Abstract
Although localized colon cancer is often successfully treated with surgery, advanced disease requires aggressive systemic therapy that has lower effectiveness. Approximately 30% to 75% of patients with colon cancer use complementary and alternative medicine (CAM), but there is limited formal evidence of survival efficacy. In a consecutive case series with 10-year follow-up of all colon cancer patients (n = 193) presenting at a San Francisco Bay-Area center for Chinese medicine (Pine Street Clinic, San Anselmo, CA), the authors compared survival in patients choosing short-term treatment lasting the duration of chemotherapy/radiotherapy with those continuing long-term. To put these data into the context of treatment responses seen in conventional medical practice, they also compared survival with Pan-Asian medicine + vitamins (PAM+V) with that of concurrent external controls from Kaiser Permanente Northern California and California Cancer Registries. Kaplan-Meier, traditional Cox regression, and more modern methods were used for causal inference—namely, propensity score and marginal structural models (MSMs), which have not been used before in studies of cancer survival and Chinese herbal medicine. PAM+V combined with conventional therapy, compared with conventional therapy alone, reduced the risk of death in stage I by 95%, stage II by 64%, stage III by 29%, and stage IV by 75%. There was no significant difference between short-term and long-term PAM+V. Combining PAM+V with conventional therapy improved survival, compared with conventional therapy alone, suggesting that prospective trials combining PAM+V with conventional therapy are justified.
Keywords
Background
Colorectal cancer, the third most common cancer in both men and women, is the second most common cause of cancer death in the United States. In 2008, 148 810 new cases were diagnosed, accounting for 9% of cancer deaths in women and 8% in men. 1 The 5-year survival is 90% for localized disease, 68% if lymph nodes are involved, but only 10% if there is evidence of metastatic spread at the time of diagnosis. 2
Surgical resection is essential. Examining at least 12 adjacent draining lymph nodes in addition to the primary pathological specimen allows the most accurate staging and defines the need for adjunctive therapy. 3 Adjuvant radiation therapy is restricted to treatment of advanced tumors located in retroperitoneal portions of the colon and for neoadjuvant and adjuvant treatment of rectal carcinomas. 4 Although three quarters of patients present when all gross tumor can be surgically removed, there is still a high risk that patients will die of metastatic disease.
5-fluorouracil (5-FU) has been used since 1957 in adjuvant treatment and in metastatic disease. Augmenting 5-FU’s efficacy with leucovorin led to widespread use of the Mayo Clinic and Roswell Park regimens for adjuvant therapy in the 1990s.5,6 Despite a decade of advances in screening and treatment, there was no apparent improvement in survival in the United States between 1986 and 1997, 7 the period when patients for this 10-year follow-up study were initially treated. At that time, median survival in patients with metastatic disease following 5-FU/leucovorin combination chemotherapy was 11 months, compared with 5 months with best supportive care. 8
Delivering 5-FU by outpatient continuous infusion pumps and combining it with other agents led to the first improvements over standard delivery by bolus injection, yielding higher response rates and prolonged survival. The topoisomerase inhibitor, irinotecan, and the third-generation platinum agent, oxaliplatin, improved responses when added to 5-FU/leucovorin in the metastatic setting.9-12 Oxaliplatin and 5-FU/leucovorin in various FOLFOX regimens improved response rates and survival when compared with 5-FU/leucovorin alone. 13 For patients with metastatic disease, sequencing FOLFOX with the irinotecan-based FOLFIRI regimen now provides potent chemotherapeutic options and prolongs survival compared with 5-FU alone.14,15 The oral 5-FU prodrug, capecitabine, further expanded choice of chemotherapy for colorectal carcinoma, also in combination with either irinotecan or oxaliplatin. 16 Monoclonal antibodies, including bevacizumab, targeting circulating vascular endothelial growth factor, and cetuximab, directed against the epithelial growth factor receptor,17,18 are now providing benefits not available when patients for this 10-year follow-up study were initially treated.
Randomized trials from China suggest that Chinese herbal therapies combined with chemotherapy reduce nausea 19 and improve survival rates in colon cancer metastatic to the liver, 20 compared with chemotherapy alone.
Randomized Controlled Trials in CAM Therapies
Randomized controlled trials are considered the gold standard in clinical trials; however, they are seldom desirable to patients; less than 5% of cancer patients participate in trials. 21 The success of recruitment in CAM trials is a little-understood question. 22 Herbal medicines are used by 30% to 75% of colon cancer patients,23-25 and the ubiquitous availability of CAM therapies may substantially reduce the acceptability of enrolling in randomized CAM trials for cancer patients. Study designs exist that can be used as alternatives to randomized trials. 26
Biases such as selection bias and confounding can be reduced in observational studies by using the marginal structural models (MSMs)27-30 and propensity score31,32 methods, which make treatment groups comparable based on the probability of having been treated given individual characteristics and can thus be thought of as standardization tools. 33 In nonrandomized data, this method can provide results comparable to randomized trials.34-36 The MSM method has not been used in studies of cancer survival 37 or Chinese herbal medicine, and the propensity score has not been used in Chinese herbal medicine studies.
