Abstract
Objectives:
This study examined the efficacy of the General Practitioner Assessment of Cognition–Chinese version (GPCOG-C) in screening dementia and mild cognitive impairment (MCI) among older Chinese.
Methods:
Survey questionnaires were administered to 293 participants aged 80 or above from a university hospital in mainland China. Alzheimer disease and MCI were diagnosed in light of the National Institute on Aging and the Alzheimer’s Association (NIA/AA) criteria. The sensitivity and specificity of GPCOG-C and Mini-Mental State Examination (MMSE) in screening dementia and MCI were compared to the NIA/AA criteria.
Results:
The GPCOG-C had the sensitivity of 62.3% and specificity of 84.6% in screening MCI, which had comparable efficacy as the NIA/AA criteria. In screening dementia, GPCOG-C had a lower sensitivity (63.7%) than the MMSE and a higher specificity (82.6%) higher than the MMSE.
Conclusions:
The GPCOG-C is a useful and efficient tool to identify dementia and MCI in older Chinese in outpatient clinical settings.
Introduction
About 47 million people around the world are affected by dementia, and this number will reach 131.5 million by 2050, largely due to global aging. 1 As advanced age is the most salient risk factor of Alzheimer disease (AD), the global prevalence of dementia among those older than 80 is about 30%. 2 In China, the aging of population and subsequent growing prevalence of AD are public concerns. According to the China National Bureau of Statistics of 2016, there were about 150.03 million people aged 65 years or older in China, accounting for 10.85% of total population. By 2050, the number of people aged 65 years or above will reach about 329 million, and the number of those aged 80 years or above will reach about 121 million. 3 Thus, China will encounter challenges to provide treatment and care for the growing population for adults aged 80 years or above among whom the dementia rate is over 3 times that of those below age 80. 1
Much of the world’s projected increase in the number of individuals with dementia is attributable to the growing incidences in low- and middle-income countries. 4 The most remarkable trend is an estimated high prevalence of dementia in East Asia. 1 ,4 The World Alzheimer Report (2015) stated that the number of individuals with dementia in China was about 9.5 million in 2015. The number will reach over 16 million by 2030, and about 28 million by 2050. 1 Lower proportionate increases in dementia cases in high-income countries may be related to their efforts in dementia prevention, education, and treatment, as well as other factors such as improvements in living conditions and chronic disease management. This suggests that it is possible to reduce the risk and progression of dementia, namely, though early diagnosis and prevention of AD. Efforts aimed at early diagnosis and prevention may lead to net savings from delayed institutionalization. 5 -7
Despite the benefits of early diagnosis and prevention of AD, assessment of dementia in primary care settings is not commonly practiced. In Hawaii, one cross-sectional study reported 26 cases of dementia among 297 Asian American outpatients aged 65 years or older. However, 17 (65%) cases of dementia were not recorded in medical histories. 8 Furthermore, more than 90.9% of the mild cognitive impairment (MCI) cases were not previously assessed or recorded. 8 Mild cognitive impairment is a stage that has a high risk of advancing to dementia. About 10% of these people with MCI progress to dementia each year, with some reverting to normal cognition. 9 -11
Lack of use of a reliable, accurate tool in primary care settings might be one of reasons why cases of dementia are not being identified. Several screening tools for cognitive function are available, such as the Mini-Mental State Examination (MMSE) and the General Practitioner Assessment of Cognition (GPCOG). The MMSE has good sensitivity and specificity for detection of moderate-to-severe dementia 12 ; however, it is not sensitive to capture the MCI, 13 and it is influenced by respondent’s educational attainment. Little is known about whether or not the GPCOG can effectively screen out dementia and the MCI in Chinese older population. This study focused on the efficacy of the General Practitioner Assessment of Cognition–Chinese version (GPCOG-C) scale in primary care settings and made recommendations for practice changes.
