Abstract
Objective
Positive human epidermal growth factor 2 (HER2) expression and its predictive clinicopathological features remain unclear in Sri Lankan gastric cancer (GC) patients. Here, we aimed to determine GC HER2 status predictors by analyzing associations between clinicopathological features and HER2 expression using immunohistochemistry (IHC) and silver in situ hybridization (SISH).
Methods
During this 4-year prospective study, clinicopathological data were collected from participants in the National Hospital of Sri Lanka. HER2 IHC and SISH were performed using commercial reagents. Using chi-square tests, associations of HER2-IHC/SISH with clinicopathological features were analyzed.
Results
Overall, 145 GC patients were included, 69 had gastrectomies and 76 had biopsies. Positive HER2 expression by IHC was associated with age <60 years, high T stage (assessed pathologically in resections and radiologically in biopsies), high nuclear grade, tumor necrosis, mitosis >5/high-power field, with additional perineural invasion and lymphovascular invasion in resections. These features, excluding lymphovascular invasion but including male sex, were associated with HER2 expression by SISH.
Conclusions
Age <60 years, high nuclear grade, tumor necrosis, and perineural invasion are associated factors of HER2 status. These could be used to triage GC patients for HER2 status testing in limited resource settings where IHC/SISH analysis is costly.
Keywords
Introduction
Gastric adenocarcinoma (GC) is the fourth most common cancer worldwide, with approximately one million new patients diagnosed annually. 1 It is among the most prevalent cancers in Eastern Asia, 2 with the highest GC incidence reported in the East Asian region and the lowest in the North American region. 3 Because most GCs are diagnosed at an advanced stage, treatment options are generally limited. Therefore, the 5-year survival rate is consistently low, being around 20% in most parts of the world.4,5 Many GC patients present at an advanced (i.e., metastatic) stage in Sri Lanka.6,7 Currently, there is no comprehensive screening endoscopy program for detecting early GC, mainly because of its associated high cost relative to the low disease incidence in this resource-constrained setting. Therefore, advanced GC is common, and treatment of these patients remains a challenge in Sri Lanka.
Targeted therapies have significantly impacted the treatment strategy for many common malignancies. At present, the biology of human epidermal growth factor receptor 2 (HER2)-positive tumors has been established for GC. 8 Trastuzumab, also called Herceptin, is a monoclonal humanized antibody directed against HER2 that has become a pivotal agent for the management of HER2-positive advanced and metastatic GC cases. 9 HER2 testing with targeted treatment would be an important aspect of GC therapy in Sri Lankan patients, with eligible GC patients being those with HER2 gene amplification. An individual’s HER2 status can be determined by evaluating HER2 protein overexpression levels by immunohistochemistry (IHC) or HER2 gene amplification by in situ hybridization (ISH).
Resource limited settings have many constraints for performing HER2 silver in situ hybridization (SISH) testing, including its costliness and lack of free availability. Hence, a GC patient’s HER2 status would likely be determined by IHC. Therefore, a study to predict HER2 status using clinicopathological parameters would be significantly helpful for such settings. Here, we examined HER2 protein expression levels by IHC and HER2 gene amplification by SISH in a cohort of Sri Lankan GC patients. The findings were correlated to the clinicopathological features of the patients to help predict the HER2 status. Overall, this study aimed to assess the potential for predicting HER2 overexpression in GC using clinicopathological features that are not currently evaluated in Sri Lankan GC patients.