Objectives
We conducted this retrospective study to examine whether Pan-Asian medicine + vitamins (PAM+V) therapy can improve survival of patients with colon cancer. Our hypotheses were that (A) long-term use of PAM+V would lead to longer survival than short-term use lasting only for the duration of chemotherapy/radiotherapy; and (B) PAM+V combined with conventional therapy would improve survival, compared to conventional therapy alone. We followed the reporting guidelines “Strengthening the Reporting of Observational Studies in Epidemiology (STROBE).” 38
Methods
Patients
The participants (n = 193) in this cohort were a consecutive case series of all patients with colon cancer seen at a clinic of Chinese medicine (Pine Street Clinic, San Anselmo, CA) during 1987-1992, who were also being treated at local hospitals by medical, surgical, and/or radiation oncologists. We followed patients for 10 years or until death or loss to follow-up. After establishing the treatment protocols, we continued in-person and/or telephone follow-up for patients who maintained care at Pine Street Clinic and telephone follow-up for those continuing at other CAM centers. Telephone follow-up for patients following at other centers typically happened every few months and was intended to maintain contact and encourage continuation of therapy. Although we found that this encouragement was beneficial in helping sustain treatment adherence, we did not design the study specifically to study this effect. Those who neither continued at Pine Street nor went to other CAM centers, and therefore for which no follow-up was possible, were considered lost to follow-up.
Treatment Rationale
The protocol was designed based on Chinese herbal strategies that guide the body through alternating cycles of attack and rebuilding. The treatment calendar is divided into 3 parts chronotherapeutically timed to complement the front-line adjunctive chemotherapy strategy.
Part I is defined as day 1 through 3 of the chemotherapy cycle. The essential effort during these 2 days is to enhance circulation because effectiveness of chemotherapy drugs depends on circulation to carry them throughout the body to reach target cells. If there are areas of the body that, for whatever reason, have less-than-optimal circulation, then, these areas may be underserved by the chemotherapy. For example, areas of the body where there have been previous surgeries, scar tissue, or other injuries are all places where blood circulation may be inhibited. In addition, areas of the body that hold tension are also areas where circulation may be especially poor. The goal in part I, then, is to use various techniques such as imagery, visualization, exercise, and Qi-Gong to improve circulation. Part I is a time to be restful, low key, and avoid stress.
Part II is defined as days 4 through 7. At this time, the chemotherapy has been successful in killing numerous cells, both cancer cells and healthy cells. These dead cells then need to be removed; however, the amount of dead cells that accumulate can often exceed the body’s natural ability to purge them from the system, which contributes to lingering chemotherapy-related toxicities. The main goal of part II, therefore, is to assist the body in discharging and cleansing, so that these dead cells and remaining toxic chemotherapy metabolites will not be in the way during the next round of therapy. In the herbal and supplemental suggestions, dosages and timing shift to adjust to this new part as well. Cleansing can also be figurative as well: cleaning out closets, completing projects, and resolving personal and business issues and relationships.
Part III is defined as days 8 through 30, before the start of the next cycle of chemotherapy. During this time the blood counts will start to come up, the physiology will begin to normalize, side effects begin to lessen, and energy starts to return. The goal at this time is to strengthen and enhance the immune system. Everything a person does should begin to reflect this, and engaging in activities that help them feel good and genuinely happy should have high priority at this time. When chemotherapy was provided on a weekly schedule, we divided the protocol into 2 parts: part I being days 1 to 3 and part II being days 4 to 7.
The strategy for patients whose conventional therapy consisted of radiation was to include protocol items for minimizing radiation side effects, minimizing antioxidant therapies during the radiation phase, and resuming antioxidants after radiation. Acupuncture was made available on an as-needed basis, but the primary treatment emphasis was on the herbal/nutraceutical elements. The herbal medicine portion of the PAM+V protocol was administered in the traditional Chinese method of giving patients dry herbs to take home and decoct.
Diet
For all patients, diet was discussed, including general nutrition advice (a natural foods variation of standard cancer patient dietary advice), with recommendations for increased fiber consumption, consumption of organic natural foods when possible, and an emphasis on personal food preparation when possible.
Exercise
All patients received exercise recommendations, beginning with minimal walking, household and garden chores, and then, where possible, proceeding to moderate exercise, including yoga, jogging, healthclub activities, and formal classes in yoga, Tai Ji, or Qi-Gong.
Human Subjects Concerns
Prior to initiating this study, we received approval from institutional review boards (IRBs) at the University of California at Berkeley (protocol #2004-6-279), Kaiser Permanente Northern California (#CN-04LKush-01-H), State of California Health and Human Services Agency (#07-08-55), and Pine Street Foundation (Independent Review Consulting, Corte Madera, CA; #EXE04-009-01).
Data Sources
Data on PAM+V-treated patients were obtained by chart review from Pine Street Clinic (San Anselmo, CA). Survival time in the PAM+V cohort was obtained directly from patient records and/or confirmed by in-person or telephone follow-up with patients’ family members. Chinese herbal medicine and vitamins were provided concurrently with chemotherapy/radiation treatment (short-term use). After completion of chemotherapy/radiation, we monitored whether or not the patients continued herbal/vitamin therapy (long-term use). Patients who were lost to follow-up and for whom treatment adherence was thus unknown were analyzed as part of the long-term adherence group, following the intention-to-treat principle. 39 Additionally, short-term versus long-term adherence was noted for both patients who after initial consultation here at Pine Street elected to continue at our center or follow up with other CAM centers. Thus, each of these 2 variables was measured separately: location (Pine Street or CAM) and duration (short term or long term).
