Abstract
Quercetin is a plant-derived flavonoid known to possess anti-inflammatory property, but its use as a chemopreventive substance has been reviewed quite recently. A number of reports have assessed the pro-apoptotic action of quercetin in cancer cells, which contributes to the chemopreventive action of food, similar to many other flavonoids. Quercetin is a forthright inhibitor of PI3K, NF-B, and other kinases involved in intracellular signaling. However, a possible selective intracellular target for quercetin has still not been focused on. The challenge is to highlight an eligible target in order to address possible natural compounds, either in food extracts or as pharmaceuticals, as real drugs against cancer.
Introduction
Quercetin (C15H10O7, 3,3′,4′,5,7-pentahydroxy-2-phenyl-chromen-4-one) is a plant-derived flavonoid known to possess anti-inflammatory properties. 1 Its use as a chemopreventive substance has recently been reviewed. 2 From a biochemical point of view, quercetin is a flavonol that is biosynthesized in plants through the phenylpropanoid pathway, starting from the amino acid phenylalanine; its closest related metabolites are naringenin and eriodictyol that lead to quercetin through a dihydroquercetin intermediate (Figure 1). Quercetin can be found in several plant-derived foods and beverages, and its main dietary sources are normally diffused in the Mediterranean area, being represented by red onion, capers, pepper, and broccoli, in addition to berries and apples, which constitute a common traditional diet in southern Europe. A growing focus on diet and the use of naturally abundant compounds as supplements is occurring nowadays, because their properties have many potential health benefits with minimal side effects. Quercetin has increased in popularity because it is a highly studied, multidimensional, bioactive compound that possesses both antioxidant properties and the ability to modulate signal transduction pathways, leading to prevention and inhibition of many inflammatory reactions. Nevertheless, substantial concern about the real possibility to use food-derived quercetin in clinics has been raised because of the complexity in intestinal absorption and bioavailability of polyphenols3,4 and because of lack of information about their pharmacokinetics, 5 although some suggestions have recently been made. 6 This issue may actually hamper experimental settings and interpretation of results. In plants, flavonoids such as quercetin exist in several conjugated forms, although most in vitro assays take into account the aglycone purified forms of these polyphenols. Some authors have suggested a fundamental role of the food matrix in ameliorating gut absorption. 7 In humans, when digested conjugated quercetin undergoes radical transformation inside the gut because of intestinal microflora, biochemical pathways such as methylation and hydroxylation lead to the formation of other flavonoids: For example, 3′-methylation of quercetin leads to isorhamnetin and 5′-hydroxylation of quercetin leads to myricetin. 1 Phenolic glycosides that reach the colon will be hydrolyzed by the gut microflora and may also undergo further metabolism and degradation. Once absorbed, dietary phenolics can be methylated (mainly at positions 3, 3′, 4′, 7) by methyl transferases of tissues to a varying degree and will be conjugated to sulfate and/or glucuronic acid prior to excretion. Following gut biochemical modification and absorption, quercetin bioavailability in vivo reaches a nanomolar range of plasma concentration, quite far from the micromolar range effective in in vitro studies. This observation, besides the complex derivatives born from gut microflora modification, may constitute a hindering issue to ascertain the potential benefit of quercetin in human cancer chemoprevention in vivo. However, as most of absorbed quercetin is hydrolyzed in its sugar link 3 and because of the complexity of the role exerted by quercetin within the cell, as recently reported for many plant phytochemicals, some authors have suggested the possibility of hormetic and synergistic effects also in the nanomolar dose range.1,8 Further insights on this topic may contribute to a clearer explanation of the role of quercetin in human biology and in cancer research in the future.
