Abstract
Cerebrovascular disease (CVD) contributes to spatial navigation deficits; however, the everyday outcomes of this association remain unexplored. We investigated whether CVD was a risk for getting lost behavior (GLB) in elderly with mild cognitive impairment (MCI) and mild Alzheimer disease (AD). Getting lost behavior was assessed using a semistructured clinical interview and was associated with white matter lesions (WMLs) in patients with MCI. Specifically, right occipital WMLs increased the odds of GLB by 12 times (P = .03) and right temporal WMLs increased the odds of GLB by 4 times (P = .01), regardless of age, gender, global cognitive impairment, and occipital or medial temporal gray matter atrophy. Hypertension increased the risk of GLB in MCI by contributing to the burden of WMLs. White matter lesions were not associated with GLB in mild AD. Our findings suggest that interventions aimed at reducing GLB in prodromal dementia may involve preventing WMLs by optimizing hypertension control.
Introduction
Getting lost behavior (GLB) is defined as the inability to find one’s way in familiar or unfamiliar environments. 1 Although GLB is observed as part of the normal aging process, 2 its prevalence is exacerbated in elderly with cognitive impairment, including prodromal dementia and early-stage dementia. 3 The consequences of GLB are severe as dementia patients with GLB experience greater rates of institutionalization, falls, and death compared to dementia patients without GLB. 3 Thus, it is critical to identify individuals at risk of GLB and to offer timely preventive treatments and precautionary safety measures. In this regard, understanding the pathophysiology underlying GLB is of paramount importance to allow early detection and effective intervention.
Getting lost behavior is a consequence of age-related decrements in cognition. 1 One modifier of these age-related effects on GLB can be cerebrovascular disease (CVD), 4 which is highly prevalent in elderly individuals. In CVD, the perfusion and oxygen supply to the brain is affected, which leads to cerebral insults such as cerebral white matter lesions (WMLs), lacunes, and perivascular spaces. 5 These CVD-related lesions are most prevalent in regions critical for wayfinding, namely the prefrontal, parietal, and medial temporal regions. 6 Among these, lacunes have been associated with reduced performance on spatial navigation tasks in patients with mild cognitive impairment (MCI). 7 These findings provide preliminary support for the vascular basis of GLB.
The most reliable MRI surrogate for CVD is WMLs, which represent demyelination and axonal loss. 8 White matter lesions are common in the aging population and are of high clinical significance as they predict cognitive decline and conversion from MCI to dementia. 9 Specific cognitive abilities that are most vulnerable to WMLs include executive functions, memory, visuospatial ability, and information processing speed, 10,11 and dysfunctions in each of these abilities play a role in GLB. 12,13 Thus, WMLs may contribute to the pathophysiology of GLB in both cognitively normal and cognitively impaired elderly; however, this hypothesis remains to be proven. Given that WMLs can be a potential target for intervention 6 to reduce GLB, identifying the role of WMLs in GLB may have a significant impact in reducing the prevalence of GLB.
White matter lesions are most prevalent in elderly with cardiovascular risk factors including hypertension, diabetes, hyperlipidemia, smoking, and obesity. 2 Chronic hypertension in particular has been shown to be detrimental to structures of the brain critical for spatial navigation 14 and as a consequence, associated with poorer performance on spatial navigation tasks in the healthy aging population. 15 Hypertension and other vascular risk factors may be a potential therapeutic target for reducing the burden of CVD on GLB. To date, the role of hypertension and other risk factors on GLB, as a function of WMLs, remains unclear.
In this study, we sought to investigate the associations of WMLs and vascular risk factors with GLB in a cohort of healthy controls (HCs), MCI, and mild Alzheimer disease (AD). We hypothesized that in HC and MCI, WMLs are a risk for GLB, independent of gray matter atrophy. We further hypothesized that the association between WMLs and GLB may be driven by vascular risk factors such as hypertension, diabetes mellitus, hyperlipidemia, and an overweight or obese BMI. In patients with mild AD, we hypothesized that WMLs may have limited association with GLB given that neurodegeneration may outweigh the impact of WMLs. 2
Materials and Methods
Participants
Patients with MCI and mild AD were recruited from a tertiary neurology center (National Neuroscience Institute, Singapore) between 2013 and 2016. Diagnosis of MCI was based on Petersen’s criteria 16 and included patients with a Clinical Dementia Rating Scale (CDR) 17 of 0.5. Mild probable AD was based on NIA-AA Criteria 18 and included patients with a CDR of 1. All diagnoses were conducted by cognitive neurologists and supported by clinical interviews, comprehensive neuropsychological assessments, and a blood test to rule out cognitive impairment due to vitamin deficiency or thyroid abnormalities. Age-matched HC from the community were recruited at the National Neuroscience Institute from 2010 to 2016 and included elderly who were “cognitively normal” as determined by a comprehensive cognitive assessment and a CDR of 0.