Data on external concurrent controls diagnosed during the same time span as the Pine Street patients and matched on cancer stage were obtained by database extraction from the California Cancer Registry (CCR; www.ccrcal.org) and Kaiser Permanente Northern California (KPNC) Division of Research Cancer Registry (www.dor.kaiser.org). We selected cases using histology codes for adenocarcinoma (8010, 8020-8022, 8140-8145, 8210-8211, 8220-8221, 8230-8231, 8260-8263), squamous cell carcinoma (8070-8078), and mucinous carcinoma (8470, 8480, 8481). 40 Patients with squamous cell or mucinous cell colorectal cancers have worse prognosis than those with adenocarcinoma. 41
Stage
For Pine Street Clinic patients, staging was identified by review of medical records related to patients’ cancer diagnosis: biopsy reports, scan reports, and physicians’ dictation notes. All patients were biopsy-proven cases. For the California Cancer Registry (CCR) controls, American Joint Committee on Cancer (AJCC) staging was used. 42 For the Kaiser controls, AJCC stage was not available for the study recruitment years, so we reconstructed AJCC stage from tumor extension, node involvement, and metastasis. 43 For a detailed case selection inventory, see Table 1.
Case Selection and Exclusion Algorithm

Abbreviations: PAM+V, Pan-Asian medicine + vitamins; CCR, California Cancer Registry; KPNC, Kaiser Permanente Northern California; AJCC, American Joint Committee on Cancer; SEER, Surveillance, Epidemiology and End Results; CAM, Complementary and Alternative Medicine.
Data Analysis
The primary end point was all-cause mortality. Our primary treatment variable of interest was long-term versus short-term herbal/vitamin therapy (yes/no); baseline covariates were stage (I, II, III, or IV), age at diagnosis, sex, histology (adenocarcinoma, squamous, or mucinous), and treatment with surgery, radiation, or chemotherapy (yes/no).
We analyzed data in 2 phases: (1) within Pine Street Clinic, comparing long-term with short-term PAM+V, and (2) comparing PAM+V users with external concurrent controls. We used the Stata statistical package (version 9.2; StataCorp, College Station, TX).
We used Cox proportional hazards regression 44 stratified by stage and adjusted by all covariates for traditional survival analysis. Time at risk was defined by month/year of diagnosis and death. For causal inference, we used Cox proportional hazards weighted by the inverse probability of each patient’s having received treatment (stabilized inverse probability of treatment weighting [IPTW]) in a MSM. The IPTW balances exposure groups with regard to their probability of having received treatment27,28,33 using logistic regression with the same covariates as used in our standard Cox regression model. We used the bootstrap 45 with 1000 repetitions for estimation of the confidence intervals about the hazard ratio (HR). For this analysis, we constructed our own program using the Stata statistical programming language.
We also investigated causal inference with the propensity score. This method begins by using the multivariate logistic regression model to estimate the probability of receiving treatment using a person’s individual disease and treatment characteristics31,32,46,47 using the same set of covariates as above. We first estimated the propensity score and divided it into 5 or more levels or strata (a method shown to remove up to 90% of bias if adequate covariate information is available) 48 and then used the propensity score as a variable in Cox regression. We used the pscore program in the Stata statistical software. 49
Results
Number of Patients
In the Pine Street cohort, we had 193 colon cancer patients (Table 1). Most patients in the Pine Street Clinic group were presenting for first-line chemotherapy. Lag time between diagnosis and initiation of PAM+V treatment averaged between 30 and 34 days. This is comparable with what is known about delays in delivery of care to those with lung cancer in conventional oncology practice: delays attributable to the patient average 18 days, and delays attributable to the health system average 62 days. 41
From the CCR, we obtained data for 11 678 patients suitable for analysis and from Kaiser Permanente Northern California, data for 1987 patients. Table 2 reports patient and treatment characteristics. Table 3 reports PAM+V protocol components and timing. Table 4 reports herbal medicines used.
Patient Characteristics by Group

Abbreviations: PAM+V, Pan-Asian medicine + vitamins; CCR, California Cancer Registry; KPNC, Kaiser Permanente Northern California.
PAM+V Protocol Timing Concurrent With Chemotherapy, 21-Day Cycle a

Abbreviations: PAM+V, Pan-Asian medicine + vitamins.
Day 1 is defined as the day of chemotherapy infusion. Protocols for patients were usually updated at quarterly (seasonal) consultation visits. Each of the herbs and supplements listed above were not all used simultaneously during the course of therapy. Items were identified if they were used for more than 3 seasonal protocol updates during the course of a patient’s treatment at Pine Street. After completion of chemotherapy, long-term treatment continues with Part III.
Chinese Herbal Medicine Formulas Used
Comparing Long-Term Follow-up With PAM+V to Short-Term Follow-up
In Kaplan-Meier analysis, in stage I and II, survival was comparable between short-term and long-term treatment groups (Figure 1). There were no deaths in stage I short-term patients (the denominator in this analysis), so that estimation of a HR was not possible. A modest but not statistically significant advantage in favor of long-term maintenance of PAM+V treatment was found by multivariate causal analysis with the propensity score in stage II (HR = 0.71; 95% CI = 0.06, 8.79) and by MSMs in stage IV (HR = 0.85; 95% CI = 0.10, 7.24). Some of our analyses, by contrast, found a nonsignificant trend toward superiority of short-term PAM+V treatment (Table 5).

