Abstract
Aims/Introduction
Vitamin D deficiency is prevalent among individuals with diabetic foot infections (DFIs) and can influence a range of patient-related parameters. Hence, we aimed to find the association of serum vitamin D levels with various clinical, biochemical, and inflammatory parameters, as well as the microbial environment in the wounds of DFI patients.
Materials and Methods
From September 2022 to March 2024, 169 DFI patients participated in cross-sectional research at the hospital. Utilising an electrochemiluminescence immunoassay, vitamin D levels were determined. Vitamin D receptor (VDR) and Cathelicidin (LL-37) were estimated using ELISA Kits. The tissue culture plate technique was used to analyze biofilm formation. Patient-related parameters were obtained from medical records.
Results
The vitamin D status of DFI patients indicated that 70.4% were deficient, 19.5% were insufficient, and 10.1% were sufficient. The median vitamin D, VDR protein, and LL-37 values were 15.3 ng/mL, 0.832 ng/mL, and 1.54 ng/mL, respectively. Biofilm formation was seen in 84.36% of bacteria from vitamin D-deficient DFI patients (P < .001). Vitamin D levels were negatively correlated with ulcer duration, hospital stay, white blood cell count, neutrophil-lymphocyte ratio, C-reactive protein, and systemic inflammation index (r = −0.787, −0.849, −0.6, −0.475, −0.402, and −0.521, respectively; P < .001). However, vitamin D levels were positively correlated with VDR and LL-37 levels (r = 0.988 and 0.944, respectively; P < .001).
Conclusions
The majority of DFI patients exhibited vitamin D deficiency, which was significantly associated with most of the clinical, biochemical, and inflammatory parameters, in addition to the microbial environment within the wound.
Introduction
The International Diabetes Federation reports that diabetes mellitus (DM) affects approximately 10% of adults worldwide, equivalent to 589 million adults aged 20-79 years. 1 Projections predict an increase to 853 million by 2050. 1 A recent study estimates that 101 million people in India, or 11.4% of the country's population, currently have DM. 2 Various complications can arise in DM patients with uncontrolled blood sugar levels, with diabetic foot ulceration being one of the most critical. 3 These ulcers are prone to the development of diabetic foot infections (DFIs), which may result in hospitalisation, lower limb amputation, disability, or even death. 4 Approximately 10%-15% of patients with DFIs require multidisciplinary treatment. Of these cases, 20% classified as moderate or severe may necessitate amputation. 5 Patients with DFIs face a 2.5-fold increase in mortality risk. 5 In 2017, a survey indicated that the cost of managing DFIs in India was approximately 1572 USD per patient. Data shows that patients with DFIs incur expenses four times greater than those without DFIs. 6
Several patient-related parameters can influence the development and severity of DFIs. These include age, gender, dietary pattern, the duration of diabetes, glycated haemoglobin (HbA1c), C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), and systemic immune-inflammation index (SII).7–13 Furthermore, hyperglycaemic state in these patients generates oxidative stress and sets off a chain of unfavourable consequences directly related to the deteriorating DFI prognosis. 3 Among these consequences are a compromised immune system, chronic inflammation, microvascular dysfunction, challenges in wound healing, and the formation of biofilms.3,14
Vitamin D, initially employed for treating nutritional rickets, is now widely used because of its numerous physiological benefits.15,16 It functions by binding to the vitamin D receptor (VDR) found in skin and immune cells. 17 Additionally, vitamin D works as an antibiotic by aiding the formation of antimicrobial peptides (AMPs) such as defensins and cathelicidin (LL-37) in different species.18,19 In humans, LL-37 is a small AMP that demonstrates proangiogenic and immunomodulatory properties. Consequently, it has the capacity to improve the recovery process of wounds. 20 Vitamin D is one of the factors that regulate VDR and LL-37 expression.21,22 Therefore, any disruptions in maintaining optimal vitamin D range could potentially impact the production of VDR and LL-37 and subsequently influence innate immunity and thereby affect wound healing.23,24 Moreover, the growing challenge of treating patients with DFI is partly due to the existence of bacteria that create biofilms. 25 Furthermore, research demonstrates that a deficiency in vitamin D correlates with elevated levels of inflammatory cytokines, reduced LL-37, and the development of biofilms.25–27 Evidence is also available on the vitamin D-LL-37 pathway's role in countering microbial protein. 28 Therefore, individuals with diabetes who have vitamin D deficiency are more susceptible to chronic and recurring DFI.29,30 Also, vitamin D levels are associated with low-cost and widely accessible inflammatory markers, including white blood cell (WBC), CRP, NLR, and SII, indicating that vitamin D may have a part in controlling inflammation.31–33
Considering the aforementioned evidence, this study aimed to estimate serum vitamin D levels in DFI patients and to determine its relationship with various clinical (age, duration of diabetes, specifics regarding their current ulcer, and hospital stays), biochemical (HbA1c, VDR protein and LL-37 levels), and inflammatory (WBC, CRP, NLR, and SII,) parameters as well as the microbial environment (biofilm) present in the wound.
