Abstract
Background. Many women use black cohosh as a natural treatment for menopausal symptoms. However, controversy exists around safety in breast cancer, because of its purported estrogenic activity. We conducted a systematic review of black cohosh use in women with or at risk of breast cancer.Methods. We searched MEDLINE, Embase, the Cochrane Library, and AMED from inception to July 2012 and October 2012 for human interventional or observational data pertaining to the safety and efficacy of black cohosh in patients with or at risk of breast cancer, including an assessment of the effect of black cohosh on estrogen responsive tissues. Results. Of 450 records, we included 26 articles: 14 randomized controlled trials, 7 uncontrolled trials, and 5 observational studies.The evidence on efficacy for ho t flashes is divided, with some benefits seen when compared with baseline, but not when compared with placebo. Two observational studies found no association between black cohosh and risk of breast cancer, whereas 2 studies reported significant reductions in risk of primary breast cancer among postmenopausal women (adjusted odds ratio = 0.47, 95% confidence interval = 0.27-0.82), and risk of recurrence (adjusted hazard ratio = 0.75, 95% confidence interval = 0.63-0.89). Seventeen trials showed no significant impact on circulating hormone levels or proliferation in estrogen responsive tissues. Conclusions. Current evidence does not support an association between black cohosh and increased risk of breast cancer. There is a lack of evidence supporting the efficacy of black cohosh for reduction of hot flashes in breast cancer patients. Given conflicting but promising results, and apparent safety, further research is warranted.
Keywords
Introduction
Breast cancer accounts for almost one third of all cancers diagnosed and is the second leading cause of cancer death among women, with nearly 40 000 attributable deaths expected in 2011 in the United States. 1 Women who are survivors of breast cancer treatment, often live with considerable adverse effects,2,3 including induction of early menopause, leading to vasomotor symptoms, mood changes, vaginal atropy, altered menses, and musculoskeletal pain.2,3 These women often resort to natural therapies for the management of such symptoms, in part because of perceived relative safety. 4 In addition, among women without a history of breast cancer, those who are entering menopause often resort to natural herbal therapies as an alternative to hormone replacement therapy. One of the reasons women choose complementary approaches to control menopausal symptoms is because of the increased risk of breast cancer, cardiovascular disease, and stroke from hormone replacement therapy.5,6 Despite widespread use, however, there is a lack of up-to-date, high-quality scientific evidence for many of these complementary therapies.
Black cohosh, also known as Cimicifuga racemosa or Actea racemosa (family Ranunculaceae), is a popular herb commonly used for a variety of women’s health concerns. 4 The German Commission E has approved black cohosh for the treatment of premenstrual syndrome, dysmenorrhea, and menopausal symptoms.7,8 Although there is little data regarding the precise rate of use among breast cancer patients, black cohosh remains one of the most controversial natural therapies used by this patient population 4 because of its purported phytoestrogenic activity as a selective estrogen receptor modulator (SERM)–like agent. In theory, phytoestrogens possess amphoteric effects on the estrogen receptor (ER). Under conditions of estrogen excess, phytoestrogens may act as estrogen antagonists through competitive inhibition of the ER, only stimulating it weakly. Under conditions of low estrogen, phytoestrogens may act as weak agonists.9-11 In cases where black cohosh contributes to a net estrogenic effect, its use may result in deleterious effects on breast cancer risk or recurrence. 12 This is of particular concern among women undergoing antiestrogen therapy. Given these conflicting data and the potential for harm, there is an urgent need for a synthesis of available evidence pertaining to the use of black cohosh and its impact on breast cancer risk. A systematic review by Walji et al 13 suggested that black cohosh has a high safety profile for use by cancer patients, however, this review has not been updated to include evidence since 2007.
Several standardized extracts of black cohosh root and rhizome are available commercially. Remifemin is a 40% isopropanolic extract by volume, standardized to contain 1 mg of triterpenes, measured as 27-deoxyacteine per 20 mg tablet. 14 The C racemosa extract BNO 1055 (Menofem/ Klimadynon) is a 58% ethanolic extract by volume, corresponding to 21.5 mg root per tablet. 15
We conducted a systematic review of black cohosh for use by pre- or postmenopausal breast cancer patients or those at risk of breast cancer. We assessed the following: impact on risk of primary breast cancer incidence, risk of breast cancer recurrence, effect on estrogen responsive tissues, and efficacy in treating menopausal symptoms following breast cancer treatment.
Methods
Search Strategy
Electronic search strategies were developed and tested through an iterative process by an experienced medical information specialist in consultation with the review team. Using the OVID platform, we searched Ovid MEDLINE, Ovid MEDLINE In-Process & Other Non-Indexed Citations, Embase Classic+Embase, and AMED (Allied and Complementary Medicine). We also searched the Cochrane Library on Wiley (including CENTRAL, Cochrane Database of Systematic Reviews, DARE, and HTA) and pertinent trial registries (eg, MetaRegister of Controlled Trials [mRCT]). The searches were initially performed on February 2, 2011 and updated on July 29, 2012. A supplementary search exploring the estrogenic effects of black cohosh was performed in the 2 OVID MEDLINE databases on October 19, 2012.