Exclusion criteria for all participants included (a) major visual impairment, such as blindness, visual agnosia, or cortical blindness, (b) a current diagnosis or history of severe neuropsychiatric conditions (eg, psychosis, depression), (c) comorbid neurodegenerative diseases (eg, Parkinson disease), (d) a history of clinical strokes, and (e) a history of drug and alcohol abuse.
The study received approval from the Institutional Ethics Committee and informed consent was obtained according to the Declaration of Helsinki from the controls and patients, or their next of kin if they were mentally incapable of giving consent.
Measures
Getting lost behavior was indexed using a semistructured clinical interview with a psychologist blinded to diagnosis. The interview queried the changes, if any, in visuospatial abilities and involved responses from both the participant and the caregiver, or family member in the case of HC. The participant was queried on whether they still travel alone, how well they can recall travel routes (including travel route to the present location), whether they make wrong turns on familiar paths and whether they have experienced getting lost in the past 6 months. Questions to caregivers/family members sought to validate participants’ responses and focused on whether the participant still traveled on their own and whether there have been instances of getting lost in the past 6 months. The presence of GLB was recorded as a yes or no by the psychologist based on clear indications that the participant was not able to orientate themselves in familiar environments or that there have been instances of getting lost. More detail is published elsewhere. 13
Vascular risk factors such as age, gender, and educational attainment were obtained from clinical interview, while history of hypertension, diabetes, hyperlipidemia, and BMI were collected from medical records.
Global cognition was indexed using the Montreal Cognitive Assessment 19 and locally validated norms. 20
MRI markers included volume of WMLs and gray matter using a 3T Siemens Tim Trio system (Siemens, Erlangen, Germany) for participants scanned before 2015 (N = 62), and a 3T Siemens Prisma system (Siemens, Erlangen, Germany) for participants recruited in 2015 and beyond (N = 101). Scanner differences have previously been shown to have negligible effects on segmented gray matter images in patients with AD scanned across 6 different scanners 21 ; thus, scanner type was not included as a confound. 3D volumetric scans were obtained using a T1-weighted magnetization-prepared rapid gradient-echo (MPRAGE) sequence (repetition time 2300 ms, echo time 2.98 ms, matrix = 192 × 256 × 256, voxel = 1.0 mm isotropic) as per the Alzheimer Disease Neuroimaging Initiative protocols (http://adni.loni.usc.edu/). Voxels of WMLs were obtained from Fluid Attenuated Inversion Recovery (FLAIR) sequences, using the corresponding MPRAGE sequence as a template. Voxel-based morphometry was obtained using the Computational Anatomy Toolbox (CAT12) package for the Statistical Parametric Mapping 12 (SPM12) software (http://www.fil.ion.ucl.ac.uk/spm) in MATLAB. Images were segmented into gray matter, white matter, and cerebrospinal fluid maps using a unified segmentation pipeline 22 including affine regularization to the International Consortium for Brain Mapping space template for East Asian brains. The generated gray matter and white matter maps were then used to generate global volumes of gray and white matter. Volumetric analysis of WMLs was done in SPM8 using an existing workflow. 23 FLAIR sequences were coregistered with their corresponding Montreal Neurological Institute (MNI)-normalized MPRAGE sequences, and normalized to MNI space. Voxels of WMLs were identified based on having an intensity of at least 1.4 times compared to the surrounding white matter, and standardized region of interest maps were used to distinguish between WMLs in the frontal, occipital, temporal, and parietal lobes, as well as between periventricular and deep WML. 24
Statistical Analysis
Statistical design
Identify the between-group (diagnostic group) and within-group (GLB group) differences in demographics, global cognition, vascular risk factors, WMLs, and gray matter volumes. Determine the correlation between GLB and both vascular risk factors and WMLs for each diagnostic group. For significant correlations between GLB and WMLs, determine the adjusted odds ratio (OR) that patients with a specific WML profile would have GLB, using logistic regression. For significant regression findings, confirm whether the relationship between GLB and WMLs were independent to gray matter using an interaction analysis. For significant regression findings, determine whether vascular risk factors were an important driver of the association between GLB and WMLs using mediation analysis.
