Abstract
Melanoma is an aggressive tumor that expresses the pigmentation enzyme tyrosinase. Tyrosinase expression increases during tumorigenesis, which could allow for selective treatment of this tumor type by strategies that use tyrosinase activity. Approaches targeting tyrosinase would involve gene transcription or signal transduction pathways mediated by p53 in a direct or indirect manner. Two pathways are proposed for exploiting tyrosinase expression: (a) a p53-dependent pathway leading to apoptosis or arrest and (b) a reactive oxygen species–mediated induction of endoplasmic reticulum stress in p53 mutant tumors. Both strategies could use tyrosinase-mediated activation of quercetin, a dietary polyphenol that induces the expression of p53 and modulates reactive oxygen species. In addition to antitumor signaling properties, activation of quercetin could complement conventional cancer therapy by the induction of phase II detoxification enzymes resulting in p53 stabilization and transduction of its downstream targets. In conclusion, recent advances in tyrosinase enzymology, prodrug chemistry, and modern chemotherapeutics present an intriguing and selective multitherapy targeting system where dietary bioflavonoids could be used to complement conventional cancer treatments.
Introduction
Melanoma and neuroblastoma are 2 aggressive tumor types that develop from cells of neural crest origin and distinctively express the immunogenic pigmentation enzyme tyrosinase.1-3 Tyrosinase is an exploitable target in melanocytic tumors for multiple reasons. Expression of tyrosinase is limited mainly to melanoma or melanocytes and its expression is noted to increase during tumorigenesis.4,5 Additionally, both primary and metastatic melanocytic lesions are usually associated with changes in tyrosinase-mediated pigmentation, which makes it an ideal immunological and molecular target when compared with other antigens.6,7 Several studies showed successful results in treating melanoma with the induction of a cytotoxic T-lymphocyte response using tyrosinase as tumor-associated antigen.6,8-11 However, apart from acknowledging the fact that tyrosinase is an attractive target for immunotherapy, we will not be further explicating on it in this review. Additionally, tyrosinase has shown promise in activating multiple protherapeutic compounds that lead to selective inhibition of DNA synthesis among other outcomes.12-14 Again, since we cannot provide a complete review of tyrosinase-activated prodrugs, see the review by Jawaid et al. 15 In this review, however, we will instead discuss a third, and more novel, ability of tyrosinase to bring about cell death. We propose a possible indirect pro-apoptotic enzymatic reaction catalyzed by tyrosinase, which leads to the activation of quinone species. Furthermore, we aim to demonstrate that quinone-forming flavonoids, such as quercetin, are prime substrates to carry out this antitumor cascade.
Overview of Melanoma and Neuroblastoma
Properties of Melanoma
Melanoma is categorized as the most aggressive form of skin cancer, and the number of cases worldwide has doubled in the past 20 years. 1 The incidence of melanoma has been increasing at a rate of 6% to 7% annually and accounts for approximately 4% of all skin cancers and 75% of deaths from skin cancer. In the United States, the incidence and mortality of melanoma continues to rise faster than that of any other cancer. 2 The frequency is almost 22 per 100,000 for males and 14 for females, which translates to approximately 59,580 new diagnoses and 7,770 deaths from melanoma in 2005. 16 In the early phase when diagnosed as a thin lesion, surgical resection serves as a cure in most of the cases and results in a 5-year survival rate of more than 80%. However, advanced stage metastatic melanoma is almost always fatal and patients display 5-year survival rates of 45% for stage III and 10% for stage IV.1,17,18 Cytotoxic chemotherapy for late and metastatic melanoma is relatively ineffective and is usually accompanied by systemic toxicities and poor quality of life in patients. The lack of effectiveness of current treatments using cytotoxic therapies prompts the investigation of modalities that can both increase the responsiveness of melanoma and keep healthy tissue toxicity to a minimum. Therefore, we will discuss strategies that selectively target malignant melanoma cells due to their specific protein expression profiles.