Methods
Study Design and Ethical Approval
A cross-sectional study was carried out with 169 DFI patients who were admitted from September 2022 to March 2024 in the Department of General Surgery of the tertiary healthcare hospital. The institutional ethics committee (IEC) of the hospital has granted approval for the study, and the Helsinki Declaration (2013) ethical principles for human biomedical research were followed. The study documentation followed the STROBE guidelines. 34
Patient Enrolment
Patients with diabetes, both male and female, aged 18 and older, diagnosed with DFI according to the IWGDF/IDSA infection grading system, and those who provided consent were included. 35 The exclusion criteria encompassed patients with hepatic and renal disorders (which affect vitamin D metabolism), coeliac disease, chronic pancreatitis, Crohn's disease, cystic fibrosis (which impacts vitamin D absorption), and peripheral vascular disease. In addition, those who received immunosuppressants, chemotherapeutic agents, or corticosteroids within the preceding 30 days, and pregnant or lactating women were also excluded.36,37 Moreover, patients on vitamin D supplements or any specific diet (fortified with vitamin D) and who did not consent to participate were excluded from the study. Additionally, patients with DFI whose wound samples indicated negligible bacterial growth were also excluded from the study. 38
In this study, a total of 424 patients were screened for eligibility. Among these patients, 154 had peripheral vascular disease, 21 had renal disorders, 9 had hepatic disorders, 57 had wound cultures that demonstrated negligible bacterial growth, and 5 patients did not give consent. As a result, all of these patients were excluded, leaving 178 who met the inclusion criteria. Furthermore, we excluded 9 patients because the preserved organisms from their wound samples displayed non-significant growth when regrown for biofilm analysis. Consequently, the final analysis included data from 169 patients with DFI. A flowchart illustrating the patient selection procedure is presented in Figure 1.
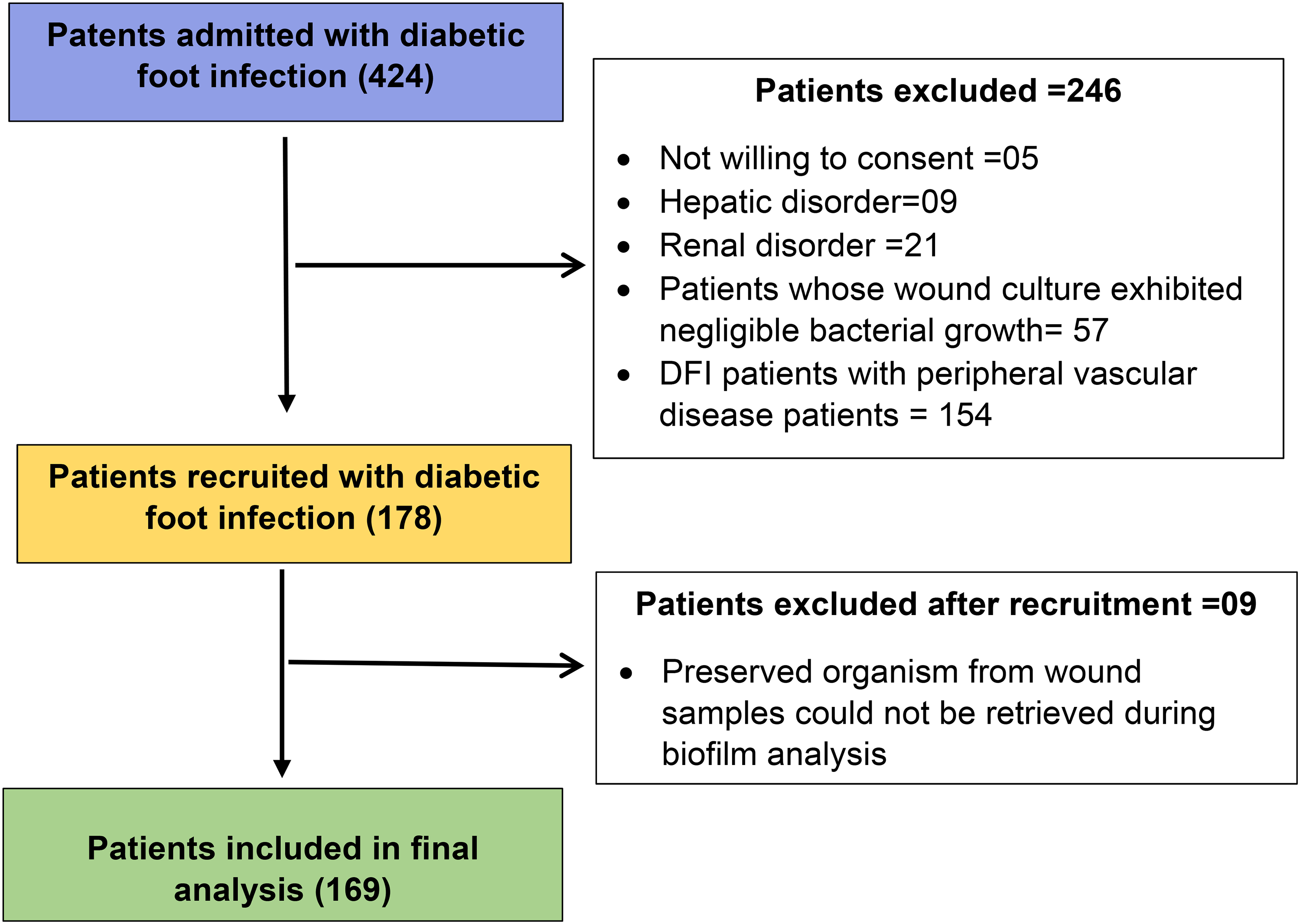
Study Flowchart for the Selection of DFI Patients.
