Abstract
Previous studies have demonstrated that exercise improves cognitive function in Alzheimer’s disease mice but the exact mechanism needs further studies. This research aimed to study the effects of aerobic treadmill exercise on epidermal growth factor (EGF) levels and learning and memory in
Introduction
Over 47 million individuals are living with dementia worldwide, and this number is anticipated to quadruple by 2050. 1 Alzheimer’s disease (AD) is a primary degenerative disease of the central nervous system (CNS) that starts in old age and dementia is its main clinical manifestation. 2 The early behavioral changes consist of mild memory disturbances, which gradually deteriorate over 3-9 years and eventually lead to severe dementia symptoms.3,4 AD is a devastating disease that places a heavy financial burden on patients, their families, and society. 5 Therefore, it is especially important to use effective methods to inhibit its development and progression.
Acetylcholine is a typical transmitter of the CNS. 6 The presence of acetylcholine synthase generally serves as a specific marker for central cholinergic neurons, and acetylcholinesterase-positive fibers are abundant in the hippocampal and dentate gyrus. 7 Studies have shown that in aging human brain tissue, acetylcholine is significantly reduced, along with the ability to use choline to produce acetylcholine. 8 It is generally accepted that there is a relationship between the degeneration of cholinergic neurons in patients with AD and the deficiency of the required neurotrophic factors in their neurons. 9 Neurotrophic factors are a class of proteins that selectively regulate neural growth and survival in the peripheral nervous system and CNS and play an important role in neuronal growth and differentiation, maintenance of normal neuronal function, as well as repair of neurological damage, and in regulating the metabolism and function of the nervous system. 10 It prevents atrophy of forebrain cholinergic neurons, increases acetylcholinesterase levels in aged rats presenting with cognitive impairment, increases cholinergic neuron function, and improves memory. 11 Epidermal growth factor (EGF), an important neurotrophic growth factor, is secreted in an endocrine manner, thus strongly promoting skin cell division and growth even with small amounts. 12 It has been shown that different sources of EGF have pro-cell proliferation and tissue growth effects. It may modulate cholinergic neurons by modifying neuronal precursor cells, resulting in elevated neuronal gene expression, decreased postsynaptic long-duration enhancement, and increased cholinergic activity, 13 thereby affecting learning and memory in patients with AD.
Physical activity appears to be the most protective factor against the negative effects of aging on health and cognition, according to several studies.14–17 Physical activity and exercise are tempting low-cost and low-risk alternative treatment strategies, so many trials are underway to explore the impact of physical activity and exercise on cognition in patients with AD. 18 Physical activity and exercise may slow down cognitive decline. 19 Currently, the research on the application of physical exercise to AD focuses mostly on appropriate physical exercise to slow down the decline in learning and memory after the onset of the disease 15 and is expected to improve the quality of life of these patients. However, few studies have been reported on the modulatory role of physical exercise in the development of the disease.
In this study, we established an aging model by subcutaneous injection of
Materials and Methods
Animals
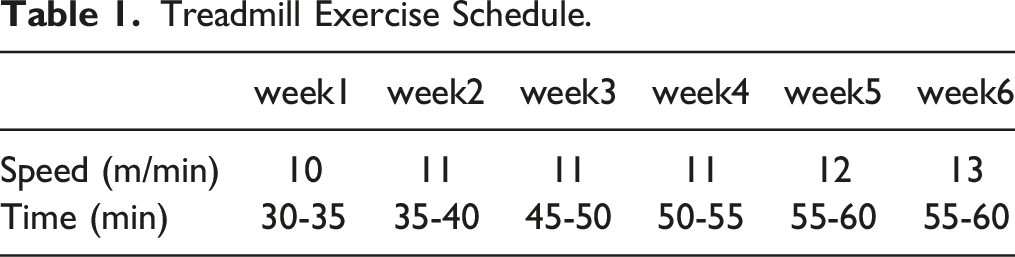
Treadmill Exercise Schedule.
Step Down Test
The test equipment is a 25 cm × 22 cm × 30 cm jumping reaction box. All sides of the box are made of opaque teal glass and the bottom is equipped with a copper grid that can be electrically stimulated (36 V). The distance between the copper bars constituting the copper grid is 1 cm. The rear corner of the box has a safety platform with a height and diameter of 8 cm.
Learning behavior test: the mice were gently put into the reaction box, after 3 min of adaptation. The mice were gently put on the platform and generally came down quickly. After three repetitions, when the mice stood on the copper fence, continuous electrical stimulation (36V) was given. The animals received an electric shock and their normal response was to jump back to the platform. Most of the mice may jump to the copper fence again and jump back to the platform quickly after receiving an electric shock. This training was repeated for 5 min, that is, the learning was completed, and the number of times each mouse received an electric shock and the reaction time were recorded.
