Abstract
Background. The bone healing after fracture had a great impact on the patients’ life quality. However, how miR-7-5p participated in fracture healing has not been investigated. Methods. For in vitro studies, the pre-osteoblast cell line MC3T3-E1 was obtained. The male C57BL/6 mice were purchased for in vivo experiments, and the fracture model was constructed. Cell proliferation was determined by CCK8 assay, and alkaline phosphatase (ALP) activity was measured by commercial kit. Histological status was evaluated using H&E and TRAP staining. The RNA and protein levels were detected via RT-qPCR and western blotting, respectively. Results. Overexpression of miR-7-5p increased cell viability and ALP activity in vitro. Moreover, in vivo studies consistently indicated that transfection of miR-7-5p improved the histological status and increased the proportion of TRAP-positive cells. Overexpression of miR-7-5p suppressed LRP4 expression while upregulated Wnt/β-catenin pathway. Conclusion. MiR-7-5p decreased LRP4 level and further activated the Wnt/β-catenin signaling, facilitating the process of fracture healing.
Keywords
Introduction
Fracture has been regarded as a vital health problem that caused high morbidity and enhanced the financial costs around the world. 1 The bone repair was a regenerative process that had a great impact on the patients’ life quality. 2 The typical characteristics of fracture healing could be identified as restoring the bone structure and cellular functions of damaged skeletal organ, which included recruitment and differentiation of stem cells and the formation of cartilaginous callus and blood vessels. 3 Over the past few decades, strategies to clinically enhance bone repair processes have been investigated, including biophysical methods (low-intensity pulsed ultrasonography and electromagnetic fields) and biological methods (bone marrow grafting, Wnt signaling family, and LDL receptor-related proteins). 4 However, to the best of our knowledge, the molecular mechanisms and gene therapy approaches have not been comprehensively explored.
Wnt was a secreted glycoprotein that participated in various cellular processes including ontogeny and morphogenesis. 5 Studies have shown that Wnt signaling was essential for bone remodeling, and it could be associated with low-density lipoprotein receptor-related protein receptor proteins (LRP) 5 binds and affected the differentiation of osteoblasts. 6 LRP4 also belonged to the LDL proteins family and highly related with LRP5. 7 A previous study reported that LRP4 independently mediated the Wnt pathway and regulated the tooth development. 8 Therefore, targeting LRP4 might be a potential approach for the molecular treatment of fracture.
MicroRNAs were conserved non-coding small RNAs involved in various cellular functions including cell differentiation, apoptosis, and proliferation. 9 Among different species of miRNAs, miR-7-5p was reported as a cancer inhibiting gene in considerable malignant tumors. 10 A recent study reported that miR-7-5p existed in the exosomes derived from bone marrow mesenchymal stem cells could significantly inhibited PI3K/AKT/mTOR pathways and further attenuated the progression of acute myeloid leukemia. 11 Moreover, miR-7-5p was found to downregulate TAF9B level and inhibited proliferation of osteosarcoma cells. 12 Using bioinformatics tools, it could be predicted that miR-7-5p potentially bound with LRP4. However, the effects of miR-7-5p on LRP4 and fracture healing have not been explored.
In this study, we hypothesized that miR-7-5p targeted LRP4 and further promoted the activation of Wnt/β-catenin signaling, which contributed to the fracture healing, aiming at providing alternative therapeutic approaches for the clinical treatment of bone fracture.
Methods
Cell Culture
The mouse pre-osteoblast cell line MC3T3-E1 was obtained from Procell Life Science &Technology Co., Ltd (Wuhan, China). The cells were cultured in α-MEM medium (Gibco, MA, USA) that supplemented with 1% antibiotics (penicillin and streptomycin) and 10% FBS. The cells were incubated at 37 °C in a humidified atmosphere containing 5% CO2. For evaluating the effects of miRNA, the miR-7-5p mimics were synthesized by Sangon Biotech Co., Ltd, Shanghai, China. The NC mimics were also obtained for negative control. When the confluence of cells achieved 70%, the transfection was implemented through Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) as per the protocol of manufacturer.
Animal Model
The male C57BL/6 mice (8-11 weeks old, 22 ± 2g) were purchased from Shanghai Lab, Animal Research Center, China. The mice were kept in the 12 h light/dark cycle animal room with free access to sufficient water and food. The fracture models of mice were constructed as previously described. 13 Briefly, the mice were anesthetized by injection of pentobarbital sodium (50 mg/kg). The lateral parapatellar approach was implemented to expose the right knee, after which the medial patella was dislocated. Subsequently, the femur intercondylar groove was exposed through flexion and a burr hole (0.5 mm in diameter) was punctured at the center part, then a needle with 0.5 mm diameter was inserted into the hole to prevent the displacement of fracture site. The fracture was created using a thin electric saw, and the wound was irrigated using saline and sutured. For evaluating the effects of miRNA, the miR-7-5p mimics or negative control mimics were injected through tail vein for continuous 3 days.
