days.
Assessment of proliferation
Cells in the logarithmic growth phase were seeded into 96-well culture plates at a density of 5 × 103 cells/well. Then, 100 µl GSI (Enzo Life Sciences, Farmingdale, NY, USA) at final concentrations of 0, 1, 5, 10 and 20 µmol/l was added to the wells. Untreated cells served as controls. A blank control consisting of the drug solvent, dimethyl sulphoxide (DMSO) and GSI without cells was also included in the assay.
After 24, 48 or 72 h, 20 µl of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) (Sigma-Aldrich, St Louis, MO, USA), dissolved in phosphate-buffered saline at a concentration of 20 µmol/l, was added to each well.
Cells were incubated for an additional 4 h at 37℃, centrifuged at 300
Each stimulation condition was performed in duplicate and repeated three times.
Cell-cycle analysis
The RPMI 8226 MM cells were treated with 0, 5 or 10 µmol/l GSI for 48 h, then collected and washed twice with cold phosphate-buffered saline (pH 7.0). Cell-cycle analysis was performed using a BD FACScan™ flow cytometer (Becton, Dickinson and Company, Franklin Lakes, NJ, USA) as described previously. 19 The cells were stained with propidium iodide solution (50 µg/ml) on ice and ≥10 000 cells were analysed for cell-cycle distribution.
Western blot analyses
Western blotting was performed as described previously. 19 Cells were treated with GSI at concentrations of 0, 10, 20 and 40 µmol/l for 48 h, or with GSI at a concentration of 10 µmol/l for 24, 48 or 72 h. Cells were then lysed in buffer (pH 7.0) containing freshly prepared protease inhibitors. The protein concentration of the cell lysates was determined using the Bio-Rad Protein Assay kit (Bio-Rad Laboratories, Hercules, CA, USA) with a bovine serum albumin standard. Proteins were subjected to sodium dodecyl sulphate–polyacrylamide gel electrophoresis, electrotransferred to polyvinylidene difluoride membranes and immunoblotted using antibodies to Notch1, Jagged1, Jagged2, phosphorylated Akt (p-Akt) and Bcl-2. The antibody dilutions used were: Notch1, 1 : 250; Jagged1, 1 : 1000; Jagged2, 1 : 1000; p-Akt, 1 : 500; and Bcl-2, 1 : 1000. Antibodies to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) at a dilution of 1 : 3000 were used as a loading control. The appropriate horseradish peroxidase-conjugated secondary antibodies were used and protein bands were visualized using enhanced chemiluminescence. Expression of Notch1, Jagged1 and Jagged2 was measured in cells treated with 0–40 µmol/l GSI for 48 h. Expression of Notch1 and Jagged2 was measured in cells treated with 10 µmol/l GSI for 0–72 h. Expression of Bcl-2 was measured in cells treated with 0–40 µmol/l GSI for 48 h. Expression of p-Akt was measured in cells treated with 0–20 µmol/l GSI for 48 h.
Notch1, Jagged1, Jagged2 and GAPDH monoclonal antibodies and secondary antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA); p-Akt and Bcl-2 antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Statistical analyses
Data were presented as mean ± SD. Intergroup comparisons were performed using Dunnett’s test, a multiple testing procedure under one-way analysis of variance. Comparisons of means between groups were performed using Student’s t-test. A P-value < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS® software, version 11.5 (SPSS Inc., Chicago, IL, USA).
Results
Proliferation and cell-cycle analysis
We found that GSI inhibited the proliferation of RPMI 8226 cells in a time- and concentration-dependent manner (Figure 1). After 24 h, cell proliferation was significantly inhibited (P < 0.05) by GSI at concentrations ≥5 µml/l (Figure 1). When compared with the control group, cells treated with 5 or 10 µmol/l GSI had a significantly higher proportion of cells in G0/G1 phase and a lower proportion of cells in S-phase (P < 0.05 for both; Figure 2).