Group differences
Between group differences were determined using analysis of variance with Tukey post hoc for continuous variables and χ2 for categorical variables. Within group differences between those with and without GLB was determined using t test for continuous and χ2 for categorical.
Correlation
A bivariate Pearson correlation determined the association between GLB, demographics, and vascular risk factors. A partial Pearson correlation, controlling for age, education, and global cognitive impairment, determined the association between regional WML volume and GLB. The left and right regions of WML were assessed given asymmetric hemispheric association with visuospatial processing. 25 All correlations with GLB corrected for multiple comparisons using false discovery rate, which uses rank ordering of P values as a step-down hypothesis testing procedure to ensure that the smallest P value is subjected to the most stringent critical P threshold. 26 False discovery rate methods ensure higher power compared to family-wise error rate procedures, which is advantageous for statistical designs that identify trends using multiple tests and further analyze the significant findings.
Regression
Logistic regression included GLB as the outcome, WML as the primary predictor, and the covariates as age, years of education, global cognitive impairment, and regional gray matter volume. The medial temporal gray matter was included as a covariate instead of temporal gray matter given we previously found it to be associated with GLB. 13 Each region of WML was assessed in an independent regression. Healthy controls, MCI, and patients with mild AD were assessed in independent analyses. Odds ratios were interpreted using Chen and colleagues 27 guidelines: OR of 1.68 (small), 3.47 (moderate), and 6.71 (large).
Interaction
Interaction analysis was conducted using Hayes 28 PROCESS macro for SPSS. In this model, the outcome was GLB, the predictor variable included regional WMLs which were significant in the partial correlation, and the interacting variable included the corresponding regional gray matter volumes.
Mediation analysis