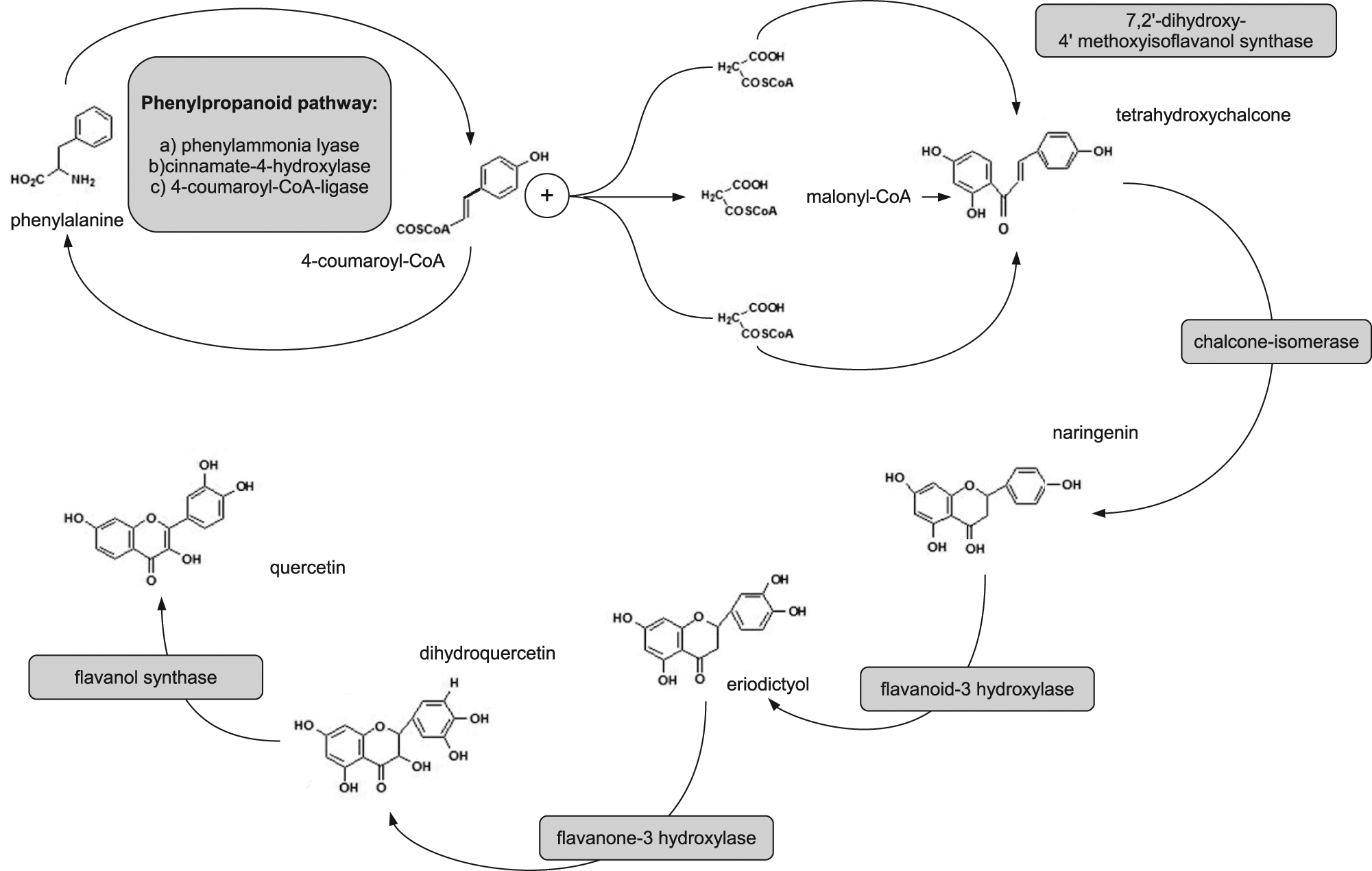
Biosynthetic pathways leading to quercetin from phenylalanine
Quercetin as a Chemopreventive Agent
Like most flavonoids, quercetin has potentially beneficial effects on health promotion and disease prevention, including tumors. The mechanism by which quercetin exerts an anti-inflammatory, pro-apoptotic, and chemopreventive action has not yet been fully understood, but several models attempting to explain its molecular action have been addressed. One of these models concerns nuclear factor-κB (NF-κB) inhibition by quercetin, also through the PI3K/Akt/IKK/NF-κB signaling axis. As it is widely acknowledged, flavonoids are naturally occurring NF-κB inhibitors.9-11 The transcription factor NF-κB has attracted widespread interest because of its unusual and complex regulation and because of the wide variety of stimuli that activate it, in addition to its apparent involvement in a variety of human diseases, the diverse genes and biological responses that it controls, and the striking evolutionary conservation of its structure and function among family members. 12 Recently, NF-κB has been shown to be a promising target of new drug discovery, thus expanding the interest to those compounds from raw natural sources.13,14 NF-κB plays an outstanding role in tumorigenesis and cancer development. The primary form of NF-κB, called RelA (the heterodimer p50/p65), is maintained in a latent form by the NF-κB inhibitor IκB. NF-κB is stimulated by phosphorylation of IκB through IκB kinase (IKK), which is constituted by the IKKα, IKKβ, and IKKγ subunits. IKKβ is the primary kinase that phosphorylates the amino-(NH)-terminus of IκB in vivo, whereas IKKγ, known as NF-κB essential modulator or NEMO, is a crucial regulatory subunit. Quercetin has been reported to possess significant antitumor activities both in vitro and in vivo, and many of these effects relate to a direct or indirect action performed by quercetin toward these molecular participants leading to cancerogenesis. Quercetin reduces IKK, 15 but this effect, while inhibiting NF-κB, may suggest different results in a pro- or anti-apoptotic direction, if one considers that NF-κB regulates p53 also by acting on p53 gene. 12 A number of reports assess the pro-apoptotic action of quercetin in cancer cells, which contributes to the chemopreventive action of food, similar to many other flavonoids.16-18 What is interesting for integrative medicine is that quercetin is a potentially genotoxic substance. Although its mutagenicity and chemical toxicity has not been confirmed in in vivo studies, it might activate p53 signal—which is negatively regulated by HDM2, a component that depends on Akt activation and which functions as an E3 ubiquitin ligase inducing p53 proteolysis 19 ; low doses of quercetin, in the context of hormesis, allow this polyphenol to act in a protective fashion. However, the role of the quercetin molecule in NF-κB–p53 cross-talk still remains obscure. A relationship between quercetin and p53 has been reviewed recently,20,21 but quercetin appears to be able to induce apoptosis regardless of the p53 signaling pathway. 21 Furthermore, this flavonoid is able to curb the PI3K/Akt/IKKα/NF-κB pathway in human salivary adenoid cystic carcinoma leading to the induction of cell apoptosis through a mitochondria-dependent mechanism. 22 In these cells, PI3K/Akt/IKKα/NF-κB signaling pathway (Figure 2) plays a role in evading cell apoptosis; quercetin promotes cancer cell apoptosis by blocking PI3K/Akt upstream of the NF-κB signaling. 22 Many flavonoids, including quercetin, are inhibitors of different PI3K isoforms and PI3K/Akt axis,23-25 so PI3K is a possible molecular target for quercetin. The fact is that quercetin is able to inhibit many other kinases and enzymes besides PI3K and NF-κB. 1 Moreover, the flavonol may act positively on kinase/suppressor factors, thus inducing indirect kinase inhibition. Quercetin does not inhibit Akt and ERK1/2 directly but is able to increase the oncosuppressor PTEN, 26 thus enhancing the inhibitory effect on PI3K function.

