Abstract
Complementary and alternative medicines are used by up to 48% of lung cancer patients but have seen little formal assessment of survival efficacy. In this 10-year retrospective survival study, the authors investigated Pan-Asian medicine + vitamins (PAM+V) therapy in a consecutive case series of all non-small-cell lung cancer patients (n = 239) presenting at a San Francisco Bay Area Chinese medicine center (Pine Street Clinic). They compared short-term treatment lasting the duration of chemotherapy/radiotherapy with long-term therapy continuing beyond conventional therapy. They also compared PAM+V plus conventional therapy with conventional therapy alone, using concurrent controls from the Kaiser Permanente Northern California and California Cancer Registries. They adjusted for confounding with Kaplan-Meier, Cox regression, and newer methods – propensity score and marginal structural models (MSMs), which when analyzing data from observational studies or clinical practice records can provide results comparable with randomized trials. Long-term use of PAM+V beyond completion of chemotherapy reduced stage IIIB deaths by 83% and stage IV by 72% compared with short-term use only for the duration of chemotherapy. Long-term PAM+V combined with conventional therapy reduced stage IIIA deaths by 46%, stage IIIB by 62%, and stage IV by 69% compared with conventional therapy alone. Survival rates for stage IV patients treated with PAM+V were 82% at 1 year, 68% at 2 years, and 14% at 5 years. PAM+V combined with conventional therapy improved survival in stages IIIA, IIIB, and IV, compared with conventional therapy alone. Prospective trials using PAM+V with conventional therapy for lung cancer patients are justified.
Keywords
Background
Lung cancer is the leading cause of cancer death in the United States, with 75% of cases being non-small-cell lung cancer.1,2 In 1988, when recruitment for the cohort described in the current study began, median survival with inoperable non-small-cell lung cancer was 6 months following etoposide/cisplatin chemotherapy and 8 months with added β- and γ-interferons. 3 Systemic therapies for advanced non-small-cell lung cancer have not substantially improved survival. Adjuvant chemotherapy reduces risk of death at 2 years by only 13% compared with surgery alone. 4 Chemoradiotherapy reduces the risk by 14% 5 ; however, adjuvant radiotherapy alone increases it by 21%.6,7 Platinum-based chemotherapy increased 12-month survival by 5% and tumor response by 62% but with greater toxicity. 8 In meta-analyses of studies, addition of platinum-based drugs to older chemotherapy regimens improved 12-month survival by only 5% (34% vs 29%; odds ratio [OR] = 1.21, 95% confidence interval [CI] = 1.09, 1.35), 8 and in individual patient data meta-analyses, 12-month survival after chemotherapy is only 29%. 9 Gefitinib did not improve survival compared with carboplatin/paclitaxel chemotherapy alone, 10 whereas erlotinib significantly improved survival. 11 Improvements in treatment are still needed.
Complementary and alternative medicine (CAM) is a broad field, including physical medicine approaches, such as acupuncture and massage, and pharmacological approaches, such as vitamins and herbal medicine. Herbal medicine is used by approximately 48% of lung cancer patients. 12 Meta-analysis of randomized trials from China suggests that herbal therapies combined with platinum-based chemotherapy increases survival in non-small-cell lung cancer by up to 42%, compared with chemotherapy alone. 13 However, evidence for survival efficacy in the Western medical literature is sparse. In a retrospective Mayo Clinic cohort study, vitamin use in non-small-cell lung cancer patients was associated with a lower risk of death (hazard ratio [HR] = 0.74; 95% CI = 0.60, 0.91), but that study relied on self-reported and proxy data on vitamins use, did not include herbal medicine, and did not examine survival within disease stage, the most important prognostic variable in non-small-cell lung cancer. 14
Randomized Controlled Trials in CAM Therapies
Although randomized controlled trials are considered the gold standard, they are sometimes unfeasible for researchers and/or undesirable to patients. Less than 5% of cancer patients participate in trials, 15 and little is known about recruitment success in CAM trials. 16 For patients, the common availability of CAM therapies may reduce the acceptability of entering randomized CAM trials. Alternatives to randomized trials exist. 17
Marginal structural model (MSM) methods for causal inference provide near-randomized comparability between treatment groups in observational studies.18,19 They reduce bias by adjusting for confounding and allow for identification of true causal effects not found through traditional association models such as Cox regression.20-23 They are a standardization tool, making treatment groups comparable based on the probability of having been treated, given individual characteristics, such as age, gender, and treatment exposure. 24 MSMs have not been applied in studies of cancer survival in general 25 or Chinese herbal medicine in particular.
The propensity score method for causal inference is another way to create near-randomized comparability between treatment groups in observational studies based on the multivariate probability of receiving treatment.26,27 It has been used in cancer survival but not in Chinese herbal medicine studies.
Objectives
We conducted this retrospective study to investigate whether Chinese herbal medicine and vitamins improve lung cancer survival and to provide a design foundation and sample size estimates for prospective trials. Our hypotheses were that (1) patients choosing long-term Pan-Asian medicine + vitamins (PAM+V) continuing beyond conclusion of standard therapy would have longer survival than patients using short-term PAM+V lasting only the duration of standard therapy and (2) PAM+V combined with conventional therapy would yield longer patient survival compared with conventional therapy alone.
Methods
Patients
Participants (n = 239) constitute in full a consecutive case series of all patients with non-small-cell lung cancer seen at a Chinese medicine center (Pine Street Clinic, San Anselmo, CA) during 1988-1993 who were also being treated at local oncology centers with surgery, chemotherapy, and/or radiotherapy. In most cases, patients were seen in conjunction with front-line chemotherapy. Patients were diagnosed by physical examination, bronchoscopy, and imaging tests such as X ray and CT scan. They were followed for 10 years or until death or loss to follow-up. We continued in-person visits and/or telephone follow-up for patients continuing at Pine Street Clinic and telephone follow-up for those continuing at other CAM centers. Telephone follow-up for patients following at other centers typically happened every few months and was intended to maintain contact and encourage continuation of therapy. Although we found that this encouragement was beneficial in helping sustain treatment adherence, we did not design the study specifically to study this effect. Those who neither continued at Pine Street nor went to other CAM centers, and therefore for whom no follow-up was possible, were considered lost to follow-up.
Human Subjects Concerns
Prior to initiating this study, we received approval from institutional review boards at the University of California at Berkeley (protocol #2004-6-279), Kaiser Permanente Northern California (protocol #N-04LKush-01-H), the State of California Health and Human Services Agency (protocol #07-08-55), and the Pine Street Foundation (Independent Review Consulting, Corte Madera, California; protocol #EXE04-009-01).
Treatment Rationale
Our treatment design was informed by Chinese herbal strategies which alternate therapeutic phases of attack and rebuilding. Treatment timing is divided into 3 parts, which are chronotherapeutically aimed at complementing the 21-day front-line adjunctive chemotherapy strategy.