Melanoma is described as a malignant tumor derived from melanocytes, which are dendritic cells originating from the neural crest. Melanoma can also originate from melanocytes in other regions of the body such as the meninges, digestive tract, eye, mucosal surfaces, or lymph nodes. 19 Neural crest cells are formed in the central nervous system and migrate from the neural crest to their target destination. 20 Changes in pigmentation occur in these cell types and arise from alterations in the expression or activity of the pigmentation enzyme tyrosinase. The transcriptional regulation of tyrosinase is influenced by external stimuli such as hair regeneration and ultraviolet exposure.21,22 Tumors of the neural crest commonly overexpress tyrosinase and is therefore a valuable tumor marker and a possible target for selective therapy. 3
Tyrosinase-Related Mechanisms
As will be discussed in this review, the p53 protein is central to apoptosis and is a major determinant of tumor response to therapies. This tumor suppressor gene also has a role in melanocyte metabolism and pigmentation. The induction of the tyrosinase gene is regulated by p53 and melanocortin peptide, melanocyte-stimulating hormone (α-MSH; Figure 1). Oxidation of phenols by tyrosinase is the basis of pigmentation reactions, but the activity can also be used to activate prodrugs. 23 Specifically, tyrosinase catalyses the hydroxylation of l-tyrosine to l-Dopa and its subsequent oxidation to dopaquinone, which undergoes a series of cascade reactions leading to the synthesis of melanin in the melanocyte.24,25 Tyrosinase is a binuclear copper-containing enzyme with a transmembrane helix closer to its C-terminal. This helix is embedded in the melanocyte cytosol and the N-terminal located inside the melanosome matrix.25,26 Sequencing studies of the promoter region of the human tyrosinase gene, and the regions regulating tyrosinase gene expression, 27 revealed the following: a TATA box 27 base pairs upstream from the transcription start site, a CAAT box 128 base pairs upstream, 5 AP-1 and 2 AP-2 sites, 2 glucocorticoid-responsive elements, 2 ultraviolet-responsive elements, and 3 Oct-1 sites. The tyrosinase promoter (Figure 1) also contains an “M box” and a binding site for the microphthalmia-associated transcription factor, which is known to activate transcription of the tyrosinase gene. 28 Products of tyrosinase oxidation, such as quinones, can induce p53. Cui et al 29 demonstrated that pro-opiomelanocortin (POMC) is a direct transcriptional target of p53, in that p53 binds directly to the POMC promoter inducing α-MSH production and POMC is directly induced with overexpression of p53 in keratinocytes. Thus, by targeting p53, one may be able to regulate tyrosinase expression in melanoma, which we will discuss in some detail below.

Tyrosinase promoter, enhancers, and protein domains
Properties of Neuroblastoma
Neuroblastoma is an embryonal tumor of the central and peripheral nervous system that originates from neural crest cells, similar to melanoma. These melanocytic tumors manifest in the adrenal glands or in the paraspinal location in the abdomen or chest,
30
and they are more common in children less than 1 year of age.
31
They account for 28% of all cancers in European and US infants and are associated with poor prognosis in children diagnosed at older age.32,33 Neuroblastoma accounts for more than 7% of malignancies in patients younger than 15 years and around 15% of all pediatric oncology deaths. Neuroblastomas express tyrosinase, like melanoma. In addition, they express a second target enzyme, tyrosine hydroxalase (TH). Melanocytes and neuroblasts can transform
Drug Therapies
Melanoma Therapies
Both single active chemotherapeutic agents and biological therapies in combination have been considered as therapeutic options for the treatment of advanced melanoma. Dacarbazine, or 5-3,3-dimethyltriazeno-imidazole-4-carboxamide (DTIC), is a single agent therapy that is approved for the treatment of melanoma with response rates ranging from only 11% to 25% 37 and 2% complete remission. 38 DTIC causes both growth arrest and cell death by methylating nucleic acids and/or direct DNA damage.37,38 Other treatment options for melanoma include temozolomide (TMZ), a DTIC derivative, and an oral DNA alkylating agent, which has been approved for the treatment of malignant gliomas. 39 TMZ is also under investigation for combination with radiation therapy in patients with metastatic melanoma in the brain. 40 At this time it appears that more effective chemotherapeutic agents are needed. Because of the current interest in combination therapies, functional foods and dietary supplements are garnering scientific interest as well as support.