Data Collection Methods
Most of the data pertaining to patient-related parameters such as age, duration of diabetes, specifics regarding their current ulcer, hospital stays due to DFI, HbA1c levels, WBC count, and CRP levels were collected from the medical records. Dividing neutrophils by lymphocytes from the differential blood cell counts test yielded the NLR values. SII value = (Platelet × Neutrophil) / Lymphocyte count.
Measurement of Serum Vitamin D Levels in DFI Patients
Electrochemiluminescence immunoassay in the COBAS 6000 analyser was used to measure vitamin D levels in 4 mL blood samples from DFI patients at the hospital. Patients with vitamin D levels below 20 ng/mL were deficient, those between 20 and 30 were insufficient, and those over 30 were sufficient. 39
Estimation of Vitamin D Receptor and LL-37 Levels in Serum Samples of DFI Patients
4 mL of blood was taken from DFI patients utilising vacutainers to estimate the levels of VDR and LL-37 using the ELISA technique. The blood sample was centrifuged at 3000 rpm for 15 min to separate serum, which was stored at −80 °C until further analysis. The human VDR kit from Elabscience, USA (catalog number E-EL-H2043) was utilised for VDR estimation. 40 For LL-37, the estimation was performed using the antibacterial protein LL-37 kit from Elabscience, USA (catalog number E-EL-H2438). 41 The procedures for estimating both VDR and LL-37 were conducted following the manufacturer's specifications.
Analysis of Biofilm Formation in Bacterial Isolates from Wound Samples of DFI Patients
200 bacterial isolates collected from wound samples of DFI patients were subjected to biofilm production test at the Department of Microbiology of the hospital. A matrix-assisted laser desorption ionisation time-of-flight (MALDI-TOF) - VITEK MS (BioMerieux, Marcy l’Etoile) system was used to identify bacterial isolates from the wounds of DFI patients. Subsequently, the VITEK 2 system (BioMérieux, Inc., Durham, NC) was used to test the samples for their antimicrobial susceptibility. 42
The tissue culture plate method was employed to assess the biofilm-producing capability of bacterial isolates. 43 A culture of bacteria was grown in 1% glucose-enriched tryptone soy broth (TSB) (HiMedia) and then transferred to a 96-well polystyrene tissue culture plate (TCP) (Tarsons, India) after being diluted 1:100 with fresh media. Strains of Staphylococcus aureus (ATCC 25923) and Pseudomonas aeruginosa (ATCC 27853) were utilised as positive and sterile TSB as a negative control. The biofilm underwent fixation (with 2% sodium acetate), staining (with 0.1% crystal violet for 5 min), and measurement utilising a micro-ELISA auto reader (Thermo Scientific, Multiskan Go). The experiment was performed three times.
The interpretation of the results was carried out in accordance with Stepanović et al 44 Average optical density (OD) values were calculated for all tested bacterial isolates and the negative control. We referred to the average OD of the bacterial isolates as (ODei), while that of the negative control was (ODnc). The results from the test were interpreted as follows: if the average ODei value was less than or equal to ODnc, it indicated that the bacteria did not produce biofilm. Conversely, if the average ODei value exceeded ODnc, it indicated that the bacteria produced biofilm. 45
Statistical Analysis
All data were analysed using Jamovi software version 2.4.8.0. The normality of the data was evaluated with the Shapiro-Wilk test. Measurement results were non-normal and provided as median (IQR). Vitamin D status in DFI patients was compared to clinical, biochemical, inflammatory, and microbiological characteristics using the Kruskal-Wallis test. Dwass-Steel-Critchlow-Fligner (DSCF) was employed for post-hoc analysis. The chi-square test analysed relationships among categorical variables. Spearman's correlation test examined DFI patients’ vitamin D levels and other parameters.
Results
Baseline Characteristics of Patients with DFI
The median age of the 169 patients with DFI was 59 years, and the majority of them were male (86.9%). Most participants (79.9%) adhered to a mixed diet, and the median body mass index was 22 kg/m², with an IQR of 18 to 26.1. Approximately 48.5% reported consuming alcohol, whereas 40.2% identified as smokers. The median diabetes duration in this cohort was 10 years. 74.6% had a prior episode of DFI, while 25.4% had a history of amputation. Among the DFI patients, merely 28.4% exhibited comprehension of appropriate foot care and hygiene protocols. The median duration of ulcers was 23 days, with patients generally encountering between 1 and 2 ulcers. The median dimensions of the ulcers were 2 cm in both length and width, and the median duration of hospitalisation for diabetic foot infection treatment was 8 days. The HbA1c level was 9%, signifying inadequate regulation of blood glucose levels. The median concentrations of VDR protein and LL-37 were 0.832 ng/mL and 1.54 ng/mL, respectively. The median serum vitamin D concentration was 15.3 ng/mL, with 70.4% of DFI patients categorised as vitamin D deficient, 19.5% as insufficient, and 10.1% as sufficient. The median WBC count was 11.8 × 10³/μL. The median counts for neutrophils, lymphocytes, and platelets were 7.3 × 10³/μL, 1.37 × 10³/μL, and 405 × 10³/μL, respectively. The median NLR was 5.27, and the median SII was 2323, signifying overall inflammation in these patients. Table 1 outlines the baseline characteristics of patients with DFI.