Strategies used a combination of controlled vocabulary (eg, cimicifuga, breast neoplasms) and keywords (actaea racemosa, black cohosh, breast cancer). Vocabulary and syntax were adjusted across databases. There were no language or date restrictions on any of the searches. Additional references were also sought through hand-searching the bibliographies of relevant items.
Specific details regarding the strategies appear in the appendix.
Inclusion Criteria
For inclusion, evidence had to come from clinical trials or observational studies in humans. Human trials had to (a) assess the safety and/or efficacy of black cohosh in breast cancer patients for the purposes of treatment or secondary prevention, or the reduction of side effects associated with chemotherapy or radiation therapy; alternately, human trials had to (b) assess the effect of black cohosh on risk of primary breast cancer, as defined by the incidence of new breast cancer cases among women without a history of previous breast cancer; (c) assess the effect of black cohosh on the following measures of estrogenic activity in target tissues: serum or urinary levels of estrogen, progesterone, follicle-stimulating hormone (FSH), or luteinizing hormone (LH); mammographic density; endometrial thickness; vaginal cytology; breast tissue proliferation; and bone metabolism. Clinical surrogate studies were included if they examined endpoints directly related to breast cancer risk or pathogenesis, or objective markers assessing healthy bodily function, such as hematological function in breast cancer patients.
Observational studies had to report on risk of primary breast cancer or breast cancer recurrence associated with black cohosh use compared with nonuse in a prospective or retrospective design. In vitro and in vivo studies were excluded due to the high risk for confounding and previous work showing a lack of correlation between preclinical and clinical results. 16
Record Screening and Selection
First pass record screening was based on title review with second pass conducted on abstracts and/or full texts where uncertainty existed. Reports published in English and German were included for full analysis if they met inclusion criteria.
Data Extraction
We piloted data extraction forms and conducted extraction in duplicate to assess interresearcher reliability (HF, RF). No major inconsistencies in data extraction were found. Extraction sheets were prepared based on the Consolidated Standards of Reporting Trials (CONSORT) statement for clinical trials and the Newcastle–Ottawa scale (NOS) for observational studies.17-19 randomized controlled trials (RCTs) were assessed for quality using the Cochrane Risk of Bias tool, 20 and observational studies were assessed for quality using the NOS. 19
Results
Of 450 records screened through 2 independent literature searches, 26 records were included for full analysis and review. Figure 1 shows a flowchart of the literature search and study selection. Of the 5 included observational studies, 2 were prospective 21,22 and 3 were retrospective;23-25 4 studies reported on risk of breast cancer occurrence or recurrence, and 1 study reported on quality of life among breast cancer survivors. These studies are described in Tables 1 and 2. Three RCTs and 2 uncontrolled trials examined efficacy for hot flashes as their primary endpoint,26-30 whereas 5 uncontrolled trials and 12 RCTs assessed the effect of black cohosh on estrogen responsive tissues.9,10,30-44 These studies are summarized in Tables 3, 4, and 5.

Literature flowchart.
Methods of Observational Studies: Black Cohosh and Risk of Breast Cancer.

Abbreviations: b/w, between; d, days; wk, week; mo, months; y, years; n/a, not applicable; NR, not reported; QoL, quality of life; iCR, isopropanolic Cimicifuga racemosa extract.
Outcomes of Observational Studies: Black Cohosh and Risk of Breast Cancer.

Abbreviations: AHR, adjusted hazard ratio; AOR, adjusted odds ratio; BC, black cohosh; CI, confidence interval; n/a, not applicable.
Black Cohosh for Hot Flashes in Breast Cancer Survivors: Methods of Human Trials.

Abbreviations: AI, aromatase inhibitors; avg, average; BC, black cohosh; BrCa, breast cancer; b/w, between; cap, capsule; d, days; wk, weeks; mo, months; y, years; ER+, estrogen receptor positive; hx, history; MRS-II, Menopause Rating Scale II; n/a, not applicable; pts, patients; R, raloxifene; st, standardized to; T, tamoxifen; tab, tablet.
Efficacy of Black Cohosh for Hot Flashes in Breast Cancer Survivors: Outcomes of Human Trials.
