Abstract
Introduction
Several microorganisms and their metabolites can be used as probiotics due to their roles in the production of gastrointestinal enzymes such as protease, lipase, lactase, B vitamins, and natural antibiotics. In fact, probiotics can inhibit at least 27 types of pathogenic bacteria in the gastrointestinal tract to decrease the incidence of gastrointestinal diseases.1,2 Specific properties of a probiotic depend on the source. Numerous strains of
Gastric cancer is a major cause of death worldwide, even though its prognosis has improved due to advances in diagnostic and surgical techniques. Although various chemotherapies have been developed to treat this disorder, many fail to completely reduce gastric carcinomas.7,8 Many human diseases occur as a result of inappropriate signaling by the mammalian target of rapamycin (mTOR). Recently, mTOR has been considered as a therapeutic target for gastric cancer because it coordinates proper cell growth and proliferation by regulating ribosome genesis and protein translation. 9 While evidence shows that LBX benefits cancer patients, we know little about the mechanism responsible, including the various signaling pathways involved.
We therefore investigated whether LBX may be a potential therapy for gastric cancer. LBX was applied to gastric cancer (KATO3) and normal (HDF) cells to determine the signaling pathways that prevent proliferation and cause apoptosis in gastric cancer cells.
Materials and Methods
Reagents and Chemicals
Dulbecco’s modified eagles medium (DMEM) and RPMI1640 were purchased from Welgene (Daegu, Korea). Cell culture dishes were purchased from NUNC (Roskilde, Denmark). Chemicals including 4-(2-hydroxyethyl)-L-piperazine ethane sulfonic acid (HEPES), phosphate-buffer saline (PBS), and L-glutamate were purchased from GIBCO (Paisley, Scotland, UK). Hoechst-33258 dye (Sigma Co, St Louis, MO) and Annexin-V-FLUOS staining dye (Roche Diagnostics GmbH, Mannheim, Germany) were used for FACS analysis. Luciferase plasmid (3x-κB and AP-1) were purchased from Stratagene (La Jolla, CA). Antibodies against ERK, phospho-ERK, NF-κB, IκB, and GAPDH were purchased from Santa Cruz Biotechnology Inc (Santa Cruz, CA). Antibodies for phosphorylated and nonphosphorylated proteins, such as p21, caspase-3, Bax, Bcl-2, PI3K, AKT, m-TOR, and p70S6 kinase, were purchased from Cell Signaling Co (Boston, MA). Whole cell lysis buffer and Hilymax/CCK-8 transfection reagent were purchased from iNtRON (Seoul, Korea) and Dojindo (Dojindo, Japan), respectively. All other chemicals used were ACS grade or better.
Preparation of Lactobacillus casei Extract
Cell Culture and Cell Viability Assay
Human gastric cancer (KATO3) and diploid fibroblast (HDF) cell lines were obtained from American Type Culture Collection (Rockville, MD) and Seoul National University, respectively. Cells were grown in RPMI1640 and DMEM supplemented with 10% (v/v) FBS (GIBCO, New York, NY) and 1% (w/v) penicillin-streptomycin (GIBCO) in a 37°C incubator with 5% (v/v) CO2 for 24 hours. KATO3 cells were seeded at 5 × 103 cells/well in a 96-well plate. After 24 hours, the cells were treated with LBX at various concentrations. The appropriate dose and time were determined by evaluating cytotoxicity. In brief, cells were treated with 0, 0.125, 0.25, 0.5, or 1 mg/mL for 24 and 48 hours. Ten microliters of cell counting kit-8 solution (Dojindo, Japan) was added for 1 hour. Cell viability was determined by measuring the absorbance at 450 nm using a microplate reader (Sunrise, Tecan, Switzerland). Each experiment was performed in triplicate.
Hoechst 33258 Staining
After treating KATO3 cells with 0.5 mg/mL LBX for 48 hours, the cells were washed twice with PBS and fixed with 3.7% (v/v) formaldehyde (in PBS) for 5 minutes at room temperature. Fixed cells were washed with PBS and stained with Hoechst 33258 (2 µg/mL) for 10 minutes at room temperature. After washing twice with PBS, apoptotic and nonapoptotic cells were analyzed using laser-scanning confocal microscopy (LSM 710, Carl Zeiss, Oberkochen, Germany) equipped with a C-Apochromat 40X/1.2 water immersion lens (405 nm argon laser/410-585 nm detection range).