Baseline Characteristics of Patients with Diabetic Foot Infections (N = 169).

BMI, Body mass index; CRP, C-reactive protein; DFI, Diabetic foot infection; HbA1c, Glycated haemoglobin; IQR, Interquartile range; NLR, Neutrophil-lymphocyte ratio; N, Number of patients; SII, Systemic immune-inflammation index; T2DM, Type 2 diabetes mellitus; WBC, White blood cells; VDR, Vitamin D receptor; LL-37, Cathelicidin.
Association of Serum Vitamin D Status with Clinical, Biochemical, Inflammatory, and Microbiological Parameters in DFI Patients
Compared to both sufficient and insufficient vitamin D groups, a significant increase in various parameters, such as number of ulcers, ulcer size (length and breadth), duration of existing ulcers, hospital stays, HbA1c levels, WBC count, CRP levels, neutrophils, lymphocytes, NLR, and SII, were observed in the vitamin D-deficient group (P < .001). On the other hand, a significant decrease in the levels of VDR and LL-37 was noted in both the vitamin D-deficient and insufficient groups compared to the sufficient group (P < .001), as shown in Table 2.
Association of Serum Vitamin D Status with Various Clinical, Biochemical, and Inflammatory Parameters in DFI Patients.
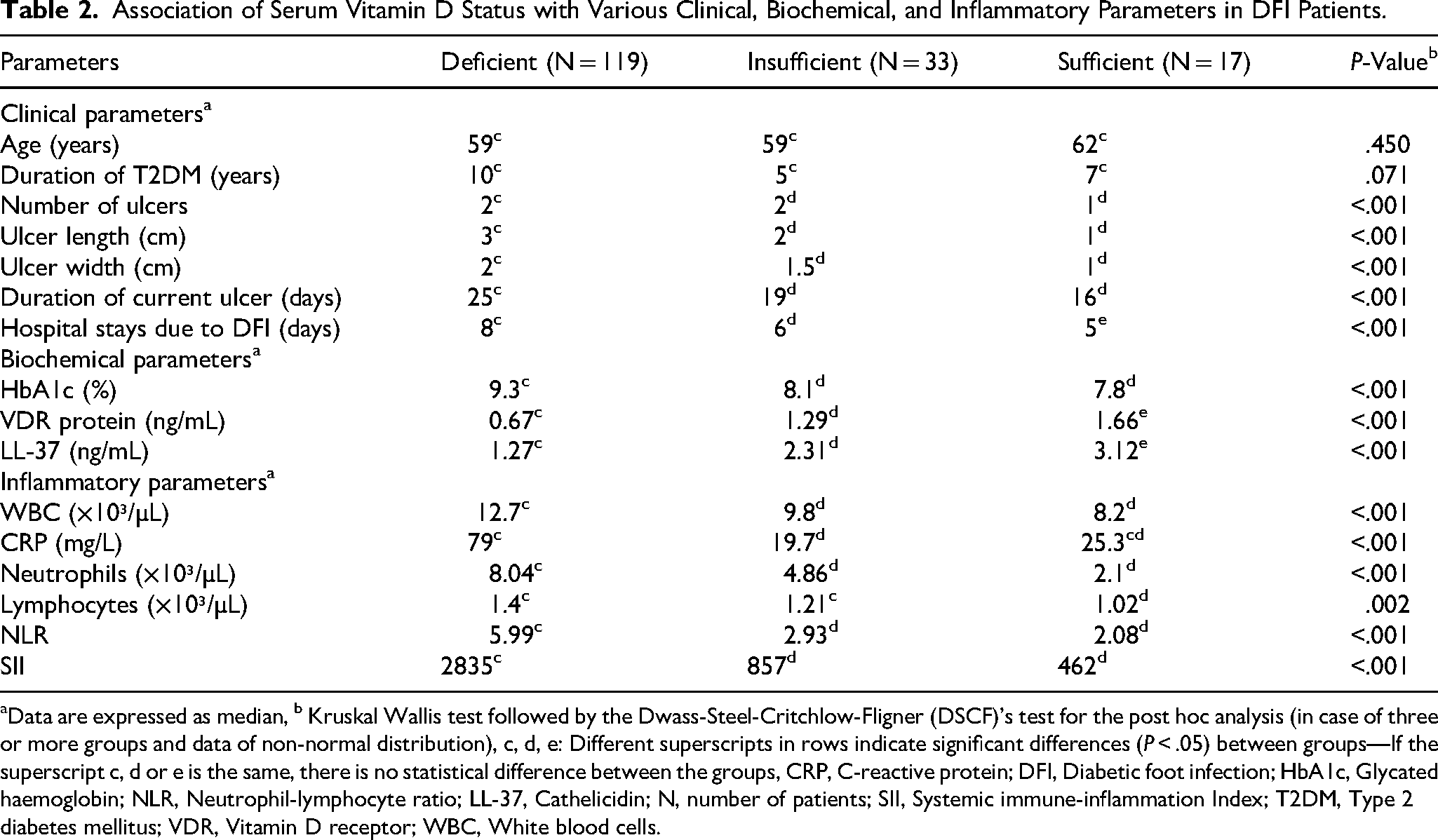
Data are expressed as median, b Kruskal Wallis test followed by the Dwass-Steel-Critchlow-Fligner (DSCF)'s test for the post hoc analysis (in case of three or more groups and data of non-normal distribution), c, d, e: Different superscripts in rows indicate significant differences (P < .05) between groups—If the superscript c, d or e is the same, there is no statistical difference between the groups, CRP, C-reactive protein; DFI, Diabetic foot infection; HbA1c, Glycated haemoglobin; NLR, Neutrophil-lymphocyte ratio; LL-37, Cathelicidin; N, number of patients; SII, Systemic immune-inflammation Index; T2DM, Type 2 diabetes mellitus; VDR, Vitamin D receptor; WBC, White blood cells.