Mediation analysis quantifies whether the association between GLB and vascular risk factors (X − Y, in Figure 1) can be explained by WMLs as an intervening variable. 28 Mediation is observed if the direct relationship between vascular risks and GLB (path c in Figure 1) reduces in significance when controlling for the mediator (path c′ in Figure 1). An independent mediation analysis was conducted for each vascular risk factor and WML region. All models were assessed using AMOS Bayesian methods with Markov chain Monte Carlo (MCMC) simulation. 29 The MCMC uses simulation to generate random samples of parameters from a probability distribution and uses these samples to approximate expectations of quantities of interest. Bayesian methods provide different inferences to conventional mixed linear models (MLMs) 30 : for instance, estimated means are analogous to regression β weights derived from MLM, the standard distance is analogous to the standard error in MLM, and 95% credible intervals are analogous to confidence intervals in MLM. A 95% credible interval that did not contain zero indicates a significant effect 30 and implies that the true estimate is likely to be contained within the credible intervals. 29 Model fit was assessed as MCMC posterior P value centering close to .5. 31
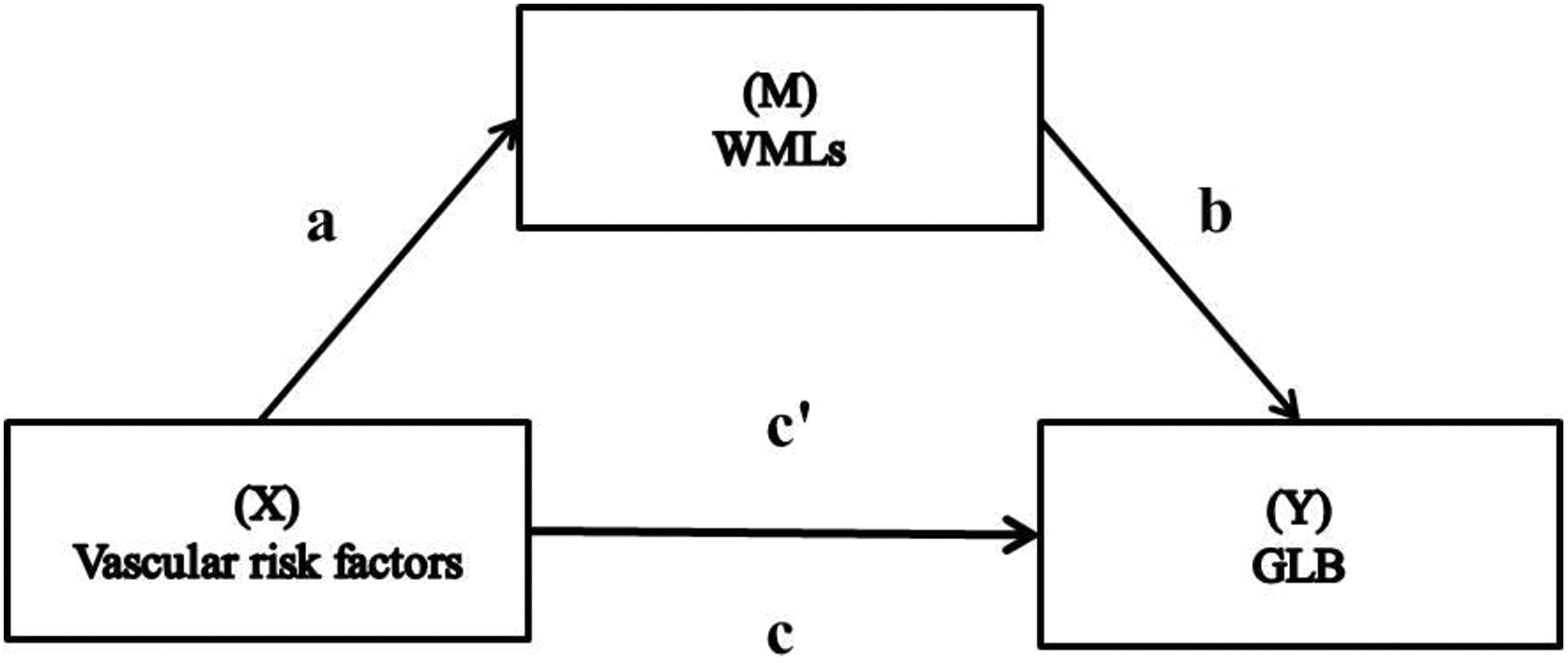
Mediation model depicts that the relationship between vascular risk factors (X) and getting lost behavior (GLB) (Y) (Path c) may be mediated by white matter lesions (WMLs) (M) (path ab → c) (Adapted from Hayes 28 ).
Results
Participants
Group differences
Table 1 indicates that compared to HC and MCI, patients with mild AD were older, F 2,176 = 28.78, P = .00; had less years of education, F 2,176 = 19.88, P = .00; and experienced a higher prevalence of GLB, F 2,176 = 4.16, P = .01; and hypertension, χ2(2, 179) = 7.64, P = .02. They also experienced a greater volume of periventricular WMLs, F 2,176 = 4.67, P = .01; and less gray matter volume, F 2,176 = 19.02, P = .00; specifically in the frontal, F 2,176 = 13.38, P = .00; parietal, F 2,176 = 12.45, P = .00; medial temporal, F 2,176 = 28.45, P = .00; and occipital regions, F 2,176 = 16.95, P = .00. Compared to HC, patients with mild AD had greater cortical WMLs in the frontal, F 2,176 = 3.38, P = .03; and occipital regions, F 2,176 = 3.54, P = .03. No differences in WML were observed between MCI and mild AD. Global cognition was significantly different among all 3 groups, F 2,176 = 139.05, P = .00. Healthy control and patients with MCI did not differ on prevalence of GLB or any of the demographic and MRI markers.
Between Group and Within Group Differences in Demographics, Cognition, Vascular Risk Factors, and MRI Markers.
