Abstract
Introduction:
Urinary incontinence (UI) is more prevalent in elderly populations with dementia than those without dementia. Alzheimer’s disease (AD) is the most common cause of dementia. Urge UI, the most common type of UI in AD patients, causes more morbidity and mortality. However, it is inconvenient to obtain the report of urodynamic study from AD patient to diagnose urinary incontinence. Nevertheless, it is easier to obtain subjective or objective questionnaires from the patients or the caregivers. The data collected from the questionnaires are used to evaluate if severity of dementia is associated with urge UI and other lower urinary tract symptoms (LUTs).
Patients and Methods:
A total of 43 AD patients were enrolled in this study, all of whom were checked post-void residual (PVR) urine amount by sonography after voiding. The severity of dementia was evaluated by questionnaire including Cognitive Abilities Screening Instrument (CASI), Mini Mental Status Examination (MMSE), Clinical Dementia Rating (CDR), and Clinical Dementia Rating Sub-of-Box (CDR-SB). The LUTs were assessed with International Consultation of Incontinence Questionnaire (ICIQ) and Overactive bladder symptom scores (OABSS) questionnaire. Independent t test and Pearson’s correlation analysis were calculated.
Results:
The average age in both AD with/without urge UI patients is 78 years old. The scores of CDR-SB, OABSS and ICIQ are significantly different in these 2 groups (p = 0.023, p = 0.003, p = 0.001; respectively). However, the neurophysiological scores of CASI, MMSE, CDR, CDR-SB is not correlated with OABSS (r = 0.047, p = 0.382; r = 0.074, p = 0.317; r = 0.087, p = 0.288; r = 0.112, p = 0.237; respectively). Interestingly, if we separate each individual symptom of OAB, there is a significant correlation between CDR-SB and urge UI score (r = 0.314, p = 0.023).
Conclusions:
Higher lower urinary tract symptom scores are noted in AD patients with urge UI. The CDR-SB score is highly correlated with urge UI in AD patients.
Introduction
With the increasing life expectancy, age-related diseases become more important in this era. Dementia, commonly seen in elder population, may impair the cognition and daily lives. 1 Alzheimer’s disease accounts for 60-80% of all dementia and the prevalence increases exponentially with age in Taiwan and across the world. 2,3 Urinary incontinence, comprised of urge incontinence, stress incontinence, overflow incontinence and functional incontinence, means an involuntary leakage of urine, defined by International Continence Society, which is commonly seen in elder patients. 4 Urinary incontinence affects one-third of women and 15%-20% older men. It is noted that urinary incontinence is prevalent in Alzheimer’s patients, both in early-stage and sever-stage disease. 5,6 Previous populational cohort studies also indicated higher risk of urinary incontinence in Alzheimer’s patients. 7 The most common UI in AD patients is urge UI according to previous study. 8 Both dementia and urinary incontinence could impair the quality of life in the elder, which increase caregivers’ burden. 9,10
Although Alzheimer’s disease is an independent risk factor of overactive bladder, only few studies have been published because of the difficulty to assess urinary incontinence in Alzheimer’s patients with severe cognitive impairment and physical disability. 11,12 The etiology of urinary incontinence in Alzheimer’s patients remains to be understood since it is thought to be multifactorial. However, the probable causes of urinary incontinence may include cognitive and functional impairment of neurodegenerative process, comorbidities and medication. 13 Previous studies pointed out that the severity of detrusor overactivity was linked to functional impairments and overactive bladder symptom score may be associated with neuropsychological parameters. 7,14 However, exclusion criteria including medication, previous pelvic surgery, previous prostate surgery, neurogenic bladder and menopause status were not considered in previous studies.
In order to clarify the association between lower urinary tract symptoms and the severity of Alzheimer disease. We aimed to specifically identify confirmed Alzheimer’s patients with overactive bladder symptoms and exclude patient with stress incontinence and parts of overflow incontinence. We assessed the lower urinary tract symptoms in AD patients and examined the correlation between neuropsychological parameters and urinary incontinence.