Memory retention test: The above test was repeated after 24 h. Mice were placed on the platform sequentially in the original order, and the time of the first jump off the platform (latency) and the number of times each mouse that received an electric shock (i.e., jumped off) within 5 min (number of errors) were recorded. The shorter the latency period and the greater the number of errors indicated poorer learning and memory. If the mice still did not jump off the platform after 5 min, the latency period was recorded as 300 s.
Number of Neurons and EGF-Positive Cells
Perfusion, Extraction, and Sectioning
The mice were placed in an anesthesia chamber, anesthetized by ether inhalation following which apical blood sampling was performed, and serum was extracted from the blood and stored in a refrigerator at low temperature for freezing until testing subsequently for biochemical parameters. A puncture needle was inserted from the left ventricle into the aorta under direct vision, and blood was released from the right atrial cutout. About 150 mL of saline was injected rapidly, and then 500 mL of 4% paraformaldehyde was drip-fed, first quickly and then slowly. This was continued for about 1 h and the head was severed to remove the brain tissue (including the hippocampus). The tissue was immersed in 4% paraformaldehyde solution and incubated at 4°C for 24 h for fixing. Subsequently, conventional alcohol gradient dehydration, transparency protocol, wax dipping, embedding, and sectioning were performed to obtain 7-micron-thick frontal sections.
Immunohistochemical Staining
After dewaxing and hydration of the paraffin sections, they were washed three times with PBS buffer (ph 7.4) for 3 min each for antigen repair. One drop of 50 μl of normal peroxidase blocking solution (Reagent A) was added to each section and incubated for 10 min at room temperature to block the endogenous peroxidase activity, and the sections were washed three times with PBS for 3 min each; the PBS solution was removed and 1 drop of 50 μl of normal non-immune animal serum (Reagent B) per section was added and incubated for 10 min at room temperature. The serum was removed and one drop of 50 μl of the primary antibody per section was added and incubated overnight for 60 min at room temperature. The sample was washed three times with PBS for 3 min each; the PBS solution was removed and one drop of 50 μl of biotin-labeled secondary antibody (Reagent C) per section was added and incubated for 10 min at room temperature. It was washed three times with PBS for 3 min each. The PBS solution was removed and the section was incubated for 10 min at room temperature. It was washed times with PBS for 3 min each. The PBS solution was removed and 1 drop of 50 μl of streptomyces anti-biotin-peroxidase solution (Reagent D) was added to each section and incubated for 10 min at room temperature. It was washed three times with PBS for 3 min each. The PBS solution was removed and 100 μl of freshly prepared DAB solution was added to each section, observed under the microscope for 3-10 min, washed with tap water, re-stained with hematoxylin, and washed again with tap water. The sections were stained blue. These sections were dehydrated and dried in gradient alcohol, made clear in xylene, and sealed with neutral gum.
Nissl Staining
After dewaxing the paraffin slices until hydrated, they were dipped in an aqueous solution of tar firm violet for 20-30 min; after washing with tap water, they were incubated with 95% alcohol, dehydrated with anhydrous alcohol, made transparent with xylene, and sealed with neutral gum.
Image Acquisition and Analysis
Two brain regions, CA1 and CA3, were analyzed from each section using the Motic Images Advanced Microscopic Image Acquisition and Analysis System, with a measurement window area of 10,232 μm2 for each brain region and three fields of view. Image analysis was performed for Nissl-stained sections, and the number of neurons in the observed region was obtained by monochromatic segmentation of the images. EGF-positive cells were counted.
Statistical Analysis
The data obtained from the experiment are expressed as mean ± standard deviation, and the SPSS 20.0 statistical software was used for statistical analysis. After verifying that the data obeyed a normal distribution, differences between groups were tested by one-way analysis of variance (ANOVA) with P < .05 regarded as a statistically significant difference.
