Abstract
Objective:
Malignant peripheral nerve sheath tumours (MPNSTs) are invasive, hard-to-treat, soft tissue sarcomas. In this study, in vitro and in vivo effects of triptolide were investigated using human MPNST cell lines.
Methods:
Cultured STS-26T and ST88-14 cells were treated with 0 - 100 ng/ml triptolide (for determination of cell proliferation by sulphorhodamine B assay), with 12.5 ng/ml or 25 ng/ml triptolide (for analysis of caspase activity, effects on apoptotic pathway intermediates [by Western blots and flow cytometry], and for measurement of vascular endothelial growth factor [VEGF] and epidermal0 growth factor receptor [EGFR] levels by enzyme-linked immunosorbent assay). A xenograft model was established by injection of STS-26T cells into nude mice, and the effects of 250 μg/kg triptolide on tumour growth and apoptosis were compared with controls.
Results:
Triptolide significantly inhibited cell proliferation and induced apoptosis in vitro, through activation of caspases, in a dose- and time-dependent manner; VEGF and EGFR levels were suppressed. In vivo, triptolide inhibited the growth of STS-26T xenografts and reduced apoptosis.
Conclusion:
Triptolide may have a therapeutic benefit in MPNST treatment.
Keywords
Introduction
Malignant peripheral nerve sheath tumours (MPNSTs) are aggressive soft tissue sarcomas with a poor prognosis. Half of all MPNSTs occur in patients with neurofibromatosis type 1 (NF1), an autosomal-dominant disorder with an incidence of one in 3500 persons. 1 Approximately 8 - 13% of patients with NF1 will eventually develop MPNSTs, which in turn are the leading cause of NF1-related mortality. 1 These tumours are characterized by invasive growth, propensity to metastasize and poor response to chemotherapy and radiation therapy. 2
Angiogenesis is essential for solid tumour growth and metastasis.3,4 Vascular endothelial growth factor (VEGF), which plays a central role in angiogenesis, is released by tumour cells and induces the formation of a network of capillary vessels via tyrosine kinase receptor-mediated signalling. 4 Blockade of VEGF may suppress angiogenesis in NF-related tumours. 5 Epidermal growth factor receptor (EGFR) is expressed at varying levels in primary MPNSTs.6,7 Research in cultured MPNST cells has indicated that inhibition of EGFR activity may decrease tumour invasiveness. 7 The VEGF and EGFR pathways share a common downstream signalling pathway, 8 and EGFR activation can induce the production of VEGF in human cancer cells.8,9
Triptolide is a diterpenoid triepoxide (molecular weight, 360) derived from Tripterygium wilfordii that has been found to have anti-inflammatory and antitumour activities.10 – 12 Studies have indicated that triptolide may induce apoptosis in a variety of cancers including those of the breast, lung or pancreas,12 – 14 via death receptors and the mitochondria-mediated pathway.15,16
Most studies evaluating potential treatments for MPNSTs have focused on compounds that block Ras apoptotic signalling. 2 Using agents such as triptolide to help characterize apoptotic signalling cascades in malignant neurofibromatosis cells may, however, provide a new approach in the control of MPNST growth. Using two cell lines derived from grade III malignant schwannomas (STS-26T and ST88-14), the present study evaluated the effects of triptolide on MPNST cell proliferation and apoptosis in vitro, and explored whether triptolide could inhibit the production of VEGF and EGFR. In addition, the effects of triptolide on tumour growth and metastasis in vivo were determined in a mouse STS-26T xenograft model.
Materials and methods
Cell Culture of STS-26T and ST88-14
Human malignant schwannoma cell lines STS-26T and ST88-14 (Georgetown University, Washington DC, USA) were maintained as monolayer cultures in Dulbecco's modified Eagle's medium (DMEM; Life Technologies, Shanghai, China) supplemented with 10% fetal bovine serum (FBS), 100 μg/ml streptomycin and 100 IU/ml penicillin. The cells were incubated at 37 °C in a humidified atmosphere of 5% CO2.