Cell Proliferation
Briefly, the cells at logarithmic growth were inoculated into 96-well plates at a density of 2 × 105 cell/mL. After various treatments, the cells were added with 10 μL CCK-8 solution (Solarbio, Beijing, China) and incubated at 37 °C for 2 h. Then the absorbance of cells was detected at 450 nm via a microplate reader (Biotek, Vermont, USA).
Histological Evaluation
The tissues were collected from mice and decalcified with 10% ethylenediaminetetraacetic acid. Then paraformaldehyde was obtained for fixation, and paraffin was used for embedding, after which the samples were prepared as 5 μm thickness sections. Subsequently, samples were deparaffinized and rehydrated and added with hematoxylin and eosin solution kit (H&E, Solarbio, Beijing, China) or tartrate-resistant acid phosphatase solution kit (TRAP, Solarbio, Beijing, China) for histological staining according to the instructions of manufacturer. For evaluating the results, the stained sections were observed using a light microscope (Olympus, Tokyo, Japan) and histological scores were evaluated. For TRAP staining, the TRAP-positive cells were counted. The results were analyzed using ImageJ software (US National Institutes of Health, Bethesda, MD, USA).
Alkaline Phosphatase Activity
For determining the activity of alkaline phosphatase (ALP), a commercial ALP assay kit (Abcam, Waltham, MA, USA) was obtained, and the experiments were performed as per the protocol. Briefly, the samples together with para-nitrophenyl phosphate solution and ALP enzyme solution were added and incubated for 60 min, after which the solution is stopped for terminating interactions. The absorbance was detected at 405 nm via a microplate reader (Biotek, Vermont, USA).
RT-qPCR
The total RNA was isolated using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). According to the instructions of reverse transcription reagent kit (Sangon Biotech, Shanghai, China), the cDNA was obtained, and the amplification was implemented on the ABI quantitative PCR system (Applied Biosystems, Waltham, MA, USA) for detecting RNA expression. The PCR conditions were as follows: denaturation at 95 °C for 10 min, followed by 35 cycles of denaturation at 94 °C for 20 s, annealing at 60 °C for 30 s and extension at 72 °C for 30 s. The relative expressions of mRNA were calculated using 2−ΔΔCt method. The primers were list as follows:
LRP4-F: 5′- AGTGACGAGCAATGCGACAT -3′
LRP4-R: 5′- ATAGGCGCACTGGAACTCTTC -3′
Wnt4-F: 5′- TCGTCTTCGCCGTCTTCTCAG -3′
Wnt4-R: 5′- GAGTCGAGTGTGGAGCAGTT -3′
GAPDH-F: 5′- AGGTCGGTGTGAACGGATTTG -3′
GAPDH-R: 5′- TGTAGACCATGTAGTTGAGGTCA -3′
Western Blotting
The total proteins were extracted using RIPA lysis buffer (Solarbio, Beijing, China). After centrifugation, the supernatant was obtained, and the concentration of proteins was detected by BCA Assay Kit (Sangon Biotech, Shanghai, China). Then proteins were separated by 10% SDS-polyacrylamide gel electrophoresis (SDS-PAGE, Sangon Biotech, Shanghai, China) gel. The proteins were transferred to polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA), and blocked with 5% non-fat milk. The samples were incubated with primary antibodies that included anti-LRP4 (ab174637, 1:1000), anti-Wnt4 (ab262696, 1:1000), anti-GSK3β (ab32391, 1:1000), anti-p-GSK3β (ab68476, 1:1000), anti-β-catenin (ab32572, 1:1000), anti-p-β-catenin (ab75777, 1:1000), and anti-GAPDH (ab8245, 1:1000) at 4 °C overnight. Next, the membrane was incubated with HRP-conjugated rabbit anti-mouse IgG secondary antibody (ab6728, 1:1000) for 2 h at room temperature. The band density was quantified using the using ImageJ software (US National Institutes of Health, Bethesda, MD, USA).
Statistical Analysis
All the experiments were performed at least 3 times and the data were presented as the mean ± standard deviation (SD). Statistical analyses were conducted using GraphPad Prism 7.0. One-way analysis of variance followed by Tukey's post-hoc test for comparison among 3 or more groups. P < .05 was regarded as a statistically significant difference.