Day 1 of the chemotherapy cycle begins part I, which continues through day 4. During these 2 days, the focus is on enhancing circulation, thus enhancing the ability of chemotherapy drugs to circulate as widely as possible through the body to reach tumor cells. Areas of the body that may have suboptimal circulation, for example, where there is tension or a history of surgery, scar tissue, or injuries, are locations where circulation may be inhibited. We use various techniques such as imagery, visualization, exercise, and Qi-Gong to improve circulation. It is also useful during part I to get extra rest and avoid stress.
Days 5 through 10 are called part II, where chemotherapy has killed both cancer cells and healthy cells, which need to be removed. The goal here is to assist the body in reducing lingering chemotherapy-related toxicities by discharging and cleansing, so that these dead cells and remaining toxic chemotherapy metabolites will not be in the way during the next round of therapy. Herbal and supplemental suggestions and lifestyle recommendations around cleansing (home, projects, and personal relationships) also are adapted to this time.
Days 11 through 20 constitute part III, in which the patient is helped to rebuild and enhance the immune system before the next cycle of chemotherapy. Blood counts, physiology, and stamina will begin to normalize, and side effects usually recede. A person’s activities should also be adapted to this, and emphasizing activities that help the individual feel genuinely happy are a premium.
The strategy for patients whose conventional therapy consisted of radiation was to include protocol items for minimizing radiation side effects, minimizing antioxidant therapies during the radiation phase, and resuming antioxidants after radiation. Acupuncture was made available on an as-needed basis, but the primary treatment emphasis was on the herbal/nutraceutical elements. The herbal medicine portion of the PAM+V protocol was administered in the traditional Chinese method of giving patients dry herbs to take home and decoct.
Diet
For all patients, we discussed diet, including basic cancer patient nutrition advice with an emphasis on natural foods, with recommendations to increase fiber and organic foods and improve personal food preparation skills.
Exercise
We advised all patients on exercise, emphasizing basic walking, household and garden chores, and then, if possible, yoga, jogging, gym activities, and training in yoga, Tai Ji, or Qi-Gong.
Data Sources
Data on PAM+V-treated patients were obtained by chart review from Pine Street Clinic (San Anselmo, CA). Survival time in the PAM+V cohort was obtained directly from patient records and/or confirmed by in-person or telephone follow-up with patients’ family members. PAM+V was provided concurrently with chemotherapy/radiation treatment (short-term use). After completion of chemotherapy/radiation, we monitored whether or not the patients elected to continue herbal/vitamin therapy (long-term use). Patients who were lost to follow-up and for whom treatment adherence was, therefore, unknown were analyzed as part of the long-term adherence group, following the intention-to-treat principle. 28 Additionally, short-term versus long-term adherence was noted for both patients who after initial consultation here at Pine Street elected to continue at our center and for those who followed up with other CAM centers. Thus, each of these 2 variables was measured separately: location (Pine Street or CAM) and duration (short term or long term).
Data on concurrent external controls diagnosed during the same time span as the Pine Street patients and matched on cancer stage were obtained by database extraction from the California Cancer Registry and Kaiser Permanente Division of Research. As the enrollment period for the Pine Street Clinic cohort began on November 1, 1988, and ended on December 1, 1993, we selected cases from the Kaiser Permanente and California Cancer Registry diagnosed between those dates. For the case selection and exclusion algorithm, see Table 1.
Case Selection and Exclusion Algorithm

Abbreviations: PAM+V, Pan-Asian medicine + vitamins; CCR, California Cancer Registry; KPNC, Kaiser Permanente Northern California; NSCLC, non-small-cell lung cancer; AJCC, American Joint Committee on Cancer; SEER, Surveillance, Epidemiology and End Results.
In Table 2 we report for each cohort the number of participants and their individual and treatment characteristics, such as which conventional treatments they received. In Table 3, we report PAM+V treatment dosage and timing. In Table 4, we report ingredients of Chinese herbal formulas used. The design principles for this whole-systems treatment approach have been described elsewhere. 29
Patient Characteristics

Abbreviations: PAM+V, Pan-Asian medicine + vitamins; CCR, California Cancer Registry; KPNC, Kaiser Permanente Northern California.
PAM+V Protocol Timing Concurrent With Chemotherapy, 21-Day Cycle

Abbreviation: PAM+V, Pan-Asian medicine + vitamins.
Day 1 is defined as the day of chemotherapy infusion. Treatment continues with part III beyond conclusion of chemotherapy.
Chinese Herbal Formula Ingredients
Stage
For external controls obtained from the California Cancer Registry, the American Joint Committee on Cancer (AJCC) staging system was used as the definition for cancer stage for lung cancer patients. 30 For external controls obtained from Kaiser Permanente Division of Research, AJCC stage was not available for the study recruitment years (1988-1993), so we used the Surveillance Epidemiology and End Results (SEER) Program Comparative Staging Guide for Cancer31,32 to manually build TNM staging from data fields for tumor extension, node involvement, and metastasis.
Analysis Methods
We planned to use traditional association models, Kaplan-Meier estimation 33 and Cox proportional hazards regression 34 as well as more modern methods—MSMs 35 and propensity score 26 —for causal inference.
Data Analysis
The primary end point was overall survival. Our primary treatment variables of interest were herbal/vitamin therapy (yes/no) and duration of herbal/vitamin therapy (long/short); baseline covariates were stage (II, IIIA, IIIB, or IV), age at diagnosis, sex, race (white/black/Asian), smoking history (ever/never), histology (adenocarcinoma/squamous), and treatment with surgery, radiation, or chemotherapy (yes/no). Time at risk was defined by dates of diagnosis and death (month/year).
We analyzed data in 2 phases: (1) within Pine Street Clinic by comparing patients maintaining long-term Chinese herbal/vitamin therapy with those using only short-term treatment and (2) comparing the Pine Street Clinic cohort with external controls.
For traditional survival analysis, we used Cox proportional hazards regression, 34 stratified by stage and adjusted by all covariates, with the stcox program in the Stata statistical package (version 9.2; StataCorp, College Station, TX), and Kaplan-Meier estimation with sts.
To investigate causal inference, we used the MSM approach, in which the Cox proportional hazards model is weighted by the inverse multivariate probability of each patient having received treatment (Inverse Probability Treatment Weighting [IPTW], stabilized). The IPTW balances treatment groups with respect to their probability of having received treatment using logistic regression with the same covariates as our standard Cox regression. We used the bootstrap 36 with 1000 repetitions for estimation of the CIs about the HR. We wrote our own program for this analysis, using the Stata statistical software and again stratified by stage.