Method
Design and Setting
This study selected Huazhong University of Science and Technology (HUST) as a research site. Huazhong University of Science and Technology is a public university located at Wuhan city in China. It is a comprehensive national university with 55 000 students and nearly 50 000 current staff members and retirees. There are about 4000 retired people aged 65 years, and more than 1300 are older than 80 years. The hospital affiliated with the HUST is a nonprofit organization that mainly serves college students, staff in service, and retired university employees. The hospital provides inpatient and primary medical services. Students, faculty, and staff are able to receive treatment in outpatient clinics, and the number of visits to such clinics totals around 370 000 a year.
Participants
For eligibility, participants must be aged 80 years or older and live in HUST communities. We excluded individuals with hearing loss, poor vision, mental illness history, developmental diseases, and long history of poison (eg, herbicides) exposure. Eligible participants were invited to receive physical examinations and cognitive tests in a face-to-face setting.
We recruited participants through 2 primary channels. Health education talks were utilized to attract patients who came to the outpatient department for routine checkups. The other approach was through phone contact using a retired employee list provided by the University Personnel Department. For each participant, we solicited a family member, including a spouse, adult child, or long-term caregiver, as his or her informant. We reached 409 eligible participants, among whom 10 individuals were excluded due to poor vision or hearing problems, and another 106 refused to join the research for various reasons (eg, inconvenience, no time, no interest). The remaining 293 individuals from the outpatient clinic comprised the final sample.
Research Procedure
This research was approved by the medical ethics committee of the HUST Hospital with the approval number of [2015] IEC (S047), and it confirmed to the principles of World Medical Association Declaration of Helsinki. All the participants provided written informed consent, and we ensured the privacy information about individual cases would not be published.
The research team was comprised of various professionals from 3 local hospitals. The primary investigators were from the HUST university hospital, including 1 senior neurological doctor, 1 resident physician, 2 psychologists, 3 psychological counselors, and 2 general physicians. In addition, consultation was sought from an expert from the Dementia Center of Wuhan Union Hospital and 2 psychiatrists from Li Yuan Hospital. Collaborators from the other 2 hospitals provided training to the student evaluation team, reviewed site assessments for quality assurance, and helped with diagnosis. After receiving training in dementia screening, 10 graduate students from Psychology and Social Work Departments of HUST conducted face-to-face interviews.
The expert from the Dementia Center of Wuhan Union Hospital was invited to train the research team on dementia screening. During the test, the student evaluation teams were randomly assigned to 2 or more groups to perform dementia screening, with a psychiatrist or a neurologist assisting with screening.
A dementia screening studio was set up in the outpatient department to conduct screening for routine visit patients aged 80 years or older. The studio was spacious, bright, and quiet to ensure that no noise from the outside environment would affect participants’ mood and concentration. Patient and researcher trust was cultivated through open communication prior to research. Participants also received regular physical examinations, which included blood pressure screenings and height and body weight measurement. Blood samples were taken to detect a complete set of homologous cysteine, folic, and acid levels. All screening processes were conducted free of charge, and participants were asked to sign an informed consent.
Assessment Tools
Participants’ demographic information was obtained through the Elderly Health Service Assessment Form and Population and Lifestyle Questionnaire. The National Institute on Aging and the Alzheimer’s Association (NIA/AA) criteria for the diagnosis of AD 14 was used as reference to identify AD and MCI. We also used cognition scales, including GPCOG-C and MMSE. In addition, Hachinski ischemia scale was used to distinguish dementia subtypes, along with a physical functioning scale and a behavioral problem scale.
