Abstract
Background/Rationale:
This systematic review aims to evaluate the efficacy of the nonpharmacological interventions reducing burden, psychological symptoms, and improving quality of life of caregivers of individuals with mild cognitive impairment (MCI).
Method:
Databases reviewed included Medline, Cochrane Library, Embase, PsycNet, AgeLine, and ProQuest Dissertations and Theses. Studies using an experimental/quasi-experimental design including nonpharmacological intervention were included. Four studies were included, and no meta-analysis was conducted.
Results:
Calendar training and note-taking (cognitive intervention) significantly decreased caregiver’s depressive symptoms and prevented worsening of subjective burden 6 months posttreatment. Daily engagement of meaningful activity combined with problem-solving therapy and educational material reduced depressive symptoms 3 months posttreatment. Moreover, educational intervention and social conversation phone calls decreased caregiver burden 3 months posttreatment.
Conclusion:
Studies suggest that nonpharmacological interventions can support caregivers of older adults with MCI, but the few published articles present some bias and are inconclusive. Randomized-controlled trials targeting specifically caregivers are needed to determine the most efficient type of interventions for those individuals.
Keywords
Introduction
Mild cognitive impairment (MCI) is characterized by deficits in cognitive functioning in different domains (ie, controlled attention, executive functioning, learning and memory, language, sensorimotor functions, and social cognition). Reduced cognitive functioning in MCI cannot be simply attributed to aging, and cognitive decline does not have a significant impact on everyday life. 1 When the domain of learning and memory is affected, more specifically when episodic memory is impacted, MCI can be considered as a probable prodromal phase of Alzheimer’s disease (AD). Although individuals with MCI are still able to function independently in most activities of daily living, they frequently require some level of assistance and support from family members, named caregivers. 2 Caregiver is defined as any family member or friend providing instrumental or psychological support to another individual, without receiving any financial compensation. 3
The relationship between an individual with MCI and his or her caregiver can sometimes be difficult, especially for the person providing assistance. Indeed, 36% of caregivers of people with MCI report a clinically significant burden. 4 This burden implies emotional, physical, social, and financial consequences. The concept of burden includes 2 dimensions: (1) objective burden refers to concrete problems resulting from daily care (eg, time per day) directed to the individual receiving care or the impossibility for the caregiver to devote time for himself or herself and (2) subjective burden refers to the caregiver’s perception of caregiving, attitudes, and feelings (eg., depression, anxiety, and guilt). 5 Objectively, caregivers of individuals with MCI assume new responsibilities such as financial planning and grocery shopping, and they entrust a binding lifestyle. 2 Subjectively, caregivers suffer when they witness their significant other having memory deficits 6 ; caregivers report increased psychological distress (ie, depressive and anxiety symptoms) 6 and a diminished quality of life (QoL). 7
Caregivers express a need to be supported when providing care and support to individuals with MCI. 8 As a result, different areas of intervention emerged to help caregivers reduce their burden and improve their QoL. Recently, cognitive, psychological, and multidimensional interventions have successfully reduced the burden of MCI caregivers. 9 -11 However, the majority of these interventions target the individual with MCI directly, and the impact of treatment on the caregiver is subsequently measured. Cognitive interventions include cognitive training, cognitive remediation, and cognitive rehabilitation. 12 The rationale of these types of interventions in the context of this systematic review is that the impaired functioning of elderly people with MCI predicts subjective burden of caregivers. 13 Therefore, it is plausible that improved functional abilities of people with MCI reduce the burden of their caregivers. Indeed, Greenaway and colleagues 9 trained individuals with MCI to use a calendar and to take notes (cognitive remediation). The impacts of this type of intervention were measured; the intervention successfully increased caregivers’ mood and had a protective effect against the caregiver’s subjective burden at 6-month follow-up. Psychological interventions include psychological support interventions, psychoeducation, psychotherapy, and respite care. 14 For example, a study using problem-solving therapy (psychotherapy) significantly reduced caregivers’ depressive and anxiety symptoms. 10 Finally, other researchers studied the effect of multidimensional interventions for individuals with MCI and their caregivers. In particular, a study using cognitive–behavioral therapy (CBT) combined with certain components of cognitive rehabilitation and psychoeducation has shown to improve caregivers’ feelings of self-efficacy 6 to 8 months postintervention. 15 In addition, a cognitive intervention combined with psychoeducational activities reduced the burden of caregivers for which individuals with MCI responded positively to the intervention. 16
Up to now, no systematic review of the literature regarding the effect of nonpharmacological interventions on MCI caregivers’ burden was done. Due to an increasing number of studies on the topic, a review of the literature is warranted to identify those interventions that show the most promise. This systematic review will clarify the efficacy of nonpharmacological interventions available to date. It also suggests clinical recommendations to improve support for caregivers of individuals with MCI, and it proposes avenues for future research on the topic.
Accordingly, the overarching objective of this systematic review is to examine the efficacy of nonpharmacological interventions (ie, cognitive, psychological, physical exercise, and nutritional interventions) on burden, psychological symptoms, and QoL of MCI caregivers. To our knowledge, there does not appear to be a previous study measuring the direct or indirect impact of nutritional or physical exercise programs on caregivers, but these 2 types of interventions were nevertheless included in the present systematic review for exploratory reasons. Since these interventions may improve cognition of individuals with MCI, they may in turn positively impact caregivers’ burden. Overall, we hypothesized that nonpharmacological interventions directly administered to the caregiver or to the individual with MCI significantly reduce caregivers’ burden and psychological symptoms as well as improve their QoL.