We also used the propensity score for causal inference, which estimates in a multivariate model the probability of receiving treatment using a person’s individual disease and treatment characteristics, applying the pscore Stata program 37 and using the same set of covariates described above. The resulting propensity score was divided into 5 or more levels or strata, and the propensity score then used as a variable in adjusted Cox regression was again stratified by stage. Subclassification of patients into 5 or more levels of the propensity score has been shown to remove 90% of bias. 38
Manuscript Preparation
In the drafting of our manuscript, we followed the Strengthening The Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 39
Results
Number of Patients
Survival time data suitable for analysis were obtained for 235 of 239 patients from Pine Street Clinic, 11 853 from California Cancer Registry, and 901 from Kaiser (Table 1).
Comparing Long-term With Short-term PAM+V Use
In adjusted Cox regression analysis, survival was significantly better for long-term versus short-term PAM+V in stages IIIB (HR = 0.17; 95% CI = 0.08, 0.36) and IV (HR = 0.28; 95% CI = 0.13, 0.60); there was a large but statistically nonsignificant survival advantage in stages II and IIIA (Table 5).
Lung Cancer Survival With Pan-Asian Medicine and Vitamin Therapy a

Abbreviations: PAM+V, Pan-Asian medicine+vitamins; HR, hazard ratio; CI, confidence interval; MSM, Marginal Structural Model; CCR, California Cancer Registry; KPNC, Kaiser Permanente Northern California.
P < .05; **P < .01; ***P < .001.
Adjusted for age, sex, race, cell type, smoking, surgery, chemotherapy, and radiation.
Propensity score built using age, sex, race, cell type, and smoking and treatment with surgery, chemotherapy, and radiation.
MSM built using age, sex, race, cell type, and smoking and treatment with surgery, chemotherapy, and radiation.
Adjusted for age, sex, race, cell type, surgery, chemotherapy, and radiotherapy.
Propensity score built using age, sex, race, cell type, and treatment with surgery, chemotherapy, and radiation.
Marginal structural models built using age, sex, race, cell type, surgery, chemotherapy, and radiation.
PAM+V compared with CCR: stage IIIA unadjusted propensity score balanced with age, sex, surgery, and radiation.
In adjusted propensity score analysis, survival was significantly better for long-term versus short-term PAM+V in stages IIIB (HR = 0.17; 95% = CI 0.08, 0.36) and IV (HR = 0.28; 95% CI = 0.12, 0.61); there was a modest but statistically nonsignificant survival advantage with long-term PAM+V in stages II and IIIA (Table 5).
In MSM analysis, there was a large but nonsignificant survival advantage to long-term versus short-term PAM+V in all stages (Table 5).
With Kaplan-Meier analysis, survival was substantially better for long-term versus short-term PAM+V in stages IIIB and IV, comparable in stage II, and with a trend toward improved survival in stage IIIA (Figure 1).
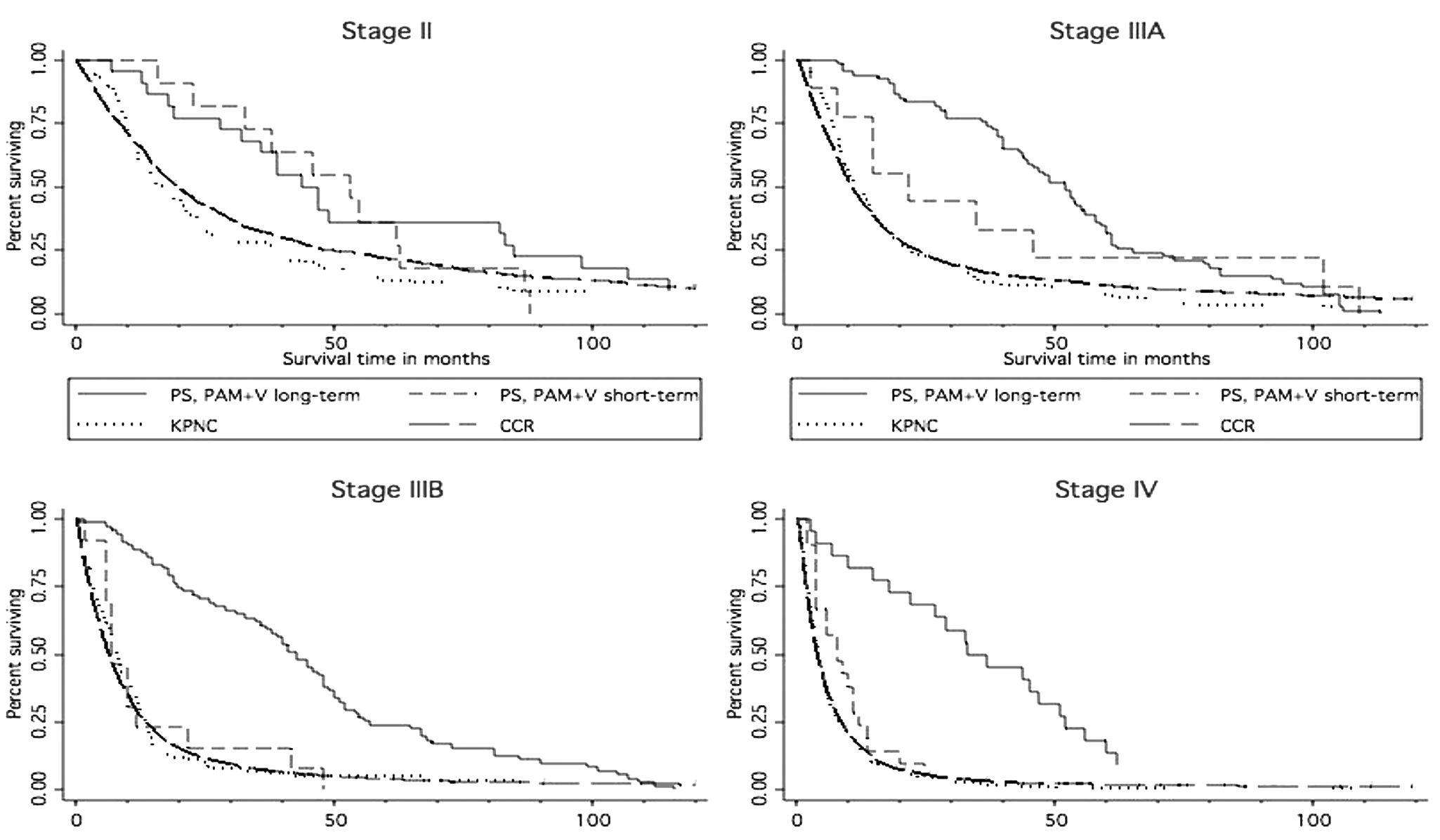
Survival in non-small-cell lung cancer, long-term versus short-term PAM+V and comparison with external controls
Comparing Long-term PAM+V-Treated Patients With Concurrent External Controls From the California Cancer Registry
In adjusted Cox regression, survival was significantly better for long-term PAM+V users versus California Cancer Registry controls in stages IIIA (HR = 0.49; 95% CI = 0.38, 0.63), IIIB (HR = 0.34; 95% CI = 0.26, 0.44), and IV (HR = 0.36; 95% CI = 0.23, 0.54) and was comparable in stage II (HR = 0.92; 95% CI = 0.41, 1.43; Table 5).