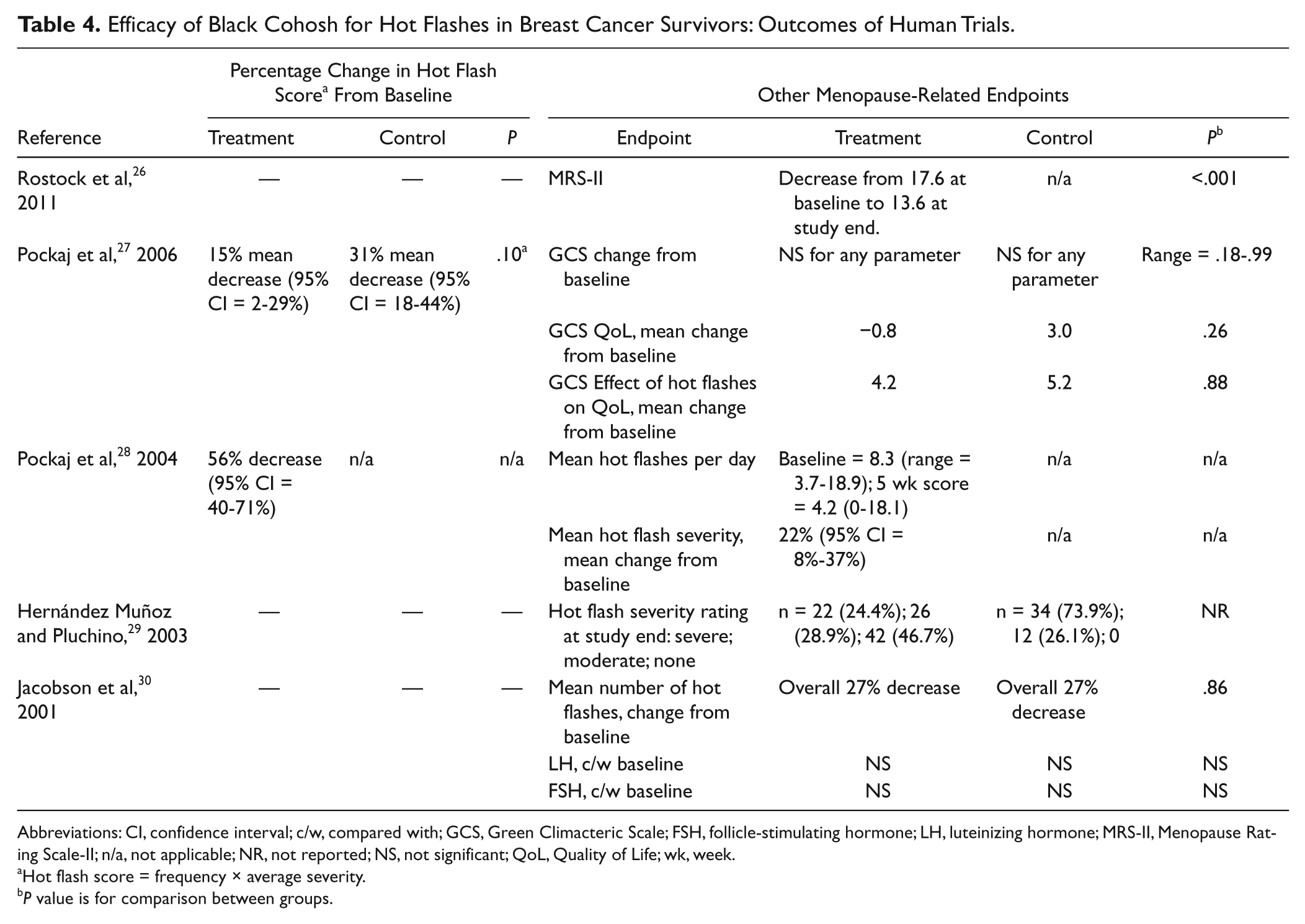
Abbreviations: CI, confidence interval; c/w, compared with; GCS, Green Climacteric Scale; FSH, follicle-stimulating hormone; LH, luteinizing hormone; MRS-II, Menopause Rating Scale-II; n/a, not applicable; NR, not reported; NS, not significant; QoL, Quality of Life; wk, week.
Hot flash score = frequency × average severity.
P value is for comparison between groups.
Estrogenic Effects of Black Cohosh in Human Studies.

Abbreviations: bALP, bone-specific alkaline phosphatase; BC, black cohosh; BL, baseline; diff, difference; HRT, hormone replacement therapy; hx, history; BrCa breast cancer; M, menopause; M-status, menopausal status; NR, not reported; NS, nonsignificant; st, standardized to.
The following are standardized and/ or proprietary black cohosh extracts: Remifemin: standardized to contain 1 mg of triterpenes, measured as 27-deoxyacteine per 20 mg tablet; isopropanolic extract 40% by volume. CimiPure: standardized to 2.5% triterpenes, 1 mg 23-epi-26-deoxyactein per 40 mg extract, per 40 mg tablet. Klimadynon/Menofem: dried aqueous and ethanolic extract, 58% extract by volume, corresponding to 20 mg dried herb per tablet.
Where the comparator is an active agent such as HRT, the effect on the selected endpoints is reported as change over time within the black cohosh group only;: for instance, the effect of 2 months of black cohosh treatment on endometrial thickness, compared to baseline. Endpoints related to the other active groups are not reported.
Risk of Breast Cancer
Observational evidence
Of the 3 studies pertaining to risk of breast cancer, 2 studies found no significant association with black cohosh use compared with nonuse among postmenopausal women with no history of breast cancer, hazard ratio (HR) = 1.17 (95% confidence interval [CI] = 0.75-1.82) and adjusted odds ratio (OR) = 0.80 (95% CI = 0.63-1.00).21,23 One study found a significant reduction in breast cancer risk associated with black cohosh. Rebbeck et al 24 reported a 53% decreased risk of primary breast cancer incidence in postmenopausal women, OR = 0.47 (95% CI = 0.27-0.82), comparing users with nonusers.