Method
Literature Review and Articles Selection
The articles were obtained using MedLine, Cochrane, Embase, PsycNet, AgeLine, and ProQuest Dissertations and Theses databases. Since the year 1999 is the year of the first publication including MCI diagnostic criteria, articles were retrieved for the period between January 1999 and September 2017. 17 The literature search using key words was conducted. The key words were combined and included (1) caregiver (or equivalent words: carer, dyad, family, friend, significant other, care-partner, partner, spouse); and (2) MCI (or equivalent words: cognitive decline, cognitive disorder, cognitive deficit, cognitive impairment, memory disorder, memory loss, memory problem, pre-Alzheimer, preclinical AD, preclinical Alzheimer, predementia state, prodromal, prodromal Alzheimer, progressive cognitive decline); and (3) nonpharmacological intervention (or equivalent words: intervention, nonpharmacological therapy, nonpharmacological treatment); or (4) cognitive intervention (or equivalent words: cognitive learning, cognitive rehabilitation, cognitive stimulation, cognitive training, memory rehabilitation, memory stimulation, memory training, neuropsychological intervention, neuropsychological stimulation, neuropsychological therapy); or (5) psychotherapy (or equivalent words: behavior intervention, behavior therapy, behavior treatment, cognitive therapy, cognitive behavior intervention, cognitive behavior therapy, psychodynamic psychotherapy) or mind-body therapy (or equivalent words: meditation, mindfulness, relaxation) or counselling or psychoeducation or respite or support; or (6) physical activity (or equivalent words: aerobic, balance, cycling, exercise, flexibility, gym, motor activity, physical endurance, physical performance, physical program, physical therapy, physical training, run, strength, swim, tai chi, walk, yoga); or (g) nutrition (or equivalent words: calcium, diet, plant extract, plant oil, regimen, vitamins); and (7) burden, caregiver health or quality of life: (or equivalent words: anxiety, depression, caregiver distress, caregiver outcome, CES-D, mood, neuropsychiatric inventory, psychiatric status rating scale, RAND 36-Item Health Survey (RAND-36), Revised Memory and Behavior Problems Checklist (RMBPC), self-care, sense of competence questionnaire, severity of illness index, stress, well-being); and (8) randomized trial (or equivalent words: controlled clinical trial, controlled study, experimental study, randomized clinical, randomized control) or quasi-experimental study or longitudinal study or pilot project or preliminary study or pre intervention (or equivalent words: pretest–posttest, pre and postintervention, pre and posttreatment, pretreatment). To complete the search, references in systematic reviews and meta-analyses on nonpharmacological interventions for individuals with MCI or caregivers were also considered.
The literature search was completed with the help of a librarian (specialized in psychological literature searches) on September 24, 2017, and 4342 articles were initially retrieved. The first author (NSD) examined titles and abstracts to exclude any articles that could not be included, leading to the selection of 193 articles. Then, the first 2 authors (NSD and PV) independently verified the admissibility of each selected article according to predetermined inclusion and exclusion criteria.
Inclusion and Exclusion Criteria
To define the inclusion and exclusion criteria, authors used the PICOS search strategy as recommended by the statement. 18 This method focuses on generating criteria according to 5 categories: P, participant; I, intervention; C, comparators (ie, control group); O,: outcome; and S, study design. No criteria for comparators category were used to promote the inclusion of relevant studies. Studies with or without control group were included. All the articles included in the systematic review had to satisfy the following criteria.
Participants
The target population included individuals with MCI (amnestic or not) and/or their caregiver. Individuals with MCI were identified according to published diagnostic criteria. 1,17,19 -23 All articles including participants with MCI for whom the cognitive deficits may be due to a psychiatric or a medical condition were excluded. Articles including participants with MCI and AD were included only if the results for each group were separately reported; the articles were excluded otherwise.
Interventions
Participants with MCI or their caregivers needed to receive a nonpharmacological intervention, defined as a cognitive, psychological, nutritional, or physical exercise program. Interventions could be administered individually to the participant or in groups. The duration of the treatment and the time of each session were not specified as exclusion criteria.
Cognitive interventions had to include a cognitive training, remediation, or rehabilitation intervention defined according to the criteria of Clare and colleagues. 12 Psychological interventions had to include support intervention, psychoeducation, psychotherapy, or respite care as defined by Sorensen and colleagues. 14 Studies including meditation and relaxation interventions were additionally included. Physical exercise interventions had to include a physical training program, a physical rehabilitation program, or recreational activities incorporating resistance, cardiovascular, balance, or stretching elements as defined by Orgeta and colleagues 24 and by van Uffelen and colleagues. 25 Finally, nutritional interventions had to incorporate any change regarding alimentation such as supplements and diets. All interventions could be administered solely or in combination (eg, cognitive intervention including psychotherapy elements).
Outcomes
Only articles reporting quantitative data regarding the efficacy of the intervention directly measured on caregivers were included. These measures included burden, psychological symptoms (ie, psychological health), and/or QoL. Burden construct had to be evaluated with tools or scales measuring objective and/or subjective burden. Results had to be reported in terms of a significant difference between the experimental group and the control group or in terms of a significant difference between pre–post measures or follow-up.
Study design
Articles had to use an experimental design (ie, randomized groups) or quasi-experimental (ie, pre–post measures with control group, post measures only with control group, or pre–post measures without control group) and needed to be written in French, English, Spanish, or Portuguese.
Article selection was compared between the first 2 authors as they performed the task independently. When discrepancy occurred, the authors discussed the results until consensus. A total of 4 articles were finally included in this systematic review (for selection process, see Figure 1). One article was excluded due to intervention type: researchers used a Reiki intervention, which is a laying hand technique administered by a peer. 26 Finally, 4 authors did not reply to e-mails requesting additional information to evaluate whether their respective articles met the inclusion criteria of the present systematic review and, consequently, their articles were excluded.
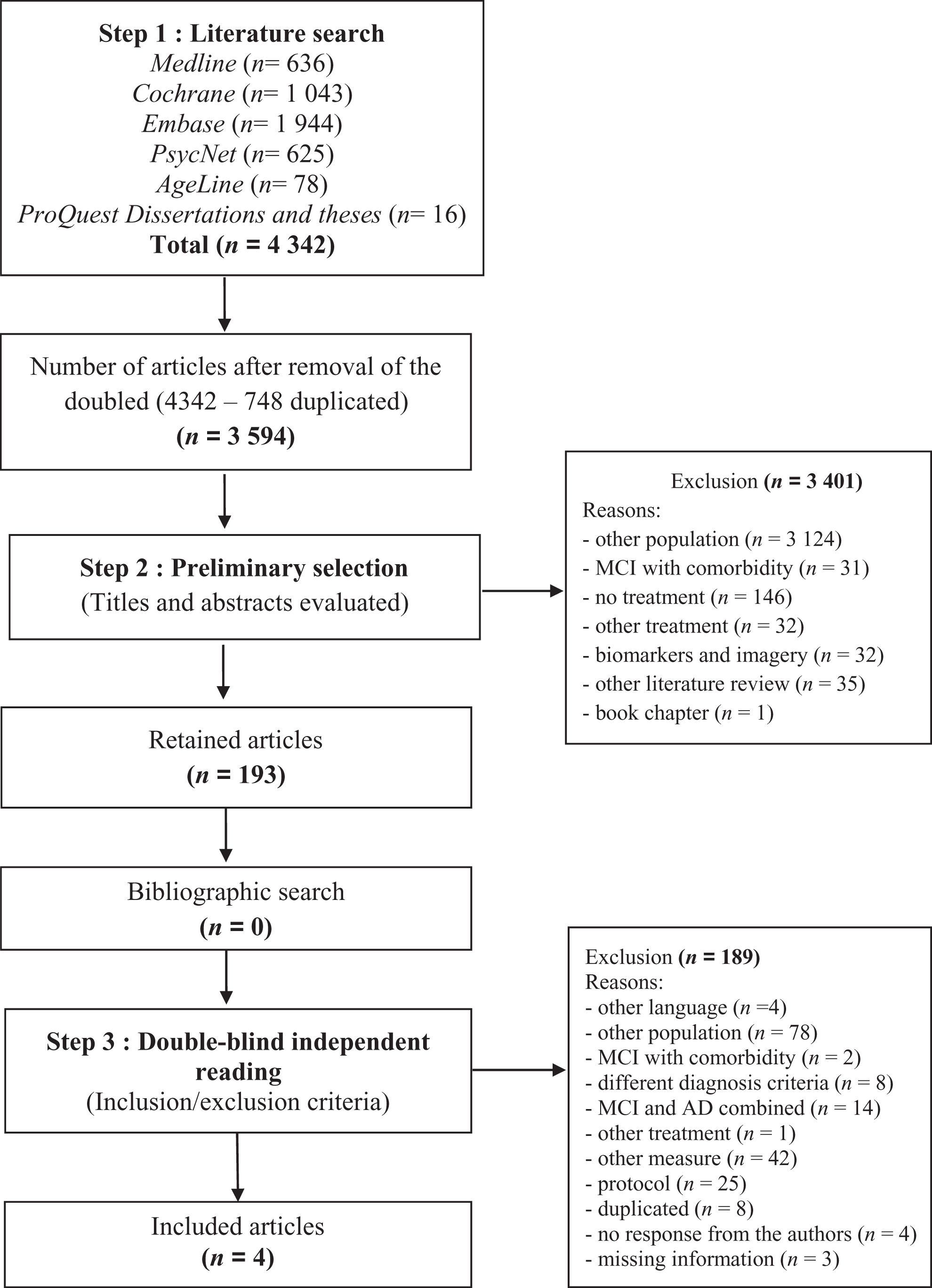
Articles selection flow diagram.