Survival in colon cancer, short-term versus long-term PAM+V and external controls
Colon Cancer Survival a
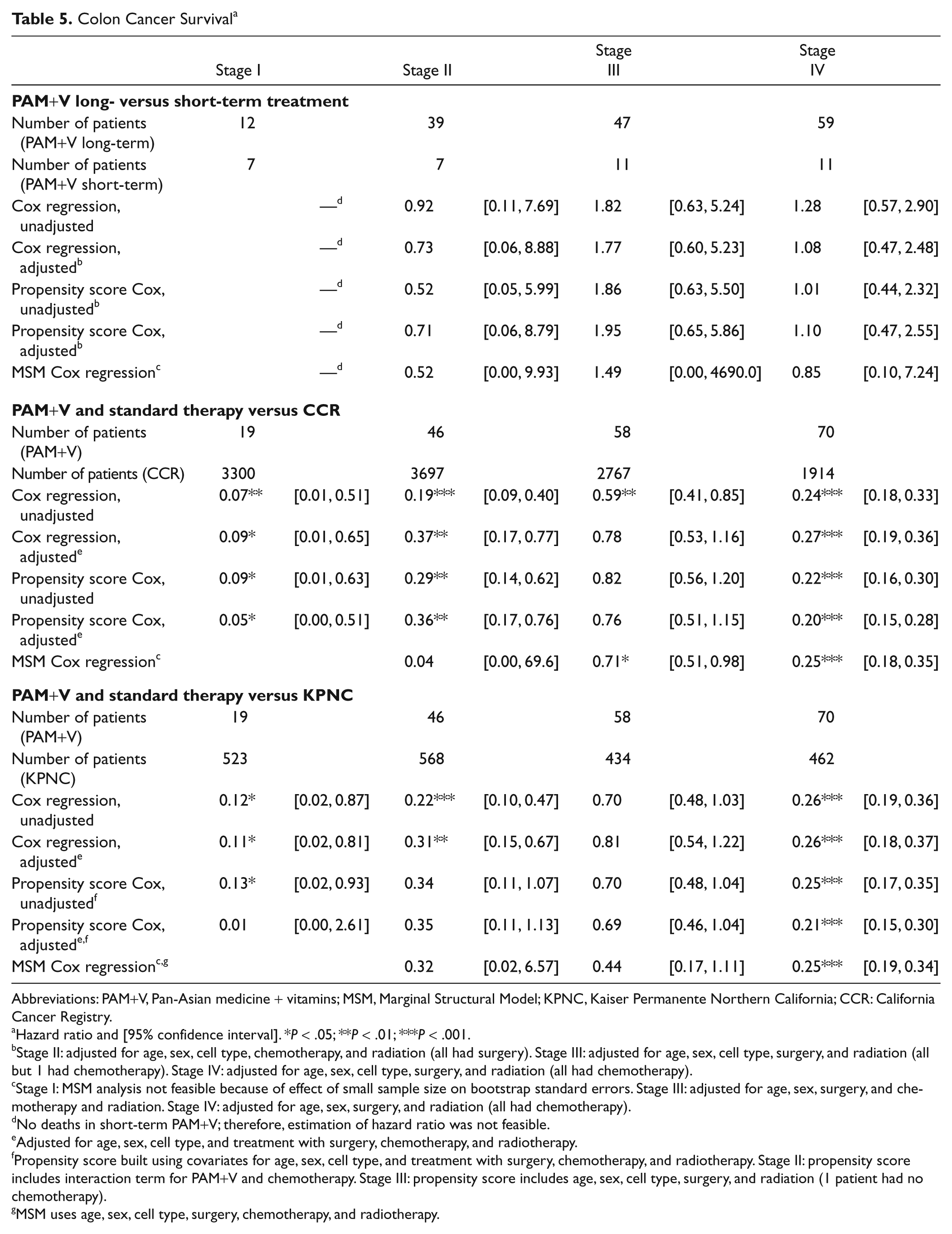
Abbreviations: PAM+V, Pan-Asian medicine + vitamins; MSM, Marginal Structural Model; KPNC, Kaiser Permanente Northern California; CCR: California Cancer Registry.
Hazard ratio and [95% confidence interval]. *P < .05; **P < .01; ***P < .001.
Stage II: adjusted for age, sex, cell type, chemotherapy, and radiation (all had surgery). Stage III: adjusted for age, sex, cell type, surgery, and radiation (all but 1 had chemotherapy). Stage IV: adjusted for age, sex, cell type, surgery, and radiation (all had chemotherapy).
Stage I: MSM analysis not feasible because of effect of small sample size on bootstrap standard errors. Stage III: adjusted for age, sex, surgery, and chemotherapy and radiation. Stage IV: adjusted for age, sex, surgery, and radiation (all had chemotherapy).
No deaths in short-term PAM+V; therefore, estimation of hazard ratio was not feasible.
Adjusted for age, sex, cell type, and treatment with surgery, chemotherapy, and radiotherapy.
Propensity score built using covariates for age, sex, cell type, and treatment with surgery, chemotherapy, and radiotherapy. Stage II: propensity score includes interaction term for PAM+V and chemotherapy. Stage III: propensity score includes age, sex, cell type, surgery, and radiation (1 patient had no chemotherapy).
MSM uses age, sex, cell type, surgery, chemotherapy, and radiotherapy.