Risk of Recurrence
Observational evidence
One study examined risk of recurrence and reported decreased risk associated with black cohosh use. Henneicke-von Zepelin et al 25 reported a 25% decreased risk of breast cancer recurrence associated with Remifemin in women with recent breast cancer and in breast cancer survivors, HR = 0.75 (95% CI = 0.63-0.89). This was despite 35.8% of Remifemin users taking tamoxifen, compared with 24.0% of nonusers. The 25% decreased risk of recurrence in the treatment group may therefore be due to either (a) use of Remifemin or (b) a higher rate of tamoxifen use. However, it does also suggest that Remifemin is unlikely to interact negatively with tamoxifen or specifically decrease its efficacy. 25
Apart from inherent methodological limitations such as retrospective design, these studies showed low to moderate risk of bias, fulfilling 6 to 8 of a possible 9 criteria on the NOS. Common inadequacies included failure to report blinded interviews to ascertain exposure (case control studies), and verifying that the outcome of interest was not present at the beginning of the study (prospective studies).
Effect on Hot Flashes or Related Symptoms
Observational evidence
One cohort study examined the association between the use of black cohosh and quality of life. Ma et al 22 found that use of black cohosh supplements among 68 women included in a 788-woman cohort was not associated with any effects on health-related quality of life or fatigue: adjusted odds ratio (AOR) better physical score = 0.66 (95% CI = 0.35-1.27), AOR better mental component score = 0.94 (95% CI = 0.52-1.73), and AOR severe fatigue = 0.78 (95% CI = 0.41-1.46). Black cohosh use was defined as ever-use compared with never use. The study was conducted prospectively from approximately 6 months postdiagnosis to 40 months postdiagnosis. Effects on survival or recurrence were not examined.
Uncontrolled clinical trials
One prospective trial examined the association of Remifemin use and hot flash severity among women with recent breast cancer diagnoses. 26 These women had “severe” hot flashes as assessed by the Menopause Rating Scale (MRS-II). Patients receiving Remifemin, one tablet twice daily (40 mg/d) plus tamoxifen (10-40 mg/d) had a significant decline in hot flash severity over the course of the study, from 17.6 at baseline to 13.6 at the end of the study (P < .001).
A second prospective trial examined the effect of Remifemin on hot flashes in 23 postmenopausal women who either had a history of breast cancer but were clinically free of disease, or who had a perceived increased risk of breast cancer. 28 Subjects received one capsule of Remifemin daily (20 mg) for 4 weeks. Six women (29%) were also taking tamoxifen or raloxifene. At the end of the study, there was a significant 56% reduction in hot flash score (95% CI = 40% to 71%), and a reduction in the mean number of hot flashes per day, from 8.3 at baseline to 4.2 at the end of the study.
Controlled clinical trials
Three RCTs investigated the efficacy of black cohosh for the reduction of hot flashes in pre- and postmenopausal women with a history of breast cancer who had completed treatment and were clinically free of disease. One study suggested benefit from black cohosh on hot flashes, 29 whereas 2 studies showed not significant effects compared with placebo.27,30 These studies are described in Tables 3 and 4.
Pockaj et al 27 conducted a crossover trial among 132 pre- and postmenopausal women with a history of breast cancer. Participants were randomized to receive 40 mg C. racemosa extract daily for 4 weeks, or placebo, following a 1-week washout period. A total of 44 patients were on hormonal therapies including tamoxifen, raloxifene, or aromatase inhibitors during the study, but these were evenly distributed between groups. Results showed no significant changes in hot flash score (P = .10) or Green Climacteric Scale scores (P value range = .18-.99) when groups were compared. There was no difference in adverse events between groups.
Jacobson et al 30 conducted a parallel-arm RCT including 85 women who had completed treatment for primary breast cancer and suffered from daily hot flashes. Randomization was stratified for tamoxifen use (n = 59). One tablet of black cohosh (uncharacterized) was given for 60 days. There was a 27% decrease in the mean number of hot flashes over the course of the study in the whole group, however, there was no significant difference in hot flash frequency between black cohosh and placebo groups (P = .86). No significant changes in LH or FSH levels were found overall or between groups. There were 3 serious adverse events: one breast cancer recurrence in a patient receiving both tamoxifen and black cohosh; one hysterectomy in a patient receiving both tamoxifen and black cohosh; and one appendectomy in a patient receiving tamoxifen and placebo. There were 10 minor adverse events with little consistency between them. No attribution to black cohosh could be inferred.
Hernández Muñoz and Pluchino 29 conducted open-label RCT comparing tamoxifen plus black cohosh with tamoxifen alone in premenopausal ER+ breast cancer survivors. 29 One capsule of black cohosh was given twice daily (40 mg/d). At study end, 42 (46.7%) of women in the black cohosh group reported no hot flashes, compared with 0 women from the control group (P value not reported). Similarly, fewer women in the black cohosh group reported “moderate” and “severe” hot flashes compared with the control group. There were 7 minor adverse events in the placebo group and 4 in the black cohosh group, but these were not described.