Data Extraction and Risk of Bias
The following information was extracted for every article included in the systematic review: (1) study (design, number of groups, and type of control group), (2) participants (type of participants, sample size, mean age and standard deviation, sex, education, nature of the relation between individuals with MCI and their caregiver), (3) intervention (type of intervention, number and duration of sessions, follow-up measures, and type of control intervention), (4) instruments or questionnaires (name of questionnaire), and (5) quantitative results (means, standard deviations, and effect sizes).
Additionally, each included study underwent an evaluation of the quality of its methodology. An evaluation of the risk of bias was performed using The Cochrane Collaboration’s tool for assessing risk of bias 27 for randomized studies, and the evaluation of nonrandomized studies was performed using the Checklist for the assessment of the methodological quality both of randomized and nonrandomized studies of health-care interventions proposed by Downs and Black. 28 This checklist includes most critical domains for the evaluation of bias and its use is recommended for systematic reviews. 29
The first 2 authors performed data extraction and evaluation of bias independently. Again, all diverging results were discussed until consensus.
Data Analysis
The articles were retained for this systematic review regardless of the results of the study. Effect sizes were calculated for all significant results when possible. Cohen d effect size with a 95% confidence interval was used to compare standardized mean scores small effect: d = 0.20 à 0.49; moderate effect: d = 0.50 à 0.79; large effect: d = 0.80 à 1.00; very large effect: d = 1.01 and more. 30
No quantitative meta-analysis of the results was performed due to the methodological heterogeneity of the selected articles.
Results
Three randomized 9,31,32 and 1 nonrandomized 11 control trials, for a total of 223 caregivers, satisfied the inclusion criteria of this systematic review. Characteristics of these 4 studies are detailed in Table 1. Measures are presented in Table 2 and obtained results are presented in Table 3.
Articles’ Characteristics.
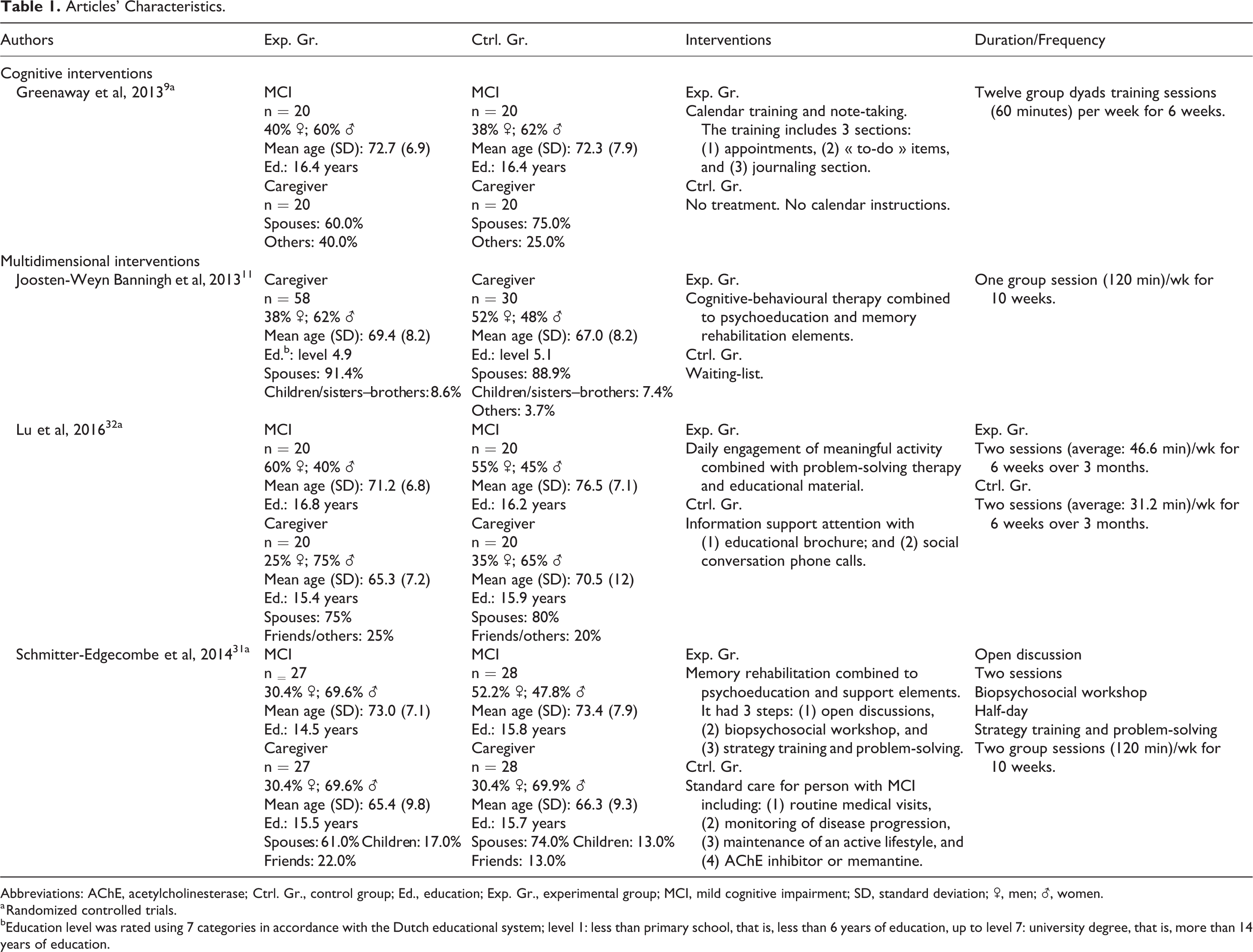
Abbreviations: AChE, acetylcholinesterase; Ctrl. Gr., control group; Ed., education; Exp. Gr., experimental group; MCI, mild cognitive impairment; SD, standard deviation; ♂, men; ♀, women.