Methods
Study setting and ethics
This prospective, collaborative study was performed at the Departments of Surgery and Pathology, Faculty of Medicine, University of Colombo, the Department of Pathology, National Hospital of Sri Lanka (NHSL), and the Department of Anatomical Pathology, Pathwest QE II Medical Centre, Perth, Australia. The study was approved by the Ethics Committees of the Faculty of Medicine, University of Colombo (Registration No: EC 11-139) and the NHSL (Reference No: AA/ETH/2012). All participants provided written informed consent before they were included in the study. All patient details were de-identified. The reporting of this study conforms to STROBE guidelines. 10
Study population
GC patients presenting to the NHSL over 4 years (April 2012 to April 2016) were studied and followed up until December 2017. All patients underwent upper gastrointestinal endoscopy and biopsy for confirmation of the diagnosis. Patients with gastro-esophageal junction (GOJ) cancers were excluded from the study. None of the patients had received neoadjuvant chemotherapy or any other type of treatment prior to biopsy/resection. Only the gastric resection specimen was included for patients who underwent curative surgery following biopsy. The endoscopic biopsy was included for patients with advanced tumors who did not undergo gastric resection. Radiological stage was assessed by contrast enhanced computed tomography of the abdomen and thorax. Radiological data were used to determine the N (nodal enlargement >1 cm) stage of patients who only had biopsies without resections and the metastasis (M) stage of all patients. The tumor (T) and nodal (N) stages of all patients were determined in accordance with the 7th edition of the TNM UICC guidelines. 11
Pathology
All tumor samples were fixed in 10% formalin for 24 to 48 hours for histopathological and IHC evaluation. Histopathological assessment was performed on hematoxylin and eosin-stained tissue sections, cut in 4-μm slices. Lauren’s classification for gastric adenocarcinoma was used for histological subtyping (diffuse, intestinal, or mixed). 12 Glandular formation and cytologic pleomorphism were considered to histologically grade tumors as well, moderately, or poorly differentiated. Tumor differentiation (grade), necrosis, mitotic count (< or >5/high-power field [HPF], field diameter 0.65 mm), nuclear grade, presence of signet ring cells, extracellular mucin, and tumor inflammation with eosinophils were assessed in all tumors and documented in a structured data sheet. Additionally, the lymphocytic response at the tumor–host interface, perineural invasion (PNI), lymphovascular invasion (LVI), muscle invasion, infiltrating tumor border, lymph node status, and pathological staging were assessed in gastric resections. A structured data sheet was used to document demographic and clinico-radiological-pathological features.
IHC
Representative formalin-fixed, paraffin-embedded (FFPE) tumor tissue sections cut at 4 μm were stained for HER2 protein expression by IHC. A polyclonal rabbit anti-human c-erB-2 oncoprotein (Dako A0485; Agilent Technologies, Santa Clara, CA, USA) and Dako Real TM Envision system were used for IHC staining. Breast cancer tissue with HER2 +3 score by IHC was used as the positive control. HER2 IHC staining was interpreted following the scheme described by Ruschoff et al. 13 IHC scoring was performed by two independent pathologists. An IHC score of 0 or +1 was considered negative for HER2 overexpression, +2 was considered positive, and +3 was considered strongly positive.
SISH
Tissue microarrays (TMAs) were prepared from 145 GC tissue blocks at the Department of Anatomical Pathology, Pathwest QE II Medical Centre, Perth, Australia. For the TMAs, two tissue cores with a diameter of 0.6 mm were extracted from each tumor using the TMA arrayer (TMA Master 1.16 SP1). The tumor cores were sequentially placed in molds, embedded in paraffin, and cooled to form the tissue array blocks. Sections (4 μm) obtained from the TMA blocks were used for SISH/IHC. The slides were stained using the Benchmark Ultraview automatic staining device (Ventana Medical Systems, Roche Diagnostics, Oro Valley, AZ, USA). HER2 SISH assessment was performed for the 145 GC cases using the INFORM HER2 dual ISH DNA Probe Cocktail (Ventana Medical Systems). This was designed to use light microscopy to quantitatively detect amplification of the HER2 gene and the centromere portion of chromosome 17 (CEP17) via two color chromogenic ISH in FFPE human GC tissues. For SISH signal counting, a discrete signal was counted as a single copy of HER2 or CEP17. HER2 SISH signals (black) are typically smaller in size and more discrete in appearance than CEP17 SISH signals (red) because of differences in target size and detection chemistry. For signal interpretation, 20 cells were counted for red (CEP17) and black (HER2) signals. HER2 gene status was classified as non-amplified (HER2/CEP17 ratio <2.0) or amplified (HER2/CEP17 ratio ≥2.0).