In adjusted propensity score analysis, survival was significantly better for long-term PAM+V users versus registry controls in stages IIIA (HR = 0.51; 95% CI = 0.39, 0.67), IIIB (HR = 0.37; 95% CI = 0.28, 0.50) and IV (HR = 0.31; 95% CI = 0.20, 0.48), and there was a nonsignificant trend toward better survival in stage II (Table 5; HR = 0.92; 95% CI = 0.59, 1.44).
In MSM analysis, survival was significantly better for long-term PAM+V users versus registry controls in stage IIIA (HR = 0.41; 95% CI = 0.28, 0.55), stage IIIB (HR = 0.44; 95% CI = 0.31, 0.57), and stage IV (HR = 0.29; 95% CI = 0.20, 0.38), and there was a nonsignificant trend toward better survival in stage II (Table 5; HR = 0.92; 95% CI = 0.41, 1.43).
In Kaplan-Meier analysis, in all stages, long-term PAM+V combined with conventional therapy yielded longer survival compared with conventional therapy alone in registry controls (Figure 1).
Comparing Long-term PAM+V-Treated Patients With Concurrent External Controls From Kaiser Permanente
In adjusted Cox regression, survival was significantly better for long-term PAM+V users versus Kaiser controls in all stages of lung cancer: stage II (HR = 0.56; 95% CI = 0.32, 0.99), stage IIIA (HR = 0.14; 95% CI = 0.08, 0.25), stage IIIB (HR = 0.23; 95% CI = 0.13, 0.40), and stage IV (HR = 0.21; 95% CI = 0.13, 0.34; Table 5).
In adjusted propensity score analysis, survival was significantly better for long-term PAM+V users versus Kaiser controls in stages IIIA (HR = 0.18; 95% CI = 0.09, 0.35), IIIB (HR = 0.52; 95% CI = 0.31, 0.88), and stage IV (HR = 0.09; 95% CI = 0.04, 0.21), and there was a small but nonsignificant trend toward better survival in stage II (HR = 092; 95% CI = 0.59, 1.44; Table 5).
In MSM analysis, survival was significantly better for long-term PAM+V users versus Kaiser controls in stages IIIA (HR = 0.40; 95% CI = 0.30, 0.54), stage IIIB (HR = 0.07; 95% CI = 0.01, 0.65), and stage IV (HR = 0.10; 95% CI = 0.01, 0.19), and there was a large but nonsignificant survival advantage in stage II (HR = 0.66; 95% CI = 0.26, 1.05; Table 5).
In Kaplan-Meier analysis, in all stages, long-term PAM+V combined with conventional therapy yielded longer survival compared with conventional therapy alone in Kaiser controls (Figure 1).
Comparing All PAM+V Users With Concurrent External Controls From the California Cancer Registry
In adjusted Cox regression, survival was significantly better for long-term PAM+V users versus California Cancer Registry controls in stages IIIA (HR = 0.49; 95% CI = 0.38, 0.64), IIIB (HR = 0.34; 95% CI = 0.26, 0.44), and IV (HR = 0.36; 95% CI = 0.24, 0.55) and was comparable in stage II (HR = 1.00; 95% CI = 0.64, 1.54; Table 5).
In adjusted propensity score analysis, survival was significantly better for long-term PAM+V users versus registry controls in stages IIIA (HR = 0.51; 95% CI = 0.39, 0.67), IIIB (HR = 0.37; 95% CI = 0.28, 0.50), and IV (HR = 0.31; 95% CI = 0.20, 0.48), and survival was comparable in stage II (HR = 0.91; 95% CI = 0.59, 1.42; Table 5).
In MSM analysis, survival was significantly better for long-term PAM+V users versus registry controls in stage IIIA (HR = 0.42; 95% CI = 0.28, 0.55), stage IIIB (HR = 0.43; 95% CI = 0.31, 0.56), and stage IV (HR = 0.29; 95% CI = 0.21, 0.38), and there was a small and statistically nonsignificant trend toward better survival in stage II (HR = 0.91; 95% CI = 0.47, 1.35; Table 5).
Comparing All PAM+V-Treated Patients With Concurrent External Controls From Kaiser Permanente
In adjusted Cox regression, survival was significantly better for long-term PAM+V users versus Kaiser controls in stage IIIA (HR = 0.27; 95% CI = 0.17, 0.43), IIIB (HR = 0.26; 95% CI = 0.16, 0.40), and IV (HR = 0.25; 95% CI = 0.16, 0.40; Table 5).
In adjusted propensity score analysis, survival was significantly better for all PAM+V users analyzed together versus Kaiser controls in stage IIIA (HR = 0.29; 95% CI = 0.18, 0.46), stage IIIB (HR = 0.34; 95% CI = 0.19, 0.59), and stage IV (HR = 0.22; 95% CI = 0.12, 0.39), and there was a strong but statistically nonsignificant effect of better survival in stage II (HR = 0.61; 95% CI = 0.35, 1.07; Table 5).
In MSM analysis, survival was significantly better for long-term PAM+V users versus Kaiser controls in stage IIIA (HR = 0.40; 95% CI = 0.30, 0.52), stage IIIB (HR = 0.14; 95% CI = 0.07, 0.28), and stage IV (HR = 0.25; 95% CI = 0.08, 0.41), and there was a large but nonsignificant survival advantage in stage II (HR = 0.66; 95% CI = 0.30, 1.03; Table 5).
Comparing Combinations of PAM+V With Chemotherapy, Surgery, and Radiotherapy
Figure 2 compares Kaplan-Meier survival curves between patients who received PAM+V combined with surgery, PAM+V alone, surgery alone, or neither PAM+V nor surgery stratified by cancer stage and using California Cancer Registry controls. Figure 3 reports all combinations of PAM+V and chemotherapy and Figure 4 all combinations of PAM+V and radiotherapy. Virtually all analyses showed a dose–response effect, with longest survival seen with the full treatment combination of PAM+V with conventional therapy and the least favorable survival response in patients receiving neither treatment, with PAM+V alone or standard therapy alone in the middle (Figures 2 to 4).