a Randomized controlled trials.
bEducation level was rated using 7 categories in accordance with the Dutch educational system; level 1: less than primary school, that is, less than 6 years of education, up to level 7: university degree, that is, more than 14 years of education.
Efficacy Measures Utilized in the Studies.
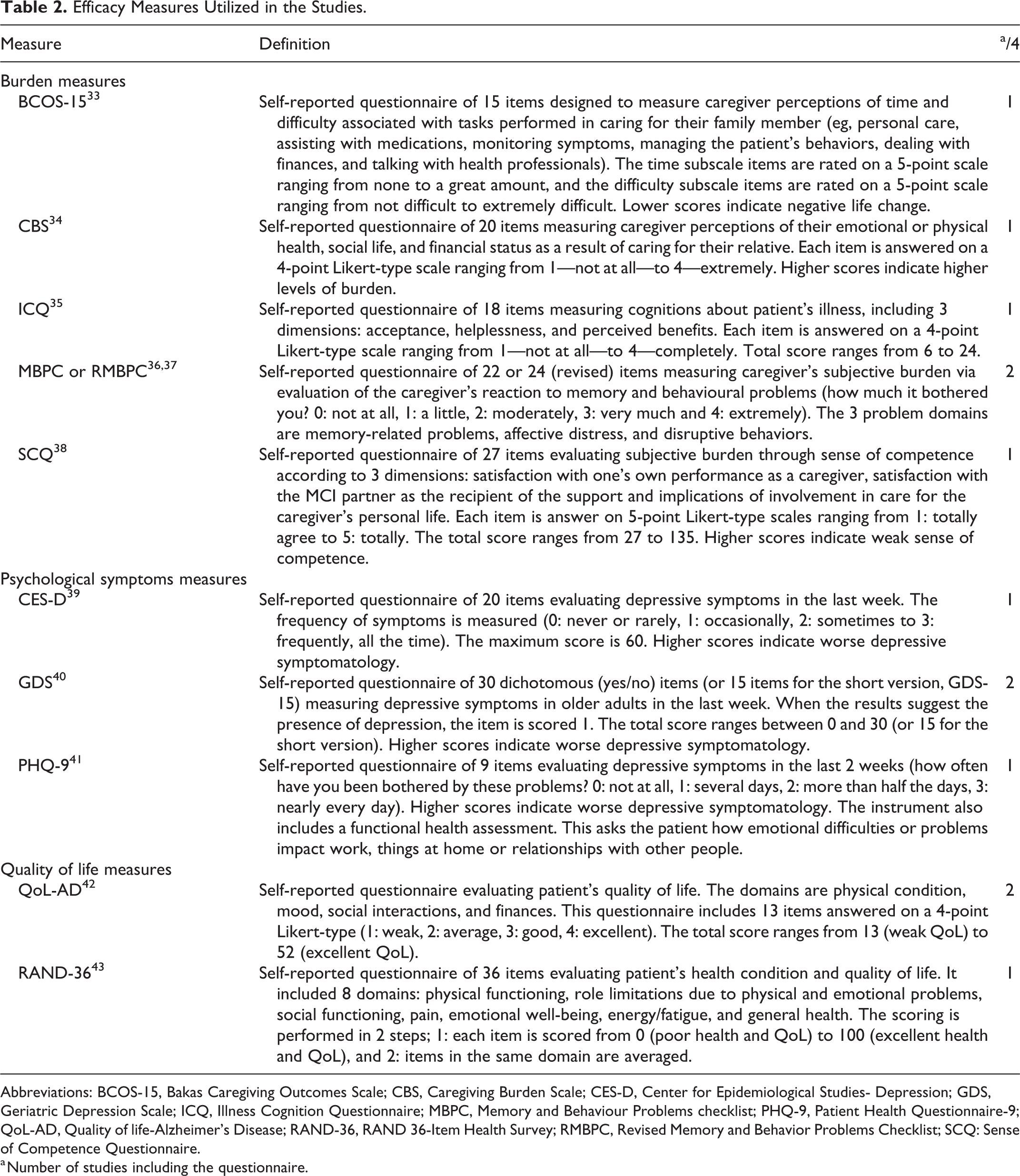
Abbreviations: BCOS-15, Bakas Caregiving Outcomes Scale; CBS, Caregiving Burden Scale; CES-D, Center for Epidemiological Studies- Depression; GDS, Geriatric Depression Scale; ICQ, Illness Cognition Questionnaire; MBPC, Memory and Behaviour Problems checklist; PHQ-9, Patient Health Questionnaire-9; QoL-AD, Quality of life-Alzheimer’s Disease; RAND-36, RAND 36-Item Health Survey; RMBPC, Revised Memory and Behavior Problems Checklist; SCQ: Sense of Competence Questionnaire.
a Number of studies including the questionnaire.
Results.

Abbreviations: BCOS-15, Bakas Caregiving Outcomes Scale; CES-D, Center for Epidemiological Studies-Depression Scale; Ctrl. Gr., control group; Exp. Gr., experimental group; GDS-15, Geriatric Depression Scale; ICQ, Illness Cognition Questionnaire; MC, mean change; NS, nonsignificant; PHQ-9, Patient Health Questionnaire-9; pre, pretreatment; post: posttreatment; QoL-AD, quality of life-Alzheimer’s disease; RAND-36, RAND 36-Item Health Survey; RMBPC, Revised Memory and Behavior Problems Checklist; SCQ, Sense of Competence Questionnaire; TS, total score; vs, versus.
Cognitive Interventions
The impact of cognitive interventions was evaluated only in a single study by Greenaway and colleagues. 9 This study included 40 older adults with MCI along with their 40 caregivers. There were no exclusion criteria to be considered for a caregiver except to be in contact at least twice a week with their program partner. A total of 67.5% caregivers were spouses, and the relationship was not specified for 32.5% of caregivers.