Association of Serum Vitamin D Status with Biofilm-Producing Bacterial Isolates in DFI Patients
The bacteria frequently detected in wound specimens from patients with DFI are methicillin-susceptible Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae. Figure 2 shows the percentage of bacterial isolates obtained from these wound samples. A significant majority of bacteria (84.36%) isolated from the wounds of DFI patients with vitamin D deficiency exhibited biofilm production (P < .001), as illustrated in Table 3.
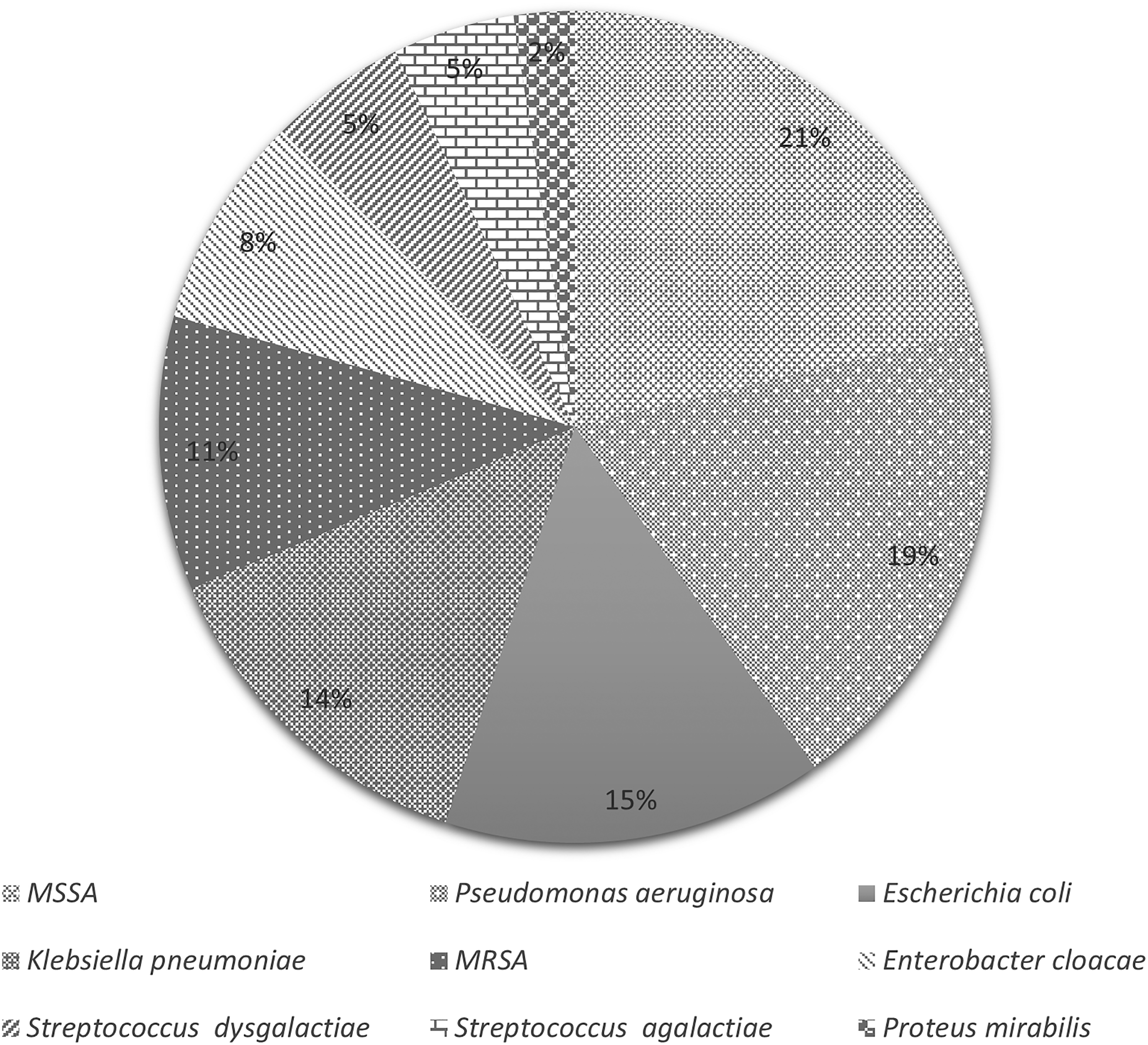
Bacterial Isolates from Wound Samples of DFI Patients.