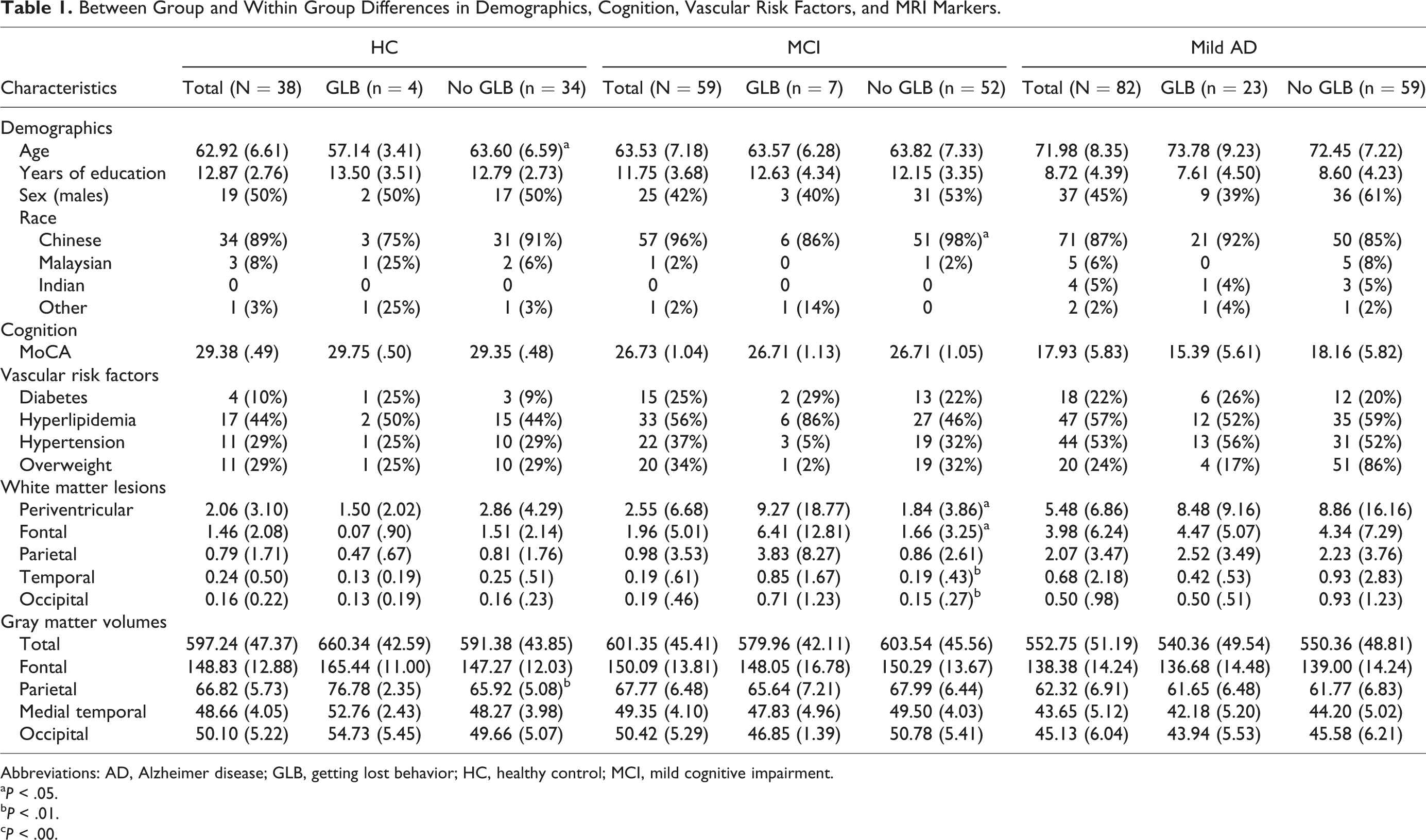
Abbreviations: AD, Alzheimer disease; GLB, getting lost behavior; HC, healthy control; MCI, mild cognitive impairment.
a P < .05.
b P < .01.
c P < .00.
Within-group differences
Within the HC group, those with and without GLB did not differ in demographics, global cognition, and vascular risk factors or WMLs (Table 1). Parietal gray matter was lower in HC with no GLB compared to those with GLB.
Within the MCI group, patients with GLB had higher total, frontal, temporal, and occipital WMLs compared to MCI with no GLB (Table 1). No differences were observed for demographics, global cognition, vascular risk factors, or gray matter volumes.
Within the mild AD group, those with and without GLB did not differ in demographics, global cognition, vascular risk factors, WMLs, or gray matter volumes (Table 1).
Associations
Correlation
In HC, GLB was not associated with demographic characteristics, vascular risk factors, or WMLs after controlling for covariates and adjusting for multiple comparisons (etable 1).
In patients with MCI, GLB was not associated with demographic characteristics or vascular risk factors, however, was associated with right and left occipital WMLs and right temporal WMLs after controlling for covariates and adjusting for multiple comparisons (Table 2).
Correlation Between GLB and Vascular Risk Factors, WMLs, and Gray Matter in Patients With MCI.

Abbreviations: GLB, getting lost behavior; MCI, mild cognitive impairment; MoCA, Montreal Cognitive Assessment; WMLs, white matter lesions.
a P < .00.
b P < .05.
cSignificant after adjusting for multiple comparisons.
d P < .01.