The authors focused on the efficacy of a calendar and note-taking rehabilitation intervention compared to a control group receiving a calendar without any specific instructions. The calendar included 3 sections (1) appointments, (2) “to-do” list, and (3) taking notes concerning important daily events and activities that could include a phone call received, thoughts, information concerning family members, and so on. Intervention group dyads received a total of twelve 60-minute training sessions over 6 weeks, with homework routinely assigned to encourage practice.
The Greenaway and colleagues’ study measured the efficacy of the cognitive intervention on subjective burden, depressive and anxiety symptoms, and QoL of MCI caregivers. 9 Change in mood (ie, depressive symptoms as measured by CES-D) was better in caregivers in the intervention group compared to the caregivers in the control group. The effect was significant at 8 weeks (medium effect size; d = 0.75) and 6 months (extra-large effect size; d = 1.03) postintervention. Compared to baseline, caregivers of the intervention group reported a decrease in depressive symptoms 6 months following the intervention (total score at CES-D; small effect size; d = 0.39), which was not reported for caregivers in the control group. Moreover, caregivers’ mood in the control group tended to get worse at 6 months postintervention (nonsignificant result; P = .06).
The intervention did not significantly impact the subjective burden (as measured by the Zarit Burden Interview), QoL (as measured by the QoL-AD), and anxiety (instrument not reported) of caregivers. However, a small effect size (d = 0.32) was reported; caregivers’ subjective burden worsened in the control group 6 months post-intervention.
Multidimensional Interventions
The 3 other studies tested the efficacy of multidimensional interventions. 11,31,32 Banningh et al 11 evaluated the efficacy of CBT combined with cognitive rehabilitation and psychoeducation components. The study included 88 caregivers (55% women; mean age = 68.2 years old), and no exclusion or inclusion criteria were specified for these participants. In the sample, 90.2% of them were spouses, 8.0% were relatives (ie, brothers, sisters, or children), and 1.8% of caregivers had an unspecified relation with the care recipient.
This intervention focused on acquisition of new strategies to cope with MCI symptoms and consequences, learning to recognize memory problems in everyday life, communication with caregivers, and self-regulation skills. The following themes were covered during the meetings: memory function, MCI as a clinical label, available resources and interventions, strategies to enhance memory, ways to recognize strain, learning to relax, the importance of undertaking pleasant everyday activities, and coping with social conflicts and worries. Participants were invited to recognize and cognitively restructure dysfunctional self-evaluations and negative social and unduly anxious cognitions. The intervention consisted of 10 weekly 120-minute group sessions. The control group consisted of a waiting list. Overall, results demonstrated that CBT combined with cognitive rehabilitation and psychoeducation components did not significantly reduce caregivers’ subjective burden (as measured by the RMBPC—frequency and hindrance subscales; the Illness Cognition Questionnaire, Sense of Competence Questionnaire), depressive symptoms (as measured by the Geriatric Depression Scale; GDS-15), or QoL (as measured by the RAND 36-Items Health Survey; RAND-36) 2 weeks postintervention.
Lu and colleagues 32 explored the benefits of the Daily Engagement of Meaningful Activities (DEMA) tool combined with problem-solving therapy that included some psychoeducation elements. This study included 40 patient–caregiver dyads (caregiver: 70% women; mean age = 67.9 years). In the sample, 77.5% of caregivers were spouses, and 22.5% were friends or others. The intervention included 2 main components: (1) engagement in meaningful activities, autonomy support, problem solving, goal achievement, and emotional needs articulation and (2) self-management tool kit including educational material to understand and deal with individuals with MCI. The DEMA was tailored according to dyads’ awareness of congruence in functional abilities, types and frequencies of meaningful activity, and barriers to engage in these activities. This intervention consisted of 6 biweekly sessions over 3 months. The average duration of each session was 47 minutes. The information support (IS) attention control group received 2 face-to-face meetings and obtained an Alzheimer’s Association MCI educational brochure, along with 4 biweekly follow-up conversation phone calls to ask questions about this brochure. Measures were administered at baseline (time 1), 2 weeks (time 2), and 3-months after the intervention (time 3).
The IS intervention demonstrated a significant moderate effect size (d = −0.50) on depressive symptoms (measured by Patient Health Questionnaire-9; PHQ-9) at time 2 and a small effect size (d = −0.46) on burden (measured by Bakas Caregiving Outcomes Scale) at time 3 relative to DEMA. A small effect size (d = 0.28) was reported regarding the reduction in caregivers’ depression in DEMA group 3 months postintervention. Subset analyses were conducted for patients with MCI and caregivers with depressive symptoms. Compared to the IS group, DEMA did not significantly impact caregivers’ depressive symptoms at time 3 or burden at time 2. The IS group had more positive life changes (less burden) at time 3 (significant moderate effect size; d= −0.77). However, caregivers in DEMA group did show a decrease in depressive symptoms 3 months after the intervention compared to baseline (significant very large effect size; d = 1.55).
Schmitter-Edgecombe and Dyck 31 included 55 older adults with MCI and their respective caregivers. No inclusion or exclusion criteria were predetermined for the caregivers. The sample included 69.8% women, mean age was 65.9 years, and mean education level was 15.6 years. Caregivers included 67.5% spouses, 15.0% children, and 17.5% friends. In this study, authors measured the efficacy of a cognitive rehabilitation focused on episodic memory combined with psychoeducation elements. Specific support intervention for older adults with MCI and their family was also added to the intervention. The intervention group was compared to a control group receiving standard care. This multidimensional intervention included 3 parts: (1) The first 2 sessions included open discussions to develop complicity and obtain information about participants’ competences and needs, (2) the next session involved a half-day biopsychosocial workshop to acquire knowledge about changes within the brain, memory deficits, and their impact on daily functioning. This workshop also targeted participants’ interest in learning new strategies to cope with memory deficits and to reduce distress and social isolation, and (3) utilization of a 3-section notebook (ie, to-do list, monthly calendar and personal notes), together with problem-solving strategies acquisition and implementation of these strategies in everyday life. This last part included 2 weekly 120-minute group meetings for 10 weeks. Standard care included routine medical visits, monitoring of disease progression, maintenance of an active lifestyle, and, in some cases, the administration of acetylcholinesterase inhibitors or memantine.
The results of Schmitter-Edgecombe and Dyck 31 revealed that their cognitive/psychoeducation intervention did not significantly impact caregivers’ depressive symptoms (as measured by the GDS-15) or QoL (as measured by the QoL-AD). Moreover, caregivers in the intervention group did not show a decrease in depressive symptoms or an increased QoL 2 weeks after intervention compared to baseline.
Nutrition and Physical Exercise
No studies retained for this systematic review analyzed the impact of nutritional or physical exercise interventions on caregivers of older adults with MCI.
Risk of Bias
Risk of bias of randomized controlled trials and nonrandomized control trials is presented in Tables 4 and 5, respectively.