Results
Step Down Test
Figures 1(A) and (B) show that the response time of mice in the D-gal group is 20.14 s, and it is significantly higher than those in the C (7.33 s), AE (6.91 s) and D-gal + AE (second) groups (P < .05). There was no significant difference among the C, AE, and D-gal + AE groups. However, the number of errors in the D-gal group was .80 times, lower compared to C (1.20 times), AE (1.20 times), and D-gal + AE (1.10 times) groups. Learning behavior test for each group of mice. (A) The number of errors. (B) The response time. *: P ; .05, **: P ; .01(C Vs D-gal); #: P ; .05, ##: P ; .01(D-gal Vs D-gal + AE).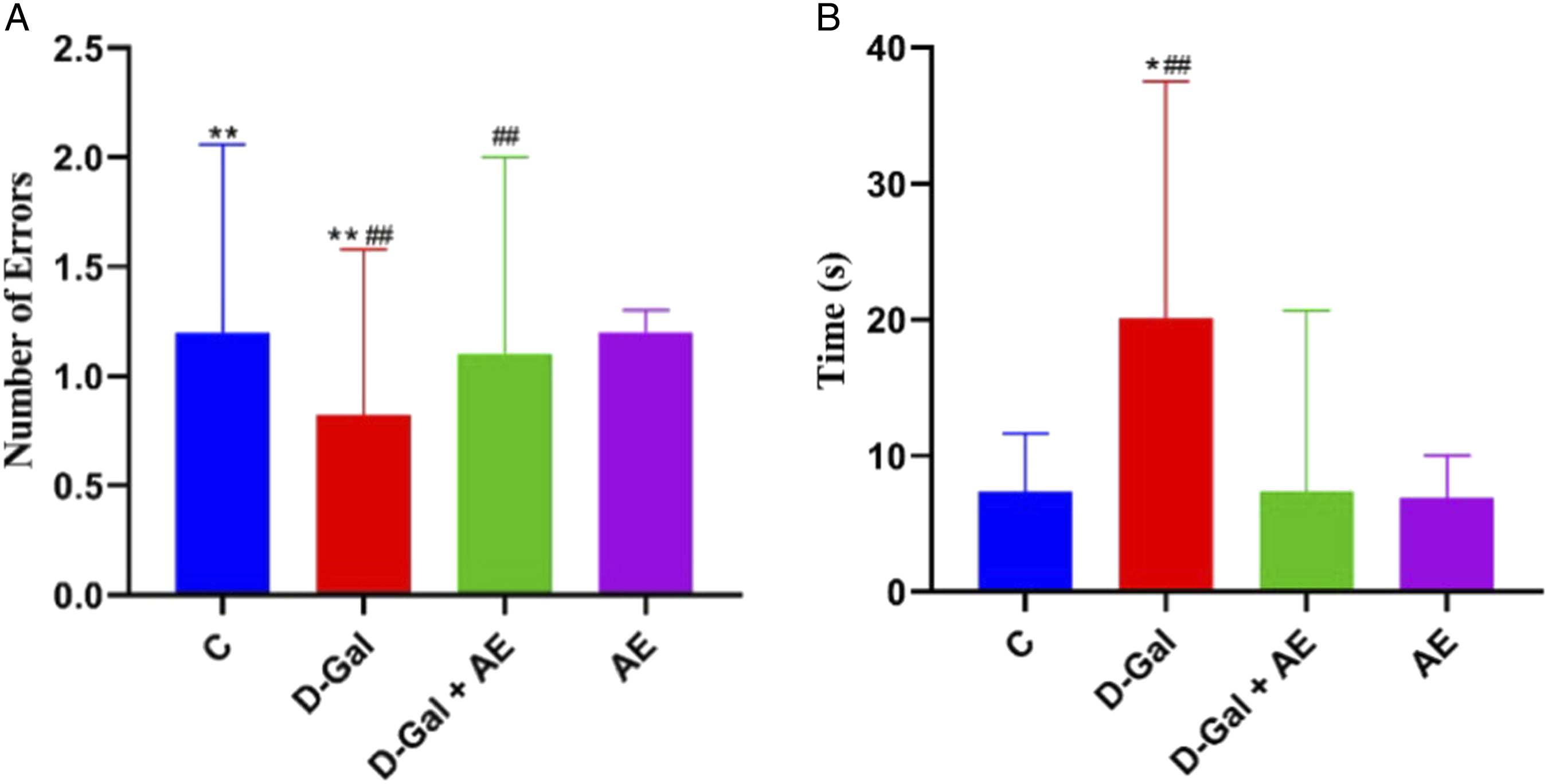
Figures 2(C) and (D) show that the latency period of mice in the D-gal group was significantly shorter than those of the other three groups (P < .05). The number of errors in the D-gal group was 2.10 times, higher than those in the C, AE, and D-gal + AE groups (P < .01); there was no significant difference among C, AE, and D-gal + AE groups. Memory retention test for each group of mice. (C) The latency period. (D) The number of errors. *: P ; .05, **: P ; .01(C Vs D-gal);
#
: P ; .05,
##
: P ; .01(D-gal Vs D-gal + AE).
Superoxide Dismutase (SOD) Activity in Mice
As shown in Figures 3(E), compared with the C group, the SOD (10.71 U/ml) activity in the D-gal group decreased (P < .01). However, the SOD activity in D-gal + AE groups was higher than in the D-gal group (P < .05). Meanwhile, there was no significant difference among the C and D-gal + AE groups. Superoxide Dismutase activity for each group of mice. (E). *: P ; .05, **: P ; .01(C Vs D-gal); #: P ; .05, ##: P ; .01(D-gal Vs D-gal + AE).