The GPCOG-C has acceptable internal consistency and test–retest reliability, 15 with exemplary sensitivity of 97.4% and specificity of 89.2% in detecting dementia found in one study. 15 It consists of 2 sections: a patient examination (GPCOG-patient) and an informant interview. The patient section has 4 items: time orientation, reporting an event, a clock drawing, and a word recall task, and the informant interview has 6 items. Items were derived from the Cambridge Cognitive Examination, 16 the Psychogeriatric Assessment Scale, 17 and the instrumental activities of daily living scale. 18 According to the sequential 2-stage scoring method, 19,20 the patient’s cognition is tested in the first step: A score of 9 indicates intact cognition and scores lower than 5 indicates impaired cognition. 19 However, the next step of an informant interview clarifies whether there is significant cognitive impairment when the patient section score fall between 5 and 8. Scores lower than 4 on the informant section indicate significant cognitive impairment. 19 In this study, the patients were considered at risk of cognitive abilities and probably MCI if they had the scores of 4 or higher in informant interview and the scores between 5 and 8 in the patient’s section. The patient section took less than 4 minutes and the informant interviews took less than 2 minutes to complete. The administration time of the GPCOG (2-stage method) was significantly shorter than that of the MMSE. 15
The MMSE is one of the oldest and is the most commonly used cognitive assessment tool. The MMSE had a good sensitivity of 81%, specificity of 76%, and a positive predictive value of 57%. 19 However, the MMSE scores were often influenced by other factors, such as age and educational level, 21 and it usually takes more than 6 minutes to complete. In this study, we used the cutoff point of 26 to detect dementia 22,23 and the screening threshold of MMSE scores (27 ≤ and ≤ 29) to distinguish MCI. 24
Hachinski ischemia scale is a simple clinical tool to distinguish different subtypes of dementia, such as AD, multi-infarct dementia (MID), and mixed (AD plus cerebrovascular disease) dementias, and the cutoff value was ≤4 for AD and ≥7 for MID. 25,26