Lung cancer survival, showing all possible treatment combinations of PAM+V and surgery, using California Cancer Registry patients as external controls

Lung cancer survival, showing all possible treatment combinations of PAM+V and chemotherapy, using California Cancer Registry patients as external controls

Lung cancer survival, showing all possible treatment combinations of PAM+V and radiotherapy, using California Cancer Registry patients as external controls
Distinguishing the Effect of Treatment From Location of Follow-up
We were also interested in whether there was a “center effect”—that is, survival differences between people continuing long-term PAM+V at Pine Street Clinic and those continuing at other CAM centers. In Kaplan-Meier analysis, we found no apparent difference in survival, with 1 exception of better survival in stage-IV patients continuing at other CAM centers, although this represents only 4 of 43 stage-IV patients (Figure 5).

Survival in non-small-cell lung cancer, comparing TCM patients initially treated at PSC with those followed up at other CAM centers
CAM Therapy in External Controls
We were fortunate to identify some records within the California Cancer Registry in which clinicians had documented patients’ use of CAM therapies (coded in the parlance of the time as “unproven therapies”) and compared survival with long-term and short-term practitioner-guided PAM+V with those of CAM users from the registry database. In Kaplan-Meier analysis, in stages II, IIIA, IIIB, and IV lung cancer, there was longer survival with long-term practitioner-guided PAM+V therapy compared with CAM use in the general population (Figure 6). In patients with stages IIIA, IIIB, and IV cancer, there was no difference in survival between short-term PAM+V users within the Pine Street cohort and CAM users in the general population, further reinforcing the benefit of long-term maintenance of adjunctive complementary therapies (although these findings on CAM use in the general population are prone to error because of misclassification bias arising from the way in which CAM use data were gathered by the California Cancer Registry (Figure 6).
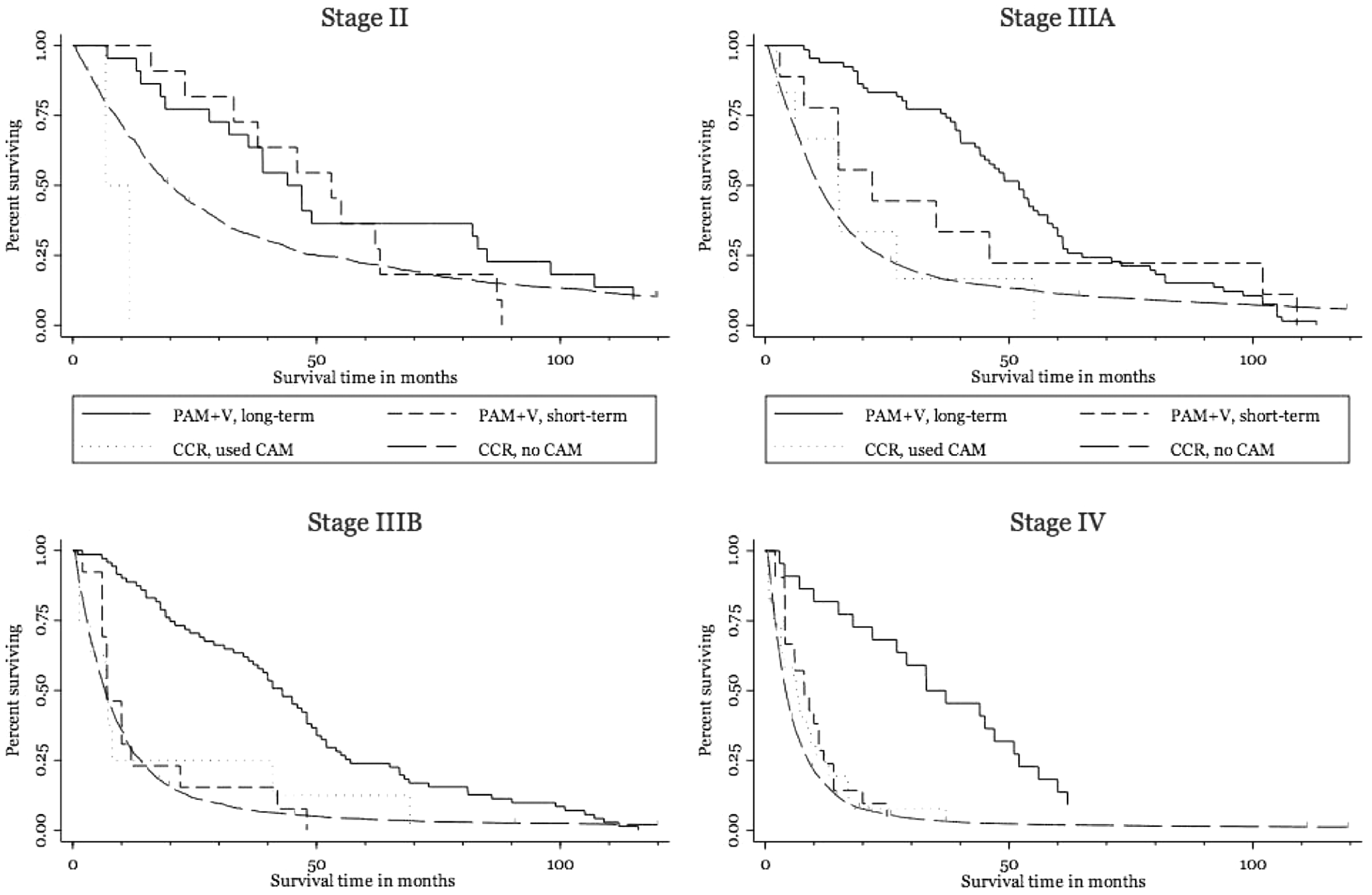