Inhibition of proliferation of RPMI 8226 multiple myeloma cells treated with different concentrations of γ-secretase inhibitor (GSI) for different time periods. After 24 h, cell proliferation was significantly inhibited (P < 0.05) by GSI at concentrations ≥5 µml/l. Percentage of cells in different cell-cycle phases in RPMI 8226 multiple myeloma cells treated with different concentrations of γ-secretase inhibitor (GSI) for 48 h. Compared with the control group, cells treated with 5 or 10 µmol/l GSI had significantly higher proportions of cells in G0/G1 phase and lower proportions of cells in S-phase (P < 0.05 for both).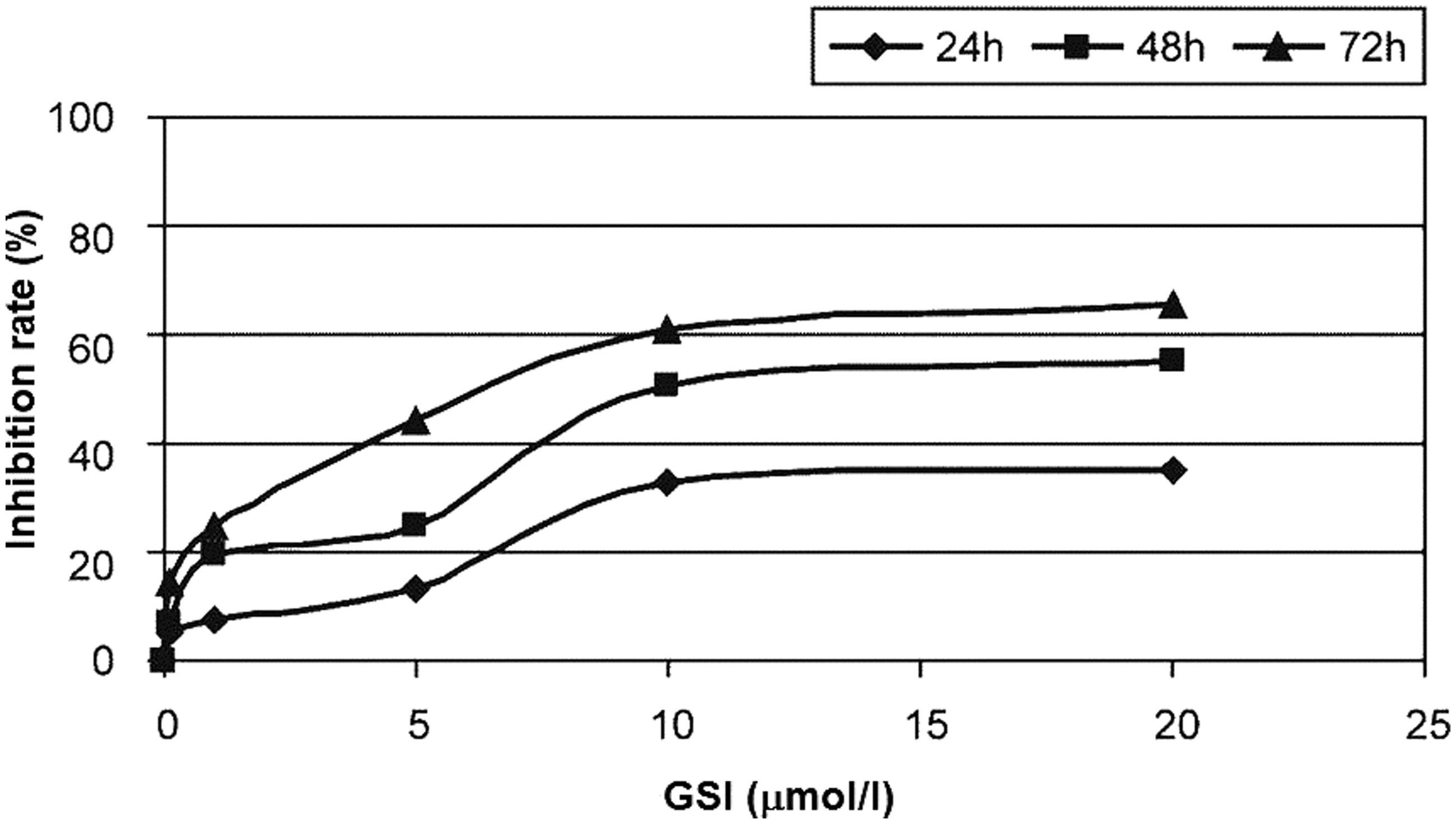