Tyrosinase-Based Therapies
The ability of tyrosinase to indirectly activate drugs has been explored. In particular, it has been demonstrated that tyrosinase is able to contribute to the oxidation of the hydrazine group in the anticancer agent procarbazine. 41 Tyrosinase is able to contribute via oxidation of substrates, namely, phenols such as quercetin, to reactive species.42,43 These reactive o-quinone species are then able to oxidize the hydrazine linker on procarbazine and initiate procarbazine’s antitumor activity. 41 Tyrosinase also mediates the release of cytotoxic agents from carbamate and urea prodrugs via a cyclization–drug release mechanism. 44 In murine models, tyrosinase-activated conditionally replicative adenoviruses have been reported to enhance prolongation of survival in mice with experimental brain tumors. 45 Thus, it is that the oxidized substrates of tyrosinase directly promote antitumor effects not tyrosinase itself. This is important because it provides a form of selectivity where only cells that express tyrosinase will be exposed to heightened levels of the oxidized or activated form of quercetin.
Potential of Tyrosinase-Activated Quercetin in Cancer Treatment
Absorption of Quercetin
Phenolic dietary compounds can produce the same cell-specific toxicities as described above but have several advantages because they (a) are readily available in food, (b) have many complementary anticancer properties, (c) can be selectively “activated” by tyrosinase, and (d) could protect some normal tissues. 46 The flavonoid quercetin fits all these parameters and will be discussed as a dietary component that can be used to target tyrosinase activity for prevention or as a complementary therapy to conventional cytotoxic therapies.
Quercetin is one of the most studied flavonols and is ubiquitously present, usually in its glycosylated form, in fruits and vegetables. 47 Daily intake is calculated to range between 6 and 31 mg/d, 48 and supplementation of aglycosylated quercetin can be increased to 2 g/d. 47 Potential health effects of bioactive compounds such as quercetin depend on their bioavailability after oral administration (Table 1). It has been illustrated that the concentration of quercetin dictates whether it will have antioxidant/anti-apoptotic properties (<10 µM), both pro-apopototic and antioxidant effects (10-100 µM), or pro-oxidant/pro-apoptotic effects (>100 µM).46,49 It is likely that the pro-apoptotic effects desired for treatment of existing cancer will only be achievable by direct administration of quercetin intravenously and only maintainable for a short period of time, 50 such as an adjuvant for chemotherapy. However, dietary administration could be of more use in preventative medicine by maintain a lower circulating concentration for a long period of time. 46