Results
As shown in Figure 1A, miR-7-5p expression was upregulated by miR-7-5p transfection, indicating the overexpression models have been successfully constructed. Overexpression of miR-7-5p promoted cell proliferation and upregulation of ALP activity compared to the control group (Figure 1B and C). Subsequently, the in vivo mice models were constructed. Results revealed that miR-7-5p expression was decreased in model group, while overexpression of miR-7-5p increased miR-7-5p level, suggesting the successful construction of in vivo models (Figure 2A). H&E staining and TRAP staining demonstrated that miR-7-5p improved histological status and increased the proportion of TRAP-positive cells (Figure 2B). In addition, the RT-qPCR results showed that the mRNA level of LRP4 was enhanced while Wnt4 was decreased in fracture model mice, whereas transfection of miR-7-5p dramatically reversed LRP4 and Wnt4 expression (Figure 3A). Consistently, the western blotting data suggested that the protein expression of LRP4 and p-β-catenin/β-catenin ratio were increased, while Wnt4 protein level and p-GSK3β/GSK3β ratio were decreased in model group by western blotting (Figure 3B and C). However, overexpression of miR-7-5p had reversed effects on protein expression. The evidence indicated that miR-7-5p might downregulated LRP4 and activated Wnt/β-catenin pathway, promoting the process of fracture healing.
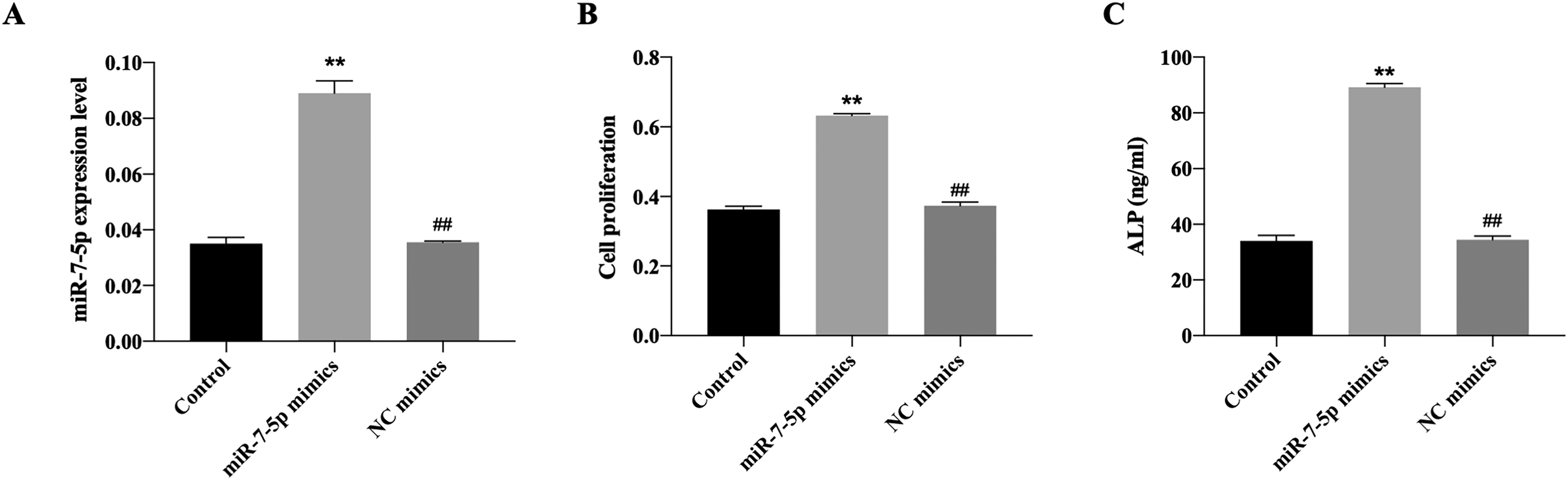
The effects of miR-7-5p on mouse pre-osteoblast cell line MC3T3-E1. (A) The expression of miR-7-5p after transfection of miR-7-5p mimics. (B) The cell proliferation detected by CCK8 assay. (C) The ALP activity determined using commercial kit. **P < .01 versus control group. ##P < .01 versus miR-7-5p mimics group.