Changes in the Number of Neurons in the Hippocampal CA1 and CA3 Regions
Figure 4(F) and (G) show the number of neurons in the hippocampal CA1 and CA3 regions in the four groups. The number of neurons in the D-gal group decreased compared to those in the C, AE, and D-gal + AE groups (P < .05). The number of neurons in the hippocampal for each group of mice. (F) The CA1 region. (G) The CA3 region. *: P ; .05, **: P ; .01(C Vs D-gal);
#
: P ; .05, ##: P ; .01(D-gal Vs D-gal + AE). 

Change in EGF-Positive Cell Number in the Hippocampal CA1 and CA3 Regions
Figure 5(H) and (I) show the number of EGF-positive cells in the hippocampal CA1 and CA3 regions of the mice in the D-gal + AE group. The number of positive cells in the C, AE, and D-gal + AE groups was greater compared to that in the D-gal group (CA1:40.30, CA340.20) (P < .05). The number of EGF-positive cells in the hippocampal for each group of mice. (H) The CA1 region. (I) The CA3 region. *: P ; .05, **: P ; .01(C Vs D-gal); #: P ; .05, ##: P ; .01(D-gal Vs D-gal + AE).

Discussion
The use of
The number of errors was found to be lower in the D-gal group relative to the C, AE, and D-gal + AE groups. This may be related to the loss of appetite and dislike toward activity observed in the mice of the D-gal group during the experiment. The learning and memory abilities and number of neurons in hippocampal CA1 and CA3 regions were lower than the C and AE groups (P < .05). The changes in the above phenomena and indicators are in line with the characteristics of dementia in AD and are consistent with the experimental results reported in another animal experimental literature,
23
indicating that
In previous studies, it was found that information that needs to be remembered generally flows from the dentate gyrus to CA3 to CA1 regions and then to the inferior peduncle as it enters the hippocampus, where additional information is input into each region of output in the last two regions. So this study focused on these two regions. Our experimental study showed that mice in the AE group and D-gal + AE group had better learning and memory than those in the D-gal group and the number of neurons in the hippocampal CA1 and CA3 regions in the D-gal + AE group was significantly greater than those in the D-gal group, which indicated that appropriate aerobic treadmill exercise can improve learning and memory and reduce the death and apoptosis of hippocampal neurons. According to previous studies, appropriate long-term aerobic treadmill exercise can promote the growth of the brain capillaries, increase blood flow, and change the efficiency of oxygen utilization,24,25 so that the brain would have sufficient oxygen.
EGF can nourish cholinergic nerve cells, promote nerve cell survival, and promote nerve cytosol and protrusion growth and development, thereby ultimately improving the morphology and function of cholinergic neurons.26,27 When the body needs to learn and remember new stimuli for cognitive activities, acetylcholine is released from the hippocampal region of the brain and actively participates in synaptic transmission to achieve processes underlying learning and memory. 28 It was found that appropriate aerobic treadmill exercise improved blood and oxygen supply to brain tissue, improved the function of the CNS, and maintained the stability of the environment in brain tissue. 29 One study found that wheel training can contribute to the growth of hippocampal dentate gyrus nerve cells in adult mice, thereby enhancing the information-processing capabilities of the brain. In addition, studies have shown that physical activities enhance RNA levels in the brain, 30 along with enhancing the oxidative enzyme system function of brain tissue. 31 All of these help in better learning and memory, as well as other brain functions.
The experimental results also showed that hippocampal EGF-positive neurons in the D-gal group were significantly lower, while those in the D-gal + AE group were significantly higher, consistent with the results of previous studies.32,33 The mechanism underlying the benefits of aerobic treadmill exercise is that it increases EGF levels in the hippocampus, however, the details are not well understood. Studies have shown that running wheel exercise promotes nerve growth in rats and reduces the threshold of long-term postsynaptic enhancement along with increasing the mRNA expression of brain-derived nerve growth factor in the hippocampal dentate gyrus.34,35 Therefore, it is speculated that increased EGF levels in the brain may also be related to the expression of its mRNA stimulated by aerobic treadmill exercise.
Based on the present finding, we believe that aerobic treadmill exercise may contribute to increasing EGF levels in aging mice. EGF may block the degeneration of neurons in the brain of mice through nutritional cholinergic neurons, to improve the morphology of neurons and improve the learning and memorizing behavior abilities in aging induced by
Conclusion
Aerobic treadmill exercise improved the scores of learning and memory-related behavioral tests and changed the morphology of hippocampal neurons in the exercise group of aging mice, suggesting that aerobic exercise may repair the damaged hippocampal function. The levels of EGF in the CA1 and CA3 regions of the hippocampus in the D-gal + AE group were significantly higher compared to those in the D-gal group. The changes in EGF levels and neuronal morphology in the D-gal + AE group may be one of the mechanisms underlying the improvement in learning and memory due to aerobic exercise in mice in the D-gal group.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sports Science and Technology Research Project of Hebei Provincial Sports (20213012).