The Alzheimer Disease Cooperative Study–Activities of Daily Living scale assessed the difficulties of performing activities. This 78-item scale ranged from self-maintenance to ability to complete instrumental activities, with higher scores indicating better cognition. The Alzheimer Disease Cooperative Study–Activities of Daily Living has broad applicability, good test–retest reliability, wide scaling to cover a range of performance, and good sensitivity to detect change in disease progression. 27
The Neuropsychiatric Inventory (NPI) was used to assess 10 behavioral disturbances occurring in patients with dementia: delusions, hallucinations, dysphoria, anxiety, agitation/aggression, euphoria, disinhibition, irritability, apathy, and aberrant motor activity. The advantages of using NPI included evaluating a wide range of psychopathology, soliciting information about behaviors that may distinguish among different etiologies of dementia, and minimizing administration time by focusing only those behavioral domains with positive responses to screening questions. 27
Overall, GPCOG is a tool to measure the patient’s cognition, along with other above-described measures on dementia types, physical function, and behavioral problems. Thus, GPCOG is a part of a full assessment of the patient’s cognition, including the exclusion of delirium, psychotropic medication use, and depression.
Results
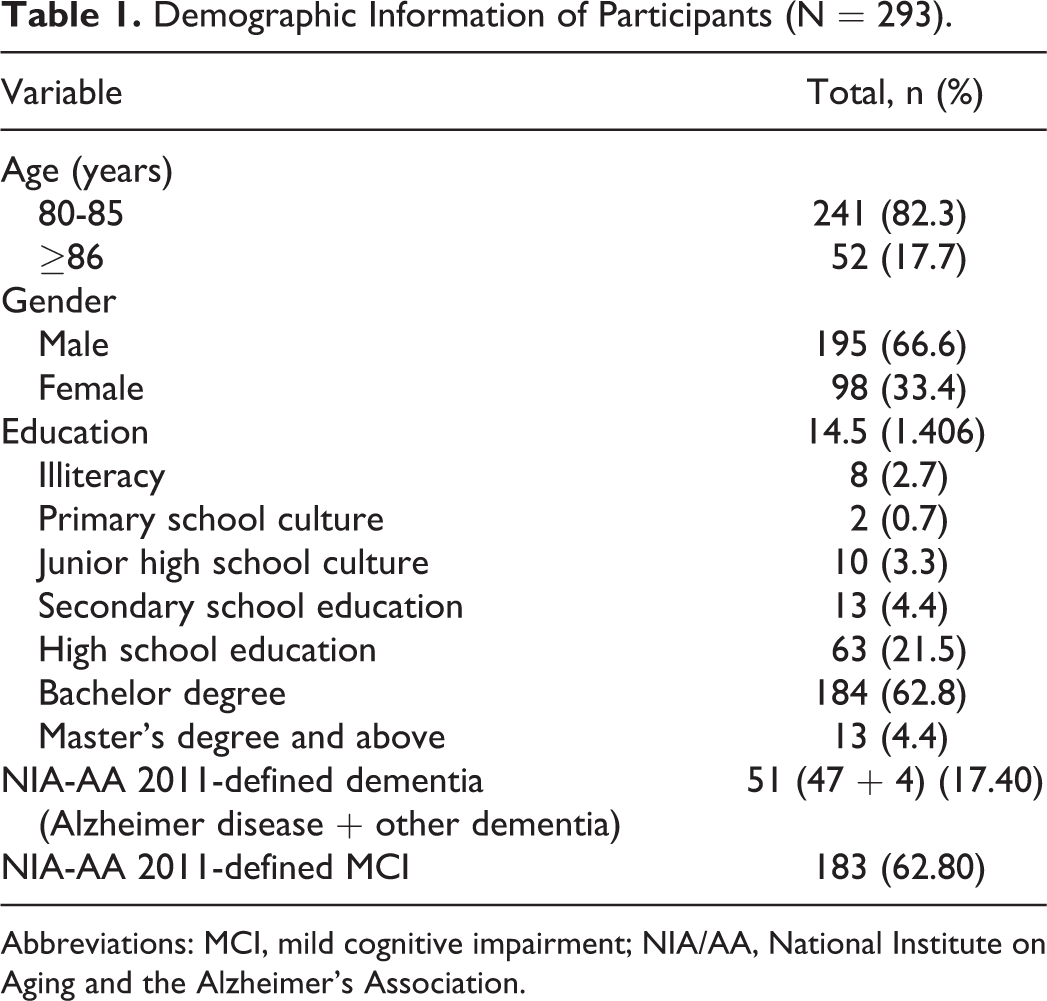
Descriptive statistics of variables used in this study are summarized in Table 1. The average age of the participants was 82.87 (standard deviation [SD] = 3.134) years. Most (82.3%) of them were 80 to 85 years old, and 66.6% of the total sample was male. According to Chinese educational habits, educational levels divided from low to advanced illiteracy, primary school education, junior high school education, technical secondary school education, high school education, bachelor’s degree, master’s degree and above, and with most of the participants (62.8%) having bachelor’s degrees.
Demographic Information of Participants (N = 293).
Abbreviations: MCI, mild cognitive impairment; NIA/AA, National Institute on Aging and the Alzheimer’s Association.
The expert from the Dementia Center of Wuhan Union Hospital and clinical neurologists from HUST university hospital applied the NIA/AA criteria for the diagnosis of AD, and 51 individuals were diagnosed with dementia: 47 with AD, 4 with other dementias, and 183 individuals with MCI (see Table 1). The mean score of Hachinski scales was low (mean = 0.54, SD = 1.172), which was consistent with our finding, indicating that most dementia type was AD.