Expression of Notch signalling proteins
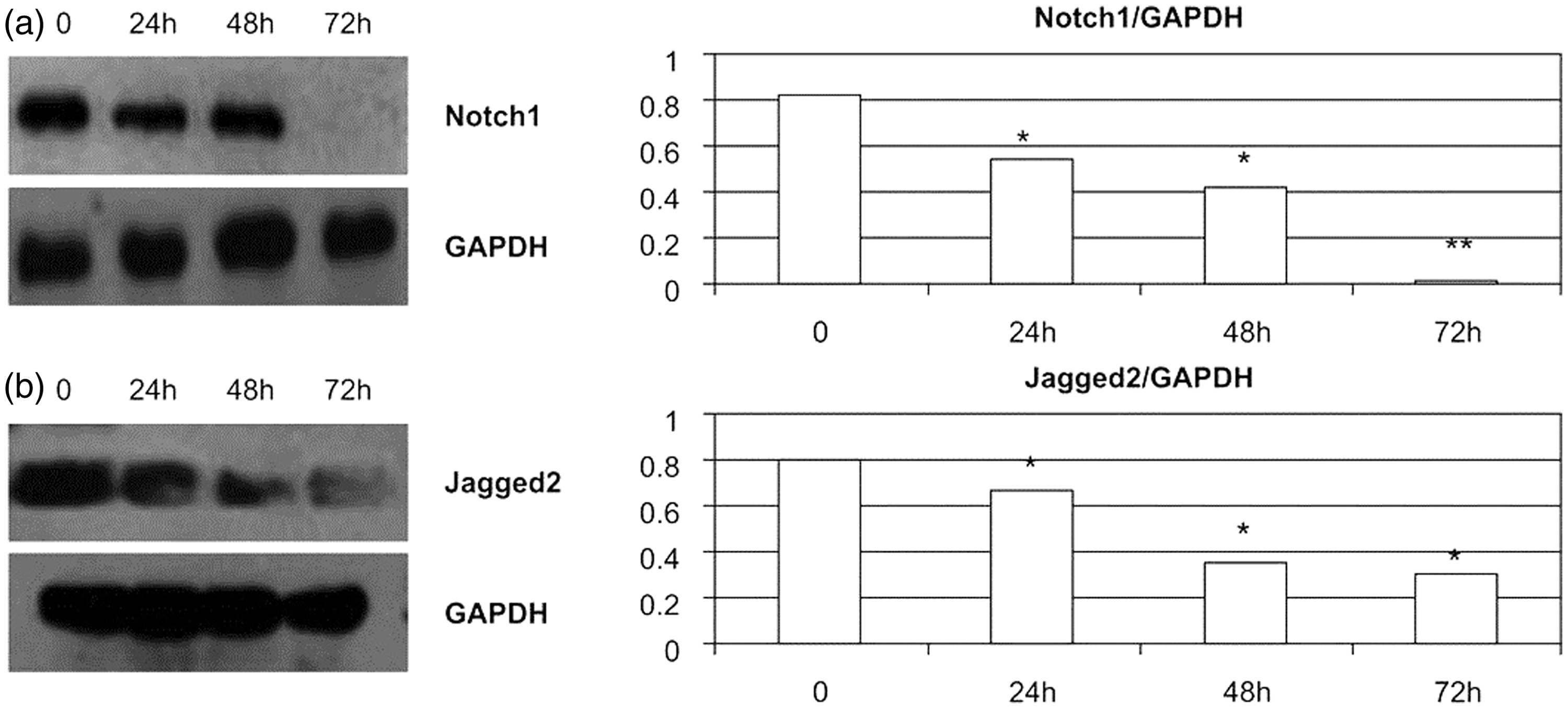
Expression of Notch1, Jagged1 and Jagged2 was reduced in a concentration-dependent manner in RPMI 8226 cells treated with GSI for 48 h (Figure 3). Treated cells showed significant differences (P < 0.05) from control cells at GSI concentrations of 20 and 40 µmol/l for Notch1 and at GSI concentrations of 10, 20 and 40 µmol/l for Jagged1 and Jagged2. In addition, expression of Notch1 and Jagged2 was downregulated in a time-dependent manner in RPMI 8226 cells treated with 10 µmol/l GSI (Figure 4); treated cells showed significant differences (P < 0.05) from control cells at all time points.