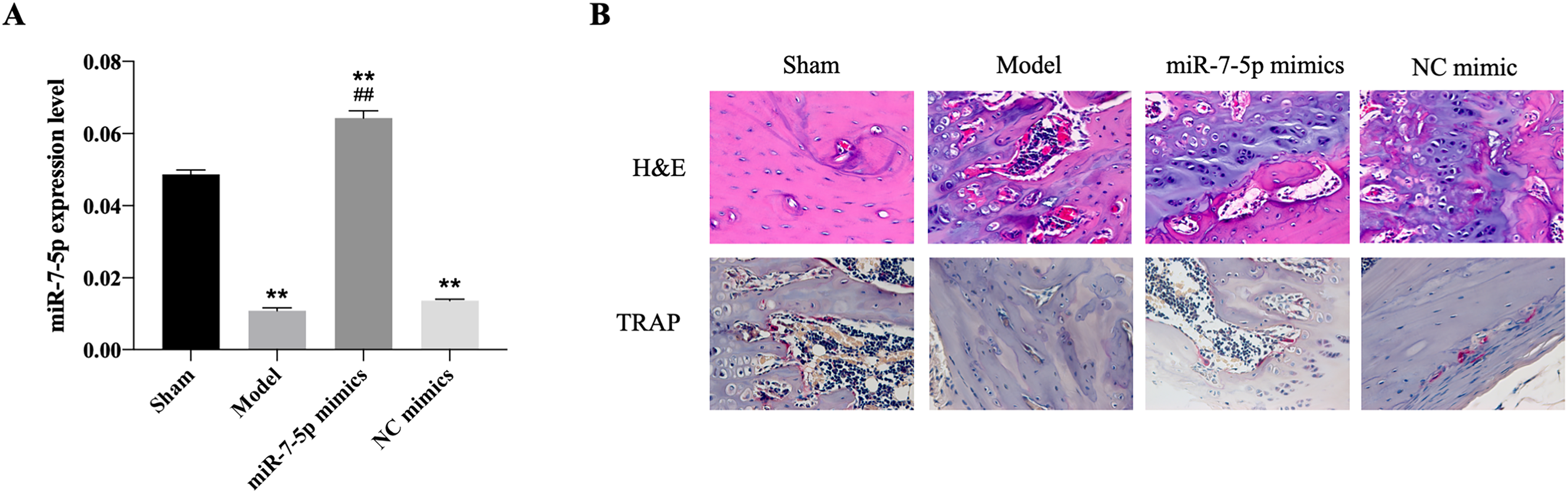
The effects of miR-7-5p on fracture healing in vivo. (A) The expression of miR-7-5p after transfection of miR-7-5p mimics. (B) The H&E staining and TRAP staining for histological evaluation. **P < .01 versus control group. ##P < .01 versus model group.
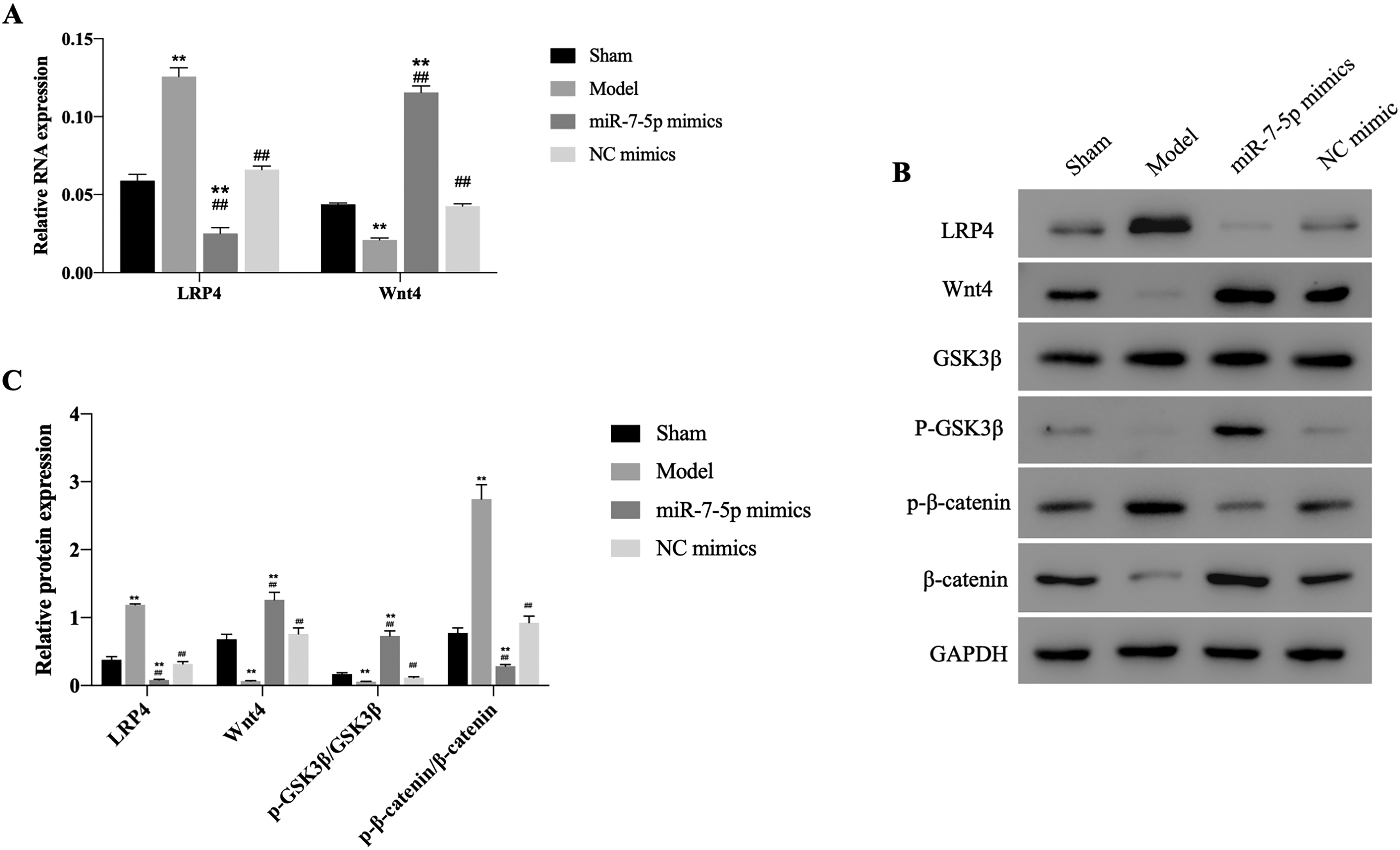
The effects of miR-7-5p on Wnt/β-catenin pathway in vivo. (A) The mRNA expression of LRP4 and Wnt4 measured by RT-qPCR. (B&C) The protein expression of LRP4, Wnt4, p-β-catenin, β-catenin, p-GSK3β, and GSK3β monitored using western blotting. **P < .01 versus control group. ##P < .01 versus model group.
Discussion
Recently, the investigations on bone healing have been rapidly developed. The periosteum and bone were disrupted when bone fracture occurred, and the proinflammatory factors were recruited by chemokines secreted from platelet degranulation. Increased levels of tumor necrosis factor-α have been observed and peaked at 24 h after bone injury. 14 Subsequently, the mesenchymal cells swelled and migrated from periosteum to form bone through endochondral ossification and reabsorption of primary bone by osteoclasts. 15 During this process, the transforming growth factor-β and BMP family was demonstrated to be of vital importance. 16 The Wnt family has also been reported to have the ability in regulating differentiation from mesenchymal stem cells into osteoblasts and involved in the cellular process of bone formation. 17 Thus, targeting and promoting various phases would contribute to the process of fracture healing.
The relationship between microRNAs and bone healing has been studied for decades. A previous study indicated that miR-144-5p presented in the exosomes derived from diabetic bone marrow-derived macrophages could impair the fracture healing through targeting Smad1, resulting in the delayed union of fractures in diabetics. 