Data analysis
SPSS Version 21 (IBM Corp., Armonk, NY, USA) was used for data analysis. The chi-square test was employed to identify associations between HER2 expression by IHC/SISH and clinicopathological features with a significance level of 5%. Haldane-Anscombe correction was used for calculating the effect measures when any cell in the contingency tables had a value of zero.
Results
Patient demographics and tumor characteristics
One hundred forty-five (145) consecutive GC patients were included in the study. Table 1 depicts the pathological characteristics and demographics of the GC study population. Many of the tumors (n = 72, 49.7%) were of Lauren’s intestinal histological subtype, most (n = 87, 60%) were located in the proximal stomach, and over 60% of both proximal and distal GC tumors presented at an advanced (stage III/IV) radiological stage. Overall, 72 (49.6%), 42 (28.9%), and 31 (21.3%) cases were intestinal, diffuse, and mixed histological subtypes, respectively. Resected gastric specimens were primarily stage II (n = 35, 50.7%) for pathological staging (Table 1). Of the samples, 69 (47.6%) were gastric resections and 76 (52.4%) were endoscopic biopsies. There was a male predominance, with a male:female ratio of 1.6:1. The mean age at diagnosis was 60.06 years (range: 32 to 82 years).
Demographics and pathological characteristics of the study population of gastric carcinoma patients.

HER2 by IHC
HER2 expression was negative (0, +1) in the majority of cases (n = 133, 91.7%). Overall, there were 8.3% (12) HER2-positive cases by IHC (Score +2, moderate positivity, n = 7; Score +3, strong positivity, n = 5). Table 2 shows the correlations between the HER2 status by IHC score and the demographic, clinical, radiological, and pathological features of the GC cases.
Human epidermal growth factor 2 (HER2) immunohistochemistry (IHC) score associations with demographics and clinical-radiological-pathological features of gastric carcinoma cases.

HPF, high-power field.
HER2 IHC expression and clinicopathological features
The comparison of clinicopathological features and HER2 IHC status by univariate analysis is shown in Tables 3 and 4. HER2-positive GC predominately occurred in those less than 60 years in age. Other clinicopathological features that were found to be significantly associated with HER2 positivity included higher T stage, higher nuclear grade, mitotic count >5/HPF, and the presence of tumor necrosis, PNI, and LVI in resections.
Comparison of clinicopathological features and human epidermal growth factor 2 (HER2) immunohistochemistry (IHC) status (N = 145; univariate analysis).

Comparisons of clinicopathological features and human epidermal growth factor 2 (HER2) immunohistochemistry (IHC) status in resection samples (N = 69; univariate analysis).

HER2 by SISH
HER2 positivity assessed by SISH was 4.8% of cases (n = 7). All IHC +3 cases (n = 5) were SISH positive. Of the seven IHC +2 cases, SISH positivity was observed in only two cases. All IHC negative (0 and +1) cases (n = 133) were confirmed by SISH as negative (Figures 1 –3, Tables 3 and 4).

Human epidermal growth factor 2 (HER2)-positive gastric carcinoma (GC). (a) Moderately HER2-positive (+2) GC analyzed by immunohistochemistry (IHC) with normal surrounding gastric tissue (20×). (b) IHC HER2+ 2 GC (40×). (c) Silver in situ hybridization (SISH)-amplified GC with adjacent non-amplified normal gastric tissues and (d) SISH amplification of IHC HER2 + 2 GC.

Human epidermal growth factor 2 (HER2)-positive gastric carcinoma by immunohistochemistry (strongly positive (+3) tumor (20×)).