Effect on Estrogen Responsive Tissues
We included 5 uncontrolled trials31-35and 12 RCTs9,10,30,36-44 assessing the effects of black cohosh on estrogen responsive tissues, including in non–breast cancer populations, described in Table 5.
Of 9 studies assessing the impact of black cohosh on serum estradiol, all reported no change or nonsignificant decreases associated with black cohosh, with the exception of one study.10,32,33,35,37,38,41,42 This study assessed black cohosh in infertile women undergoing clomiphene citrate therapy, and the result was increased estradiol on the day of human chorionic gonadotropin injection (P < .001); however, this was deemed a therapeutic outcome by study authors. 36
Of 11 studies assessing impact of FSH and/or LH concentrations, only one study reported a decrease in FSH due to 2 perimenopausal women in the study who ovulated while in the study; with the exclusion of these 2 outliers, this study too reported no change in FSH or LH. 35 All other studies reported no impact on FSH or LH level, with the exception of Duker 43 , who reported a significant decrease in LH only (P < .05). 10,30,32,35,37,38,41-44
Of 3 uncontrolled trials assessing mammographic density, all 3 studies reported no significant changes overall with black cohosh use.31,34,35 In a large trial of 400 participants, the authors reported an increase observed in one participant only, with higher density, after using black cohosh for the 1-year treatment period. 35
Of 9 trials assessing endometrial thickness or incidence of hyperplasia, most found no change attributed to black cohosh. Two RCTs of infertile women on clomiphene citrate found that the addition of black cohosh to their fertility regime resulted in significantly greater endometrial thickness on the day of human chorionic gonadotropin injection mid-cycle (P < .001), however, this was deemed therapeutic.36,37 One RCT conducted in peri- and postmenopausal women reported a borderline significant decrease in endometrial thickness (P = .05). 39 One RCT reported a mean 0.5-mm increase, which was, however, deemed “not clinically relevant.” 40 The remaining studies reported no change,9,34,35,41 including one study that reported no cases of endometrial hyperplasia among a population of 351 subjects taking black cohosh for 1 year. 38
Two uncontrolled trials assessed markers of breast tissue proliferation, including nipple aspirate volume, pS2 levels in serum and nipple aspirate fluid, and Ki-67 expression.33,34 One study found nonsignificant decreases in serum pS2, nipple aspirate pS2, and no change in nipple aspirate volume. 33 The other study found no change in the number of Ki-67+ cells. 34
Three RCTs assessed vaginal cytology.10,38,42 None of the 3 studies found significant changes in the number of parabasal cells present or in cell maturation index; however, one study found a trend toward an increase in the number of superficial cells present (P = .0542). 10
Three studies found evidence of potentially beneficial effects on bone tissue.9,10,35 One uncontrolled trial found that use of black cohosh increased CrossLaps, a marker of bone resorption, ~96% in women with low levels at baseline, but decreased CrossLaps ~26% in women with high baseline levels; black cohosh increased osteocalcin (bone formation) between 5% and 36% in both groups. 35 Two RCTs found no change or a decrease in CrossLaps, and both found increased levels of bone-specific alkaline phosphatase, a marker of bone formation (P = .05 and P = .01).9,10
Risk of Bias
Randomized controlled trials assessing efficacy for hot flashes and assessing impact on estrogen responsive tissues were assessed for risk of bias using the Cochrane Risk of Bias tool. 20 Overall, the studies showed a moderate risk of bias, with the most common deficiency being unclear adequate allocation concealment, poor description of randomization methods, and an inability to assess for inclusion of complete outcome data (attrition bias) in approximately 50% of the studies. Since the outcomes assessed as part of the estrogenic effects analysis use objective testing methods, there is a low risk of detection or performance bias. Scoring results are depicted in Figures 2 and 3.

Risk of bias graph.

Risk of bias summary.
Discussion
Our review suggests that current evidence does not support an association between use of black cohosh and increased risk of breast cancer. Of 4 studies examining the impact on breast cancer risk, 2 studies found no significant association,21,23 and 2 reported an inverse relationship such that black cohosh use was associated with significantly reduced risk of primary breast cancer incidence or breast cancer recurrence, including the study of black cohosh combined with tamoxifen.24,25 With respect to hot flashes, current evidence is conflicting, with 2 placebo-controlled studies showing no significant effects,27,30 and 1 study comparing black cohosh plus tamoxifen to tamoxifen alone showing benefit. 25 A large placebo effect due to expectation bias may be at play, a real possibility especially given the importance of subjective outcomes in these studies; in addition, one study showing benefits between groups was an open study design, which may introduce bias. 29 However, equally plausible is that black cohosh does have some utility in this patient population.
A number of limitations within the included studies deserve consideration. Among observational studies, there is considerable variation in the dose of black cohosh considered to be the threshold that determines a “user”. Examples of this threshold point include a single day per week for at least 1 year out of the past 10 years 21 ; at least 3 times per week for 1 month 24 ; and simply having taken black cohosh (frequency not reported) for at least 3 months. 23 Whether the findings of these studies would translate to patients taking black cohosh on a daily basis for several months or even years is not clear. Furthermore, the impact on risk associated with breast cancer and potential benefit for hot flashes are both susceptible to effects introduced by such differing dose and duration schedules. Another potential source of bias is the retrospective design of several of the included studies (n = 3).