Intracellular signaling map of PI3K/Akt/IKK/NF-κB pathway with main related cross-talks. Major points affected by quercetin are indicated. Activations are indicated by arrows whereas dashed lines indicate inhibitory ways. For further explanation see text
Akt has a well-documented pro-survival function. 27 The kinase activity of Akt is elicited through the phosphoinositide-3-OH kinase (PI3K) and PI3K-dependent kinase 1/2 (PDK 1/2) signaling pathways: inhibition at this level may therefore lead to the inactivation of Akt. 28 Quercetin dependent inhibition of cell survival, cancerogenesis, and tumor development as well as induction of apoptosis might appear related to upstream PI3K inactivation, as quercetin is a direct antagonist of PI3K—like wortmannin—29,30but actually many other downstream components are involved in the chemopreventive and antitumoral action of this flavonoid, and the complete picture is not yet fully understood. 31 Overexpression or constitutive activation of Akt has been associated with tumorigenesis in a number of studies.32-34 The PI3K/Akt/NF-κB axis in the signaling pathways leading to tumorigenesis and the possible chemopreventive action of quercetin in this network are summarized in Figure 1. One of the key factors in cancerogenesis is represented by IκB kinase. 35 The role of its subunits IKKα and IKKβ is not indifferently related to the role of quercetin within the PI3K/Akt/IKK/NF-κB signaling. Phosphorylation of Ser536 in RelA by IKKβ represents the mechanism that links PI3K/Akt to NF-κB signaling. Possible inhibition of Akt by affecting upstream PI3K function may lead to suggest these following scenarios: (a) Akt is not able to phosphorylate IKKβ, which in turn cannot phosphorylate Ser536 in RelA and activate NF-κB, leading to the inhibition of NF-κB mediated cancerogenesis and tumor progression and (b) Akt is not able to activate IKKα leading to inhibition of cyclin D1-dependent cell cycle, by suppressing the activity of IKKα on estrogen receptor α (ER-α) and its cofactor SCR3 in a NF-κB independent way or also by affecting β-catenin and cyclin D1. 12 Nevertheless, several issues raise the suggestion that quercetin may actually exert a pleiotropic action on multiple targets involved in cell signaling. Some authors have recently investigated the anti-proliferative effects of quercetin on activated hepatic stellate cells showing that the flavonol caused G(1) arrest with increased levels of p53, CIP1/WAF1 (p21), and p27 (KIP1), a decrease in most cyclins (D1, D2, A, and E), induction of apoptosis through a Fas/FasL mechanism, and a reduction in the level of inflammatory cytokines (CXCL10, for example). 36
Insights Into The Role of Quercetin
In this articulated network of protein-to-protein cross-talks, what is the main role of quercetin and, if possible, its ruling target? In addition to the molecular form by which quercetin acts in vivo, its actual bioavailability, to the role of concentration ranges and hormetic/synergistic or biphasic mechanisms, this question is hampered by a great number of other critical and difficult issues. A cross-talk between NF-κB and the tumor suppressor factor p53 regulating apoptosis has been reported.12,37 NF-κB can induce anti-apoptotic genes that antagonize the pro-apoptotic function of p53. NF-κB, or the IκB family member BCL-3, can also induce the expression of HDM2 and reduce p53 protein levels; so the role of p53 in this context is highly complex and it is probably early to indicate p53 as a good target for quercetin in fighting against tumors. A competition may occur between p53 and RelA for binding to shared coactivator proteins, for example, cyclic-AMP responsive element binding (CREB)-binding protein (CBP) or p300; by contrast, cooperative pathways between p53 and NF-κB were also reported.12,38 In human cervical cancer (HeLa) cells quercetin induces apoptosis by promoting p53 activity and inhibiting NF-κB: quercetin provoked a p53/p21 mediated cell cycle arrest in G2/M, 39 a result that confirmed previously reported evidence in other tumors. 