Flow Cytometric Analysis Using PI–Annexin-V staining
Cells were treated with LBX for 48 and 72 hours, harvested, washed twice with PBS, and centrifuged at 2000 rpm for 5 minutes at 4°C. Then the cells were incubated with 0.2 mg/mL Annexin-V FLUOS and 1.4 mg/mL propidium iodide (PI), a DNA stain, for 15 minutes at room temperature. To determine the phenotype of cells at 48 and 72 hours, cells were treated with 0.5 mg/mL (IC50 value) LBX. Measurements were conducted using a FACS Calibur flow cytometer (Becton Dickinson Immunocytometry Systems, San Jose, CA) at 488 nm excitation with 530/30 nm band pass filter to detect Annexin-V and a 670 nm high pass filter to detect PI. FACS data were analyzed using WinMDI V2.9 software.
Luciferase Reporter Assay
KATO3 cells were transfected using Hilymax (Dojindo, Japan) according to the manufacturer’s instructions. For the luciferase assay, cells were cultured in 24-well plates in RPMI1640 for 18 hours before being transiently transfected with 3x-κB (NF-κB-Luc plasmid) and AP-1 (AP-1-Luc plasmid). Cells were transfected for 4 hours, incubated overnight in media. Cells were treated with 0.5 mg/mL LBX for 24 hours. Promoter activity was measured using a Dual Luciferase Assay Kit (Promega, Madison, WI), measuring both luciferase and renilla luciferase with a luminometer (Promega, Seoul, Korea). Cells were lysed and assayed for NF-κB and AP-1 reporter activities, which were corrected for constitutive β-galactosidase luciferase expression. Normalized values were calculated by dividing the luciferase activity by the renila luciferase activity.
Western Blot Analysis
Western blots were conducted as described previously. 10 The protein concentrations of cell lysates were measured using a BCA protein assay kit. Equal amounts of protein from cell lysates were resolved on sodium dodecyl sulfate-polyacrylamide gel and transferred to Immobilon nitro cellulose membranes. Blots were blocked with 5% (w/v) nonfat dried milk and 0.1% (v/v) Tween 20. Blots were incubated overnight at 4°C with primary antibodies against ERK (1:5000), phospho-ERK (1:1000), NF-κB (1:1000), IκB (1:500), p21 (1:1000), Caspase-3 (1:500), Bax (1:1000), Bcl-2 (1:1000), PI3K (1:1000), AKT (1:1000), phospho-AKT (1:1000), mTOR (1:1000), p-mTOR (1:1000), p70S6 kinase (1:1000), phospho-p70S6 kinase (1:1000), and GAPDH (1:1000). Blots were washed and incubated with anti-rabbit secondary antibody conjugated to horseradish peroxidase (1:2000, Invitrogen, Carlsbad, CA). The immune complexes were visualized using an enhanced chemiluminescence (Millipore, Billerica, MA) system according to the manufacturer’s protocol.
Statistical Analyses
Graph-Pad Prism (GraphPad, San Diego, CA) was used for statistical analyses. Student’s
Results
LBX Inhibits the Growth of Gastric Cancer Cells
To investigate the effect of LBX on proliferation of gastric cancer cells, KATO3 cells were treated with LBX, and the growth was measured using an MTT assay. Treating KATO3 cells with LBX for 24 hours increased growth, but the difference between the treated and control cells was not statistically significant (data not shown). By 48 hours, LBX significantly inhibited proliferation of KATO3 cells (Figure 1A). LBX, however, did not affect the proliferation of normal human HDF cells at 48 hours (Figure 1B). Thus, LBX was specifically effective for gastric cancer cells. To quantify the effective LBX concentration against gastric cancer, KATO3 cells were treated with various LBX concentrations (0, 0.125, 0.25, 0.5, and 1 mg/mL) for 48 hours. As shown in Figure 1A, LBX inhibited KATO3 cell growth in a dose-dependent and time-dependent manner. At 0.5 mg/mL, LBX inhibited approximately half of KATO3 cells and was thus considered the concentration of 50% inhibition (IC50).

MTT assay of KATO3 and HDF cells treated with LBX
LBX-Induced Apoptotic Changes in KATO3 Cells
To observe morphological changes, KATO3 cells treated with 0.5 mg/mL LBX were analyzed using phase contrast microscopy. As shown in Figure 2A, KATO3 cells were clearly decreased in size and number after incubation with LBX for 48 hours. Most KATO3 cells were viable after treatment with LBX for 24 hours, although some cells were poorly attached. By 48 hours, however, most cells began to shrink and their nuclei were fragmented, suggesting apoptosis (Figure 2B).

Morphological changes in KATO3 cells treated with 5 mg/mL LBX
The effect of LBX on cell death was analyzed quantitatively and qualitatively using flow cytometry after Annexin-V and PI staining (Figure 3). Flow cytometry can differentiate between viable cells, those undergoing later stages of apoptosis (Annexin-stained, nondisrupted cells), and those undergoing necrosis. KATO3 cells appeared shifted from live cells to those in the stage of late apoptosis. Without LBX, 84.17% of KATO3 cells were alive after 48 hours. In the absence of LBX, 1.2% and 8.84% of KATO3 cells were in early and late apoptosis, respectively. After treating cells with LBX (0.5 mg/mL) for 72 hours, 11.55% of cells were in early apoptosis (Annexin V-stained cells) and 85.62% of cells were in late apoptosis. Thus, LBX induced most cells to undergo apoptosis in a dose-dependent and time-dependent manner.