Achievable Quercetin Concentration in Humans and Rats
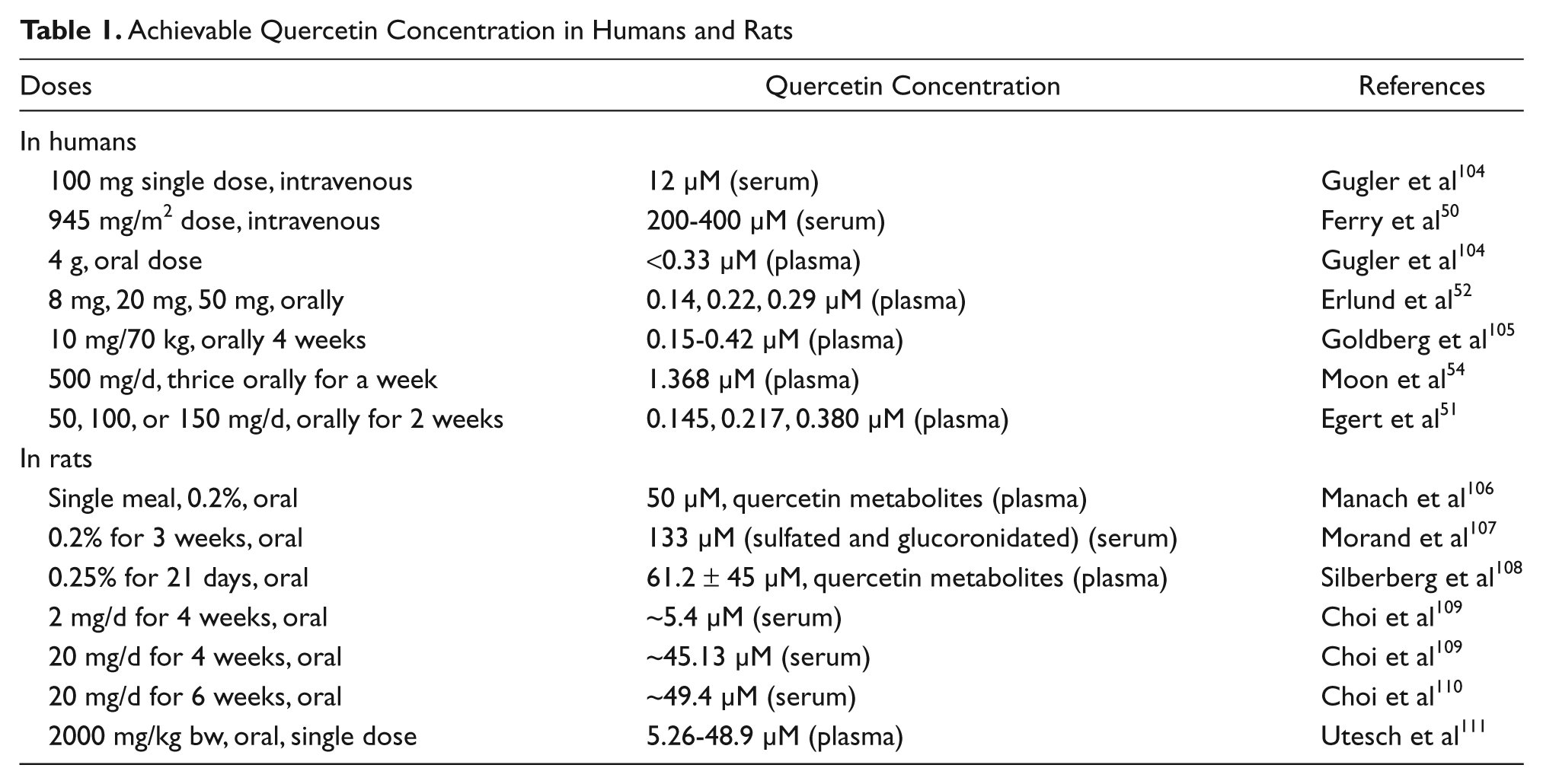
In plants and plant-derived foods, quercetin is mainly present as various glycosides. 47 Data on the bioavailability of quercetin aglycone or quercetin glycosides vary substantially based on different species (Table 1). Although the exact mechanisms are still being explored, it is generally accepted that quercetin absorption, digestion, and bioavailability depend on the following factors: the type and location of glycoside bound, other intestinal contents at the time of ingestion, and postdigestion conjugation forms. 47 Despite challenges in determining how quercetin reaches circulation, it is clear that it is does so (Table 1). Perhaps the most robust examples have come from studies on orally administered aglycosylated quercetin in humans. These studies show an increase in quercetin conjugates and metabolites (isorhammetin and tamarixetin) in blood measurements.47,51
Metabolism of Quercetin
Although the pharmacokinetics of quercetin have not been extensively studied, most of the results obtained so far are promising. Intravenous administration of quercetin from a phase I clinical trial demonstrated median clearance estimate of 0.28 L/min/m2, and antiproliferative effects were noted. 50 The half-lives of orally administered quercetin metabolites have been observed to be 15 to 28 hours,52,53 whereas quercetin aglycone’s half-life is only 3.5 hours in plasma. 54 This decreased half-life of free quercetin may be circumvented by conjugation of quercetin into one of its metabolites by xenobiotic metabolizing enzymes before entering circulation. Then, once reaching the target tissue, quercetin may return to its aglycosylated form and/or be activated to its pro-oxidant form by enzymes such as tyrosinase. 46
Since we are proposing that quercetin be used in conjunction with other chemotherapy, it is important to address the issues of drug–nutrient interactions. Quercetin is able to modulate the activity and/or expression of phase I and II drug-metabolizing enzymes, which could lead to significant changes in the metabolism of coadministered drugs. 55 Studies looking only at chemotherapeutic drugs and quercetin have shown that this bioactive exerts a beneficial synergistic effect when coadministered. 56 However, to ensure safety, further studies are needed to determine the effects of quercetin on a greater diversity of anticancer drugs and nonrelated drugs that may be prescribed to patients during cancer therapy, such as anticoagulants and antidepressants.