40 The way by which quercetin induces apoptosis in cancer cells is therefore more complex than the inhibition of PI3K/Akt/IKK/NF-κB axis and may occur regardless of p53 involvement.21,39 Nevertheless, other ways are not less complicated. Without considering the role of quercetin in the oxidative pathway, the flavonol is a direct inhibitor of GSK-3β, a component of GSK-3β/β-catenin signaling pathway, whose inhibition leads to the suppression of cell cycle and induction of apoptosis. 40 GSK-3β is a key regulator of the NF-κB activity, as its inhibition might be addressed to counteract those cancers with constitutively active NF-κB.40,41 Also for the GSK-3β/β-catenin pathway, encouraging suggestions have been made as a possible forthcoming target but researchers have to face these controversial issues. The place of quercetin within the crowded population of intracellular kinases and signaling proteins, therefore, appears unsettled. Solving this molecular issue is necessary to design reliable quercetin-derived molecules that are able to counteract cancer development and metastasis; nowadays, apart from numerous attempts to recognize and apply nature-derived molecules, selected for their efficacy and bioavailability, the fight against cancer using natural products is mainly confined to preventive medicine. The process of carcinogenesis is complex and not easy to eliminate and certainly identifying food and food components with biological properties, collectively called nutraceuticals, which can hinder such alterations and prevent the inactivation of tumor-suppressor genes is a very promising area for cancer prevention. 42 A very wide range of intracellular components can be affected directly or indirectly by quercetin, and trying to identify a strategic target for cancer treatment is like looking for a needle in a haystack. Maybe, possible suggestions could come from considering two important hallmarks of quercetin as a chemopreventive substance, namely, that it is a genotoxic compound and a phytoestrogen. For example, as a moderately and well tolerated genotoxic compound in vivo, 43 quercetin belongs to that wide range of plant-derived molecules that contributed to the conservation, during evolution, of p53 superfamily, having the task of inducing apoptosis in response to xenobiotic genotoxic agents: 44 current strategies in targeting p53 in cancer have recently been attempted.45,46 Deepening the role of plant-derived products from dietary intake in the context of the molecular regulation of apoptosis may result in a possible comprehension of their role in animal cells. As a phytoestrogen, it may induce apoptosis47,48 either by extrinsic or intrinsic pathways, involving NF-κB and the PI3K/Akt signaling pathways 26 : This evidence has been reported, however, mostly for breast cancer.
Conclusions
In conclusion, although a huge stack of evidence and hypotheses is still being collected, many other insights are needed because of the increasing number of polyphenolic compounds that are meeting and invading many research areas with promising expectations. Several plant-derived molecules, which are widely present in the human daily diet, are proving feasible tools in fighting against tumors by inhibiting the growth of cancer cells with an ability to act as chemopreventive compounds. Their cancer-preventive effects have been attributed to various mechanisms, including the induction of cell-cycle arrest and/or apoptosis as well as antioxidant functions. The challenge is to highlight an eligible target in order to address possible natural compounds, either in food extracts or as real drugs against cancer. This goal is being enriched by promising results and models but we are still very far from an effective solution to design meaningful and cost-effective clinical trials.49,50
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