Cell Proliferation Assay
The effect of triptolide on cell proliferation was evaluated using a sulforhodamine B (SRB) colorimetric assay. 17 Briefly, STS-26T and ST88-14 cells (1 × 103 cells/well) were treated with 0 – 100 ng/ml triptolide (99.9% purity; Institute of Medical Research, Fuzhou, China) or vehicle control (0.01% dimethyl sulphoxide [DMSO]) for 48 h. In a separate experiment, cells were treated with 25 ng/ml triptolide or control for 0 – 72 h. Cell morphology was analysed using an Invertoskop 40C inverted microscope (Carl Zeiss, Thornwood, NY, USA). After morphological analysis, cells were fixed with 10% trichloroacetic acid for 2 h at 4 °C and then stained with 70 μl 0.3% SRB for 30 min (also at 4°C). The number of cells was estimated by measuring the optical density at 490 nm (OD490). Percentage survival was defined as 100 × (T – T0)/(C – T0), where T is the OD490 value after drug exposure, T0 is the OD490 value prior to drug exposure, and C is the OD490 value at the end of the experiment in cells that had not been treated with vehicle or triptolide.
Colony Formation Assay
The STS-26T and ST88-14 cells (30 000 cells) were suspended in 0.36% agarose (1 ml) in 10% FBS and DMEM containing triptolide (12.5 or 25 ng/ml) or 0.01% DMSO, then poured onto a layer of 0.6% solid agarose in 10% FBS and DMEM in six-well plates. Colonies > 60 μm in diameter were counted 2 weeks later using an OMNICON™ Image Analysis System (Imaging Products International, Chantilly, VA, USA).
Analysis of Apoptosis
The STS-26T and ST88-14 cells were treated with 0 – 100 ng/ml triptolide or 0.01% DMSO for 48 h, or 25 ng/ml triptolide or control for 0 – 72 h. Cells (2 × 105) were then harvested and stained with 20 μg/ml Annexin V™ (Molecular Probes, Eugene, OR, USA) at room temperature for 30 min before washing in 0.01 mol/l phosphate-buffered saline (PBS; pH 7.2). Staining with 50 μg/ml of propidium iodide (Molecular Probes) was then carried out at room temperature, immediately before analysis using a flow cytometer (FACS™, Becton Dickinson, San Jose, CA, USA). Each sample was analysed twice and all experiments were repeated in triplicate. The percentages of cells in the high-red, low-red and high-green regions were measured under the different treatment regimens.
Determination of VEGF and EGFR Levels
Supernatants (100 μl) were collected from cultured STS-26T and ST88-14 cells treated with 12.5 ng/ml or 25 ng/ml triptolide, or 0.01% DMSO, for 48 h. Concentrations of VEGF and EGFR were determined in supernatants using commercially available enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems, Minneapolis, MN, USA), according to the manufacturer's instructions.
Analysis of BCL-2, Cytochrome C, Caspase-3, -8 and -9 PARP
Whole-cell lysates from STS-26T or ST88-14 cells (80% confluence) treated with triptolide (12.5 ng/ml or 25 ng/ml) or control were harvested with lysis buffer (1% Triton X™-100, 0.5% sodium deoxycholate, 0.5 μg/ml leupeptin, 1 mM ethylenediaminetetra-acetic acid, 1 μg/ml pepstatin, and 0.5 mM phenylmethanesulphonyl fluoride; pH 7.4). Lysates were then centrifuged at 12 000
For reprobing, blots were stripped with a buffer containing 50 mM Tris-HCl (pH 6.8), 2% SDS and 0.1 M β-mercaptoethanol (pH 2.5 – 3). Glyceraldehyde 3-phosphate dehydrogenase was used as an internal control.
Determination of Caspase-3, -8 and -9 Activities
Activities of caspase-3, -8 and -9 were measured using commercially available fluorescent substrate assay kits, according to the manufacturer's instructions. The caspase-3 assay kit was purchased from Molecular Probes (Eugene, OR, USA); the caspase-8 and -9 assay kits were purchased from BioVision (Mountain View, CA, USA). Briefly, 1 × 106 STS-26T or ST88-14 cells treated with triptolide (12.5 or 25 ng/ml) for 24, 48 and 72 h were collected and incubated in cold lysis buffer (pH 7.4) for 30 min at 4 °C. Cell lysate (50 μl) was then incubated with 50 μl 2 × reaction buffer (pH 7.5; BioVision) for 2 h at 37°C with DEVD-R110 (a caspase-3 substrate), IETD-AFC (a caspase-8 substrate) or LEHD-AFC (a caspase-9 substrate), which release fluorescence after their cleavage.