Polyphenol Activation
Tyrosinase is able to convert monophenols and o-diphenolic compounds to o-quinones, which are naturally cytotoxic and can be exploited (Figure 2). 57 Nontoxic phenol and catechol prodrugs have been shown to form toxic quinones, after oxidation by tyrosinase (Figure 2), within the vicinity of melanoma tumors. 58 Tyrosinase-mediated quercetin activation is a good example of this mechanism. Prodrugs, such as N-acetyl-4-S-cysteaminylphenol, have been shown to cause tyrosinase-dependent apoptosis. 59 Riley et al 6 studied the phenolic compounds that are readily oxidized by tyrosinase and suggested that its major cytotoxic action is because of the depletion of thiols. This suggests that tyrosinase is able to pair enzymatic depletion of thiols with activated quercetin-mediated mechanisms in order to more effectively promote cell death.

Strategies for tyrosinase-based therapies in tumors with null or mutant p53
Tyrosinase Uses Quercetin as a Substrate in Melanogenesis
Quercetin can act as a substrate for tyrosinase-related enzymes. 8 Quercetin has fewer hydroxyl groups than some other related compounds, making it easier for quercetin to cross the cell membrane and interact with enzymes. 60 Once inside the cell, quercetin-mediated cytotoxicity involves the oxidation of quercetin into electrophilic o-quinone compounds by enzymes such as peroxidases and tyrosinase, which are expressed in melanoma cells. Additionally, the now activated quercetin can further isomerize into quinone methides, which are more toxic than the parent compound itself. 8 In particular, Awad et al 61 established that quinone methide–GSH adducts form specifically in a melanoma cell line known to contain significant amounts of tyrosinase. Additionally, a time and dose dependent increase in melanin content, which is indicative of tyrosinase action, was observed after addition of 1 to 20 µM of quercetin to cultured HMVII melanoma cells. 62 Treatment of 10 µM of quercetin for 3 to 5 days caused an induction in tyrosinase activity, and treatment for 7 days increased tyrosinase expression as well as melanin synthesis in a 3-dimensional human epidermal model. 63 We now understand that quercetin is acted on by tyrosinase and forms an electrophile, which results in an increase in melanogenesis.
Potential Molecular Mechanisms of Quercetin-Mediated Treatments
Interaction With p53
It has been shown that the chemotherapeutic-enhancing properties of quercetin are linked to p53. 43 Tumors cells with wildtype p53 were sensitized to DTIC-mediated apoptosis by quercetin. In contrast, p53 mutant cells treated with quercetin and DTIC showed less sensitization but still did undergo apoptosis more often than controls. 43 Presumably this occurs due to the tyrosinase-activated quinone form of quercetin increasing the amount of ROS in tumor cells. This oxidative stress subsequently stimulates p53 and apoptosis. 64 This makes quercetin an ideal choice for co-chemotherapeutic treatment of tyrosinase-expressing tumors.
The tumor suppressor gene p53 continues to be a focal point in the study of the predisposition, progression, and prognosis of cancer. Because p53 is primarily wildtype in melanoma, modulation of p53 transcriptional activity and its downstream mediators are desirable therapeutic targets to increase damage and induce apoptosis in melanocytic tumors. 25 The p53 protein, which is present in cells at low levels, has a short half-life under basal conditions and is rapidly upregulated in response to stress. Through a feedback mechanism the protein is degraded by murine double minute 2 (MDM2), an E3 ubiquitin ligase, in the proteosome following ubiquination.65,66 Stabilization of p53 occurs after posttranslational modification events such as phosphorylation or by binding to proteins such as NAD(P)H quinone oxidoreductase 1 (NQO1). The activity of p53 is also central to tyrosinase-based therapies through apoptotic signaling mechanisms and gene transcription (Figure 3). Last, and perhaps most notably, conditions that induce p53 have been shown to induce tyrosinase. Transcriptionally activated p53, determined by activation of a downstream p53 target, was shown to increase tyrosinase mRNA expression. 67

Involvement of p53 in signal transduction events in tyrosinase-based therapies
As evidenced above, p53 is both relevant and central to tyrosinase-based therapies. Although many melanomas are wildtype p53, they may also express deletion mutants of p53 family members that interfere with p53’s ability to act as a transcription factor (Figure 3). One example is the p73 deletion mutant, ΔNp73. The p73 gene is a homologue of full-length p53 and is involved in the transactivation of p53 target genes. It can thereby work with p53 to induce apoptosis or inhibit cell proliferation. 68 The transcriptionally active form, or TAp73, is the active isoform of p73 and is frequently expressed in human tumors. 69 The terminally truncated form of p73 (ΔNp73) can antagonize p53.