In patients with mild AD, GLB was associated with cognitive impairment and medial temporal gray matter; however, the associations did not survive multiple comparison correction. No associations were observed between GLB and vascular risk factors or other WMLs after controlling for covariates and adjusting for multiple comparisons (etable 2).
Regression
For patients with MCI, GLB was associated with right occipital WMLs with an OR of 12.15 (b = 2.49, standard error (SE) = 1.72, 95% confidence interval [CI]: 0.42-34.99; P = .03) and right temporal WMLs with an OR of 4.65 (b = 1.54, SE = .91, 95% CI: 0.78-28.02; P = .01). Getting lost behavior was not associated with left occipital WMLs (P > .05).
Interaction
In patients with MCI, GLB was not associated with the interaction between right occipital WMLs and right occipital gray matter, nor right temporal WMLs and right medial temporal gray matter, after controlling for covariates (P > .05). This indicates that the association between GLB and both occipital and temporal WMLs was independent to occipital and temporal gray matter, respectively.
Mediation
In patients with MCI, right occipital and right temporal WMLs were mediators for the indirect relationship between hypertension and GLB (Table 3). That is, hypertension was associated with the prevalence of GLB by contributing to the burden on right occipital and right temporal WMLs. Furthermore, an overweight/obese BMI was indirectly associated with less prevalence of GLB, as mediated by right temporal WMLs. No effect was observed with diabetes mellitus or hyperlipidemia.
The Association Between Vascular Risk Factors and GLB as Mediated by WMLs in Patients With Mild AD.

Abbreviations: AD, Alzheimer disease; CI, credible interval; GLB, getting lost behavior; M, mediator (WMLs); MCMC, Markov chain Monte Carlo; P, posterior distribution; SD, standard distance; X, predictor variable (vascular risk factors); Y, outcome (GLB); WMLs, white matter lesions.
a P < .05.
Discussion
We demonstrated that WMLs were a large risk of GLB in patients with MCI, independent of gray matter atrophy. Specifically, WMLs localized in the right occipital region increased the odds of GLB by 12 times, while right temporal WMLs increased the odds of GLB by 4 times in MCI, regardless of age, gender, severity of global cognitive impairment, and gray matter atrophy. We further observed that hypertension increased the risk of GLB by contributing to the burden of right occipital and temporal WMLs. White matter lesions were not related to GLB in HC or patients with mild AD. These findings support a vascular pathogenesis of GLB that is unique to prodromal dementia.
White matter lesions compromise the integrity of white matter pathways that are crucial for information transfer between cortical–cortical and cortical–subcortical regions. 6 This is particularly detrimental for wayfinding behavior as wayfinding relies on interactions between multiple cognitive functions, including top–down modulatory effects of executive functions on visuospatial processing. 13 Given that compromised white matter pathways contribute to a disconnection syndrome, 32 our findings suggest that GLB in MCI may be a consequence of a disconnection syndrome caused by CVD. We further propose that disconnection in the right occipital and temporal regions pose the greatest risk for GLB, which is consistent with the right hemispheric dominance for visuospatial information processing. 25
The role of occipital and temporal regions in GLB is consistent with previous findings that the allocentric occipital–temporal pathway is the most affected in individuals with MCI and GLB, compared to the egocentric occipital–parietal pathway. 33 The allocentric pathway encodes information about the location of one object with respect to another object, while the egocentric pathway encodes information regarding objects in relation to one’s body. A critical region of the allocentric pathway is the hippocampus, which functions as a cognitive map of objects in the environment. 4 Allocentric deficits are a normal part of the aging process due to age-related decline in hippocampal volume 13 ; however, allocentric deficits are exacerbated in patients with MCI. 34 Our findings extend our understanding of how allocentric deficits contribute to GLB by indicating that WMLs in occipital–temporal pathways may be a key pathogenesis of the association between allocentric deficits and GLB. We note that we did not have allocentric-specific cognitive assessments, only an assessment of global cognition, which was not associated with GLB in MCI. Future research may benefit from investigating performance on allocentric assessments (spatial encoding and retrieval) and its association with GLB and WML in occipital and temporal pathways.