Survival in non-small-cell lung cancer, comparing patients treated with long-term and short-term PAM+V with CAM users and nonusers from within the CCR database
Validity of External Controls
To confirm the validity of external controls, we compared survival rates from our analysis among California Cancer Registry and Kaiser controls with those reported by other authors also using SEER data and found that they were comparable (Table 6). For example, in stage IIIB and IV patients treated with chemotherapy, median survival in California Cancer Registry controls was 6.9 months and in Kaiser controls 6.1 months, compared with 6.8 months in other studies also using SEER data during the same time period. 40
Absolute Survival Rates for Examining the Validity of External Controls
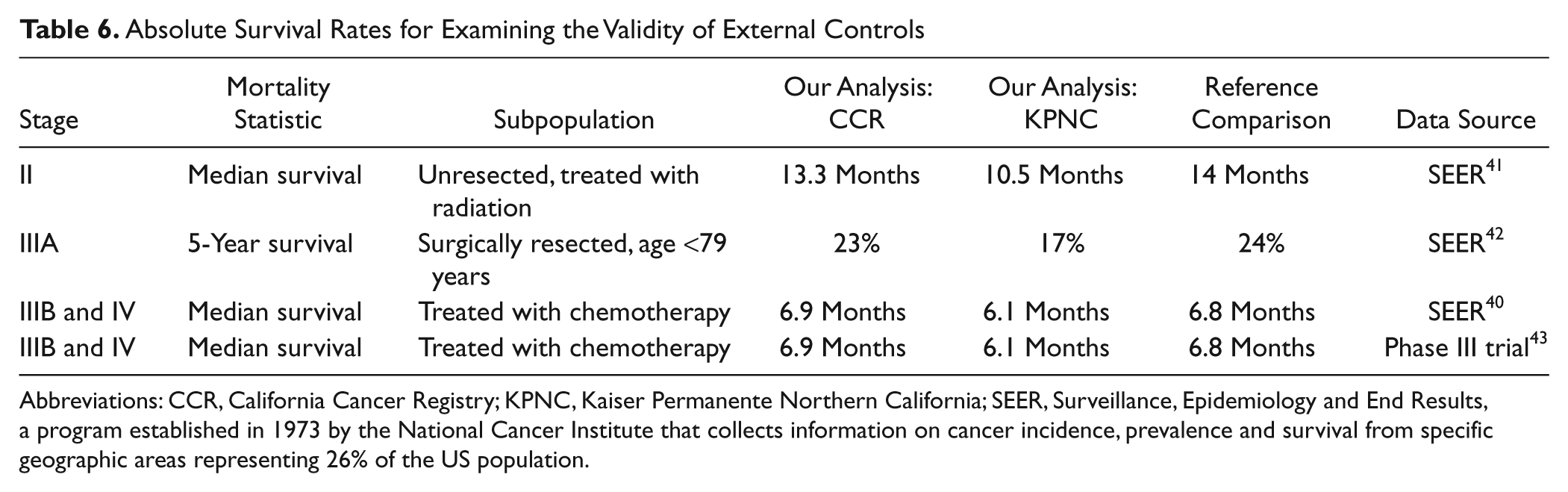
Abbreviations: CCR, California Cancer Registry; KPNC, Kaiser Permanente Northern California; SEER, Surveillance, Epidemiology and End Results, a program established in 1973 by the National Cancer Institute that collects information on cancer incidence, prevalence and survival from specific geographic areas representing 26% of the US population.
Crude Survival Rates: Stage II
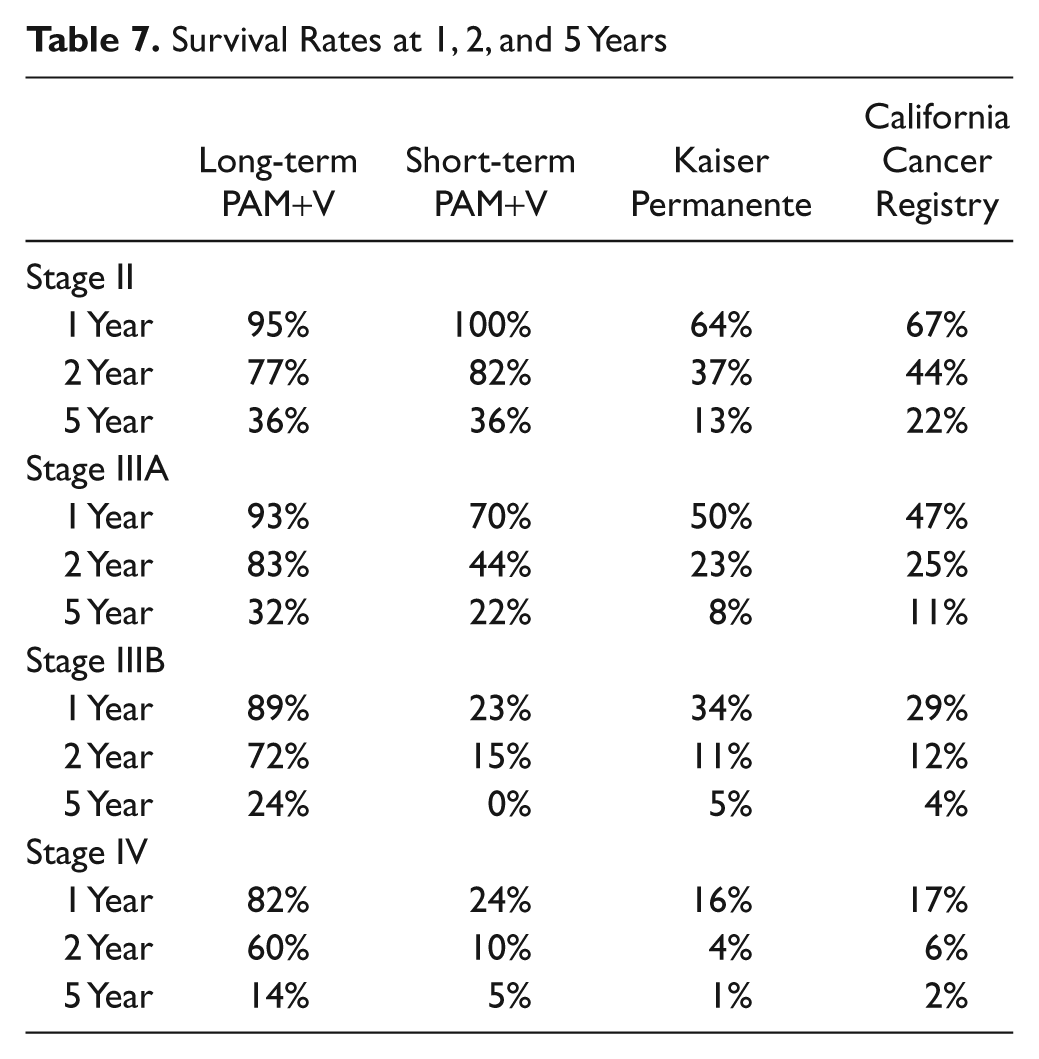
Survival at 1 year was 95% in the long-term PAM+V group, 100% in the short-term PAM+V group, 64% in the Kaiser controls, and 67% in California Cancer Registry controls. Survival at 2 years was 93% in the long-term PAM+V group, 70% in the short-term PAM+V group, 50% in Kaiser controls, and 47% in California Cancer Registry controls. Survival at 5 years was 36% in both long-term and short-term PAM+V groups, 13% in Kaiser controls, and 22% in California Cancer Registry controls (Table 7).
Survival Rates at 1, 2, and 5 Years
Crude Survival Rates: Stage IIIA
Survival at 1 year was 93% in the long-term PAM+V group, 70% in the short-term PAM+V group, 50% in Kaiser controls, and 47% in California Cancer Registry controls. Survival at 2 years was 83% in the long-term PAM+V group, 44% in the short-term PAM+V group, 23% in Kaiser controls and 25% in California Cancer Registry controls. Survival at 5 years was 32% in the long-term PAM+V group, 22% in the short-term PAM+V group, 8% in Kaiser controls, and 11% in California Cancer Registry controls (Table 7).