Our risk of bias assessment revealed that with respect to hot flashes, the only controlled trial that reported a marked benefit with black cohosh treatment also demonstrated a high risk of bias. 29 Two RCTs with low risk of bias showed no significant difference on hot flashes compared with the control group.27,30 Common deficiencies that may introduce bias to the findings reported in our review included lack of blinding and inadequate reporting of withdrawals in approximately 50% of the included RCTs. Use of objective testing to assess the impact of black cohosh on estrogen responsive tissues minimizes the risk of detection and performance bias among these studies. Among observational studies, those with better rankings on NOS scoring showed significantly reduced risk of breast cancer or recurrence,24,25 compared with those with poorer scores who showed no significant effects.21,23
Among RCTs, the cross-over study by Pockaj et al 27 included only a 1-week washout between treatments which may have been insufficient to allow for the complete washout of a hormonally or centrally active agent. 27 Moreover, both placebo-controlled trials27,30 demonstrated considerable placebo effects of 31% 27 and 27% 30 . This is important because the endpoints measured in these studies were inherently subjective (ie, self-reported frequency and severity of hot flashes). This appears to be true across studies of black cohosh for hot flashes in general, and not limited to those in breast cancer patients, as found in a recent yet to be published systematic review by Flower et al (personal communication, November 2011). Finally, this appears to be a consistent problem in hot flashes research, since other investigators have reported that the placebo effect can be as high as 25% on average, with up to a 75% reduction in hot flashes in 15% of women.45,46
Our review suggests that black cohosh has limited estrogenic activity. Black cohosh does not appear to possess classical estrogenic activity, as measured by breast and uterine tissue proliferation, but may possibly have nonclassical activities as seen by its effects on bone metabolism. Our review shows that black cohosh has no consistent pattern of influence on serum hormone levels (estradiol, FSH, LH) or the following estrogen responsive tissues: endometrial tissue, breast tissue, or vaginal tissues. Black cohosh does seem to stimulate bone formation and may inhibit bone breakdown in women with high bone turnover.
Mechanism of Action
It has been widely noted that although early preclinical studies indicated that black cohosh constituents were able to bind the ER in vitro, this is not supported by newer evidence.14,47 Several recent studies have shown a lack of estrogenic activity as mediated through the ER by black cohosh,14,48-50 whereas others show outright antagonism of estrogen-induced activities, including the proliferation of MCF-7 breast cancer cells and estradiol-induced gene expression. 51 These findings may collectively be due to differences in the ability of black cohosh to activate ER-α compared with ER-β.ER-α is the classical ER, distributed in the uterine and other estrogen sensitive tissues where it exerts proliferative effects, whereas ER-β has been associated with antiproliferative effects and modulates the responsiveness of target tissues to estradiol.52,53 Black cohosh has been shown to increase expression of ER-β gene within the uterus (P value not reported). 54 It has been suggested that black cohosh may possess SERM-like properties. 54
Black cohosh may also act on central estrogen (or other) receptors at the level of the hypothalamic GnRH pulse generator, where it has been suggested, though not proven, that ER-β may play a role.54,55 Menopause in humans and in animal models is characterized by exaggerated LH pulsatility, which is thought to affect the nearby hypothalamic thermoregulatory and other centers, causing typical vasomotor symptoms; negative feedback exerted by estrogen on the hypothalmus and LH pulsatility is a major mechanism for the reduction of hot flashes. 54 In one human study, black cohosh was able to reduce LH secretion in menopausal women, 43 though other studies included in our review failed to confirm this.30,42
Finally, black cohosh appears to affect other signalling systems. Dopaminergic and serotonergic systems have been proposed as possible pathways affected by black cohosh. Borrelli et al 14 suggests that effects such as inhibition of ER-positive cell proliferation (nonestrogenic-like) and bone sparing effects (estrogen-like) may be in part attributable to dopaminergic signaling, since these effects are inducible by other dopamine agonists. 14 The dopaminergic-based hypothesis is also supported by a study in which black cohosh extract increased prolactin, an effect that was then reduced by a D2 antagonist.14,56Jarry et al 47 have demonstrated D2 receptor binding by the black cohosh extract BNO 1055; dopamine has been shown to mediate ER transcription, an effect that has been also been observed after black cohosh administration. 14