Bias of Randomized Controlled Trials.
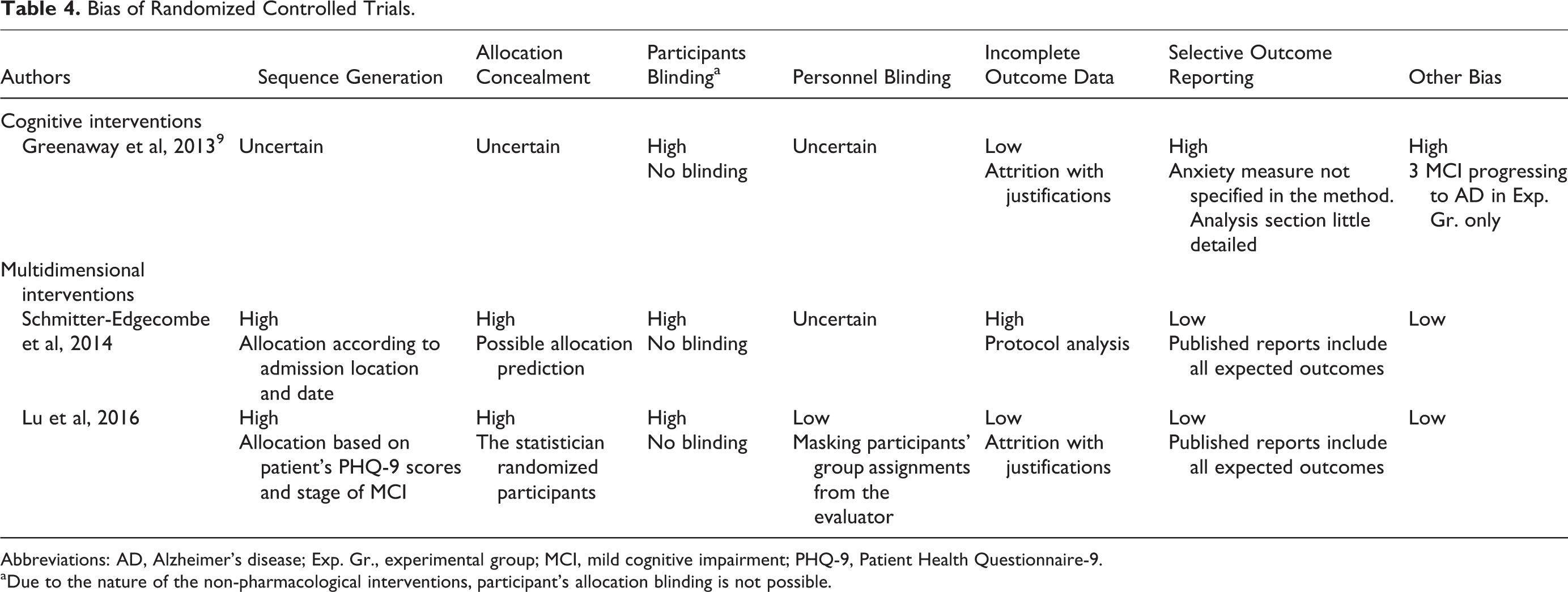
Abbreviations: AD, Alzheimer’s disease; Exp. Gr., experimental group; MCI, mild cognitive impairment; PHQ-9, Patient Health Questionnaire-9.
aDue to the nature of the non-pharmacological interventions, participant’s allocation blinding is not possible.
Nonrandomized Control Trial Bias’s Risk.
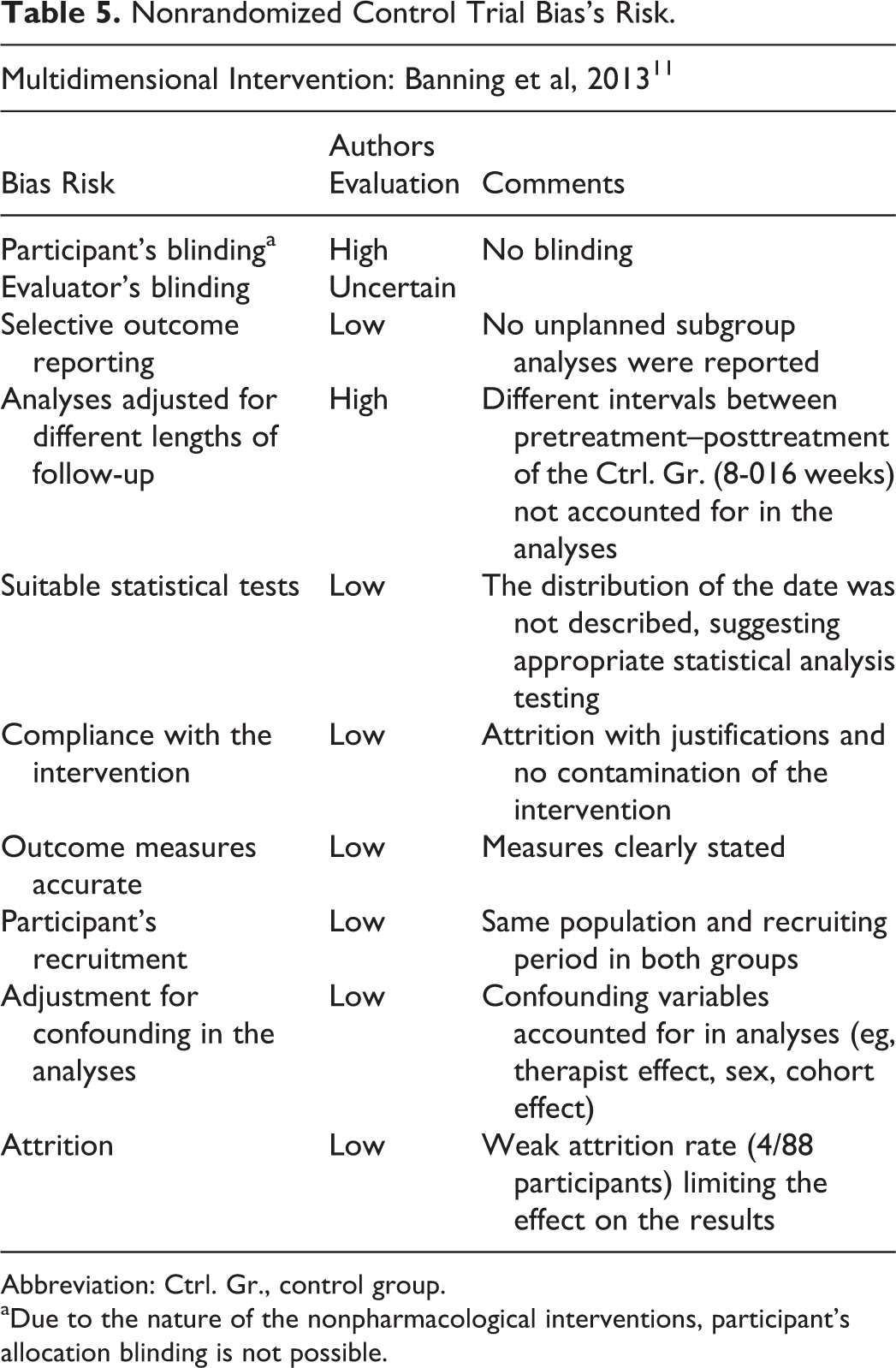
Abbreviation: Ctrl. Gr., control group.
aDue to the nature of the nonpharmacological interventions, participant’s allocation blinding is not possible.