Nude Mice Xenograft Models
Xenograft models were established in 5 – 6-week-old female athymic nude mice (weight, 18 – 22 g) injected with STS-26T, ST88-14 or T265 cells. Mice were kept under 12-h light/dark cycles and were allowed free access to food and water. Briefly, cells (5 × 106) were cultured in 100-mm2 flasks and trypsinized at 80% confluency. Cells were then centrifuged at 1500
The animal protocol was approved by the Committee for Animal Care and Use, Fujian Medical University, Quanzhou, China (No. 81071968).
Determination of Apoptosis by Tunel Assay
Terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate (dUTP) nick-end labelling (TUNEL) assay was performed using an ApopTag® in situ staining kit (Chemicon, Temecula, CA, USA) according to the manufacturer's instructions. Tumour tissues were fixed in 10% formalin for 48 h before embedding in paraffin wax. Specimens were then cut into 10-μm sections, placed onto Superfrost™-plus slides, deparaffinized in xylene, rehydrated in graded ethanol and stained with methyl green. Apoptotic cells were manually counted in four random fields from eight independent tumours, from each of the treatment groups (triptolide and control), using light microscopy. The percentage of apoptotic cells was calculated by dividing the number of TUNEL-positive cells by the total number of cells.
Statistical Analyses
All statistical analyses were carried out using SigmaPlot (Systat® Software, San Jose, CA, USA). Data were expressed as mean ± SD and were analysed with either a two-sample Student's t-test or a one-way analysis of variance. A P-value < 0.05 was considered statistically significant.
Results
Triptolide decreased cell viability in STS-26T and ST88-14 cells in a concentration- and time-dependent manner (Figs 1A and 1B). Results from the flow cytometric analysis demonstrated that triptolide induced apoptosis in a concentration- and time-dependent manner in STS-26T and ST88-14 cells (Figs 1C and 1D). At concentrations of 12 ng/ml and 25 ng/ml, triptolide inhibited the ability of malignant schwannoma cell lines to form colonies on soft agar (Fig. 1E).
Cell viability of the human malignant schwannoma cells (STS-26T and ST88-14) following treatment with: (A) 0 – 100 ng/ml triptolide (TPL) for 48 h or (B) 25 ng/ml TPL for 0 – 72 h, as determined by sulphorhodamine B colorimetric assay. Apoptosis of STS-26T and ST88-14 cells following treatment with: (C) 0 – 100 ng/ml TPL for 48 h or (D) 25 ng/ml TPL for 0 – 72 h, as determined by flow cytometric analysis of Annexin V™ and propidium iodide staining. (E) Ability of STS-26T and ST88-14 cells to form colonies (> 60 μm diameter) on soft agar following treatment with TPL (12.5 ng/ml or 25 ng/ml) for 2 weeks. Data presented as mean ± SD of three independent experiments. *P < 0.05 versus control (two-sample Student's t-test)
Triptolide activated caspase-3, -8 and -9 in a dose- and time-dependent manner in STS-26T and ST88-14 cells (Fig. 2). Western blot analysis showed that treatment of STS-26T and ST88-14 cells with increasing concentrations of triptolide for 48 h was associated with an increase in the cleavage of caspase-3, -8, -9 and PARP, and an increase in the production of cytochrome c (Fig. 3). Triptolide treatment also dose-dependently inhibited production of Bcl-2.