Interestingly, the prevalence of GLB did not differ between HC and patients with MCI, nor did the volumes of WMLs or gray matter. Despite this, WMLs were only associated with GLB in MCI. It is possible that MCI patients had a small burden of AD pathology which may have interacted with WMLs to manifest GLB. Although we observed WMLs were associated with GLB independent of gray matter atrophy, the possible amyloid in MCI may have been a catalyst for WMLs to become a risk for GLB. Meanwhile, for patients with AD, the amyloid burden, and consequently severe gray matter atrophy, may confound the impact of WMLs on GLB. Consistent with this, MCI and mild AD exhibited similar volumes of WMLs; however, WMLs were not associated with GLB in mild AD. Further research is warranted in MCI and mild AD to investigate how amyloid moderates the association between WMLs and GLB.
One modifiable risk factor driving the association between WMLs and GLB in MCI was hypertension. This is a clinically important finding given hypertension is prevalent in 70% of the aging population. 35 Chronic hypertension has detrimental effects on cognitive decline and is a particular risk for executive dysfunctions, 36 which has previously been shown to be a key deficit involved in GLB. 13 One mechanism by which hypertension contributes to cognitive impairment is through CVD, 13 which is consistent with our finding that WMLs mediated the effect of hypertension on GLB in MCI. Other mechanisms by which hypertension may affect cognition may involve blood–brain barrier dysfunction or the formation of free oxygen radicals. 37 We also found an overweight/obese BMI was negatively associated with GLB, which may have been a function of immobility. Diabetes and hyperlipidemia were not associated with GLB, despite previous research indicating that diabetes and hypertension exert the same strength of effect on cognitive impairment. 38
Implications
Our findings suggest that interventions for GLB in patients with MCI may involve reducing WMLs and hypertension. This is particularly promising given WMLs are potentially reversible with optimal blood pressure control 39 or lifestyle changes such as exercise. 40 As we observed large effect sizes between WMLs and GLB, we propose that slowing the progression of WMLs may lead to an observable benefit on the prevalence of GLB in MCI.
Strengths and Limitations
One strength of our study is that we demonstrated the association between WMLs and GLB independent of gray matter atrophy in 3 different ways: Firstly, volumes of gray matter were consistent across MCI patients with GLB and no GLB, despite volume of WMLs being greater in patients with GLB; secondly, WMLs and gray matter did not interact to affect GLB suggesting their effects are not dependent on each other; and thirdly, WMLs were still significantly related to GLB after controlling for regional gray matter volumes. However, we do acknowledge that WMLs are dynamic and may interact with other pathophysiological processes such as secondary axonal degeneration and amyloid pathology. 2 Although we observed that WMLs did not interact with gray matter to affect GLB, future longitudinal research with microstructural imaging is required to determine whether WMLs and other pathologies interact to affect GLB. In this study, we indexed CVD as WML given it is the most reliable MRI surrogate for CVD 8 and a common target for intervention. 6 Our significant findings support investigation into the impact of other CVD markers and GLB, including lacunes and microbleeds. A further limitation is the modest sample size; however, the large effect size observed between WMLs and GLB in MCI indicates a larger sample size may provide limited additional benefits or findings.
Conclusion
To our knowledge, we provide the first evidence that WMLs are a primary pathophysiology of GLB in prodromal dementia. Theoretical implications alter our understanding of GLB as a disconnection syndrome caused by CVD. Interventions aimed to prevent WMLs by optimizing hypertension control as well as lifestyle modification may be beneficial in reducing GLB.
Supplemental Material
Supplementary_material_Final - Cerebrovascular Disease Is a Risk for Getting Lost Behavior in Prodromal Dementia
Supplementary_material_Final for Cerebrovascular Disease Is a Risk for Getting Lost Behavior in Prodromal Dementia by Chathuri Yatawara, Kok Pin Ng, Levinia Lim, Russell Chander, Juan Zhou and Nagaendran Kandiah in American Journal of Alzheimer's Disease & Other Dementias
Footnotes
Authors’ Note
C.Y. contributed to the study conception and design, analysis of data, and drafting the manuscript. K.P.N. contributed to the conception and design of the study and revising the manuscript for intellectual content. L.L. and R.C. contributed to acquisition of data and drafting the manuscript. J.Z. contributed to revising the manuscript for intellectual content. N.K. contributed to the conception and design of the study and revising the manuscript for intellectual content.
Acknowledgments
The authors thank the team at the National Neuroscience Institute for assistance with data collection and supporting this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A*STAR Biomedical Research Council, Singapore (BMRC 04/1/36/372 to J.Z. and N.K.).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