Comparing PAM+V-Treated Patients With Concurrent External Controls
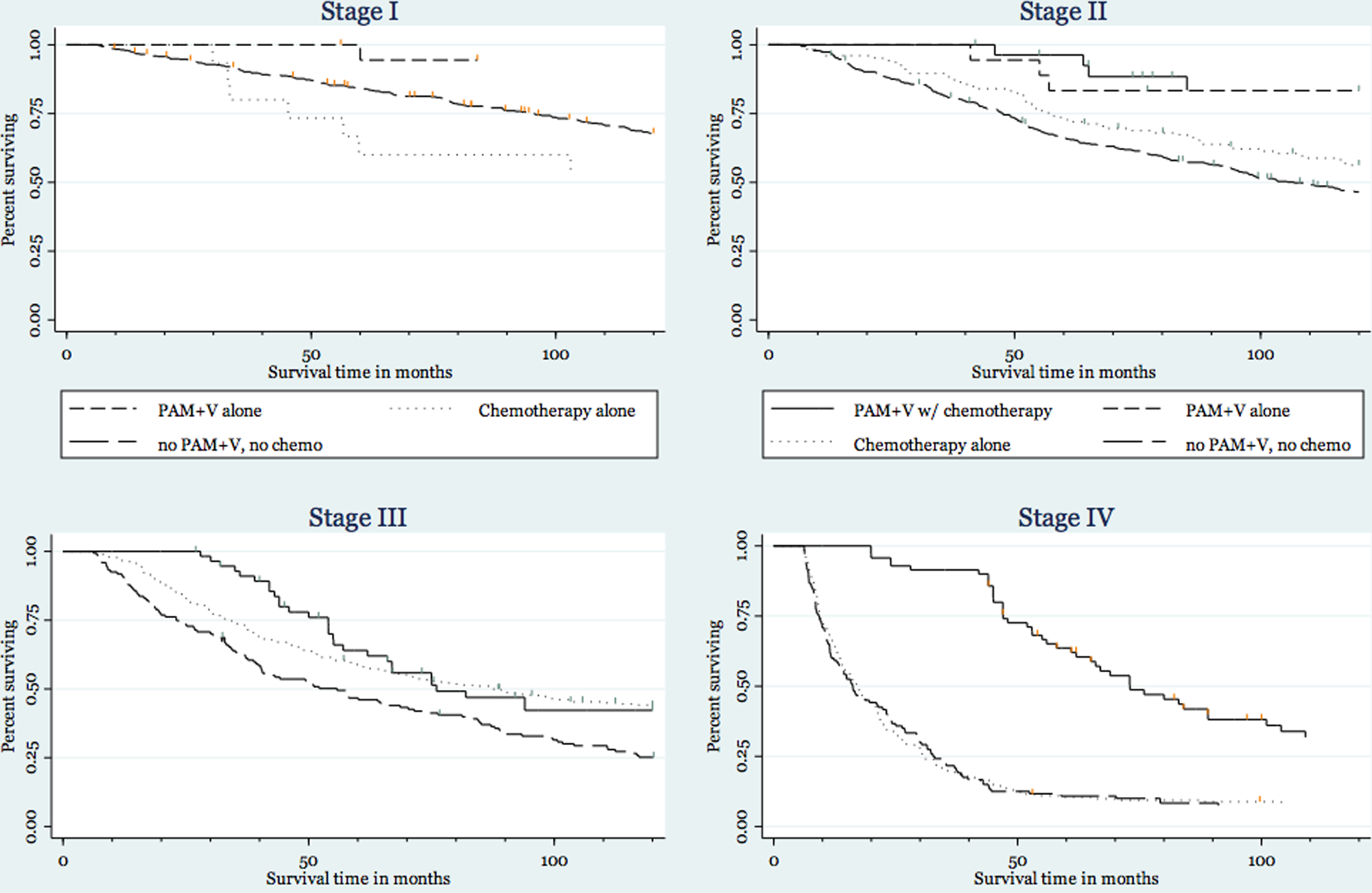
In Kaplan-Meier plots, we found that in patients with all stages of colon cancer, treatment with both short-term and long-term PAM+V resulted in longer survival than was seen in concurrent external controls from both Kaiser Permanente and CCR (Figure 1).
Using concurrent external controls from the CCR, we found significantly better survival with PAM+V combined with conventional therapy compared with conventional therapy alone: using traditional adjusted Cox analysis, in stage I (HR = 0.09; 95% CI = 0.01, 0.65), stage II (HR = 0.37; 95% CI = 0.17, 0.77), and stage IV (HR = 0.27; 95% CI = 0.19, 0.36); and using MSMs, in stage III (HR = 0.71; 95% CI = 0.51, 0.98) and in stage IV (HR = 0.25; 95% CI = 0.18, 0.35). There was a strong but nonsignificant trend toward better survival (Table 5) in stage II (HR = 0.04; 95% CI = 0.00, 69.6) and, using propensity scores, in stage I (HR = 0.05; 95% CI = 0.00, 0.51), stage II (HR = 0.36; 95% CI = 0.17, 0.76), and stage IV (HR = 0.20; 95% CI = 0.15, 0.28).
Using concurrent external controls from Kaiser Permanente, we found significantly better survival with PAM+V combined with conventional therapy when compared with conventional therapy alone (Table 5): using traditional adjusted Cox analysis, we found better survival in stage I (HR = 0.11; 95% CI = 0.02, 0.81), stage II (HR = 0.31; 95% CI = 0.15, 0.67), and stage IV (HR = 0.26; 95% = CI 0.18, 0.37) and a nonsignificant trend toward better survival in stage III (HR = 0.81; 95% CI = 0.54, 1.22); using MSMs analysis, there was significantly longer survival in stage IV (HR = 0.25; 95% CI = 0.19, 0.34) and a nonsignificant trend toward better survival in stage II (HR = 0.32; 95% CI = 0.02, 6.57) and stage III (HR = 0.44; 95% CI = 0.17, 1.11). Also, using adjusted propensity score analysis, there was significantly longer survival for PAM+V-treated patients, compared with concurrent external controls in stage IV (HR = 0.25; 95% CI = 0.15, 0.30).