Analysis of the cell death pathway triggered by triptolide (TPL; 12.5 or 25 ng/ml) in the human malignant schwannoma cell lines STS-26T and ST88-14. Caspase activity was determined using a tetrapeptide chromogenic substrate: DEVD-R110 for caspase-3 in (A) STS-26T and (B) ST88-14 cells; IETD-AFC for caspase-8 in (C) STS-26T and (D) ST88-14 cells; or LEHD-AFC for caspase-9 in (E) STS-26T and (F) ST88-14 cells. Data presented as mean ± SD of three independent experiments. *P < 0.05 versus control (two-sample Student's t-test) Effect of triptolide (TPL, 12.5 ng/ml or 25 ng/ml) on apoptotic factors involved in death receptor and mitochondrial pathways in lysates of human malignant schwannoma cell lines. Western blot analysis showing effects of TPL following 48 h of treatment on caspase and poly (ADP-ribose) polymerase (PARP) cleavage, cytochrome c and B-cell lymphoma 2 (Bcl-2) in (A) STS-26T and (B) ST88-14 cells. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) used as internal control

Control-treated STS-26T and ST88-14 cells secreted high levels of VEGF and EGFR into the cell culture medium (Fig. 4) that peaked at 72 h. Levels of VEGF and EGFR were, however, higher in STS-26T cells compared with ST88-14 cells. Triptolide treatment inhibited the release of both VEGF and EGFR in a concentration- and time-dependent manner in both cell lines (P < 0.05).
Determination of vascular endothelial growth factor (VEGF) and endothelial growth factor receptor (EGFR) secretion in human malignant schwannoma cell lines STS-26T and ST88-14, treated with 12.5 ng/ml or 25 ng/ml triptolide (TPL) or 0.01% dimethyl sulphoxide (control) for 48 h, as determined by enzyme-linked immunosorbent assay. VEGF concentrations in (A) STS-26T and (B) ST88-14 cells; EGFR concentrations in (C) STS-26T and (D) ST88-14 cells. Data presented as mean ± SD of three independent experiments. *P < 0.05 versus control (two-sample Student's t-test)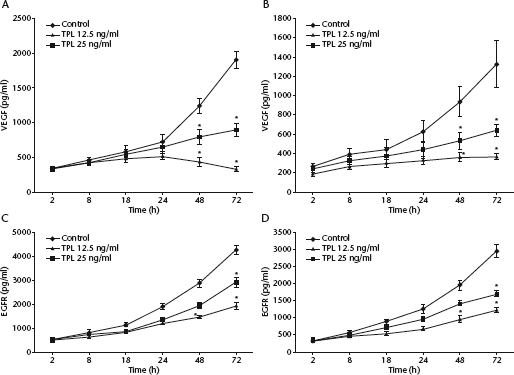
Tumours developed in nude mice injected with STS-26T cells but not in those injected with ST88-14 or T256 cells (data not shown). Triptolide (250 μg/kg) significantly inhibited tumour growth compared with control (Fig. 5A; P < 0.05) and reduced tumour weight (Fig. 5B; P < 0.05). The percentage of apoptotic cells in tumours from the STS-26T xenograft model, as determined by TUNEL assay, was increased in triptolide- compared with control-treated animals (Figs 5C and 5D; P < 0.05).
Effect of triptolide (TPL, 250 μg/kg) in the nude mouse STS-26T tumour xenograft model. TPL suppressed (A) tumour growth and (B) reduced tumour weight 44 days after injection of cells. (C) Photomicrographs showing apoptosis in tumour sections from STS-26T xenografts treated with TPL, as determined by terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate (dUTP) nick-end labelling (TUNEL) assay. (D) Percentage of apoptotic cells in tumour sections from STS-26T xenografts treated with TPL, calculated by dividing the total number of TUNEL-positive cells in each field by the total number of cells. Eight tumours were analysed from each group. *P < 0.05 versus control (two-sample Student's t-test)
Discussion
No effective treatments for MPNSTs have been found to date and excision of the tumour does not always prevent local recurrence; metastases to the lung, liver and brain are common. As such, there is a medical need for new therapeutic options to treat these malignancies. Previously published studies have demonstrated that triptolide has activity against a variety of cancers through death receptor and mitochondrial apoptotic pathways.15,16
Stimulation of the death receptor pathway results in caspase-8 activation, which in turn initiates direct cleavage of downstream effector caspases, such as caspase-3 and caspase-7. 18 The mitochondrial apoptotic pathway is initiated by mitochondrial release of apoptotic factors including cytochrome c, which triggers caspase-3 activation through the formation of the cytochrome c/Apaf-1/caspase-9– containing apoptosome complex. 19 Linkage of the two pathways occurs via the cleavage of Bid (a proapoptotic member of the Bcl-2 family) by caspase-8, thereby initiating a mitochondrial amplification loop. 19 Activated Bid is translocated to the mitochondria, resulting in the release of cytochrome c and subsequent caspase-9 activation. 20