Expression of (a) Notch1, (b) Jagged1 and (c) Jagged2 Notch signalling proteins detected by Western blotting in RPMI 8226 multiple myeloma cells treated with different concentrations of γ-secretase inhibitor (GSI) for 48 h; data are given relative to the expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). *P < 0.05; **P < 0.01, compared with 0 µmol/l GSI. Expression of (a) Notch1 and (b) Jagged2 Notch signalling proteins detected by Western blotting in RPMI 8226 multiple myeloma cells treated with 10 µmol/l of γ-secretase inhibitor (GSI) for different time periods; data are given relative to the expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH).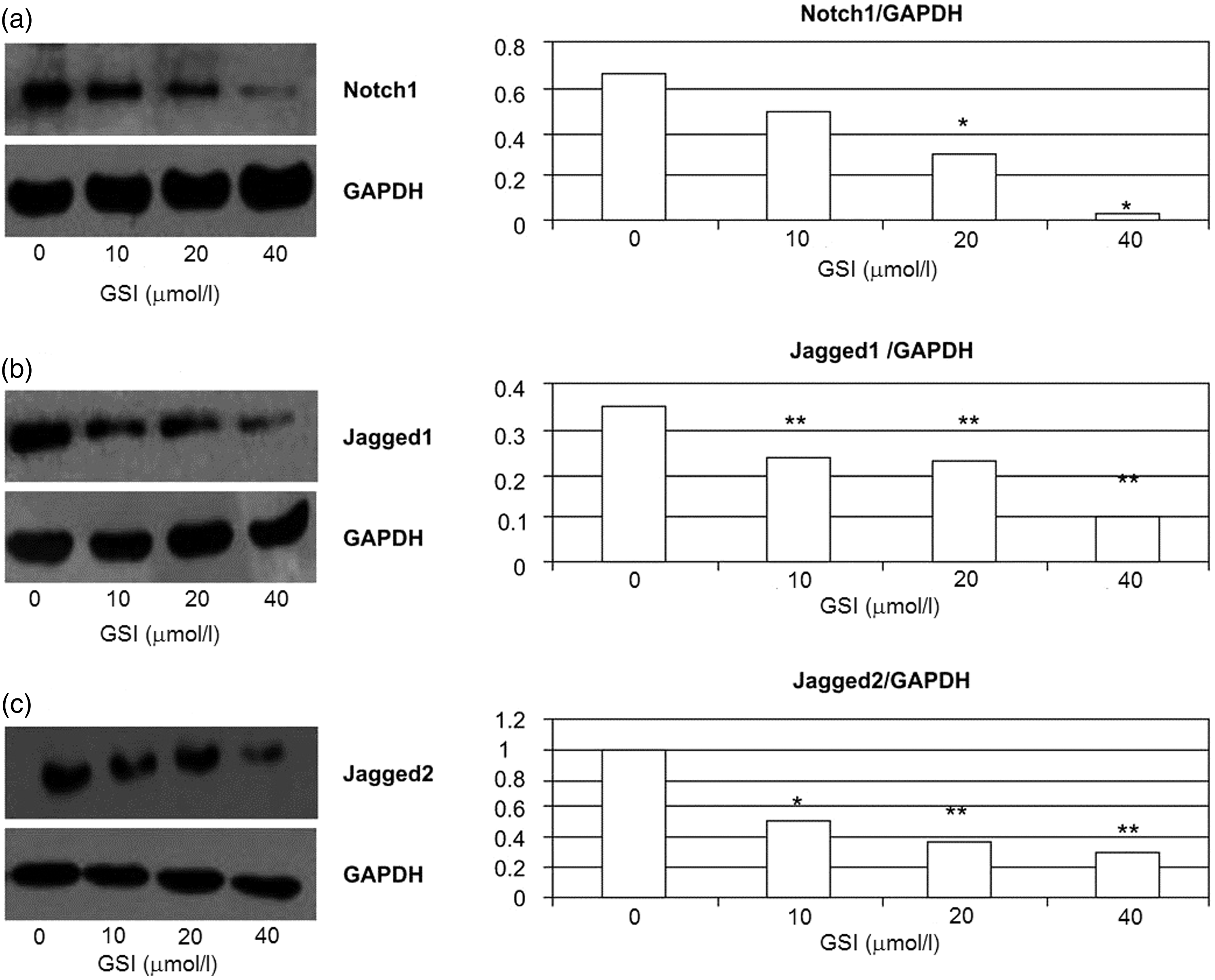
Expression of Bcl-2 and p-Akt
Expression of Bcl-2 and p-Akt was reduced in a concentration-dependent manner in RPMI 8226 cells treated with GSI for 48 h (Figure 5). Treated cells showed significant differences (P < 0.05) from control cells at GSI concentrations ≥10 µmol/l.
Expression of (a) Bcl-2 and (b) p-Akt proteins detected by Western blotting in RPMI 8226 multiple myeloma cells treated with different concentrations of γ-secretase inhibitor (GSI) for 48 h; data are given relative to the expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). *P < 0.05 compared with 0 µmol/l GSI.