Material and Methods
Patient Collection
We enrolled 43 patients diagnosed Alzheimer’s diseases with one or more components of lower urinary tract symptoms in our neurology department from September 1st, 2017 to October 10th, 2018. Ethical approval was obtained from the Institutional Review Board and informed consent was obtained from each patient. This study was approved from our Institutional Review Board (KMUH-IRB-SV(I)-20180057). Patients with other dementia, severe dementia with behavioral disturbance or inability to communicate were excluded. Apolipoprotein E4 protein real time test was also performed for parts of the patients in this study.
All subjects underwent a thorough history taking, physical examination, urinary diary, and ultrasound post-void residual exam. In current study, patient who had stress incontinence and overflow incontinence were excluded. Cognitive tests were performed using the MMSE, CDR and CASI. Lower urinary tract symptoms were assessed using OABSS and the International Consultation of Incontinence Questionnaire – Short Form (ICIQ-SF). Brain computed tomography (CT) or magnetic resonance image (MRI) was also performed on each patient. Periventricular hyperintensity (PVH) and Deep white matter hyperintensity (DWMH) were further evaluated based on the brain CT and MRI images by 2 neurologists.
Statistical Analysis
Univariate analysis included student t test and chi-square test were used to evaluate the differences in gender, age, education time, comorbidities, cognitive test scores and lower urinary tract symptoms scores between urge UI + and urge UI − patients. Pearson’s correlation analysis was used to analyze the relationship of 2 independent variables. All statistical calculations were analyzed using SPSS version 19.0 (SPSS Inc., Chicago, IL, USA). A 2-tailed p-value lower than 0.05 was considered to be statistically significant.
Results
Characteristics of Alzheimer’s Disease With/Without Urge Incontinence
A total 43 patients (males 13 and females 30), with confirmed Alzheimer’s disease and with one or more lower urinary tract symptoms, were enrolled in this study. We divided the patients into 2 groups, one with urge incontinence and the other group without urge incontinence. Table 1 summarizes the comparison of clinical characteristics in Alzheimer’s disease patients between urge UI + and urge UI − patients. The average cognitive score of MMSE is 18.69 and 19.13 in urge UI+ and urge UI− patients, respectively. No statistical differences in age, gender, education time, neuropsychological tests (MMSE, CASI, CDR), white matter change in image study (PVH and DWMH), post residual void (PVR) were noted. However, among neuropsychological tests, CDR-SB increased significantly with urge urinary incontinence (p < 0.05). Differences of dyslipidemia, OABSS and ICIQ sum were also seen in these 2 groups (p < 0.05).
Characteristics of Alzheimer’s Disease With/Without Urge Incontinence.

Correlation Analysis of Overactive Symptoms and Neuropsychological Test Scores
In correlation analysis, no statistically significant differences were noted between OABSS and all neuropsychological tests (MMSE, CASI, CDR, CDR-SB) (Table 2). Nevertheless, individual symptom score of urge incontinence was highly correlated with CDR-SB scores (r = 0.314, p < 0.05). No correlations were found between other individual symptom scores and other neuropsychological test scores (Table 3).
Correlation Between Neurophysiologic Scores and OABSS.

Correlation Between Individual OAB Symptom Score and Neurophysiologic Parameters.