Association of Serum Vitamin D Status with Biofilm-Producing Bacterial Isolates in DFI Patients.
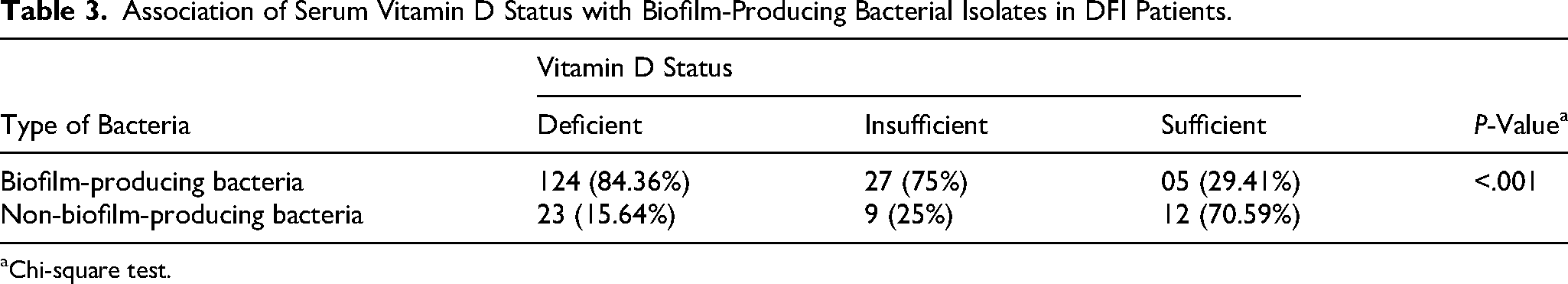
Chi-square test.
Correlation of Vitamin D Concentrations with Patient-Related Factors in DFI Patients
Vitamin D levels exhibited a significant negative correlation with ulcer duration, hospital stay due to DFI, WBC count, NLR, CRP, and SII (r = −0.787, −0.849, −0.6, −0.475, −0.402, −0.521, respectively; P < .001), while demonstrating a significant positive correlation with VDR and LL-37 levels (r = 0.988 and 0.944, respectively; P < .001). Additionally, HbA1c levels and lymphocyte counts exhibited a significant weak negative connection with vitamin D concentrations (r = −0.327; P < .001 and r = −0.18; P = .027, respectively). The age did not exhibit a significant correlation (r = 0.011; P = .883) with vitamin D concentration, as shown in Table 4.
Correlation of Vitamin D Concentrations with Patient-Related Factors in DFI Patients.
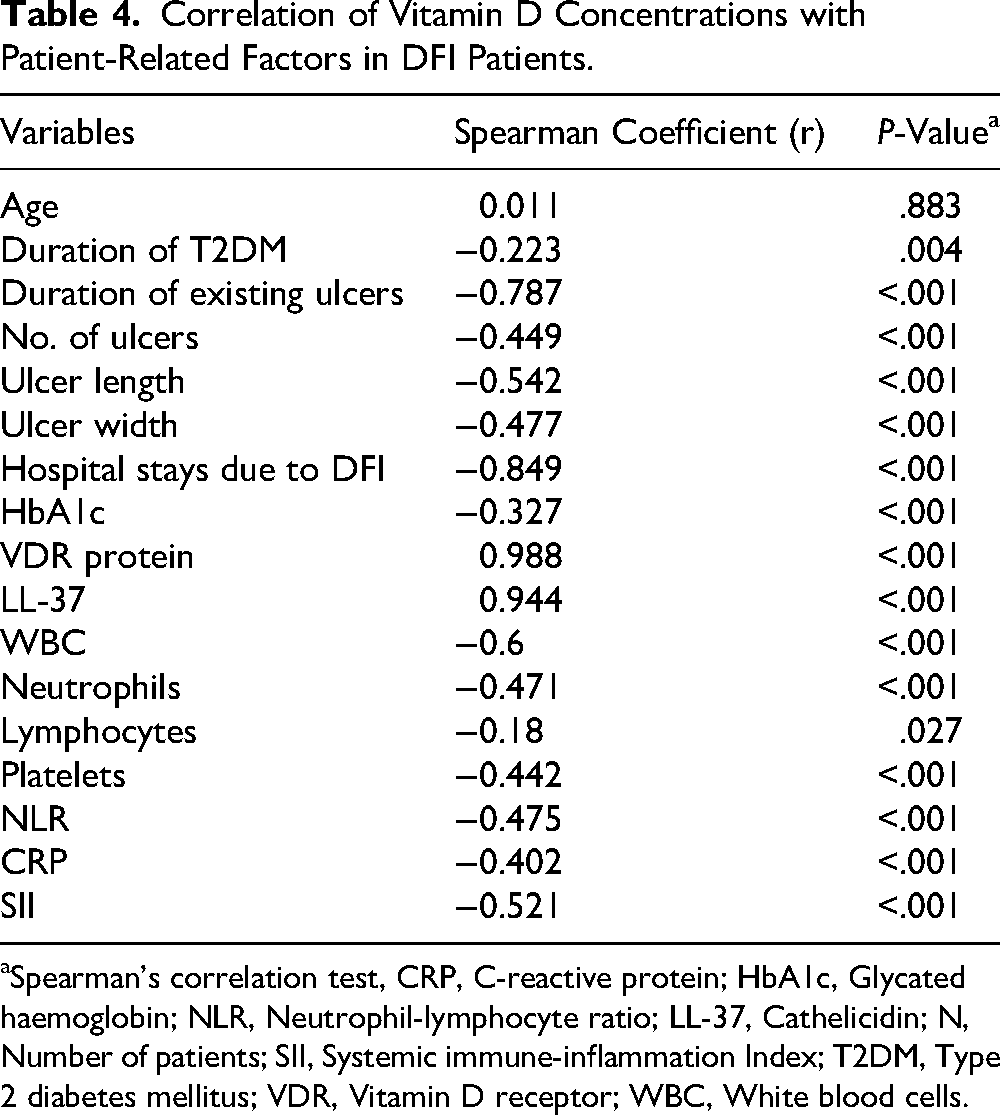
Spearman's correlation test, CRP, C-reactive protein; HbA1c, Glycated haemoglobin; NLR, Neutrophil-lymphocyte ratio; LL-37, Cathelicidin; N, Number of patients; SII, Systemic immune-inflammation Index; T2DM, Type 2 diabetes mellitus; VDR, Vitamin D receptor; WBC, White blood cells.
Discussion
The study's primary outcome revealed that, among 169 DFI patients tested for vitamin D levels, a significant majority were vitamin D-deficient (70.4%). This confirms past studies on vitamin D deficiency as a significant risk factor for developing DFI.3,29,46–48 Nonetheless, certain studies have indicated insufficient -linkage between vitamin D and DFI, which could be due to variations in the methodologies employed for assessing vitamin D levels.7,49,50 In our study, we employed an electrochemiluminescence immunoassay and assessed vitamin D levels, while the other investigations assessed through the ELISA technique.
A study conducted in China reported that patients with unhealed ulcers were vitamin D-deficient compared to those with healed ulcers, though levels were not statistically significant. 51 Similarly, the current study findings revealed that vitamin D-deficient patients with DFI had significantly bigger ulcer sizes (both in length and breadth), longer durations of ulcers, and extended hospital stays. However, a study conducted in Brazil demonstrated that patients with vitamin D-sufficiency exhibited similar ulcer characteristics to those with vitamin D deficiency. 52 The researchers suggested that deficiency of this vitamin might not be the primary cause of ulcers but rather a consequence of other factors. Nevertheless, this deficiency could still hinder the healing process. 52 Their trial results did not reveal a significant difference in ulcer size between the vitamin D groups and the placebo group after treatment; however, there was a trend suggesting potential statistical significance in wound size changes following vitamin D therapy. 52