With respect to serotonin, black cohosh has shown 5-HT7 receptor binding and activation in vitro.57,58 The recently identified black cohosh compound, N-ω-methylserotonin, has been shown to bind the 5-HT7 receptor with strong affinity, induce cAMP as a second messenger, and block serotonin reuptake, suggesting serotonergic activity for black cohosh. 58 Burdette et al 50 found that black cohosh bound strongly to 3 receptor subtypes (5-HT 1A, 1D, and 7), thereby acting as a partial agonist. Since the 5-HT 1A and 7 receptor subtypes are present in the hypothalamus, it has been proposed that serotonergic activity in the hypothalamic thermoregulatory centers may be responsible for the reduction in hot flashes.50,59
Safety: Hepatotoxicity
The safety of black cohosh has become controversial recently following several case reports of hepatotoxicity. 60 Although we did not systematically assess its impact on liver function, among the studies we included, no impact on liver function or symptoms suggestive of impaired liver function were reported. The issue of the potential hepatotoxicity has been reviewed in much more comprehensive detail elsewhere60-64 and a systematic review by others of our team addressing this question will be published imminently (Flower et al). In a meta-analysis of 5 RCTs, no effects were seen on alanine aminotransferase, aspartate aminotransferase, and gamma glutamyl transpeptidase (n = 1117). 62 A causality analysis of 69 cases of reported hepatotoxicity concluded that all reports were subject to confounding factors, such as “poor case data quality, uncertainty of [black cohosh] product, quality, and identification, undisclosed indication, insufficient adverse event definition” etc. 60
Tamoxifen and Aromatase Inhibitors
The potential for interactions with tamoxifen, in particular aromatase inhibitors, must also be considered. Five studies in our review (4 trials and 1 cohort study) included patients who were receiving both tamoxifen and/or raloxifene.25,27-30 None of the trials reported the impact of the combined therapy on risk of recurrence; however, the cohort study by Henneicke-von Zepelin et al 25 suggested that taking black cohosh reduced risk of recurrence by 25% (adjusted hazard ratio = 0.75, 95% CI = 0.63-0.89) in the treatment group, 35.8% of which was taking tamoxifen. 25 No consistent serious adverse events related to the combination of black cohosh and tamoxifen was reported by any of the trials.27-30
An animal study comparing the antitumor effects of formestane with or without the addition of 60 mg/kg isopropanolic black cohosh extract found that the addition of black cohosh had no effect on formestane-induced tumor reduction or reduction of serum estrogen levels. 65 A second study examining black cohosh with tamoxifen in a model of endometrial cancer found similar results; unlike the endometrial estrogen agonist tamoxifen, “black cohosh did not further growth or metastasizing potential of the primary tumor.” 66 There were no detectable supportive or antagonistic effects between the 2 treatments. 66
In humans, one study reported a statistically significant inhibition of CYP 2D6 by black cohosh: (difference = −0.046; 95% CI = −0.085 to −0.007).67,68 This study used a very high dose of black cohosh (>1000 mg), however, and the magnitude of the effect seen (approximately 7%) “did not appear to be clinically relevant,” 68 which cases some doubt on its clinical applicability. Other studies failed to confirm this effect. 69 Nonetheless, this finding is worth noting, given that tamoxifen, an SERM, is primarily metabolized by CYP 2D6. 70 Other inhibitors of CYP 2D6, such as selective serotonin reuptake inhibitors have been shown to reduce serum levels of tamoxifen’s active metabolites, notably endoxifen, by up to 50%. 70 Theoretically, black cohosh might have lesser such effects, though this has not been directly studied. Black cohosh does not appear to affect the following enzymes: CYP 1A2, CYP 2E1, CYP 3A4, CYP 3A5, or Pgp.67,68,71,72 Conversely, the aromatase inhibitor anastrozole, which is primarily used in postmenopausal women, 73 is metabolized primarly by CYP 3A4 and also to some extent by CYP 3A5, CYP 2C8, and UGT1A4 74 suggesting that this drug is less likely to be affected by a pharmacokinetic interaction with black cohosh. The other third generation aromatase inhibitors including letrozole and exemestane are also not affected by CYP 2D6. 75
Conclusion
Black cohosh does not influence circulating levels of estradiol, FSH, or LH, or appear to exert estrogenic effects on breast, endometrial, or vaginal tissues. There is mixed evidence regarding the efficacy of black cohosh for the reduction of hot flashes in breast cancer survivors. Based on preliminary observational data (n = 4), black cohosh does not appear to adversely impact the risk of breast cancer recurrence or incidence in women with or without a history of breast cancer. More evidence is required to confirm these early findings before the question of black cohosh’s safety and efficacy in this population can be conclusively answered. Given the lack of adequate safe therapies, particularly hormone replacement therapy, and the unlikely impact of black cohosh on breast cancer risk or recurrence, the use of black cohosh for hot flashes by women surviving breast cancer may be warranted in some cases provided that there are no allergic contraindications.