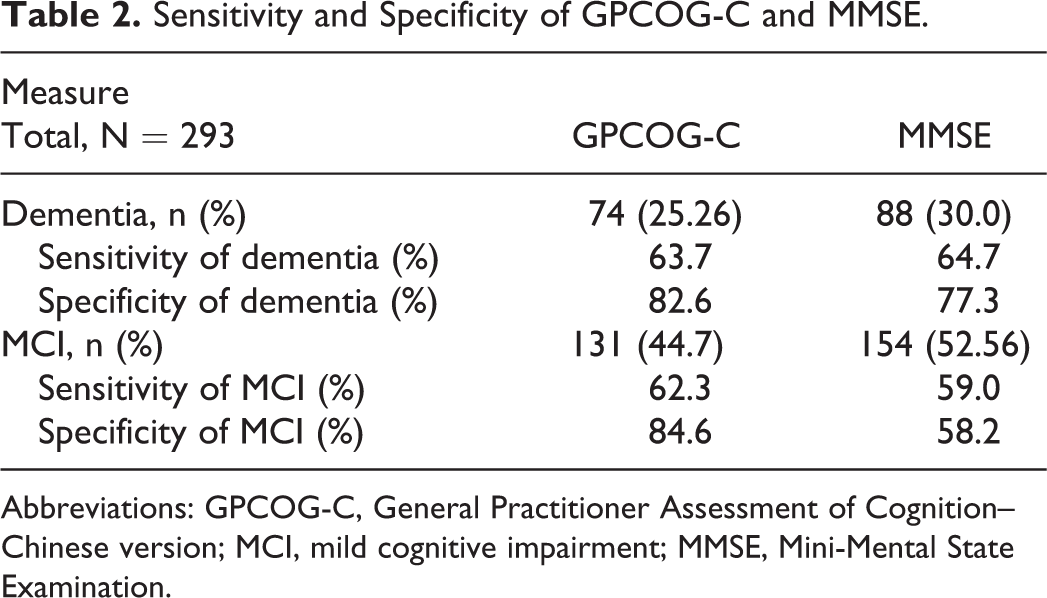
Using GPCOG-C, 131 people had MCI. Among them, 114 were diagnosed as MCI according to the NIA/AA 2011 diagnostic criteria. As shown in Table 2, GPCOG-C has the sensitivity of 62.3%, and the specificity of 84.6% to screen the outpatients with MCI. Thus, GPCOG-C has comparable efficacy as the NIA/AA criteria when screening outpatients with MCI. For identifying dementia, GPCOG-C had the sensitivity of 63.7% and specificity of 82.6%. The GPCOG-C meets the NIA/AA criteria well when screening outpatients with dementia. Its sensitivity was slightly worse, but specificity was better than that of the MMSE.
Sensitivity and Specificity of GPCOG-C and MMSE.
Abbreviations: GPCOG-C, General Practitioner Assessment of Cognition–Chinese version; MCI, mild cognitive impairment; MMSE, Mini-Mental State Examination.
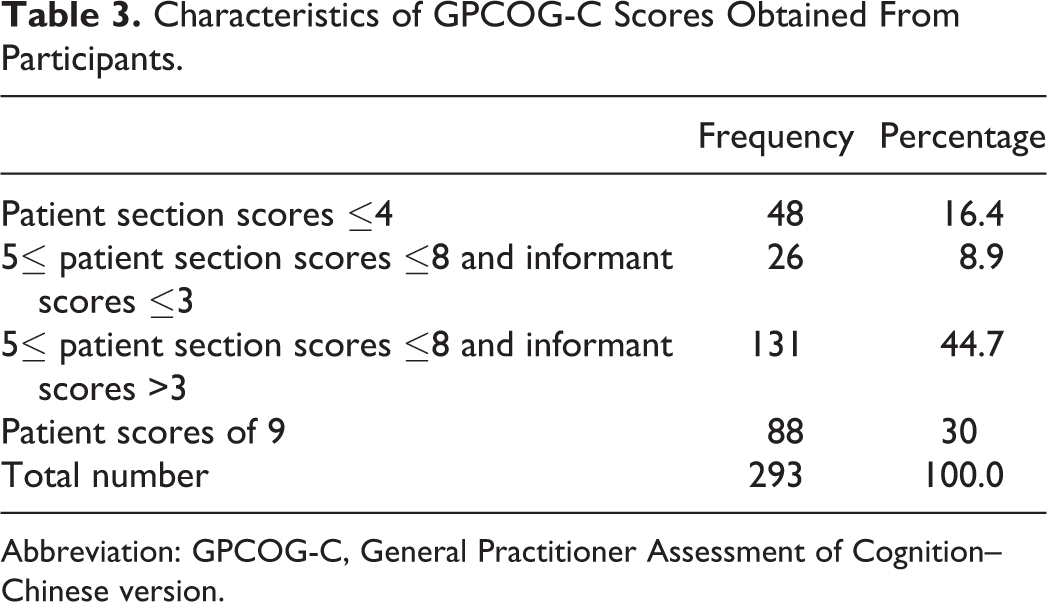
As shown in Table 3, according to the GPCOG-C, 157 (53.6%) participants had the patient scores between 5 and 8 on the cognitive testing. It implies that only 53.6% of participants need to complete GPCOG-C informant section because informant questioning is not necessary for those with score below 5 or above 8.