Crude Survival Rates: Stage IIIB
Survival at 1 year was 89% in the long-term PAM+V group, 23% in the short-term PAM+V group, 34% in Kaiser controls, and 29% in California Cancer Registry controls. Survival at 2 years was 72% in the long-term PAM+V group, 15% in the short-term PAM+V group, 11% in Kaiser controls, and 12% in California Cancer Registry controls. Survival at 5 years was 24% in the long-term PAM+V group, 0% in the short-term PAM+V group, 5% in Kaiser controls, and 4% in California Cancer Registry controls (Table 7).
Crude Survival Rates: Stage IV
Survival at 1 year was 82% in the long-term PAM+V group, 24% in the short-term PAM+V group, 16% in Kaiser controls, and 17% in California Cancer Registry controls. Survival at 2 years was 60% in the long-term PAM+V group, 10% in the short-term PAM+V group, 4% in Kaiser controls, and 6% in California Cancer Registry controls. Survival at 5 years was 14% in the long-term PAM+V group, 5% in the short-term PAM+V group, 1% in Kaiser controls, and 2% in California Cancer Registry controls (Table 7).
Discussion
Principal Findings
In our retrospective survival analysis for non-small-cell lung cancer, we found a substantial and statistically significant survival advantage to combining PAM+V with conventional therapy compared with treatment with conventional therapy alone; this was strongest in advanced lung cancer—stages IIIA, IIIB, and IV. We also found significant benefits of long-term PAM+V use compared with short-term use discontinued after adjuvant chemotherapy and radiotherapy. MSM and propensity score analyses found results usually consistent with multivariate Cox regression analysis. Propensity score analysis had a stronger ability to detect a survival benefit, particularly in subanalyses with small sample sizes.
Strengths of the Study
To our knowledge, this is the first use of causal inference using the MSM approach in cancer survival or in any CAM therapy. It is also the first use of the propensity score in any Chinese herbal therapy.
We intentionally used specific herbal and vitamin products commonly available in the commercial markets and free of proprietary constraints. In this way, we hoped to maximize the accessibility and applicability of our treatment approach for clinicians and researchers.
We found consistency between the different multivariate analyses methods used: traditional Cox regression, MSM Cox regression, and propensity score Cox regression. Propensity score analysis was best able to detect a survival advantage, followed by MSMs.
Lead time (the delay between diagnosis and initiation of PAM+V treatment) averaged between 22 days and 28 days. This is comparable with what is known about delays in delivery of care to those with lung cancer in conventional oncology practice: delays attributable to the patient average 18 days, and delays attributable to the health system average 62 days. 44 To strengthen our inference, we conducted a sensitivity analysis in which we dropped all PAM+V-treated patients with lead time greater than 60 days. We also conducted a conservative analysis where we excluded all KPNC external controls who survived for less than 60 days. In both these sensitivity analyses, we found differences of only a few percentage points in the HRs, which did not change our inferences.
Patients’ use of imagery, visualization, exercise, and Qi-Gong was an integral part of the treatment design, and they were encouraged to learn and practice these approaches as much as possible; these are the kinds of approaches studied in whole systems research, where the emphasis is on the net effect of the holistic protocol design. Teasing apart the relative efficacy of each individual PAM+V component could be pursued in a future prospective study designed to answer this question. Holistic protocols like PAM+V can also be studied using an approach called pragmatic trials, which evaluate a therapy as it is used in normal practice (as compared with the more fixed and constrained approach used in most clinical trials). 45
Limitations of the Study
These are observational data, retrospectively gathered from medical records and cancer registry databases. There may have been unmeasured differences between patients choosing PAM+V versus those who did not, which could have additionally contributed to differences in survival. We did not have data that would allow us to control for possible confounding by socioeconomic status, which is associated with increased use of CAM, 46 better access to conventional cancer therapy, 47 and increased cancer survival. 48
In the California Cancer Registry and Kaiser, we did not have available data on smoking, which is an important prognostic factor in lung cancer survival. Although some studies have shown that smoking history confers less favorable survival in people diagnosed with lung cancer,49,50 others have shown no significant differences in survival, 51 particularly in people with advanced disease. 52 We did, however, have smoking history data available for those in the PAM+V cohort. This allowed us to include smoking as a variable in all our multivariate survival analyses comparing long-term versus short-term PAM+V treatment.
We did not have prospectively gathered data available for analysis and were therefore unable to account for time-dependent confounding, which is often present in longitudinal studies and capably handled by MSM analysis. Early papers on the MSMs stated the assumption of no unobserved confounders, although this assumption is unverifiable and has recently been called into question.53,54
We did not have data on CAM use by external controls in the Kaiser data. Nevertheless, even if the proportion of CAM use among Kaiser members was as high as the 26% to 30% reported in the literature, 55 we anticipate that this would have biased our results toward the null and that the true survival benefit of PAM+V use may even be stronger than we estimated. To strengthen our inference, we also conducted a conservative analysis where we excluded all KPNC external controls who survived less than 2 months and found only a difference in the HR of a few percentage points (which caused no change in our inference).
The existing and newly developed conventional therapies for lung cancer, which were in use during the time span of data collection for this study, are known to have significant toxicities.56-58 Enhancing patient compliance with and tolerance of conventional therapies is an often-reported goal of many CAM treatment programs. We did not include that outcome as a part of the current study. Nevertheless, we are starting to see outcomes data on the contributions of Chinese herbal therapy to chemotherapy compliance in a meta-analysis we are currently conducting.
Causal Inference Methods Used in Our Study
This study uses 2 newer analysis methods for what is called causal inference: the propensity score and the MSM. Causal inference attempts to look more closely into the data being analyzed to uncover causal relationships. This is not always possible with traditional statistical methods, which can usually conclude only that the observed association between 2 variables is not a result of chance and cannot identify which variable causes the other.
Both the propensity score and MSM methods begin by using multivariable analysis to identify for each individual in the study, based on their personal and treatment history, that that individual chose one treatment or another. The propensity score and MSM estimate that score with similar but slightly different statistical approaches. The propensity score is used as a variable in the Cox regression. The MSM is similarly used to weight the Cox regression.
In practice, both methods of causal inference will balance the treatment groups, so that patients in each group are comparable with respect to confounding variables. The primary practical difference between the MSM and propensity score methods is that the MSM can adequately handle the effects of treatment variables that vary over time, based on the outcomes of treatment. In the case of our study, however, this difference is not relevant because our study was of the point treatment design, where variables are measured only at baseline.
Possible Mechanisms
The design of the treatment protocol used in this study was based on treatment strategies from traditional Chinese medicine, combining a large number of different herbal and vitamin substances into 1 integrated treatment strategy. The timing for each protocol item described below is listed in Table 3.