In the present study, most inflammatory parameters, including WBC, CRP, NLR, and SII, were significantly elevated in the vitamin D-deficient group compared to the other vitamin D groups. Notably, the most significant differences in these parameters were observed primarily between the vitamin D-deficient group and the vitamin D-insufficient group. This supports earlier preclinical and clinical research showing vitamin D's anti-inflammatory benefits on inflammatory indicators.53–55
A northern Chinese study found that vitamin D deficiency indirectly reduced VDR transcription. 56 Additionally, Mandal et al found that low levels of vitamin D3 are associated with extremely low expression of VDR in individuals exhibiting complicated symptoms of leprosy. 23 These findings are consistent with our results, as we observed significant reductions in the concentrations of VDR and LL-37 in the vitamin D-deficient group, followed by the insufficient group. While the highest levels were found in the vitamin D-sufficient group. A study in tuberculosis patients found no definitive correlation between levels of vitamin D and LL-37. 57 However, in septic and non-septic ICU patients, a correlation between vitamin D status and levels of LL-37 was established. 58 The conflicting results indicate that differences in underlying illness conditions may influence the possible relationship between serum vitamin D and LL-37 levels. Another key finding was that HbA1c levels were significantly higher in the vitamin D-deficient group compared to the sufficient group. A similar trend has also been noted in studies conducted in the United States as well as in China.59,60 Vitamin D's role in glycaemic control and its indirect effects on wound healing may explain this difference. Vitamin D plays a crucial role in enhancing the body's ability to use insulin efficiently and regulate blood sugar levels. 61 This can assist in the management of diabetes and its related problems, like DFIs, by strengthening the immune system, decreasing inflammation, and facilitating tissue repair.62–64
Our study reveals that a significant majority of bacteria (84.36%) isolated from the wounds of DFI patients with vitamin D deficiency exhibited biofilm production. Individuals with diabetes frequently experience a persistent hyperglycaemic condition, fostering an optimal environment for microbial proliferation. 65 The bacteria frequently found in wound samples from DFI patients included methicillin-susceptible Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae. These microorganisms thrive and form biofilms.7,25 Biofilms typically consist of extracellular polymeric substances that are generated by stationary microorganisms. 66 Increased glucose levels promote the production of extracellular polymeric substances, leading microorganisms to shift to a sessile state and integrate into the biofilm matrix, which obstructs antibiotic penetration and diminishes treatment efficacy. 67 While a recent study indicates that vitamins D and K1 can effectively combat biofilms produced by specific Gram-negative bacteria, suggesting their potential efficacy in improving therapies for infections brought on by strains that are resistant to multiple drugs and biofilm-forming bacteria. 68 Nonetheless, additional in vivo research is required to substantiate these promising findings.