Human epidermal growth factor 2 (HER2)-positive gastric carcinoma by silver in situ hybridization (SISH). This is tumor is strongly positive by HER2 immunohistochemistry (+3). SISH showed strong amplification with nuclear clumps (20×).
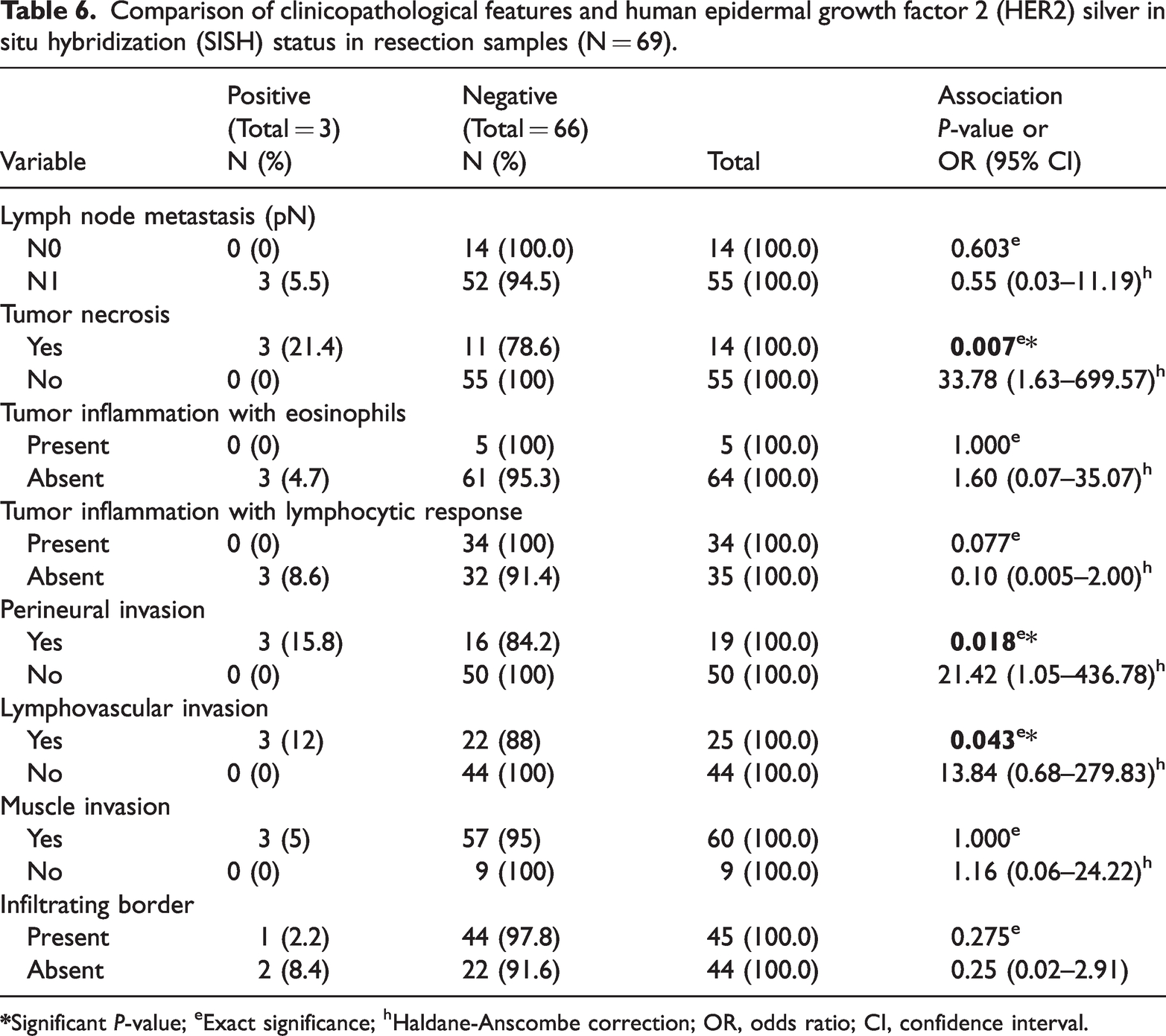
The results of the univariate analysis of the correlations between clinicopathological features and HER2 status by SISH are shown in Tables 5 and 6 The clinicopathological features found to be significantly associated with HER2 positivity included age less than 60 years, male sex, higher T stage (>T3), higher nuclear grade, higher mitotic count (>5/HPF), extracellular mucin, and the presence of tumor necrosis, PNI, and LVI in resections.