Characteristics of GPCOG-C Scores Obtained From Participants.
Abbreviation: GPCOG-C, General Practitioner Assessment of Cognition–Chinese version.
Discussion
The oldest-old Chinese people are not screened for dementia or the MCI on a regular basis in primary care settings due to the lack of social cognition and medical resources. Regular outpatient checkups are a feasible way to identify and reach out to patients with cognitive problems. The US Preventive Services Task Force states that “clinicians should assess cognitive function whenever cognitive impairment or deterioration is suspected, based on direct observation, patient report, or concerns raised by family members, friends or caretakers.” 28 However, many patients and their families consider cognitive decline a natural part of aging process, and 90.9% of the mild dementia cases were overlooked. 8 Mild cognitive impairment is an unstable state with a high likelihood of advancing to dementia. The risk of dementia was elevated in MCI cases (hazard ratio [HR]: 23.2) compared with cognitive normal individuals. 29 About 38% of MCI participants reverted to cognitive normal (175.0/1000 person-years), but 65% of them subsequently developed MCI or dementia; the HR was 6.6 compared with cognitive normal individuals. 29 Mild cognitive impairment can be regarded as a precursor state for dementia, and its identification could lead to secondary prevention strategies. 30
Moreover, evidence of behavioral symptoms or increased dependence in activities of daily living is required to identify AD or a related dementia. Yet, the patient or an informant may not voluntarily offer this information unless prompted by a provider. 8 This lack of awareness among patients and family members calls for an urgent effort from health-care providers to offer dementia screening and associated consultations.
Early identification allows patients and their families to prepare for a future when they are diagnosed with dementia. Generally, patients with AD will live between 3 and 20 years from time of diagnosis, with an average of 8 years. 31 If patients are identified in the early stages of cognitive decline, they will be able to participate in discussions regarding financial planning, preferences for end-of-life care, and future living arrangements. Early screening and intervention for MCI may be helpful to slow down the progression of dementia. 32 Thus, it is necessary to regularly screen for dementia and the MCI in primary care settings. Yet, despite the benefits, dementia screening may have some adverse psychological effects on the participants that we should be cautious about. Feelings of depression, desperation, and hopelessness may occur among patients and/or family members. Therefore, assessment of emotional changes after diagnosis should be conducted, and counseling should be provided if needed. 33
The GPCOG-C has similar advantages of the original English version and is an efficient, simple, and time-saving instrument for dementia screening. 15 , 19 In addition, it fits primary care settings. The 2 parts of GPCOG—patient section and informant interview section—are integrated. Therefore, the scale not only reflects patients’ cognitive changes but also assesses the daily life of patients from the perspectives of caregivers.
In this study, our sensitivity and specificity were slightly lower than findings from Li et al’s study. 15 This may be due to different sample characteristics as in our sample most were highly educated. Participants were deemed as MCI if they had the scores of 4 or more in the informant interview and the patient’s section scores of 5 to 8. The conclusion was based on our clinical experience and some existing evidence. One study in Italy suggests that if informant section had a score of 3 or more, they recommended follow-up examinations advised in 6 months. 23 This implies that this group cannot be considered cognitive intact, rather an at-risk group.
Most researchers and clinicians conventionally believe that patients with a score of 4 or more in the informant section as cognitively intact. We tend to challenge this assertion for 2 reasons. First, this conclusion was not explicitly endorsed by the designer of the GPCOG. 19 Second, based on our clinical experiences, a large proportion of patients with the scores of 4 or more were eventually diagnosed with MCI. In this study, the findings confirmed our assumption. Using our GPCOG-C standards for MCI, 131 people had MCI. Among them, 114 were diagnosed with MCI using the NIA/AA 2011 diagnostic criteria. That largely supports our claim that patients with 4 or more scores in the informant section and 5 to 8 scores on patient section are cognitively at-risk, in other words, MCI.