To confirm the validity of our external controls, we compared survival rates among both groups of external controls identified in this study with the rates reported by other authors also using Surveillance Epidemiology and End Results (SEER) Cancer Registry data. 50 In our study, we estimated (Table 6) that 30-month survival in stage I controls from CCR was 86% and from Kaiser Permanente (KPNC) was 90% (compared with other authors who estimated 96%). 50 The 30-month survival in stage II controls from CCR was 78% and from KPNC was 83% (compared with 89%). 50 The 30-month survival in stage III controls from CCR data was 64% and from KPNC was 73% (compared with 73%). 50 The 30-month survival in stage IV controls from CCR was 16% and from KPNC was 20% (compared with 17%). 50
Absolute Survival Rates for Examining the Validity of External Controls

Abbreviations: CCR, survival estimates from our analysis of California Cancer Registry data; KPNC, survival estimates from our analysis of Kaiser Permanente data.
Crude Survival Rates: Stage I
Survival at 1 year was 100% in both the long-term and short-term PAM+V groups, 95% in the Kaiser controls and 93% in CCR controls. Survival at 2 years was 100% in both the long-term and short-term PAM+V groups, 92% in Kaiser controls, and 88% in CCR controls. Survival at 5 years was 100% in both the long-term and short-term PAM+V groups, 81% in Kaiser controls, and 74% in CCR controls (Table 7).
1-, 2-, and 5-Year Survival Rates

Crude Survival Rates: Stage II
Survival at 1 year was 100% in both the long-term and short-term PAM+V groups, 93% in Kaiser controls, and 89% in CCR controls. Survival at 2 years was 100% in both the long-term and short-term PAM+V groups, 86% in Kaiser controls, and 81% in CCR controls. Survival at 5 years was 92% in the long-term PAM+V group, 86% in the short-term PAM+V group, 65% in Kaiser controls, and 63% in CCR controls (Table 7).
Crude Survival Rates: Stage III
Survival at 1 year was 100% in both the long-term and short-term PAM+V groups, 91% in Kaiser controls, and 83% in CCR controls. Survival at 2 years was 100% in both the long-term and short-term PAM+V groups, 78% in Kaiser controls, and 69% in CCR controls. Survival at 5 years was 61% in the long-term PAM+V group, 80% in the short-term PAM+V group, 52% in Kaiser controls, and 48% in CCR controls (Table 7).
Crude Survival Rates: Stage IV
Survival at 1 year was 100% in both the long-term and short-term PAM+V groups, 44% in Kaiser controls, and 40% in CCR controls. Survival at 2 years was 93% in the long-term PAM+V group, 100% in the short-term PAM+V group, 25% in Kaiser controls, and 20% in CCR controls. Survival at 5 years was 60% in the long-term PAM+V group, 82% in the short-term PAM+V group, 7% in Kaiser controls, and 8% in CCR controls (Table 7).
Examining Treatment Combinations
Because treatments with conventional therapies were important variables in our analysis, we also sought to report survival differences between various combinations of these therapies. Figure 2 compares survival curves between patients who received PAM+V in combination with surgery with those receiving PAM+V alone, surgery alone, or neither PAM+V nor surgery. These survival curves are stratified by cancer stage and use the CCR database records as external concurrent controls. Figure 3 similarly reports all 4 possible treatment combinations of PAM+V and chemotherapy, Figure 4 for PAM+V and radiotherapy, and Figure 5 for all possible combinations of PAM+V and any standard therapy. In virtually all these survival curve plots, we found a dose–response effect, in which the most substantial survival benefit was realized by patients receiving the full treatment combination; the least favorable survival response was seen in patients receiving neither treatment, and PAM+V alone or standard therapy alone were typically in the middle.

Colon cancer survival, showing all treatment combinations of PAM+V and surgery, using California Cancer Registry patients as external controls
Colon cancer survival, showing all treatment combinations of PAM+V and chemotherapy, using California Cancer Registry patients as external controls

Colon cancer survival, showing all treatment combinations of PAM+V and radiotherapy, using California Cancer Registry patients as external controls

Colon cancer survival, showing all treatment combinations of PAM+V and any conventional therapy, using California Cancer Registry patients as external controls
Comparing PAM+V-Treated Patients Followed at Pine Street Clinic With Those Followed at Other CAM Centers
We were also interested in determining whether there was a center effect—survival differences based on location of follow-up. In the Kaplan-Meier life table analysis, we found no apparent survival difference between patients who continued at Pine Street Clinic for long-term PAM+V and those continuing at other CAM centers (Figure 6).

Colon cancer survival, comparing PAM+V patients initially treated at PS with those followed up at other CAM centers
Examining CAM Use in External Controls
We were fortunate to identify some records within the CCR in which clinicians had documented patients’ use of CAM therapies (coded in the parlance of the time as “unproven therapies”). We therefore compared the survival of the patients who used both long-term and short-term PAM+V with that of CAM users and nonusers from within the CCR database (Figure 7). In Kaplan-Meier analysis, we found that in stage IV colon cancer, there was substantially increased survival with both long-term and short-term practitioner-guided PAM+V therapy, compared with CAM use in the general population. Furthermore, in those stage-IV CCR patients, there was no apparent survival difference between CAM users and nonusers. In patients with stage III colon cancer, there was similarity between the survival of PAM+V-treated patients and CAM users from the general population, particularly after about 5 years’ time. These results for CAM use and survival in the CCR external concurrent controls should be interpreted with caution because of likely reporting bias and/or measurement error among those controls.