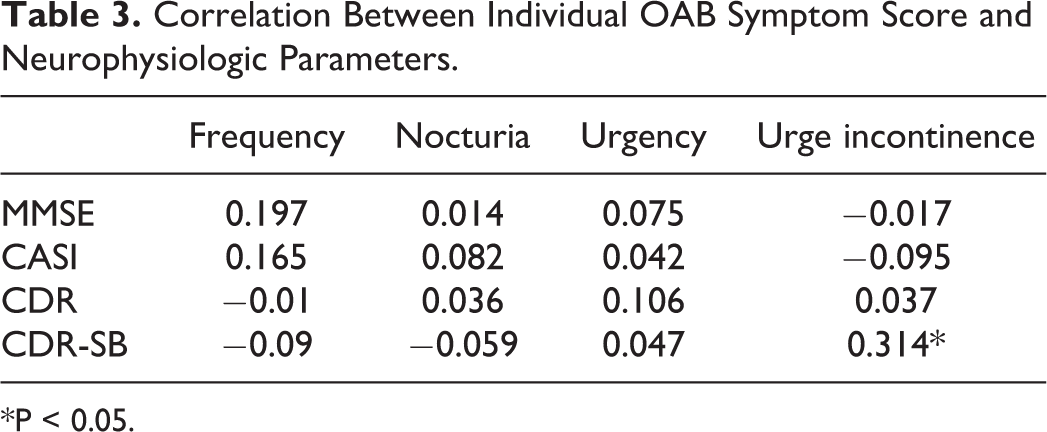
*P < 0.05.
Discussion
Overactive bladder is a troublesome disease that impairs quality of life in elders, especially in Alzheimer’s patients. Also, it could increase caregivers’ burden and economic issues in these families. 15 Few studies have been demonstrated the association between urinary incontinence and Alzheimer’s diseases. 7,8 In this study, we excluded stress incontinence and overflow incontinence because structural anomalies and neuropathic bladder could lead to difficulty in interpreting of the results. We also excluded previous pelvic surgery such as sling surgery in women and prostate surgery in men. Although prostate size and menopause may be risk factors in OAB, 16,17 all patients’ post-void residual amount were evaluated, and all female patients were in menopause status. As for medication, all patients were taking acetylcholinesterase inhibitors and all patients were treatment-naïve of lower urinary tract symptoms.
All 43 patients had one or more lower urinary tract symptoms and 13 of them had urge incontinence score >1, about 30%. Previous studies showed that all kinds of incontinence in dementia range from 11% to 90%, depending on patient sources from outpatient clinics or institutional samples, respectively. 18,19
Although incidences of OAB and AD increase with aging, there are no statistical differences in age between AD with urge UI and without urge UI in present study. The result was consistent with previous questionnaire-based correlation studies that there is no age difference between AD patients with OAB and without OAB as well as with UI and without UI. 8,14 Also, no statistical differences are found in comorbidities such as hypertension and diabetes mellitus. However, high prevalence of dyslipidemia is seen in AD patient with urge incontinence though patient number is limited.
We also analyzed the neuropsychological tests including MMSE, CASI and CDR to determine the relationship between severity of Alzheimer’ disease and lower urinary tract symptoms. The confirmed diagnosis of Alzheimer’s diseases was mostly based on cognitive impairment and dementia symptoms by neurologists and few patients were confirmed by Apolipoprotein E gene tests. The percentage of Apolipoprotein E (ApOE4) gene positive was about 15% in urge UI group and 20% in without urge UI group.
Since the white matter disease (WMD) may be overlapping with Alzheimer’s disease 20 and WMD could cause lower urinary tract symptoms, 12 we also analyzed white matter changes of PVH and DWMH in these patients. Besides, frontal white matter hyperintensity of brain MRI may predict probable lower urinary dysfunction in AD patients. 21 However, white matter changes of PVH and DWMH in our study have no significant difference between urge UI+ and urge UI−. One of the possible reasons is that PVH and DWMH are against whole white matter instead of frontal white matter and the other reason is that not all patient underwent brain MRI.