For the study of Greenaway et al, 9 the risk of bias associated with sequence generation and allocation sequence concealment were judged uncertain as a result of the absence or improper description of the methods. Due to the nature of the intervention, blinding of participants to the allocated interventions was impossible, and bias was judged high. The available information regarding evaluator blinding was not sufficient, and bias was therefore judged uncertain. Attrition rate and justification of attrition were explained for both the groups, suggesting a low risk of bias for incomplete outcome data. However, the authors reported results for a measure of anxiety, which was not mentioned in the method beforehand. Therefore, reporting bias for selective outcome was judged to be high. Finally, 3 participants in the intervention group developed AD before beginning the program, leading to uneven groups at baseline.
In the study of Joosten-Weyn Banningh, 11 the information regarding blinding of evaluators was not sufficient, and the bias risk was uncertain. Participants’ blinding was not possible, and risk of bias was high. Duration variability across pre–post intervention periods of the control group was not considered in the analyses. All other possible sources of bias were judged low (ie, participants’ recruitment, outcome measures, selective outcome reporting, appropriate statistical analyses, confounding variables, attrition, and compliance with the intervention).
Lu and colleagues 32 described a nonrandomized component in their sequence generation process (ie, a sequence generated based on the patient’s PHQ-9 score and stage of MCI), resulting in a high bias associated with the sequence generation. Evaluators were blind to the participant’s group assignments. The attrition rate and justification of attrition were explained for both the groups, suggesting a low risk of bias for incomplete outcome data. Every initially planned outcome measure was reported in the results. This study seemed free of any other source of bias.
Finally, Schmitter-Edgecombe and Dyck 31 described a nonrandomized component in their sequence generation process (ie, a sequence generated by some rule based on the date and location of admission), and bias was thus high. Evaluators were blind to the study hypotheses, but the article did not report if they were blind to group allocation. The risk for blinding of outcome assessment was then judged uncertain. Per-protocol analysis was used, thus excluding many participants. Bias associated with incomplete outcome data was high. Finally, every initially planned outcome measure was reported in the results. This study seemed free of any other source of bias.
Discussion
Main Results
The goal of this systematic review was to determine whether nonpharmacological interventions (ie, cognitive, psychological, nutritional, and physical interventions) offered to people with MCI or to their caregiver are efficient in reducing the caregiver’s burden and psychological symptoms and/or to improve the caregiver’s QoL. Four clinical trials focusing on cognitive interventions and multidimensional interventions (including elements from both cognitive and psychological intervention) were included in the systematic review, 9,11,31,32 3 of which were randomized control trials. 9,31,32 Overall, the nonpharmacological interventions included in this study did not have an impact on burden, but 2 studies showed an effect in reducing depressive symptoms of caregivers.
Cognitive Interventions
A single clinical trial using a cognitive intervention was included. 9 The study showed small to large effect-sizes in reducing depressive symptoms of caregivers in the intervention group, whereas caregiver’s mood tended to worsen after 6 months (nonsignificant results), and their subjective burden became significantly worse. These results are important since depressive symptoms may exacerbate burden of caregivers and reduce their QoL. A reduction in depressive symptoms reflects the caregiver’s more optimistic and hopeful feelings with regard to their situation, and perhaps feel less tired and irritable, which are all positive effects of this intervention. However, the cognitive intervention did not have a significant impact on caregivers’ other outcome measures. According to the authors, a possible explanation for 3 results is the inclusion of 3 of 18 participants with MCI who progressed to AD (representing 16.7% of the participants in the intervention group) by the time the authors began the intervention. Since caregiver’s burden tends to increase in the progression from MCI to AD, the data of these 3 caregivers may have contributed to the nonsignificant results. Additionally, considering the weak compliance rate 6weeks after the program and 6 months after the intervention in the intervention group, the omission of booster sessions may have contributed to nonsignificant subjective burden results.
Three main observations can be reported as follows: (1) calendar training and note-taking reduce depressive symptoms of caregivers of people with MCI on the medium term (6 months); (2) this type of intervention slows down the worsening of caregivers’ subjective burden over the medium term (6 months); and (3) caregivers receiving this cognitive intervention differed from those in the control group on subjective burden and depressive symptoms measures several months postintervention. Considering that 10% to 15% of people with MCI progress to dementia each year 21 and that caregivers’ subjective burden is associated with the care recipient’s functional deficit, 13 caregivers’ well-being may be more likely to worsen if no cognitive intervention is offered to the care recipient.
However, cognitive interventions may not be sufficient to help caregivers of patients with MCI in the short term, because these interventions do not target precisely caregivers’ needs for information about the disease, support, counselling, and respite care. 2 Moreover, no significant results were observed previously on caregivers’ objective burden, depressive symptoms, and QoL, following a cognitive training intervention in patients with dementia. 44 In brief, cognitive interventions are not useless, but should be combined with other approaches in order to alleviate caregivers’ burden or improve their QoL and well-being. Yet, these results are based on a single cognitive intervention, so generalizability cannot be made. Nevertheless, this study had low risk of bias associated with incomplete outcome data which strengthen the results.
Multidimensional Interventions
Three clinical trials evaluating the efficacy of multidimensional interventions (eg, CBT combined with cognitive rehabilitation and psychoeducation elements, 11 cognitive rehabilitation targeting episodic memory combined with psychoeducation and support elements, 31 and utilization of DEMA along with problem-solving therapy and educational component 32 ) were included in the present systematic review. The results of the multidimensional interventions were mixed: 2 studies 11,31 did not show significant results on any of the variables of interest, and 1 study 32 reported significant changes 3 months after the intervention.