This study has certain limitations as it was carried out at a single centre with a limited sample size, hindering the ability to establish causality. Additionally, the TCP method employed for the analysis of bacterial biofilms was performed in vitro, which leaves the implications of biofilm presence in vivo still ambiguous. Moreover, numerous limitations inherent in vitamin D research complicate the establishment of definitive conclusions regarding the association between vitamin D status and the risk of diabetic complications. These are considerable differences seen among the studies, including variabilities in patients’ demographics and genetic, baseline vitamin D status, doses of vitamin D supplements, and primary endpoint selection. 69 It is important to note that the Endocrine Society no longer endorses specific serum vitamin D levels to define sufficiency, insufficiency, and deficiency. 70
Current clinical guidelines provide only limited support for vitamin D supplementation in the prevention of diabetes among individuals with prediabetes. 70 Collectively, the study findings showed that the majority of DFI patients exhibited vitamin D deficiency. This significantly impacted most of the clinical, biochemical, and inflammatory parameters, in addition to the microbial environment within the wound, leading to less favourable outcomes in DFI patients. The research highlights the importance of regularly checking vitamin D levels in patients with DFI and providing vitamin D supplements to address deficiencies. This approach aims to prevent recurrent infections and reduce the risk of amputations. Considering the clinical implications of this study, subsequent investigations should focus on implementing real-time testing to evaluate how the restoration of vitamin D levels affects the patient-related parameters in DFI patients. Therefore, the study suggests implementing a rigorously controlled, extensive, multicentric randomised trial utilising vitamin D supplements to assess the impact of restoring vitamin D levels on the outcome measures in patients with DFI.
Footnotes
Acknowledgments
The authors would like to express their gratitude to the Department of Health Research, Ministry of Health & Family Welfare, New Delhi, India, for providing funding through the grant-in-aid scheme (No.R.11014/31/2023-GIA/HR). Additionally, we thank the Manipal Academy of Higher Education, Manipal, India, for offering research support. We also appreciate the Department of Microbiology at Kasturba Medical College, Manipal, for providing the laboratory facilities necessary for sample processing. Dr. Saleena Ummer Velladath contributed to the research while affiliated with Manipal Academy of Higher Education, Manipal. Dr. Velladath‘s current affiliation mentioned is for correspondence purposes only. No foreign collaboration, funding. or material transfer were involved in this work.
ORCID iDs
Ethics Approval & Trial Registry Details
The study received approval from the Institutional Ethics Committee of Kasturba Medical College & Kasturba Hospital, Manipal, India, with reference no. IEC 676/2020 and was executed in accordance with the Helsinki Declaration 2013. Written informed consent was obtained from the patients after providing patient information sheets and ensuring the patients understood the objectives and purpose of the study. The study has been registered in the Clinical Trial Registry of India (CTRI) with registration number CTRI/2021/02/031281. Animal studies: N/A.
Authors' Contributions
S.S.M. and R.B. were involved in the conceptualization and design of the study. R.B. was involved in data collection for this study. R.B., S.J.K., S.S.P., B.B., and S.V. were involved in case diagnosis and data interpretation for this article. R.B., B.B., and S.V. were involved in processing the samples. V.K.S.G. and V.K. were involved in data analysis. R.B. and S.S.M. were involved in manuscript writing. S.S.P., S.U.V., G.S.R., R.C.H., M.R., C.M., and M.M. were involved in critically reviewing this article. S.S.M., S.U.V., M.R., B.B, C.M., and R.C.H. were involved in the acquisition of the funding for the study. All the authors approved the final draft of the manuscript.
Declaration of Conflicting Interests
The authors declare no conflict of interest. The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony or patents received or pending, or royalties. The funding sources were not involved in the study design, the collection, analysis, and interpretation of data in the writing of the report, or in the decision to submit the article for publication.
Data Availability Statement
Upon reasonable request, the corresponding author will provide the supporting data for the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant-in-aid (GIA) grant (No. R.11014/31/2023-GIA/HR), Department of Health Research, Ministry of Health & Family Welfare, New Delhi, India.