In regard to screening dementia, most studies compared the efficiency of the GPCOG with the MMSE. This study indicated that the GPCOG-C had better sensitivity and specificity in screening MCI compared with the MMSE.
A systematic review evaluating 28 screening tools in 7 Asian languages suggested that the MMSE is the most widely used tool and has good sensitivity (82%-92%) and specificity (62%-100%) in detecting dementia. 34 Most studies indicated similar diagnostic properties for the GPCOG total score compared to the MMSE. 19,23 In this study, the GPCOG-C had the sensitivity of 63.7% and specificity of 82.6% to detect dementia, similar to the originally reported score by the designer of GPCOG. 19 The misdiagnosis rate was almost 20%, as a preliminary screening. Those people screened as dementia (regardless true positives and false positives) using the GPCOG-C in primary care will receive a referral within a week to a nearby comprehensive hospital—Wuhan Union Hospital. They will be carefully examined again for a final diagnosis by one geriatric neurologist expert with a small amount of cost. These false positives will be corrected by the expert.
Moreover, the efficiency of the GPCOG was independent of the patient’s gender and emotional status, and relatively independent of age and educational level. 23 It is designed specifically for primary care settings, and its 2-stage administration enhances efficiency, 23 and it takes 4 to 6 minutes to complete.
In contrast, MMSE takes 5 to 10 minutes to administer. Its efficacy is subject to the influence of patient’s age, education level, and ethnicity/culture. 21,35 -37 Also, the MMSE’s cutoff points are different, and the tool is inconvenient for long-term continuity management. 38 This study showed that only 53.6% of all GPCOG-C administrations required the informant section to be completed. Compared to the MMSE, the 2-stage procedure of the GPCOG-C was more time-efficient. The administration of GPCOG takes less time while demonstrating the comparable accuracy of screening dementia.
We attempted to compare GPCOG-C, and the Montreal Cognitive Assessment (MoCA), a commonly used tool to assess cognition although not used in this study. Compared to MoCA, GPCOG-C has similar advantages in its implementation. A systemic review indicated that MoCA has acceptable sensitivity (68.7%-93%) and specificity (63.9%-100%) in detecting the MCI. 34,39 However, compared to findings of the systematic review, GPCOG-C had similar specificity of 84.6% and slightly worse sensitivity of 62.3% compared with MoCA. Nonetheless, GPCOG-C might be a more attractive assessment tool for providers, given how quick the assessment process is. The MoCA is a 1-page, 30-point test administered approximately in 10 to 15 minutes, 40 while, in contrast, the GPCOG-C can be completed in about 4 to 6 minutes. Also, MoCA performance was found to be influenced by education, gender, and age, 40 while the GPCOG-C was unaffected by these factors. 20,23
Overall, GPCOG-C is economical, simpler, and more time-efficient than many other cognitive assessment tools. It is also unaffected by age, gender, education, regional culture, and depression. 20,23 Thus, GPCOG-C can be regarded as a better screening tool in detecting dementia and the MCI compared with other tools. However, the findings should be interpreted in the context of limitations. It is unfortunate that we did not include the MoCA in the screening scales, which we suggest be added in future studies. Although we provided standard training to all research team members, different capabilities across individual research assistants might influence results. Finally, most of the screened people had higher education with an undergraduate education or above being the average, which may have caused a slight ceiling effect.
Conclusion
It is necessary to identity dementia and the MCI on a regular basis in primary care settings for timely diagnosis and treatment. This study suggests that GPCOG-C is a well-designed screening tool that effectively identifies dementia and the MCI in the oldest-old and highly educated Chinese population. With an average assessment time of 4 to 6 minutes, it takes less time to screen for dementia and MCI, as an early caution to the families who have a relative with dementia. Providing families with such guidance and education may lower the economic costs associated with caregiving, save on medical resources, and reduce societal burden. All of this makes the GPCOG-C feasible and ideal to use in China’s busiest geriatric outpatient clinics.
Footnotes
Authors’ Note
Fang Xu and Juan Juan Ma contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