Footnotes
Appendix
Search Strategy
|
|
|
|---|---|
|
|
|
| Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations and Ovid MEDLINE(R) <1948 to Present> |
Cimicifuga/ (320) (actaea racemosa$ or cimicifuga$ or black cohosh$ or black bugbane$).tw. (460) 1 or 2 (514) exp breast neoplasms/ (175475) ((breast or mammary) adj2 (neoplasm$ or cancer$ or tumour$ or tumor$)).tw. (148217) 4 or 5 (211801) 3 and 6 (74) |
| AMED (Allied and Complementary Medicine) <1985 to January 2011> |
Cimicifuga/ (11) (actaea racemosa$ or cimicifuga$ or black cohosh$ or black bugbane$).tw. (93) 1 or 2 (93) exp breast neoplasms/ (855) ((breast or mammary) adj2 (neoplasm$ or cancer$ or tumour$ or tumor$)).tw. (1251) 4 or 5 (1251) 3 and 6 (8) |
| EMBASE Classic+EMBASE <1947 to 2011 January 26> |
Cimicifuga/ (189) Cimicifuga racemosa/ (361) Cimicifuga racemosa extract/ (777) (actaea racemosa$ or cimicifuga$ or black cohosh$ or black bugbane$).tw. (675) or/1-4 (1197) exp breast cancer/ (212993) ((breast or mammary) adj2 (neoplasm$ or cancer$ or tumour$ or tumor$)).tw. (188742) 6 or 7 (270446) 5 and 8 (259) |
|
|
|
| Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations and Ovid MEDLINE(R) <1946 to Present> |
Cimicifuga/ (367) (actaea racemosa$ or cimicifuga$ or black cohosh$ or black bugbane$).tw. (536) 1 or 2 (594) exp breast neoplasms/ (196668) ((breast or mammary) adj2 (neoplasm$ or cancer$ or tumour$ or tumor$)).tw. (171410) 4 or 5 (240389) v3 and 6 (83) 201102* or 201103* or 201104* or 201105* or 201106* or 201107* or 201108* or 201109* or 201110* or 201111* or 201112* or 2012*).ed. (1432704) 7 and 8 (7) |
| AMED (Allied and Complementary Medicine) <1985 to July 2012> |
Cimicifuga/ (16) (actaea racemosa$ or cimicifuga$ or black cohosh$ or black bugbane$).tw. (100) 1 or 2 (100) exp breast neoplasms/ (958) ((breast or mammary) adj2 (neoplasm$ or cancer$ or tumour$ or tumor$)).tw. (1378) 4 or 5 (1378) 3 and 6 (8) ("201102" or"201103" or "201104" or "201105" or "201106" or "201107" or "201108" or "201109" or "201110" or "201111" or "201112" or "201201" or "201202" or "201203" or "201204" or "201205" or "201206" or "201207").up. (16906) 7 and 8 (0) |
|
|
|
| Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations and Ovid MEDLINE(R) <1946 to Present> |
Cimicifuga/ (377) (actaea racemosa* or cimicifuga* or black cohosh* or black bugbane*).tw. (550) 1 or 2 (609) exp Estradiol/ (72289) (estradiol* or aerodiol or estrace or estraderm or oestradiol or ovocyclin or vivelle).tw. (75097) 50-28-2.rn. (71427) exp Estrone/ (8393) (estrone or estrovarin or folliculin or kestrone or unigen or wehgen).tw. (7471) 53-16-7.rn. (8178) exp Estriol/ (5657) (estriol or estetrol or epiestriol or ovestin).tw. (3718) 50-27-1.rn. (5641) exp Progesterone/ (61718) (progesterone or pregnenedione).tw. (64316) 57-83-0.rn. (50682) exp Hormones/bl, me, ur [Blood, Metabolism, Urine] (425371) exp Breast/cy, de, me, pa, ph, pp, se, ul [Cytology, Drug Effects, Metabolism, Pathology, Physiology, Physiopathology, Secretion, Ultrastructure] (17388) exp Nipple Aspirate Fluid/ (29) ((breast* or mammary or nipple* or mammogra*) adj5 (cytolog* or chang$3 or dense* or density or growth* or increas* or volume* or aspirat* or proliferat*)).tw. (39800) exp Menstrual Cycle/de, me, ph [Drug Effects, Metabolism, Physiology] (10559) (menstrua* adj5 (chang* or differen* or frequen* or length* or duration*)).tw. (4399) exp Endometrium/cy, de, me, pa, ph, pp, ul [Cytology, Drug Effects, Metabolism, Pathology, Physiology, Physiopathology, Ultrastructure] (18688) (endometri* adj5 (cytolog* or chang* or differen*)).tw. (6596) exp Leiomyoma/me, pa, ph, pp, ul [Metabolism, Pathology, Physiology, Physiopathology, Ultrastructure] (6292) ((leiomyoma* or fibroid* or fibroma* or fibromyoma or angiomyoma* or angioleiomyoma*) adj5 (cytolog* or chang* or differen*)).tw. (954) exp Vagina/cy, de, me, pa, ph, pp, ul [Cytology, Drug Effects, Metabolism, Pathology, Physiology, Physiopathology, Ultrastructure] (7256) exp Vaginal Smears/ (18786) ((vaginal or pap or cervical or Papanicolaou) adj (smear* or test*)).tw. (12023) exp Estrogens/ (138364) (estrogenic* or antiestrogenic* or anti-estrogenic*).tw. (11373) exp Receptors, Estrogen/ (36013) ((estrogen* or estradiol*) adj3 receptor*).tw. (38870) exp Estrogen Receptor Modulators/ (29280) (SERM or SERMS).tw. (1458) or/4-34 (686756) 3 and 35 (183) limit 36 to human (144) (publisher or in process or pubmed-not-medline or in-data-review).st. (1397825) 36 and 38 (8) 37 or 39 (152) |

Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by a Knowledge Synthesis grant from the Canadian Institutes of Health Research (CIHR). Deborah A Kennedy was supported by a career grant from the SickKids Foundation.