18 Xin et al showed that miR-214 level was decreased in the patients with fragility fracture, while overexpression of miR-214 significantly promoted osteoblasts proliferation and suppressed cell apoptosis through downregulating Sox4. 19 Liu et al reported that transfection of miR-21 in mice treated with fracture operation dramatically improved the histological status of bone tissues and activated PI3K/Akt signaling pathway. 20 Moreover, a recent study indicated that overexpression of miR-25 could increase the level of β-catenin, activate osteoblast differentiation, and enhance the fracture energy and stiffness of bone in Sprague-Dawley rat. 21 Consistently, we found a significant reduction in miR-7-5p levels in mouse models of fractures. The miR-7-5p overexpression dramatically alleviated the histological status, as well as downregulated LRP4 expression and activated Wnt/β-catenin pathway.
In conclusion, the present study demonstrated miR-7-5p decreased LRP4 level and upregulated Wnt/β-catenin signaling, indicating a positive effect on fracture healing. Evidence suggested that miR-7-5p might be a potential molecular target for clinical treatment of fracture healing. However, further investigations on the RNA networks and clinical experiments are needed to comprehensively understand the mechanisms of fracture healing.
Footnotes
Acknowledgments
We would like to acknowledge everyone for their helpful contributions to this paper.
Author Contributions
Each author has made an important scientific contribution to the study and has assisted with the drafting or revising of the manuscript.
Availability of Data and Materials
The data are available upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
The ethical approval was obtained from the Ethic Committee of Ganzhou People's Hospital.
Consent to Publish
All of the authors have consent to publish this research.