Comparison of clinicopathological features and human epidermal growth factor 2 (HER2) silver in situ hybridization (SISH) status (N = 145).

Comparison of clinicopathological features and human epidermal growth factor 2 (HER2) silver in situ hybridization (SISH) status in resection samples (N = 69).
Discussion
This is the first study conducted in a Sri Lankan setting that reports the potential predictors of HER2 status using SISH. Here, we found 4.8% of cases to be HER2-positive by SISH and 8.3% to be HER2-positive by IHC. Age less than 60 years, higher T stage (>T3), higher nuclear grade, mitotic count >5/HPF, and the presence of tumor necrosis, PNI, and LVI in resections were predictors of both IHC and SISH HER2 positivity. Additionally, male sex was a predictor of SISH HER2 positivity. This study helps identify potential predictors of HER2 status in lower-to-middle income settings, like Sri Lanka, where further confirmatory testing by ISH is not routinely available.
Notably, most studies that analyzed HER2 expression levels and clinicopathological features involved patients who had undergone curative resections.9,13,14 Recent advances in understanding the GC disease process from both biological and genomic perspectives have brought target-oriented therapy for advanced GC cases into clinical research and practice. The present study included a significant number of participants with advanced GC. It is essential to explore this subgroup of patients with advanced GC because they are the targeted subset that is eligible for trastuzumab.
HER2 testing in GC is an evolving area of clinical practice that has particular relevance to Asia-Pacific countries, which face a high incidence of this disease. 3 Data on HER2 expression in Sri Lankan GC patients are very limited. An earlier study conducted and published in Sri Lanka found a HER2 overexpression rate of 9% by IHC. 6 In the present study, we determined HER2 positivity rates using IHC and SISH, both of which were lower than 9%. The reasons for this are likely multifactorial. In most studies,9,14 the study sample was a mixture of GOJ and gastric carcinomas. Generally, GOJ tumors are reported to have higher HER2 positivity. 15
Until recently, no significant relationships between clinicopathological features of age, sex, pTNM, differentiation, or location with HER2 positivity have been documented in the literature.14,16–19 According to many studies,20–23 the intestinal type showed a higher rate of HER2 positivity than the diffuse type. In contrast, a recent meta-analysis 24 concluded that HER2-positive expression was associated with male sex, intestinal type, and well/moderate cell differentiation. This meta-analysis involved 15 studies (original articles), including 5990 gastric resections, that were analyzed to identify the clinicopathological factors associated with HER2 positivity. No relationship was observed between HER2 positivity and depth of tumor invasion, venous invasion, or lymphatic invasion. In the present study, younger age (<60 years) was significantly associated with both IHC (P = 0.005, OR = 0.14, 95% CI = 0.03–0.67) and SISH (P = 0.045, OR = 0.12, 95% CI = 0.02–1.06) HER2 positivity. This requires further examination with prospective analytical studies.
Both IHC and SISH HER2 positivity were significantly associated with higher T stage (T3, T4) (P = 0.032 for IHC, P = 0.015 for SISH). Sex (P = 0.043) also showed a significant association with HER2 positivity by SISH.