Whereas the endogenous TAp73 is upregulated in response to DNA damage or treatment with chemotherapeutic drugs and provides antitumor activity, the upregulation of ΔNp73 promotes resistance to these drugs. 70 The functionality of the p53 family members primarily depends on the nuclear localization of p73. Studies demonstrate that the export of the truncated form, ΔNp73, to the cytoplasm is a major inducer of p53 functionality. 71 In earlier studies with hepatocellular and cholangiocellular carcinomas, p73 the inhibitory protein was reported to be located in the nucleus.72,73 It also found to be localized in the nucleus of undifferentiating neuroblastomas. 74 Recent reports indicate that quercetin can induce the redistribution of ΔNp73 into the cytoplasm and restore p53 activity. 75 Therefore, in addition to direct cytotoxic effects quercetin may have important modulator effects on inhibitory protein of p53 activity.
NQO1 and ROS
NQO1-mediated stabilization of p53 is an important factor in tyrosinase targeted therapies because NQO1 is under the control of the antioxidant response element, which is activated by ROS and electrophiles, such as activated quercetin. 42 It was observed that inhibition of NQO1 activity led to a marked destabilization of p53 as well as a profound decrease in apoptosis in response to ionizing radiation. 76 Metodiewa et al 77 also demonstrated that NQO1 functions in the regeneration of the parent compound quercetin from the activated o-quinone to enhance the cytotoxic properties of quercetin (Figure 4). The subsequent regeneration of tyrosinase-activated o-quinone back to the parent compound allows for a self-propagating stabilization of p53 and apoptotic induction. Furthermore, this regeneration of quinones allows for subsequent binding to GSH and GSH depletion-related cell death.42,78 Studies by our group in DB-1 melanoma cells revealed that quercetin oxidation caused a decline in the cellular bioreductive potential, an increase in ROS, and an induction of NQO1 in tyrosinase overexpressing cells. Therefore, ROS generated by tyrosinase-activated quercetin leads to increased NQO1, decreased GSH, and increased p53-mediated apoptosis in tumor cells. 42

Regeneration of the parental quinone compound
ATM, Bax, p73, and Quercetin
Quercetin also promotes an ataxia telangiectasia mutated (ATM)–dependent DNA damage response in tumor cells, which leads to the activation and phosphorylation of p53, as well as the activation of downstream mediators of p53.43,79 Specifically, phosphorylation of p53 on serine 15 by ATM was greatly enhanced by activated quercetin in cells that overexpressed tyrosinase. 43 This led to an increase in sensitivity and apoptosis. 43 Phosphorylation of p53 at serine 15 is thought to interfere with degradation of p53, leading to stabilization and increased transcriptional activity. Bcl-2 associated X protein (Bax), a related pro-apoptotic protein, has also been shown to increase in activity following quercetin treatment, especially in cells overexpressing tyrosinase. 65 Bax is a member of the Bcl-2 family and is a multiple domain protein that promotes an apoptotic response following transcription by p53. 65 Increased Bax mRNA production was also demonstrated in tyrosinase overexpressors, indicating gene transcription. 42 Taken together, tyrosinase-activated quercetin is able to induce apoptosis via induction of ATM, which activated p53 and then p53 activates Bax.
Recently, it has been found that cell death of chemoresistant DB-1 melanoma cells was achievable at lower dosages of TMZ only when 75 µM quercetin was administered after TMZ. The mechanism elucidated involves increased phosphorylation of ATM and increased activation of p53. 75 It should be noted that TMZ treatment alone leads to increased levels of an isoform of p73, which localizes to the nucleus. The accumulation of p73 in the nucleus impairs p53’s transcriptional activating abilities. 80 The addition of quercetin to the treatment regimen leads to movement of p73 out of the nucleus and allows for p53-mediated apoptosis to occur. Given that tyrosinase is overexpressed in melanoma and leads to increased p73 activation, quercetin is an ideal candidate for treatment of this cancer. 75
PUMA, Noxa, and Quercetin
Other important products that could be triggered by quercetin-induced p53 in melanoma are the proapoptotic proteins p53 upregulated modulator of apoptosis (PUMA) and Noxa. Both these proteins have been identified as p53-inducible genes. As we have previously discussed, p53 is induced by quercetin in melanoma cells. 42 PUMA is a BH3-only bcl-2 family member and is localized in the mitochondria to mediate p53-induced apoptosis. 81 PUMA binds Bcl-2-xL, a downstream anti-apoptotic protein, causing the release of Bax. Bax then translocates to the outer mitochondrial membrane inducing the release of cytochrome c from the mitochondria. Cytochrome c then activates procaspase 3 and 9, which is the committed step in the apoptotic cascade.81-83 Antisense inhibition of PUMA showed a decrease in p53-induced apoptosis. 82 Moreover, PUMA knockouts can recapitulate the apoptotic dysfunction and deficiencies that p53 knockouts exhibit.81,82 It was also observed that melanoma tumors transfected with PUMA do not proliferate as fast as negative controls. 84 This confirms the important role that quercetin and p53-inducible genes can play in the programmed cell death of melanocytic tumors.