Discussion
Many studies have shown that overexpression of signalling proteins such as Notch1, Notch2 and Jagged1 is associated with abnormal cell proliferation in myeloma cells.18,20,21Therefore, the Notch signalling pathway has become an attractive candidate for therapeutic intervention in MM.5,20,22,23 Inhibition of Notch1 and Jagged2 expression using phenylhexyl isothiocyanate, a histone deacetylase inhibitor, has been shown to suppress proliferation and induce apoptosis in a concentration- and time-dependent manner in myeloma cells.18,20 Several studies have examined the effects of blocking the Notch signalling pathway by targeting the c-secretase protein complex to inhibit the activation of Notch receptors. 24 It has been shown that inhibition of the c-secretase complex prevents the growth of T-cell acute lymphoblastic leukaemia cell lines, leading to apoptosis.22,25,26 The Notch signalling inhibitor GSI has been reported to suppress the growth of several types of tumour cells including sarcoma,27,28 medulloblastoma, 29 ovarian cancer, 30 prostate cancer 31 and breast cancer.32,33 In the present study, GSI decreased the expression of Notch1, Jagged1 and Jagged2 in a time- and concentration-dependent manner in RPMI 8226 MM cells. Furthermore, GSI-mediated inhibition of Notch signalling suppressed cell proliferation by inducing G0/G1 cell-cycle arrest. These results are in agreement with other studies demonstrating the antiproliferative and proapopotic effects of GSI in MM cells. Jundt et al. 20 found that GSI treatment decreased Jagged1-mediated cell proliferation by 40–70% in myeloma cell lines. Nefedova et al. 18 reported that GSI treatment inhibited cell proliferation and induced apoptosis in MM cell lines, and significantly improved the antitumour effects of doxorubicin and melphalan in in vivo mouse models of human MM. Together, these findings suggest the potential therapeutic benefit of GSI in MM.
In the present study, the underlying molecular mechanisms governing the antineoplastic actions of GSI were investigated by measuring its effects on Bcl-2 and p-Akt. GSI-mediated antiproliferative effects were associated with downregulation of the antiapoptotic protein, Bcl-2. This is consistent with the results of Nefedova et al., 18 who reported that the antiproliferative and proapoptotic effects of GSI treatment were associated with downregulation of Hes1, a Notch1 mediator, and the antiapoptotic proteins Bcl-xL and Bcl-2. Furthermore, GSI-mediated apoptosis was mediated by upregulation of the proapoptotic protein Noxa. 18 Downregulation of Bcl-xL and Bcl-2 and upregulation of Noxa were also associated with increased sensitivity of MM cells to chemotherapy. 18 Whether the combination of GSI with chemotherapy could have synergistic activity against MM requires further investigation.
The phosphoinositide 3-kinase (PI3K)/Akt pathway has been implicated in the oncogenic effects of the Notch signalling pathway. 34 The PI3K/Akt pathway plays a key role in promoting cell proliferation by affecting various effector molecules, including apoptotic proteins and cell-cycle regulatory proteins.35,36 Several studies have reported interaction between the Notch and PI3K/Akt signalling pathways. Liu et al. 37 found that the Notch signalling pathway activated the Akt signalling pathway and improved the survival rate of melanoma cells. Palomero et al. 38 reported that Hes1 negatively regulated phosphatase and tensin homologue to induce PI3K/Akt upregulation in pre-T acute lymphoblastic leukaemia cells. Thus there is evidence to suggest the reciprocal regulation of Notch and PI3K/Akt signalling pathways: Notch1 not only induces Akt activation but the Akt pathway also induces Notch1. In the present study, GSI-mediated antiproliferative effects were associated with downregulation of p-Akt in MM cells. Nefedova et al. 18 also showed that GSI treatment decreased Akt protein expression in MM cells. Further investigation of the molecular mechanisms underlying GSI activity will be valuable in understanding Notch pathway regulation.
In summary, in the present study GSI was shown to inhibit MM cell proliferation in a time- and concentration-dependent manner by inducing G0/G1 cell-cycle arrest. GSI treatment effectively suppressed the expression of the Notch signalling proteins Notch 1, Jagged1 and Jagged2, and was associated with downregulation of p-Akt and Bcl-2. Therefore, inhibition of the Notch signalling pathway by GSI may be a promising therapeutic approach for the treatment of MM.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.