Flow cytometry of PI–Annexin-V to quantify LBX-induced apoptosis in KATO3 cells
LBX Reduces NF-κB Activity
Since LBX induces apoptosis in gastric cancer cell, we studied which downstream effectors mediate this death. We transfected 3x-κB-Luc and AP-1-Luc plasmids for 4 hours to generate high expression in KATO3 cells. The promoter activities were measured using a luminometer (Figure 4A and B). A control reporter expressing renilla luciferase from a CMV promoter (CMV RL-Luc) was transfected to measure background levels. Treating cells with 0.5 mg/mL LBX significantly decreased NF-κB activity. LBX reduced luciferase expression from the NF-κB promoter by 50%. AP-1 promoter activity, however, was increased slightly by 5%, though the effect was not statistically significant. It was notable that transcription factor NF-κB was required to maintaining cellular signaling; however, this transcription factor was cosuppressed by LBX. This suggests that LBX blocked the normal signal transduction of transcription factor NF-κB. Thus, LBX may regulate the downstream signaling of NF-κB to inhibit gastric cancer.

Promoter activity assay in KATO3 cells
LBX Reduces the ERK Signaling
Mitogen-activated protein kinase (MAPK) with extracellular signal-regulated kinases (ERKs) transduces signals from membrane receptors to affect the expression of a target gene. This phosphorylation cascade is deregulated in many cancers, including gastric cancer. 11 In general, phosphorylated ERK is translocated to the nucleus, where it phosphorylates AP-1, stimulating the transcription of target genes. When KATO3 cells were treated with LBX, ERK expression did not change (Figure 5A), though the expressions of NF-κB, IκB, and p21 were decreased in a time-dependent manner (Figure 5B). The decreased NF-κB expression is consistent with the decreased NF-κB promoter activity described above (Figure 4A).
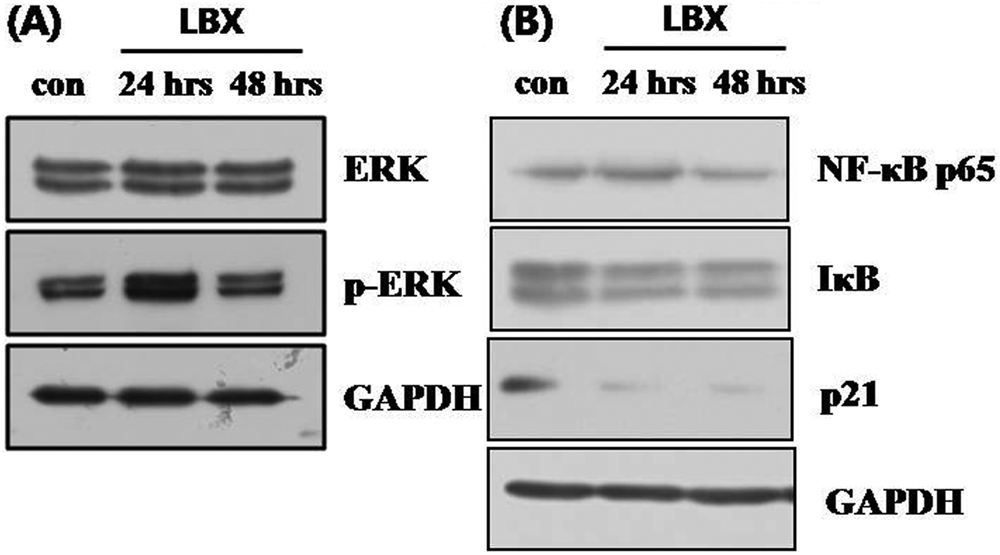
Western blots of MAP kinases and transcription factors in LBX-treated cells
LBX Suppresses the Apoptosis Linked to NF-κB and mTOR
To evaluate whether LBX induces apoptosis through caspases, we measured caspase-3 expression using Western blot analysis. Pro-caspase-3 expression (35 kDa) was decreased over time during LBX treatment, whereas that of active caspase-3 (17 kDa) was increased (Figure 6A). Expression of another pro-apoptosis marker Bax was slightly decreased, but that of Bcl2 did not change. The p21 is responsible for inhibiting cell growth and committing cells to apoptosis. 12 The p21 expression was significantly decreased after 48 hours of LBX treatment. Thus, LBX inactivates p21 signaling to prevent gastric cancer growth. The mammalian target of rapamycin (mTOR) is an unconventional protein kinase central to controlling cancer cell metabolism, growth, and proliferation. mTOR, a member of the phosphoinositide-3-kinase-related kinase (PIKK) family, plays central roles in cell growth and stress-related signaling. Phosphoinositide 3-kinase (PI3K) activates AKT via mitogenic stimuli, 13 which drives several cancer-related cellular responses, including increased cellular growth and proliferation, a shift to glycolytic metabolism, and increased cell migration. The PI3K and phospho-Akt levels were decreased after treatment with LBX for 48 hours. Indeed, the mTOR phosphorylation and p70S6K phosphorylation downstream of mTOR were concomitantly decreased during LBX treatment (Figure 6B). Ratios of phosphorylated AKT, mTOR, and S6K to inactive signaling proteins were decreased as the incubation time was made longer (Figure 6C).