Colon cancer survival, comparing patients treated with long-term and short-term PAM+V with CAM users and nonusers from within the California Cancer Registry database
Discussion
Principal Findings
In this retrospective cohort survival analysis, we found significant survival benefits to combining PAM+V with conventional therapy compared with conventional therapy alone in colon cancer patients in stages I, II, and IV. There was a trend toward superior survival in stage III, probably because of small sample size in the PAM+V group. Long-term PAM+V treatment showed no survival advantage over short-term therapy. MSMs and propensity score analyses, both showed results similar to multivariate Cox regression analysis.
Strengths of the Study
Our article is the first where modern methods—the propensity score and MSMs—are used for causal inference in a survival analysis of Chinese herbal medicine in colon cancer. Our use of modern methods for causal inference can balance such differences between treatment groups by making these groups comparable based on their individual and treatment history characteristics. When adequate measurement of covariates is present, they can help control for up to 90% of differences between treatment groups. Although articles on the MSMs have stated the assumption of no unobserved confounders, this assumption is unverifiable in practice.51,52
In our study, we used specific herbal and vitamin products commonly available in commercial markets. Our purpose was to maximize the accessibility of our treatment approach for other clinicians and researchers.
Limitations of the Study
These observational data are prone to selection bias because patients choose the treatment they receive, in contrast to a randomized trial where choice is assigned. We did not have data that would allow us to control for possible confounding by socioeconomic status (SES). SES is associated with increased use of CAM, 53 better access to conventional cancer therapy, 54 and increased cancer survival. We also did not have data on race within the PAM+V-treated cohort. Colon cancer survival has been shown to be lower in blacks than whites; however, recent work has shown that this may be largely a function of SES, rather than race specifically.55,56
It is also possible that the efforts required of patients to follow through with the highly structured PAM+V protocol such as ours may in some unknown way have contributed to these survival differences. The relative effects on patient survival of granules/tinctures versus dry herb decoctions or of highly structured versus less-structured treatment protocols would be an informative end point to design into a prospective follow-up study specifically seeking to answer such a question.
In the CCR and Kaiser Permanente Northern California data sets, we did not have available data on smoking, an important risk factor for colon cancer which worsens survival57-59 and contributes to more aggressive cancer cell growth. 60 Although cancer registry data have limitations, the data that are available from these 2 registries are generally considered to be of high quality.
We had no data on CAM use by external controls in the Kaiser Permanente database, so we were not able to account for this confounding variable in our analysis, which compared PAM+V-treated patients with external concurrent controls. However, even if it were as high as reported in the literature, this would have biased our results toward the null, and the true survival benefit of PAM+V would be even stronger than what we estimated.
There may also have been other factors that could explain in part the survival differences of short-term over long-term PAM+V; for example, patients choosing short-term PAM+V therapy might have “felt healthier” and thus not maintained treatment. Our results showing longer survival with short-term use of PAM+V than with long-term use were contrary to our expectations. This could suggest that a sufficient survival benefit could be obtained by short-term use of PAM+V lasting only the duration of conventional therapies. However, there were substantially fewer short-term PAM+V patients in stages II, III, and IV—between 7 and 11 in each stage; it is possible that these few individuals in each stage had other unknown characteristics or factors that could explain their longer survival. This observation could be further tested and validated in follow-up prospective trials specifically designed to compare short-term versus long-term use of PAM+V.
Similarly, patients choosing PAM+V may have been healthier overall than the external controls and thus able to maintain treatment. Additionally, patients choosing PAM+V may have already been following better diets, which is known to improve colon cancer survival. 61 More patients in the Pine Street Clinic groups received chemotherapy, although we controlled for this factor by including it as a variable in the multivariate analysis. Other research on the use of CAM therapies by cancer patients suggests that sicker patients are more likely to choose PAM+V therapy. 24 To put this statement in context, it is also important to contrast it with the finding that in conventional cancer therapy, sicker patients are less likely to receive chemotherapy. 62 To strengthen our inference, we also conducted a conservative analysis where we excluded all cases in the KPNC external controls who survived less than 6 months and found only a small difference in the HR of a few percentage points.
Patients’ use of imagery, visualization, exercise, and Qi-Gong was an integral part of the treatment design, and they were encouraged to learn and practice these approaches as much as possible; these are the kinds of approaches studied in Whole Systems research, where the emphasis is on the net effect of the holistic protocol design. However, our study would have been improved if we had included monitoring for compliance of each of the treatment protocol parts. Teasing apart the relative efficacy of each individual PAM+V component could be pursued in a future prospective study designed to answer this question. Holistic protocols like PAM+V can also be studied using an approach called Pragmatic Trials, which evaluate a therapy as it is used in normal practice (as compared with the more fixed and constrained approach used in most clinical trials). 63
The existing and newly developed conventional therapies for colon cancer in use during the time span of data collection for this study are known to have significant toxicities.64-66 Enhancing patient compliance with and tolerance of conventional therapies is an often-reported goal of many CAM treatment programs. We did not include that outcome as a part of the current study; this question would be best studied in a prospective design also gathering data on quality of life.
Causal Inference Methods Used in Our Study
This study uses 2 newer analysis methods for what is called causal inference: the propensity score and the MSM. Causal inference attempts to look more closely into the data being analyzed to uncover causal relationships. This is not always possible with traditional statistical methods, which can usually conclude only that the observed association between 2 variables is not a result of chance and cannot identify which variable causes the other.