The intestinal subtype has been demonstrated in multiple studies to be the pathological feature that is invariably associated with HER2 positivity,9,14,25–33 including the ToGA trial. 9 In the present study, HER2 positivity was observed by SISH equally in the intestinal and diffuse types. One mixed tumor showed positive HER2 expression in the intestinal component. Therefore, HER2 expression was more commonly observed in the intestinal type cases, which is compatible with the previous findings. In our study, 72 (49.6%), 42 (28.9%), and 31 (21.3%) cases were intestinal, diffuse, and mixed histological subtypes, respectively. Interestingly, nearly 29% of tumors in the present study were diffuse, and the intestinal type was not significantly associated with HER2 overexpression.
According to Taghavi et al., 34 HER2 overexpression has no impact on disease prognosis, while other literature has provided contradictory evidence.35,36 Only a few studies have assessed the association of PNI with HER2 in GC, which concluded that there is no significant association between PNI and HER2 status.37–42 PNI is an underexplored phenomenon in GC, and its clinical significance remains controversial. 43 In the present study, PNI and high nuclear grade were significant predictors of HER2 expression by IHC and SISH. Most large-scale studies23,24,44 have not explored the association of tumor necrosis, PNI, or higher nuclear grade with HER2 overexpression. Tumor necrosis was also a significant predictor of HER2 expression in the present study. This highlights the necessity for further analytical studies regarding these predictors.
There are several methodological limitations to this study. First, the number of HER2-positive IHC cases was not sufficient to employ multivariate analysis to obtain cofounder-adjusted clinicopathological estimates of HER2 IHC. Therefore, this analysis was restricted to bivariable analysis. Second, the study included a notable number of biopsies, as the number of gastric resections was relatively low because of the advanced stage of disease presentation in this setting. This was considered during data analysis and interpretation.
Conclusions
Our results suggest that LVI (P = 0.043), tumor necrosis (P = 0.007), and PNI (P = 0.018) in resections are the histopathological factors associated with HER2 positivity by SISH. Combined with the demographic associated factor of age, these three factors could potentially be used as screening parameters for HER2 testing in limited resource settings and would be of value for future patient management. With further robust evidence generated from future analytical studies, incorporation of these features to develop a scoring system to predict HER2 positivity is possible and would be cost effective for limited resource settings. An accurate and reliable scoring system, together with clinical information, may help us to better determine whether a patient with GC is a potential candidate for HER2-based targeted therapy.
Research Data
Research Data for Human epidermal growth factor receptor-2 gene expression positivity determined by silver in situ hybridization/immunohistochemistry methods and associated factors in a cohort of Sri Lankan patients with gastric adenocarcinoma: a prospective study
Research Data for Human epidermal growth factor receptor-2 gene expression positivity determined by silver in situ hybridization/immunohistochemistry methods and associated factors in a cohort of Sri Lankan patients with gastric adenocarcinoma: a prospective study by Duminda Subasinghe, Nathan Acott, Pasyodun Koralage Buddhika Mahesh, Sivasuriya Sivaganesh, Sithum Munasinghe, Mariyan Priyanthi Kumarasinghe, Dharmabandu Nandaveva Samarasekera and Menaka Dilani Samarawickrema Lokuhetty in Journal of International Medical Research
Footnotes
Acknowledgements
The authors thank Mrs. G. K. Wijesinghe (staff technical officer) for technical assistance with laboratory work and Dr. Medhavini Dissanayake and Dr. Sameera Ravishan for assistance with data collection.
Author contributions
DS and MDSL designed the study with contributions from PKBM, SM, MPK, SS, and DNS. DS was involved in laboratory work, data collection and analysis, and writing the manuscript. NA supervised SISH laboratory work. MPK supervised and provided expertise in SISH interpretation. PKBM and SM provided expert statistical input. MDSL critically evaluated and edited the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was funded by The National Research Council, Sri Lanka (NRC grant 11-100) and The National Science Foundation (NSF, Grant No. OSTP/2016/03).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