Western blot analysis of apoptosis proteins and mTOR-related proteins in cells treated with LBX
Discussion
Probiotics have low or no pathogenic effects and are beneficial to the health of the host.
14
Our study showed that LBX reduced cell viability, most strongly in gastric cancer (KATO3) cells (Figure 1A). Using the MTT assay, we determined the IC50 of LBX to be 0.5 mg/mL, a concentration that had no toxic effects on normal (HDF) cells (Figure 1B). In particular, LBX changed the size and number of KATO3 cells and the induced nuclear condensation and DNA fragmentation in a dose-dependent and time-dependent manner (Figure 2A and B). According to FACS analysis, LBX transformed normal cells (untreated; 84.2% normal, 1.2% early apoptotic) into mostly apoptotic cells (treated; 75.9% normal, 15.1% late apoptotic at 48 hours and 2.3% normal, 85.6% late apoptotic at 72 hours; Figure 3). These results indicate that LBX inhibits gastric cancer by promoting apoptosis. KATO3 cells treated with LBX decreased pro-caspase-3 (expression 35 kDa) and increased active caspase-3 (17 kDa). Caspases shows the signal transduction by proteolytically cleaving downstream caspases and substrate proteins. 32 Thus, LBX promoted apoptosis by activating caspase-3 apoptosis (Figure 6A).
LBX decreased Bax expression but did not affect that of Bcl-2. Therefore, it is not clear how LBX controls the Bax/Bcl-2 ratio. Caspase-3 is critical for Fas-mediated cell death, and p21, located in the mitochondria, can inactivate this death. 33 LBX may induce extrinsic and intrinsic apoptosis by modulating the Bax/Bcl-2 ratio. Interestingly, LBX decreased the level of p21, leading to cell death (Figure 5B). Thus, LBX prevents cancer by promoting extrinsic apoptosis and regulating transcription through multiple related signal pathways.
The intracellular signaling pathways that regulate apoptosis include NF-κB and mitogen-activated protein kinases (MAPKs).
34
Because NF-κB and MAPK signaling regulate inflammation, cell proliferation, and apoptosis,
The mTOR pathway is integral to the promotion of cellular growth. This pathway involves 2 functional complexes, TORC1 and TORC2, defined by their association with raptor or rictor, respectively, and their inhibition by short-term rapamycin. mTOR signaling is complicated as it is regulated by cellular energy levels, mitogens, and nutrients.35-37 TSCs, especially TSC2, connect PI3K/Akt and mTOR signaling. Excess PI3K/AKT signaling, an mTOR activator, is a feature of most, if not all, cancers. 38 AKT is activated by phosphoinositide 3-kinase (PI3K) and causes several cancer-related responses, including increased cell growth and proliferation, a shift to glycolytic metabolism, and increased cell migration. The levels of PI3K and AKT were decreased after treatment with LBX for 48 hours. Indeed, p70S6K phosphorylation, controlled by mTOR, was decreased during LBX treatment (Figure 6B). LBX was shown to downregulate the PI3K, Akt, mTOR, and p70S6K signaling cascade by inhibiting NF-κB directly or indirectly. Thus, a small molecule that controls NF-κB and mTOR signaling may be a candidate therapy to prevent gastric cancer.
In summary, LBX induced gastric cancer apoptosis by activating caspase-3. While LBX did not directly induce apoptosis through the intrinsic pathway, it decreased the level of p21 attenuating proliferation of gastric cancer cells. Most notably, LBX reduced PI3K expression and mTOR signaling. Therefore, LBX may have multiple roles in the regulation of mTOR and ERK signaling and inactivation of NF-κB, processes that are associated with apoptosis in gastric cancer. These varied properties of LBX may be the result of small molecules present in
Footnotes
Authors’ Note
Authors Ik-Soon Jang and Jong-Soon Choi contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by a Korea Basic Science Institute NAP grant (T3178B) to J. S. Choi and by the Mid-career Researcher Program through an NRF grant funded by the MEST (No. No RO1-2008-000-20253-0).