Both the propensity score and MSM methods begin by using multivariable analysis to identify for each individual in the study, based on their personal and treatment history that that individual chose one treatment or another. The propensity score and MSM estimate that score with similar but slightly different statistical approaches. The propensity score is used as a variable in the Cox regression. The MSM is similarly used to weight the Cox regression.
In practice, both methods of causal inference will balance the treatment groups, so that patients in each group are comparable with respect to confounding variables. The primary practical difference between the MSM and propensity score methods is that the MSM can adequately handle the effects of treatment variables that vary over time, based on the outcomes of treatment. In the case of our study, however, this difference is not relevant because our study was of the point treatment design, where variables are measured only at baseline. However, the MSM approach is sometimes unstable when the sample size is small, leading to large variance in the statistical estimator.
Possible Mechanisms
Several plausible mechanisms for these findings may be considered, based on what is known about the specific components of this combination treatment approach with respect to various effects.
General cancer cell cycle effects
Propolis is an antioxidant-rich resin gathered by honeybees from the sap of trees and has been shown to induce colon cancer cell apoptosis, inhibit colon cancer cell growth,67,68 induce G(0)/G(1) arrest, 69 and inhibit angiogenesis, tumor invasion and metastasis. 70 Folic acid and its analogues may prevent and inhibit the development of colorectal tumors.71,72 N-acetyl cysteine reduces the proliferative index in patients with prior colonic polyposis. 73 Co-enzyme Q-10 may prevent premalignant lesions in the colon. 74 Curcumin appears to be synergistic with FU in inhibiting colon cancer cell growth 75 and may enhance 5-FU and oxaliplatin by modulating epithelial growth factor receptor, 76 induce apoptosis of colon cancer cells, 77 and be chemopreventive against colon cancer in humans. 78 Fish oil reduces proliferation in upper-crypt cells of patients with a history of colonic polyps, 79 inhibits colon tumors, 80 in combination with fiber reduces aberrant crypt foci formation, 81 and has chemopreventive effects in colon cancer. 82 Whey suppresses angiogenesis and tumor development and induces apoptosis. 83 Shark cartilage is antiangiogenic, 84 although 1 human study found no benefits. 85 Ganoderma stimulates colon cancer cell apoptosis 86 and prevents colon cancer cell development.87-89 Coix inhibits colon cancer cell growth. 90
Chemotherapy-related effects
Folic acid and its analogues are used in combination chemotherapy. 91 Green tea may enhance antitumor efficacy of anthracyclines, cisplatin, and irinotecan. 92 Melatonin enhances the efficacy of cisplatin, anthracyclines, and 5-FU. 93
Reduction of chemotherapy toxicity and side effects
The green tea plant has been shown to have neuroprotective 94 and cognition-enhancing properties. 95 N-acetyl cysteine may be liver protective,96-98 reducing chemotherapy toxicity.99-101 Co-enzyme Q-10 may prevent FU-related cardiotoxicity 102 and hepatotoxicity. 103 Lactobacillus reduces the frequency of diarrhea and abdominal discomfort from 5-FU-based chemotherapy. 104
Reduction of radiation toxicity and side effects
The Chinese herbal medicines Radix angelicae and Radix paeoniae have been shown to protect intestinal cells from irradiation-induced damage. 105 Components found within ginseng root protects against DNA damage from radiation exposure 106 and reduces radiation-induced enteritis. 107 Radix atractylodes and Poria cocos reduce radiation injury to healthy cells. 108 In a published case study, an herbal formula containing ginseng was used to successfully resolve radiation-induced enteritis. 107 Panax ginseng protects against radiation-induced lethality, lipid peroxidation, and DNA damage. 106 Melatonin reduces radiation-induced oxidative organ damage in rats by increasing malondialdehyde levels and myeloperoxidase activity and increasing glutathione levels. 109
More general systemic effects such as immune system modulation
Royal jelly is the nutrient-rich nectar produced by worker honeybees raising the queen bee and may be immunomodulatory and myeloprotective 110 and may increase cellular differentiation. 111 Thymic protein induces T-cell maturation. 112
Unanswered Questions and Future Research
An important unanswered question is patient or treatment factors that might explain the modestly better survival with short-term rather than long-term PAM+V treatment in stages III and IV colon cancer. Our observational results showing improved survival with PAM+V treatment combined with conventional therapies suggest that prospective intervention trials are warranted.
Footnotes
Acknowledgements
To my instructors for providing guidance, to Dr Ly Suhuai and Michael Broffman for inspiration, to Louise Estupinian for assistance in records acquisition, to the kind members of Statalist for their advice, and to my family for their loving support [MM].
Author contributions are as follows: Conception and design; M. F. McCulloch, M. D. Broffman, J. M. Colford Jr. Administrative support; M. D. Broffman, L. Kushi, J. Gao, J. M. Colford Jr. Provision of study materials or patients; M. F. McCulloch, M. D. Broffman, L. Kushi. Collection and assembly of data; ; M. F. McCulloch, M. D. Broffman, J. Gao. Data analysis and interpretation; M. F. McCulloch, M. van der Laan, A. Hubbard, D. Abrams, J. M. Colford Jr. Manuscript writing; M. F. McCulloch, D. Abrams, J. M. Colford Jr.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Pine Street Foundation, a University of California at Berkeley Epidemiology Block Grant Award, and a University of California at Berkeley Research Fellowship.