We hypothesize that triptolide may activate the death receptor pathway of apoptosis initially and then activate the mitochondrial pathway through Bid cleavage. Research has already demonstrated that triptolide mediates activation of caspase-8 and -9 in pancreatic and cervical cancer cell lines. 15 Similarly, results from the present study using MPNST cell lines suggest that the effects of triptolide may be mediated by death receptor signalling, since triptolide increased the activity and cleavage of caspase-3, -8 and -9 in vitro. The present article is the first report demonstrating that death receptor signalling may be involved in triptolide-mediated MPNST cell death. Furthermore, the present study also indicated that the mitochondrial pathway might also play an important role in this process. Triptolide treatment led to a loss of mitochondrial potential and an increase in cytochrome c production. Overexpression of antiapoptotic proteins, such as Bcl-2, may stabilize the outer mitochondrial membrane to prevent cytochrome c release following a variety of insults, including treatment with chemotherapeutic agents 21 and triptolide.16,22 In the present study, triptolide increased the levels of proapoptotic proteins (such as caspases and PARP) but reduced the levels of Bcl-2 protein in both MPNST cell lines. These findings suggest that triptolide is able to inhibit Bcl-2 production in some tumour cell lines.
Results from the present study demonstrated that triptolide-induced apoptosis occurred as early as 24 h and peaked between 48 and 72 h, in both the p53-negative STS-26T cells and the wild-type ST88-14 cells. 23 Although p53 is essential for apoptosis caused by DNA damage, 24 these data demonstrate that triptolide induces apoptosis in MPNST cells regardless of p53 status.
Monoclonal antibodies and tyrosine kinase inhibitors directed at VEGF and EGFR signalling have been used in the treatment of advanced nonsmall cell lung cancer. 9 Anti-VEGF agents have also been shown to be one of the most successful treatments against NF1-related tumours. 5 Indeed, it has been reported that the experimental VEGF receptor 2 inhibitor, SU5416, inhibits tumour angiogenesis and overall growth of NF1 neurogenic sarcomas. 25 In the present study, triptolide inhibited the production of VEGF and EGFR in STS-26T and ST88-14 cells, indicating that triptolide may be a therapeutic candidate for MPNST treatment.
The present study also examined the antitumour effects of triptolide in a xenograft model of MPNST. Three MPNST cell lines (STS-26T, ST88-14 and T265) were injected into nude mice; tumours developed in animals injected with STS-26T cells, but not ST88-14 or T256 cells. Results from the ELISA showed that STS-26T cells secreted higher levels of VEGF and EGFR than ST88-14 cells under the same culture conditions, possibly accounting for the tumourigenesis of the STS-26T cells. Consistent with the in vitro findings of the present study, triptolide was shown to inhibit the growth of STS-26T tumours, reduce tumour weight and increase the number of apoptotic cells in the xenograft model. These data suggest that tumours formed by STS-26T represent a good model to study the efficacy of potential agents against malignant Schwann cells. Triptolide showed therapeutic potential for controlling this disease in the in vivo model. Despite these results, the unique target of triptolide for the induction of apoptosis in MPNST cells remains unclear and further investigation is required to determine if triptolide interferes with Ras activity.
In conclusion, the present study demonstrated that triptolide can inhibit the proliferation of STS-26T and ST88-14 cells, and may induce apoptosis via death receptor and mitochondrial signalling pathways. Furthermore, triptolide decreased the levels of the antiapoptotic factor Bcl-2, as well as levels of VEGF and EGFR. Data from an in vivo STS-26T xenograft model were consistent with in vitro findings, as triptolide was found to reduce tumour growth and increase apoptotic cell numbers. Taken together, evidence from the present study indicates that triptolide may have a therapeutic role in MPNST treatment.
Footnotes
Acknowledgements
The authors received grant support from the National Natural Science Fund of China (No. 81071968), the Key Programme of Scientific Research of Fujan Medical University (No. 09ZD014) and the Fujan Provincial Natural Science Foundation of China (No. 2008J0079). Human malignant schwannoma cell lines STS-26T and ST88-14 were provided by Professor George H DeVries of Georgetown University, Washington, DC, USA.
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