P21 and Senescence
In addition to cytotoxicity, replicative senescence induced by quercetin activation could be an important mechanism for improving therapy. Replicative senescence is defined by the phenomenon in which cells undergo a finite number of cellular divisions and enter into a permanent state of cell cycle arrest. Rapid senescence can occur following cellular stresses, such as oncogene activation, DNA damage, and oxidative stress. Integral players in senescence are p53 and the retinoblastoma protein (Rb), which are both activated at the beginning of senescence. On activation of p53 in response to damage or cellular stress, activation of an important cell cycle regulatory protein, p21, occurs. 36 P21 prevents entry into S phase, sustains G2 arrest, prevents re-replication of DNA, and activates Rb. 36 Induction of p53 with activated quercetin, followed by subsequent activation of p21, could induce cell cycle arrest or senescence, in addition to the apoptotic cascade demonstrated with Bax activation. It has been shown that quercetin in addition to the induction of apoptosis can also work through inhibition of proliferation.85,86
Neuroblastoma as a Model
Role of P53 in Neuroblastoma
Inactivation of p53 is prevalent in solid neuroblastic tumors and is a likely mechanism in cytoplasmic sequestration of p53. Cytoplasmic p53, which is unable to play its key role as a vital transcription factor, and defective translocation have been observed in the majority of primary undifferentiated neuroblastoma tumors.35,36 The inactivation of p53 provides a good model to study the effects of activated quercetin in an aberrant p53 tumor type. Initial response to chemotherapy is common in these tumors, unlike many adult malignancies. However, continued therapeutic response is the treatment barrier and is likely due to inactivation of p53 after cytotoxic therapy. Following initiation of treatment, inactivation of p53 via cytoplasmic retention attenuates tumor response to DNA damage and cell cycle arrest. 35 This leads to the decrease in sensitivity to chemotherapy seen in neuroblastoma over time and indicates a role of wildtype p53 in tumor death. 87
Quercetin-Mediated p53-Independent Apoptosis
Activated quercetin-derived quinones are able to mediate cell death with or without induction of p53.88,89 Among the P53-independent apoptotic pathways, quercetin is hypothesized to bring about endoplasmic reticulum stress via induction of heat shock proteins (HSPs). 90 The potential mechanisms by which tyrosinase-activated quinones can bring about cell death make them exceptionally versatile options to pair with existing neuroblastoma chemotherapeutics. We will discuss specific possibilities and mechanisms in the review below.
Noxa and PUMA can also be regulated independently of p53 in response to proteasome inhibitors or compounds that induce ROS/endoplasmic reticulum stress or the unfolded protein response (UPR). 91 It has even been purposed that PUMA or Noxa may be more effective at inducing apoptosis than p53 in melanoma.92,93 Quercetin is also thought to be involved in the induction of this p53-independent UPR pathway. 90 This alternate pathway is especially pertinent when addressing melanocytic tumors who have dysregulation or inactivation of p53. Activated quercetin and other polyphenols can inhibit the endoplasmic reticulum function preserving HSP family members and induce endoplasmic reticulum stress. 90 In response to ROS the endoplasmic reticulum transmembrane protein kinase, PKR-like endoplasmic reticulum kinase (PERK), activates the activating transcription factor 4 (ATF4) and then a cascade of proteins are activated leading to apoptosis (Figure 2). Specifically, CCAAT/enhancer binding protein homologous protein (CHOP) and Gadd34 are activated in this process. 94 PERK may also inhibit the anti-apoptotic protein bcl-2. CHOP, as well as caspase 12, may also in part activate PUMA and NOXA independently of p53 in response to endoplasmic reticulum stress. 94 PUMA and Noxa have also be shown to be transcriptionally regulated by the transcription factor E2F, and E2F1 expression is p53 independent. 95 In response to endoplasmic reticulum stress, E2F1 has been shown to induce expression of Noxa and PUMA in neuroectodermal tumors and mediate crosstalk between the endoplasmic reticulum and the mitochondria, with Noxa serving to activate Bak, leading to mitochondrial permeability, cytochrome c release, and apoptosis. 96 Thus, quercetin can act to provoke apoptosis with or without the presence of nonfunctional or deleted p53 and subsequent PUMA-induced apoptosis may be mediated through TAp73. Increased expression of ΔNp73alpha and beta isoforms decreases PUMA-mediated apoptosis. 97 As described quercetin could cause redistribution of ΔNp73 and increase cell death. The potential for activated quercetin to enhance this process should be explored.