Several plausible mechanisms for these findings may be considered, based on what is known about the specific components of this combination treatment approach with respect to various effects.
General cancer cell cycle effects
Green tea has been shown to reduce the risk of lung cancer in several case control studies, 59 notably in smokers. 60 N-acetyl cysteine inhibits tumor growth61-63 and metastasis. 64 Curcumin is extracted from the culinary spice turmeric (Curcuma longa) and inhibits cell invasion and metastasis, 65 inhibits NF-κB and COX-2 in human non-small-cell lung cancer cells, 66 and has antiproliferative 67 and apoptosis-stimulating functions. 68 Garlic stimulates apoptosis in non-small-cell lung cancer cells. 69 Fish oil possesses anti-inflammatory and antitumor activity.
Chemotherapy-related effects
Green tea enhances the antitumor efficacy of anthracycline, cisplatin, and irinotecan chemotherapy. 70 Green tea also contains the polyphenol epigallocatechin-3-gallate, which inhibits telomerase and induces apoptosis in drug-resistant lung cancer cells. 71
Reduction of chemotherapy toxicity and side effects
The green tea plant contains
Reduction of radiation toxicity and side effects
Components found within the ginseng root protect against DNA damage from radiation exposure, 82 potentiate the therapeutic effect of radiation on lung cancer cells, 83 and reduce radiation-induced enteritis. 84 Cordyceps protects against radiation-induced bone marrow and intestinal injuries, 85 accelerates leukocyte recovery, stimulates immune lymphocyte proliferation, and improves animal survival after radiation exposure. 86 The Chinese herbal medicines Radix angelicae and Radix paeoniae protect intestinal cells from irradiation-induced damage. 87 Furthermore, Radix angelicae inhibits radiation-induced pulmonary fibrosis, with the mechanism being downregulation of proinflammatory cytokine expression. 88 Radix atractylodes and Poria cocos reduce radiation injury to healthy cells. 89 Radix rehmannia and Radix ligustici reduced radiation pneumonitis in a study of 46 individuals with non-small-cell lung cancer (incidence of radiation pneumonitis was 26% in controls and 10% in the herbal group). 90 Gingko biloba and Panax ginseng protect against radiation-induced lethality, lipid peroxidation, and DNA damage. 82 Docosahexaenoic acid, an n-3 polyunsaturated fatty acid found in fish oil, in combination with radiation significantly increased oxidative stress and cell death in a lung cancer cell line when compared with either treatment alone. 91 It accomplishes this by increasing heme oxygenase levels in lung endothelial and fibroblast cells and by blocking radiation-induced reactive oxygen species. Curcumin, an active component of the curry seasoning turmeric, reduced radiation-induced pulmonary fibrosis in mice and increased their survival, without reducing the tumor cell kill rate of radiation therapy. 92 Caffeic acid phenylethyl ester, a component of propolis, reduced radiation-induced lung injury in a rat model through its antioxidant effect. 93 Vitamin E provided immediately after radiation therapy in a rat model protected against radiation-induced pulmonary fibrosis. 94 Melatonin reduces radiation-induced oxidative organ damage in rats by increasing malondialdehyde levels, myeloperoxidase activity, and glutathione levels. 95
More general systemic effects such as immune system modulation
N-acetyl cysteine enhances the functionality of peripheral blood mononuclear cells. 96 Thymic protein is obtained from a cloned cell line of thymic epithelial origin and induces T-cell maturation. 97 In lung cancer patients, fish oil increases appetite and body weight, reduces fatigue, improves muscle strength and lowers C-reactive protein levels. 98 Melatonin is a sleep-regulating hormone shown to improve survival99,100 and tumor response 101 in lung cancer patients when combined with conventional therapies.
An immune-enhancing Chinese herbal formula was also used, which combines herbal medicines traditionally recognized as tonifying blood yin (nourishes bone marrow), tonifying Spleen yin/qi (boosts cellular immunity), harmonizing Liver qi (balances liver detoxification pathways), and tonifying Kidney yang (improves dispositional optimism). The ingredients are shown in Table 3.
Summary and Future Directions
Although previous work has examined the survival benefit of Chinese herbal therapies in combination with standard chemotherapy for lung cancer, 13 there is little available evidence for whole-systems combination CAM therapies. In this retrospective survival analysis comparing patients treated with long-term versus short-term PAM+V therapy, there was a substantial, statistically significant, survival advantage in stages IIIA, IIIB, and IV and a modest but nonsignificant advantage in stage II. PAM+V combined with standard therapy, compared with standard therapy alone, achieved significantly better survival in stages IIIA, IIIB, and IV disease and conferred a modest but nonsignificant advantage in stage II; these results were stronger when comparing long-term users of PAM+V with external controls.
It is important to emphasize in both analyses that the survival advantages reflect the benefit of using PAM+V combined with, rather than instead of, conventional therapy. Our results suggest that prospective trials for advanced non-small-cell lung cancer patients using PAM+V and vitamin therapy, in a whole-systems approach combined with conventional therapies, are justified.
Footnotes
Acknowledgements
To my instructors for providing guidance, to Dr Ly Suhuai and Michael Broffman for inspiration, to Louise Estupinian for assistance in records acquisition, to the kind members of Statalist for their advice, and to my family for their loving support [MM].
Author contributions are as follows: Conception and design; M. F. McCulloch, M. D. Broffman, J. M. Colford Jr. Administrative support; M. D. Broffman, L. Kushi, J. Gao, J. M. Colford Jr. Provision of study materials or patients; M. F. McCulloch, M. D. Broffman, L. Kushi. Collection and assembly of data; M. F. McCulloch, M. D. Broffman, J. Gao. Data analysis and interpretation; M. F. McCulloch, M. van der Laan, A. Hubbard, A. Kramer, J. M. Colford Jr. Manuscript writing; M. F. McCulloch, J. M. Colford Jr. The collection of cancer incidence data used in this study was supported by the California Department of Public Health as part of the statewide cancer reporting program mandated by California Health and Safety Code Section 103885; the National Cancer Institute’s Surveillance, Epidemiology and End Results Program under contract N01-PC-35136 awarded to the Northern California Cancer Center, contract N01-PC-35139 awarded to the University of Southern California, and contract N02-PC-15105 awarded to the Public Health Institute; and the Centers for Disease Control and Prevention’s National Program of Cancer Registries, under agreement #U55/CCR921930-02 awarded to the Public Health Institute. The ideas and opinions expressed herein are those of the author(s) and endorsement by the State of California, Department of Public Health, the National Cancer Institute, and the Centers for Disease Control and Prevention or their contractors and subcontractors is not intended nor should it be inferred.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Pine Street Foundation, a University of California at Berkeley Epidemiology Block Grant Award, and a University of California at Berkeley Research Fellowship.