Mini Mental Status Examination (MMSE) is a questionnaire to measure cognitive function with a top score 30 points. The exam takes 5-10 minutes to complete and the questions include registration, attention, calculation, recall, language, ability to follow simple command and orientation. Previous study showed that nocturia is associated with MMSE score. 22 However, other studies failed to prove the correlation. 23 In the present study, no obvious statistical differences were noted in urge UI+ and urge UI− groups. MMSE is a short test but it is affected by educational level, age and insensitivity to disease progression. 24
Cognitive Abilities Screening Instrument (CASI) has a score ranging from 0 to 100. The exam takes 15-20 minutes to complete the questions involving attention, concentration, orientation, short-term memory, long-term memory, language abilities, visual construction, list-generating fluency, abstraction, and judgment. It is a useful tool in screening and monitoring disease that could provide clinical profiles of cognitive impairment. 25 No previous study correlated CASI with urinary incontinence, probably because the test was so time-consuming that most dementia patients could not fully answer all the questions. In present study, no significant correlation between CASI and urge incontinence was noted.
Clinical Dementia Rating Sum of Box (CDR-SB) is a good surrogate marker for disease progression in dementia patients and is highly correlated with CSF marker and glucose metabolism in AD. 26,27 It could capture the cognitive and functional impairments of the AD patient with less ceiling and floor effects. 28 Even mild dementia symptoms could be captured by using CDR-SB scores. 29 These properties may contribute to better correlation between CDR-SB and urge incontinence. Our results showed high correlation between urge UI and CDR-SB which is consistent with previous study. 23
The pathophysiology of urinary incontinence has not been well established. It is thought to be multifactorial because one factor is often related to another. First of all, detrusor overactivity is one of the proposed mechanism that is induced by loss of inhibitory function due to injury or degeneration of anteromedial frontal cortex in patients with AD. 30 Second, the cognitive function impairment was thought to be the main reason in AD patients with UI. Difficulties of recognition to empty the bladder, to identify the toilet, to remove clothes and to use the toilet were found in AD patients. 19
Third, previous observational studies showed that the functional impairments including inability to move at will, uncoordinated movement, difficulties with access to toilet and toilet posting are risk factors of UI. 31,32 Last but not the least, psychological condition including anxiety and depression, and Diabetes Mellitus might also cause UI. 10,30,31 The medication such as acetylcholinesterase inhibitors could affect bladder storage function by altering muscarinic receptors in detrusor muscle, aggravating UI in AD patients. 33,34
There are several limitations that must be addressed in the present study. Although it is a small cohort study with limited patient numbers, the small sample size is considered acceptable resulting from some reasons, A) this study is focused on patients with Alzheimer’s disease only, B) inclusion / exclusion criteria is strict, C) workup is comprehensive including brain imaging (CT or MRI) for each patients. Second, we only specified in urge incontinence because it is the most common urge incontinence in AD patients with lower urinary tract symptoms, and this makes our cohort more homogeneous. Also, we strictly excluded individuals with anatomical abnormalities and other associated factors that may contribute to urge incontinence. To explore the relationship between other type of UI and cognitive function, further longitudinal study is required. Third, there might be a certain selection bias because AD patients do not always complain urinary symptoms. Nevertheless, patients with severe dementia or inability to communicate or behavioral disturbance were excluded. Finally, elder patients usually take various medications that could aggravate voiding functions. We recorded that all the patients taking cholinesterase inhibitors in this study but did not fully assess other medications. Nevertheless, the AD patients in this study were treatment-naïve of lower urinary tract symptoms and post-void residual urine amounts were examined.
Conclusion
In conclusion, urge incontinence is the most common symptom in AD patients with overactive bladder. We demonstrated high correlation between urge UI and CDR-SB. Patients with more severe symptoms of dementia tend to have higher probability of urge incontinence. Further prospective cohort may provide more correlations between dementia and overactive bladder symptoms.
Footnotes
Authors’ Note
Che-Wei Chang and Hsiang-Ying Lee analyzed and interpreted the patient data and were major contributor in writing the manuscript. Han-Yuen Yang collected and interpreted patient data. Yung-Shun Juan revise the manuscript. All authors read and approved the final manuscript. All data generated or analyzed during this study are included in this published article. Ethics approval and consent to participate: Consents from all patient were obtained and were approved by Institutional Review Board in Kaohsiung Municipal Ta-Tung Hospital (KMUH-IRB-SV(I)-20180057).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