Inactivated p53 and Small Molecule Therapy
Circumventing p53 inactivation has become of interest in treating neuroblastoma. Small molecule MDM2 antagonists have demonstrated restoration of the p53 pathway. 98 MDM2 upregulation in neuroblastoma is thought to have a role in the translocational dysfunction and inactivation of p53. Van Maerken et al 98 investigated the use of nutlin-3, an MDM2 antagonist, in neuroblastic cell lines and saw induction of nuclear relocalization of p53 and activation of downstream p53 mediated genes. Activity of p53 was measured by induction of p21, and cells treated with nutlin-3 showed activation of p21 and subsequent increase in number or cells arrested in G1 and decreased the fraction of cells in S phase. Activation of pro-apoptotic proteins, Bax and PUMA, following nutlin-3 treatment was also seen and apoptosis was induced. This indicates that there was reactivation of p53-mediated apoptosis. 98 Therefore, the use of agents that can induce a p53-independent cell death would be useful in treating neuroblastoma. There are reports of neuroblastoma with tyrosinase activity. 99 Quercetin would be ideal because it can cross the blood–brain barrier and can induce expression of tyrosinase in these tumors. Coupled together with its antitumor properties and ability to induce p-53 independent cell death, quercetin could complement traditional therapies.
Conclusion
As we have described above, quercetin is an ideal candidate for targeting melanocytic tumors because of their unique expression of tyrosinase.3,4 Tyrosinase is able to activate quercetin, which can form a reactive o-quinone.57,58 This o-quinone can then trigger p53-dependent apoptosis and cell cycle arrest.42,57,85,86 Additionally, the reactive o-quinone can increase ROS and trigger endoplasmic reticulum stress–mediated cell death.76,85,90,100
Although quercetin is unlikely to be potent enough of antitumor treatment on its own, it is an ideal candidate for use in combination with traditional therapies. Research done with quercetin and other co-chemotherapeutic agents shows potential thus far. 43 We acknowledge, however, that the area of quercetin and drug interactions does need to be further explored and especially studied in oncology patients who may be prescribed multiple drugs at one time. Additionally, as with many bioactive compounds, sufficient target tissue concentrations will likely only be achieved by intravenous administration. 50 This could conceivably be achieved in the health care setting by simultaneous administration of quercetin with other chemotherapeutic agents. Given that tyrosinase is highly expressed in tumors of melanocytic subtypes, one would expect that activated quercetin would reach higher therapeutic effectiveness in these tumors after intravenous administration. Even in oncogenesis preventative medicine (ie, lower dosage, oral quercetin supplementation for antioxidant effects), one would expect that tissues expressing tyrosinase would selectively accumulate relatively elevated levels of oxidized quercetin that could aid in targeted melanoma prevention.
Promising work by Altaner et al 7 has suggested that tyrosinase gene expression can use prodrug activation not only for melanoma treatment but also could be applied to nonmelanocytic tumors. This is achievable in cells that are transfected with tyrosinase/tyrosinase mutants. 7 Additionally, extensive work by Russo et al101-103 has revealed that the combination of quercetin with specific antibodies leads to CD95 and tumor necrosis factor–related apoptosis-inducing ligand-mediated apoptosis in leukemia cell lines. So the future for quercetin as a co-chemotherapeutic may involve more than just melanocytic tumors.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