According to Banningh and colleagues, 11 the inefficacy of the cognitive–behavioral intervention combined with elements of cognitive rehabilitation may be explained by the measures being collected at an early stage of MCI, hindering the quantification of change regarding subjective burden, mood, and QoL. As the care recipients included in this study received the MCI diagnosis no longer than 16 weeks prior to starting the intervention, it is possible that their caregivers were positioned in an early adaptation phase. This phase is characterized by the identification of stress factors, the elaboration of strategies to adapt to the new situation, and the increase in distress and anxiety. 45 Accordingly, the participants may have been more likely to report lower levels of burden and depressive symptoms. In fact, the baseline mean GDS-15 score for the caregivers was very low in the study of Banningh and colleagues. 11 Although the intervention addressed important issues, it was somewhat unlikely to reduce depressive symptoms (or burden) in persons who were not (or mildly) depressed preintervention. Moreover, the care partners included in this study reported numerous examples illustrating adapted behaviors and appraisals of stressors; 11 caregivers were more alert to stress factors and progressively adapted their behaviors to the problems they encountered. Finally, Banningh and colleagues 11 proposed that the interval between the end of the intervention and the post-treatment measures (ie, 2 weeks) was too short to detect a significant change in the psychosocial measures.
In the study of Schmitter-Edgecombe and Dyck, 31 no significant results of the intervention were detected on objective and subjective burden, depressive symptoms, and QoL scales for caregivers of older adults with MCI, regardless of an improvement in functional abilities among persons with MCI. This study utilized a cognitive rehabilitation method along with psychoeducation components. Perhaps, these results may be explained, since this type of intervention does not specifically target the psychological symptoms and the burden of the caregivers. Moreover, although the memory abilities of the care receiver improved, there was no significant improvement in overall performance of activities of daily living by the care receiver, which may perhaps translate into a similar perceived burden and stable psychological symptoms by the caregivers. The problem described in the previous paragraph regarding the measurement of depression also arose in this study; it is possible that the absence of significant results in depressive measures was due to a floor effect at baseline mean GDS-15 score of the groups ranging from 1.5 to 2.9;. 11,31 Considering that the presence of psychological symptoms was not an inclusion criterion for caregivers, it is difficult to evaluate the short-term effect of the multidimensional intervention on a sample with very few or no psychological symptoms. Furthermore, it is possible that teaching new skills to caregivers would be more useful and efficient with the worsening of cognition of the care recipient because caregivers in this situation need to apply their skills more often. Consequently, the addition of follow-up measures in future studies may detail more accurately the utilization frequency of the taught strategies and their efficacy according to the evolution of cognitive functions among the care recipients.
For the study of Lu and colleagues, 32 DEMA’s caregiver showed a significant reduction in depressive symptoms only 3 months after the intervention (no effect 2 weeks posttreatment). The effect of this intervention appears to be delayed, as one adapts to the new strategies learned in the intervention and then applying them, emphasizing the important of having follow-up measures. Moreover, compared to DEMA, IS intervention seems more effective in reducing burden 3 months posttreatment.
Limitation
The absence of significant results of the interventions on caregivers’ burden, psychological symptoms, and QoL may be partly explained by methodological limitations that reduced the possibility of detecting an effect. First, there were measurement limitations that may explain the inconsistency in the results of the previous studies. For example, 3 different questionnaires were used to evaluate the construct of depressive symptoms of caregivers in the 4 studies included in this review. Additionally, the omission of clear inclusion and exclusion criteria for the minimum level of burden or depression in the caregivers (especially for studies without follow-up measures) may have impeded the detection of significant intervention effects. There is limited generalizability of the results of the 4 published studies due to the fact that recruitment was performed in memory, neurology, or neuropsychology clinics. 9,11,32 Another important problem is the omission of long-term follow-up measures assessing the efficacy of the intervention to prevent the worsening of the caregiver’s burden and psychological symptoms or QoL. 11,31 Also, objective and subjective burdens were not measured consistently in all clinical trials. Considering that they are influenced by different factors and therapeutic elements, 5 it is required to evaluate the effect of the intervention on both measures independently. Finally, the cognitive intervention developed by Greenaway and colleagues 9 did not respond to caregivers’ specific needs because it did not include psychoeducation, support, or counseling elements. 2
To summarize, it is important to remain cautious while interpreting data about the efficacy of nonpharmacological interventions to support caregivers of people with MCI. The number of studies meeting the inclusion criteria of this systematic review was quite small, and due to the small sample size of the previous studies as well as the clinical and methodological variability between them, a meta-analysis was not conducted.
Caregivers
Since this study found only 4 articles meeting the criteria, we may infer that nonpharmacological interventions that successfully reduce burden in caregivers of older adults with dementia, would perhaps be efficient in caregivers of people with MCI. Meta-analyses previously conducted suggest that cognitive–behavioral and respite care interventions have little success in reducing burden of caregiver. 46 Coping strategies intervention has shown to reduce burden of caregivers of people with dementia. 47 Perhaps this type of intervention would also be more successful in reducing burden of caregivers of people with MCI; future studies should test this hypothesis. However, we hypothesize that these effects would only be detected if caregivers present with significant burden or depressive symptoms at baseline.
Recommendations
Regardless of the number of articles included, some recommendations can be suggested for clinical practice. Particularly, training using calendar and note-taking seems effective in reducing caregivers’ depressive symptoms in the intermediate term (6 months) and in delaying the worsening of their subjective burden. It also appears that combining psychological elements to a cognitive intervention can maximize success or efficacy for caregivers of individuals with MCI. 14 Daily engagement of meaningful activity combined with problem-solving therapy and educational material reduce depressive symptoms 3 months post-treatment. Moreover, educational intervention and social conversation phone calls should be offered to reduce caregiver burden. 32 Finally, care must be taken not to overload caregivers with the intervention.
The clinical practice guide named Guideline on supporting people with dementia and their careers in health and social care 48 suggests developing programs that will include psychoeducation interventions, support groups, problem-solving strategies, and information on available resources. These types of programs are more efficient for prevention and alleviation of burden among caregivers of patients with dementia. One could hypothesize that these programs would also be relevant for MCI caregivers, which Lu’s study supports.
Future research may benefit from a standardized definition of the concepts of “caregiver” and “burden” in the context of MCI and suggest measures to quantify the efficacy of interventions on caregivers. These definitions would standardize clinical trials and increase the possibilities of achieving a meta-analysis. Additionally, randomized control trials with larger sample size are required to determine the most efficient interventions. Future studies should focus on identifying the therapeutic components of the interventions (and their combinations) to maximize support for caregivers of older adults with MCI. In addition, future nonpharmacological interventions should specifically and directly target caregivers’ subjective and/or objective burden. Finally, the systematic inclusion of control groups and long-term follow-up measures are essential to quantify the immediate and long-lasting benefits of the interventions. They should also be carried out while considering the worsening of cognition of the care recipient with MCI.
Footnotes
Acknowledgments
The authors would like to thank Christine St-Pierre for her precious advice concerning research methods, as well as Drs Linda Garand and Birgitte Schoenmakers who provided additional information to verify the inclusion of their article in the present systematic review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Carol Hudon was supported by a salary award from the Fonds de recherche du Québec – Santé.
